Fluoroboric Acid: Navigating Chemical Legacy and Modern Industry
Historical Development
Looking back, the journey of fluoroboric acid runs deep with chemical heritage. Chemists as early as the 19th century started probing the boundaries of mineral acids, searching for reactivity and stability. The odd coupling of boron and fluorine emerged from tireless lab work, introducing an acid with capabilities that gave rise to fresh industrial chemistry. Early work focused on reactivity and the puzzles of complex