Digging Into 1,1,2-Trichlorotrifluoroethane: Past, Present, and What Comes Next
Historical Development
Kickstarting the story with the old days, 1,1,2-Trichlorotrifluoroethane—known for years under the banner of CFC-113—sits right in the middle of a sweeping arc: industry convenience meets environmental wake-up call. Chemists first got serious about this compound before World War II, exploring new ways to keep machinery running smoothly and electronics squeaky clean, even as the groundwork got laid for large-scale chemical production. The boom after the war supercharged its use in degreasing and solvent tanks around the world. As the decades rolled, the stuff got a reputation for being an efficient cleaner—fast, strong, and able to power through grease without messing around with the metals or plastics underneath. The industry didn’t blink much about what happened to the vapors that floated off into thin air because the focus was always on keeping equipment running and processes humming. No one outside research labs talked much about the ozone back then. This attitude defined production and usage for a long stretch, right up until the late 1970s, when the tide started turning: science showed that halogenated compounds like this didn’t just fade away, they drifted to the upper atmosphere and helped break down ozone, opening society’s eyes to bigger consequences than spot-free parts.
Product Overview
You walk into any shop where folks fix up electronic equipment or precision machinery, and they’ll tell you how CFC-113 helped keep their work top-notch. It’s a colorless, almost odorless liquid at room temperature. As a solvent, it clears grime and grease without melting plastics—a big plus, since a lot of alternatives will eat away at wiring insulation or delicate components. Over years of use, people found not many chemicals could replace it with the same balance of effectiveness and low flashpoint, which meant workers stood less chance of fire risk during a job. This single trait kept it popular in military and aerospace operations where reliability matters more than anything.
Physical & Chemical Properties
This compound boils off just above normal room temperature—about 48 degrees Celsius—with a density heavier than water and a low surface tension. Such attributes add up to a formula that can sneak into tight spaces and strip away oil, dust, or soldering flux without getting trapped in little crevices. On top of that, it mixes well with other solvents and doesn’t cause corrosion, which puts it on a short list of “go-to” candidates for complex assembly lines and sensitive cleaning. The chemical backbone, a trichlorotrifluoroethane, brings a stiff stability. This stability, for better or worse, is also why it stays in the environment so long.
Technical Specifications & Labeling
Guidelines for handling don’t just come from industrial wisdom—they’re codified through decades of testing. Labels carved out for CFC-113 warn about volatility and stress the need for good ventilation in closed rooms. While many folks never saw direct harm coming from brief handling, workers running cleaning tanks learned early to gear up with gloves and goggles and keep the air flowing. It stays in steel drums or pressure tanks; sunlight and flames spell trouble because the stuff can break down into nastier byproducts. Modern labeling also flags its ozone impact, as required by regulatory agencies after more of the science hit mainstream industry.
Preparation Method
Big plants churn out CFC-113 through halogen exchange reactions. Industrial chemists use trichloroethylene, treat it with hydrogen fluoride, and coax it through heated, controlled chambers to swap hydrogen for fluorine atoms. Refrigeration pioneer Thomas Midgley Jr. set the stage for this kind of synthesis long ago, but making this compound on a scale to feed global cleaning and electronics demands meant consistent engineering and robust controls. These plants ran for decades without pause, sometimes right outside city limits, until the calls for phase-out started echoing in the halls of government.
Chemical Reactions & Modifications
1,1,2-Trichlorotrifluoroethane stands out for how little it likes to react under typical shop or lab conditions. Its halogen-heavy structure locks it down, keeping it from bursting into flames or breaking apart too easily. If you push it with high heat, strong acids, or UV, it does let some pieces go—sometimes spawning more reactive (and hazardous) offshoots like phosgene or hydrochloric acid vapor, which is why engineers built scrubbers and safety systems into degreasing installations. This stubbornness, for years, made folks see it as a technical solution—until they realized that the same stability lets it build up in the stratosphere.
Synonyms & Product Names
This compound wears a handful of different hats. Chemists might call it CFC-113, or sometimes R-113 in refrigeration circles. It appears on safety sheets as 1,1,2-Trichlorotrifluoroethane. Trade names like Freon-113 and Arklone were splashed across drums and cleaning systems in factories worldwide. Anyone working with spare parts or electronics in the late 20th century probably saw all of those names printed on cans or cleaning baths. The different names pointed to similar chemistry, always circling back to the recognizably sharp capabilities of that three-chlorine, three-fluorine carbon skeleton.
Safety & Operational Standards
Folks working with CFC-113 knew to keep it out of their lungs and away from open flames; its vapors can push out oxygen in tight rooms and mess with your central nervous system if you breathe too much. OSHA and NIOSH drew lines on how much should hang in the work air. Industrial job sites kept detectors humming, set up closed systems with recovery for used solvent, and trained techs to handle spills fast. On top of the workplace rules, the Montreal Protocol lined out a cold, hard stop—no more routine releases of CFC-113, pushing industry veterans to rethink entire production lines and cleaning routines.
Application Area
For years, electronics assembly lines swore by CFC-113. It washed away solder paste, oils, and fingerprints from circuit boards without lifting the copper tracks or blurring screen printing. Aircraft makers counted on it to get components flight-ready and clean, where a drop of oil out of place can spark a major problem. Laboratories leaned on it for delicate processes where residue just wasn’t acceptable. In my own hands-on work, trying replacements almost always meant juggling new hazards—flammability, residue, or material damage. Nothing made the pivot away from CFC-113 easy; alternative blends each brought fresh headaches, from regulatory hoops to disappointing results on the bench and factory floor.
Research & Development
As environmental watchdogs and policy makers tightened the rules, research in universities and chemical companies shifted. The goal turned from simply cleaning better to doing so without wrecking the ozone layer—or creating new headaches with toxicity and cost. Teams tested hydrofluorocarbons and newly mixed alcohols and glycol ethers, all in the hunt for that rare compound that checked safety, power, and price. Personally, I watched as some manufacturers turned back to old-fashioned mechanical cleaning—ultrasonic baths paired with detergents—and still, the quest for a “drop-in” replacement keeps simmering. Most matches still fall short on a few of the original’s unbeatable traits, whether due to flammability, cost, or environmental trade-offs.
Toxicity Research
Medical and environmental health crowd knew early that chronic, high-dose vapor exposure could make workers dizzy, give headaches, or, in rare cases, hit the heart’s rhythm with enough exposure. For most shop workers, acute danger sat low unless the air got thick with it, but studies flagged CFC-113 as a long-sticker in the body’s fat and the earth’s reservoirs. Chronic studies in mice and rats pointed toward subtle organ changes, nudging the chemical further onto “use-with-care” lists. Levels found in closed offices and machine shops dropped sharply after the phase-out, proving that regulations made a dent. Still, research keeps tracking environmental and health effects decades after it left the supply chain.
Future Prospects
CFC-113 reflects a crossroads for modern chemistry: how good intentions can sow long-term trouble without careful long-term thinking. Regulations shut down its main markets, but legacy uses pop up now and then—in specialty aerospace or old refrigeration cycles, sometimes under controlled, “critical use” exemptions. Future development circles around two tracks: finding safer, greener solvents for advanced tech, and cleaning up soil and groundwater at old manufacturing sites. Younger chemists work smart to test new blends with minimal global warming and ozone impacts. The steady push to close the cycle—from greener production to final disposal—drives today’s research. As I see younger colleagues in the field, it’s clear that the industry’s been handed a lesson: early wins need a long view, considering not just what works in the lab or factory but how it fits the planet’s bigger story.
What are the main uses of 1,1,2-Trichlorotrifluoroethane?
A Closer Look at Where This Chemical Shows Up
Most people have never heard of 1,1,2-Trichlorotrifluoroethane, but this chemical built a solid niche in industry long before most of us worried about ozone layers. Over the years, I’ve watched it move from labs and factories into hot debates about health and the environment.
Crucial Tool in Cleaning Electronics
Anyone who’s ever popped the cover on an old computer or television might have depended, without knowing it, on this compound. In electronics shops, workers often use it as a solvent to wash away oils, solder flux, and dust from circuit boards. These boards are sensitive—harsh cleaners destroy fragile connections in seconds. Trichlorotrifluoroethane cleans deeply but leaves plastic and metal untouched. Once upon a time, you could find it in nearly every repair department and electronics factory. I remember the sharp, slightly sweet smell from old maintenance closets. It got things cleaner than just about anything else.
Key Player in Refrigeration and Aerosols
Chlorofluorocarbons powered much of the cooling tech that made modern living possible. 1,1,2-Trichlorotrifluoroethane acted as a refrigerant in some refrigeration systems, especially before safer and more environmentally friendly alternatives came along. Besides cooling, it provided the pressure behind many aerosol products. Shampoos, cleaning sprays, shaving creams—many of those classic cans delivered steady, reliable sprays because of its vapor pressure and stability. The chemical let companies put almost any mixture in a can and guarantee a smooth, even spray.
Role in Specialty Metalwork
Metal manufacturing pushed for solvents that cleaned without scratching sensitive surfaces. This chemical answered that call in places like aerospace and precision engineering. It stripped away oils and buffed materials such as titanium and stainless steel. Some labs still reach for it during preparations for sensitive scientific and aerospace applications, but safety guidelines grow stricter every year.
Concerns for Health and Planet
Here’s the hitch: The same stability and persistence that make it great for cleaning also let it hang around in the environment. Scientists and regulators began linking extended use to serious harm, especially to the ozone layer. This connection triggered bans and restrictions across the world. Health-wise, breathing in high amounts can bother lungs and hurt nerves. I’ve known technicians who struggled with dizziness and headaches after working unprotected around strong fumes. Even with proper ventilation, regular exposure builds risk over time.
Looking for Alternatives and Solutions
Shops and factories have chased down replacements like hydrofluorocarbons and natural cleaning agents. Switching keeps workplaces safer and avoids long-term damage to the planet. Still, some industries struggle—the alternatives just don’t match its cleaning power in every case. Upgrading equipment can eat into tight budgets, especially for small businesses. Companies read labels more carefully and check safety data before ordering in bulk. Governments backed stronger rules while funding research into new, greener options.
Chemical use often tells the story of a trade-off: performance versus safety. Experience in repair shops and labs shows that modern industry can live without 1,1,2-Trichlorotrifluoroethane, but it takes real commitment and caution. The health of workers and the planet rests on finding better solutions and taking exposure seriously, even for those stubborn jobs that seem to demand old-school answers.
Is 1,1,2-Trichlorotrifluoroethane hazardous to health?
Getting to Know an Old-School Solvent
1,1,2-Trichlorotrifluoroethane has been an old stand-by in industrial circles. It found its way into degreasers, refrigerants, and lab cleaners. Plenty of mechanics and lab techs, like me back in my undergraduate research days, remember that sharp, peculiar smell. You’d open a canister, and the vapor would just hang in the air. I never thought much of it until later, when safety data sheets started piling up in the lab.
Health Concerns
Breathing in the vapor or touching the liquid may hit harder than you’d guess. According to research from the U.S. Environmental Protection Agency, this chemical can cause headaches, dizziness, and nausea. The Material Safety Data Sheets warn about respiratory tract irritation and potential effects on the central nervous system. Years ago, a coworker told me he’d get lightheaded after standing near the sink where this solvent evaporated. Back then, fume hoods stayed off half the time because nobody wanted the noise.
There’s something worth paying attention to: chronic exposure. Even if you don’t get sick that day, repeated exposure may stress out your liver and kidneys. Lab animals exposed to this solvent got chemical damage in dose-dependent studies. The World Health Organization flags it on their list of occupational hazards, not on the level of benzene, but still nothing to take lightly.
Environmental Fallout
Chemicals like 1,1,2-Trichlorotrifluoroethane have another big problem. They play rough with the ozone layer. Their days in refrigerants and aerosols led to bans under the Montreal Protocol. Before that, nobody cared about the ceiling over our heads. Once those holes started showing up in the news, regulators and scientists paid more attention. Scientists linked this solvent, along with other CFCs, to ozone depletion. Losing ozone means more UV radiation, an uptick in skin cancer, and higher risks for everyone spending time outdoors.
Regulation and Workplace Change
Some governments stepped in and banned this chemical from most commercial uses. I remember the scramble switching to safer alternatives—everything got relabeled and waste drums filled up with “old stock.” These new solvents aren’t perfect, but they don’t punch holes in the atmosphere at the same rate and come with stricter workplace rules. Modern safety guidelines recommend gloves, goggles, and plenty of open-air ventilation. No more free-flowing solvent down the old drains.
What Can We Do?
Open conversations with employers or educators about chemical safety go a long way. If you handle this stuff, use proper PPE and solid ventilation. Substitution stands out as the best fix—replace the hazardous solvent where possible. Push for clear information from suppliers and transparent safety training. If you work in a place that still uses 1,1,2-Trichlorotrifluoroethane, ask about safer alternatives, and point to available government guidelines.
Years after my own lab days, I still see former teammates at the reunion swap stories about those old solvents. The consensus always sounds the same: treat chemicals with respect, push for safer workspaces, and refuse to shrug off the risks just because something’s been around awhile.
How should 1,1,2-Trichlorotrifluoroethane be stored and handled?
Why Handling This Chemical Matters
Few people working outside of certain industries have come across 1,1,2-Trichlorotrifluoroethane, but for those who do, safety has to come before convenience. This compound has, over the decades, shown up in laboratories and industrial applications, mostly as a solvent and refrigerant. The risks aren't always obvious until you’ve seen what can happen when procedures get skipped. Back in my lab days, complacency could turn a routine job into a real mess — not just for equipment, but for health as well.
Storage Tips That Make a Difference
Storing this chemical means finding a cool, well-ventilated spot with locked access. You don’t want it anywhere near heat sources, flames, or direct sunlight. The vapor isn’t just flammable, it displaces oxygen and causes dizziness or worse in closed spaces. Good air flow means any leaking fumes don’t build up and create a risk you won’t see coming. Steel drums or approved chemical containers with secure seals keep leaks from turning into emergencies. In the past, I’ve seen colleagues rely on makeshift containers — that short-cut never ends well.
Safety isn’t just about the physical storage setup. Signs, labels, and up-to-date inventory lists make sure everyone in the building knows what they’re working with and what’s nearby. The EPA and OSHA regulations get updated from time to time, but they always keep the same core: keep incompatible chemicals apart, double-check the seals, and never assume a container got closed properly by the last shift. Even after years working with these substances, I always run a quick inspection at the start of a shift — just one missed sign of corrosion could spell trouble.
Personal Protection Still Comes First
Handling the chemical means gloves, goggles, and lab coats at a minimum. Polyethylene aprons or even face shields help when splashing is possible. Friends of mine skipped gloves for “just a quick job.” The chemical burns that can follow are not worth the risk. It’s important to remember that vapors can build up, so splash-resistant goggles protect the eyes when chemical-resistant gloves shield your hands.
Never work alone with volatile chemicals. Having someone nearby means you have backup in case of a leak or spill. In my own experience, buddy systems prevent small accidents from becoming big ones. Make sure showers and eyewash stations are close — and tested regularly — because chemicals don’t wait until you’re ready for an emergency.
Preventing Spills and Responding Right
Drips or leaks happen even in the best setups. Absorbent pads and neutralizing agents should sit close by. The right spill kit matters more than good intentions — it only takes one attempt to mop up a chemical with paper towels to realize it just spreads the hazard. I’ve seen teams freeze up during a spill, so regular drills give everyone that reflex to reach for the right gear rather than panic.
Waste disposal rules mean no shortcuts. Regulations on trichlorotrifluoroethane waste keep the stuff out of water and air and stop environmental damage. Most cities have strict drop-off points set up, with consequences for trying to pour it down a drain. I’ve watched facilities caught out by small leaks get hit with big fines later. Staying ahead on paperwork and tracking every ounce used or disposed saves headaches and helps protect the neighborhood.
Learning From Others’ Mistakes
Mistakes around chemicals like this don’t just harm people; they shut down operations, trigger investigations, and damage trust with local communities. At every training, the cautionary tales come up — not to scare, but to show these risks are real. All those procedures, from label checks to air monitoring, grew out of lessons learned the hard way.
What are the environmental impacts of 1,1,2-Trichlorotrifluoroethane?
A Chemical From Yesterday, an Issue for Today
People used 1,1,2-Trichlorotrifluoroethane because it could clean electronics and act as a refrigerant. It came in handy where companies wanted to avoid fires or reactions with other chemicals. Folks often didn't think much about what happened to it once it left the factory or sprayed from a can. Now, folks know it shows up in places where it shouldn't—air, water, even soil. Looking around at old research and recent environmental reports, this chemical stands out for more than one reason.
Ozone Depletion: An Invisible Threat
Most people hear about ozone when sunscreen commercials roll. Scientists realized CFCs and related chemicals, such as 1,1,2-Trichlorotrifluoroethane, break down the ozone layer in the upper atmosphere. Ozone keeps out sunburn and cancer-causing ultraviolet rays. Once this chemical drifts up into the stratosphere, sunlight splits it up, freeing chlorine atoms. Each loose chlorine atom wrecks a lot of ozone molecules before drifting away. The result? Folks and wildlife face more UV rays in places where the ozone layer thins out, especially near the poles and at high altitudes. Skin cancers in people climb, and plankton in the ocean take a hit, hurting the whole food web.
Lingering in Air and Water
1,1,2-Trichlorotrifluoroethane hangs around for a long time. Its chemical bonds make it tough to break down. This means it can travel long distances on the wind, ending up as far away as the Arctic. Rain washes some of it from the air, where it leaks into lakes or streams. The U.S. Environmental Protection Agency and European agencies both list it as a persistent pollutant for this reason alone. Wildlife and water supplies face risks not just near factories, but even where nobody used this chemical.
Replacing One Problem with Another
After people woke up to the ozone-damaging problem, global agreements like the Montreal Protocol went into action. Companies stopped making and using most CFCs and related compounds, including 1,1,2-Trichlorotrifluoroethane. Still, old air conditioners and electronics, along with some industrial processes, kept leaking small amounts for years. Replacing banned chemicals sometimes opened the door to new issues. Some alternatives warm the planet even faster than the old chemicals did, since they trap heat in the atmosphere.
Personal Responsibility and Clean-Up
Old cans of electronics cleaner or outdated AC units tucked away in garages still release fumes if tossed carelessly. Some towns hold hazardous waste days so people can drop off chemicals for safe disposal. Simple steps, like bringing old products to these events, help stop more harmful releases. Countries also work through international agreements, which push manufacturers to phase out harmful chemicals and switch over to less dangerous substitutes. By sharing facts and paying attention to local waste rules, anyone can pitch in to keep these old pollutants from coming back.
Looking Ahead
Lessons from this chemical remind folks that not everything invented to solve a problem fades away quietly. Careful regulation and honest talk about safer options matter. Researchers keep a close eye on substitutes to keep from repeating old mistakes. Sometimes doing the right thing just means double-checking what lands in the trash, reading product labels, or reaching out to community disposal programs. Real progress on environmental hazards starts at ground level and spreads outward, neighbor by neighbor.
Are there any alternatives to 1,1,2-Trichlorotrifluoroethane for industrial purposes?
The Need to Swap Out 1,1,2-Trichlorotrifluoroethane
Industries grew up leaning on fluorinated solvents like 1,1,2-trichlorotrifluoroethane, but heavy baggage has followed this compound. Used in electronics cleaning, refrigeration, and precision degreasing, the chemical gained a reputation as convenient—until research showed it damages the ozone layer and lingers too long in air and water. As someone who’s watched lab processes evolve over years, I see fewer workers reaching for this old staple. Most are now checking for safer choices, especially with tighter regulations coming from both national laws and international treaties like the Montreal Protocol.
Alternatives Taking Over Shop Floors
Companies have moved towards other chemicals, but that switch can get complicated. One group of replacements includes hydrofluorocarbons (HFCs), such as HFC-134a. This chemical avoids ozone destruction but doesn’t come without a price: it still contributes to global warming. I’ve talked to maintenance teams who switched to hydrocarbons, like n-propane or isobutane, in some refrigeration setups. These don’t harm the ozone, and their lower cost appeals, but there’s a risk. They burn easily, so ventilation and safety upgrades become non-negotiable.
For cleaning electronics or high-precision metal parts, alcohols and ketones sometimes do the job. Isopropyl alcohol works fine for many pieces but can strip coatings if workers aren’t careful. Acetone cleans quickly but evaporates before anyone’s finished a thorough wipe-down. I’ve seen more managers testing aqueous cleaning systems, where water mixed with soaps strips away residue. These setups usually run at higher temperatures and might need extra equipment, but they can save money on chemical disposal and keep air quality better inside the building. Speaking from experience, switching to water-based cleaning forces a look at every step in the workflow—not all parts survive the switch without rust or residue.
Supporting Safer Solutions with Real Action
I often hear complaints about regulatory red tape when phasing out problem chemicals, but putting off changes only gambles with health and legal trouble. Many operations are stuck weighing system modifications against chemical risks. Here, the people on the floor—technicians and engineers—call the shots. Trial-and-error matters more than textbook promises. Industry reports show that once teams get hands-on training with alternative solvents, they start to trust new routines.
Research funding matters, too. Projects under the U.S. Environmental Protection Agency’s SNAP program have been steering both manufacturers and end-users toward better substitutes. The industry doesn’t just swap one chemical for another overnight. Benchmarking requires real-world data on cleaning power, toxicity, workplace exposure, and pollution.
Building Better Habits for Cleaner Industry
It helps to remember that even the best substitute won’t fix sloppy habits. Training workers to handle new solvents—reviewing safety data sheets, checking progress, and sharing feedback—counts as much as picking the right formula. Some industries re-use and recycle solvents on-site, cutting costs and waste. Others run closed-loop systems that keep emissions from slipping into the environment. These strategies show up most in firms that view chemical choices as part of broader sustainability plans, not just a quick compliance fix.
Relying on legacy chemicals like 1,1,2-trichlorotrifluoroethane brought short-term answers but long-lasting trouble. By listening to experts on the ground and supporting clear-eyed research, industries can let go of the past and build safer, more responsible processes for everyone involved.
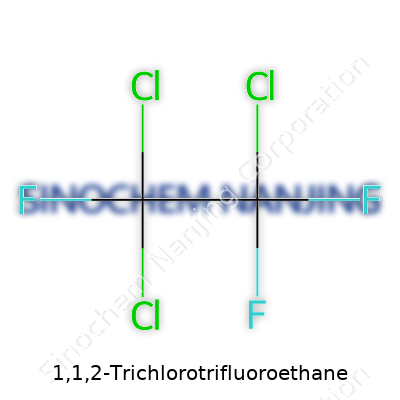
| Names | |
| Preferred IUPAC name | 1,1,2-Trichloro-1,2,2-trifluoroethane |
| Other names |
CFC-113 Freon 113 Arklone Fluorocarbon 113 Genklene Halocarbon 113 Trichlorotrifluoroethane Methylchloroform Refrigerant 113 Ucon 113 |
| Pronunciation | /ˌwʌnˌwʌnˌtuː traɪˌklɔːrəˌtraɪˌflʊərəˈɛθeɪn/ |
| Identifiers | |
| CAS Number | 76-13-1 |
| Beilstein Reference | 3581446 |
| ChEBI | CHEBI:39146 |
| ChEMBL | CHEMBL2105931 |
| ChemSpider | 9474 |
| DrugBank | DB01362 |
| ECHA InfoCard | 03f0c0b3-238d-4a53-ab0d-7b521d06cc1b |
| EC Number | 205-553-2 |
| Gmelin Reference | 104176 |
| KEGG | C14357 |
| MeSH | D014265 |
| PubChem CID | 6577 |
| RTECS number | KI9625000 |
| UNII | K77I6FE458 |
| UN number | UN1022 |
| CompTox Dashboard (EPA) | DTXSID9020221 |
| Properties | |
| Chemical formula | C2Cl3F3 |
| Molar mass | 187.377 g/mol |
| Appearance | Colorless liquid |
| Odor | Faint ethereal odor |
| Density | 1.563 g/cm³ |
| Solubility in water | 4.4 g/L (30 °C) |
| log P | 2.80 |
| Vapor pressure | 72.6 mmHg (20°C) |
| Acidity (pKa) | ~15.0 |
| Basicity (pKb) | pKb: 17.44 |
| Magnetic susceptibility (χ) | −0.82×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.364 |
| Viscosity | 0.65 mPa·s (25 °C) |
| Dipole moment | 0.30 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 380.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1000 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2226 kJ/mol |
| Pharmacology | |
| ATC code | D08AX01 |
| Hazards | |
| Main hazards | Harmful if inhaled, causes skin and eye irritation, may cause drowsiness or dizziness, and is a suspected carcinogen. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P210, P261, P271, P304+P340, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 38°C (100°F) |
| Autoignition temperature | 770°C |
| Lethal dose or concentration | Lethal Concentration: LC50 (rat, inhalation): 38,000 ppm/4H |
| LD50 (median dose) | LD50 (median dose): 7.5 g/kg (oral, rat) |
| NIOSH | SN8750000 |
| PEL (Permissible) | 1000 ppm |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 2000 ppm |
| Related compounds | |
| Related compounds |
Trichlorofluoromethane Dichlorodifluoromethane Chlorotrifluoromethane 1,1,1-Trichloro-2,2,2-trifluoroethane 1,2-Dichlorotetrafluoroethane Chlorodifluoromethane Chlorofluorocarbons |

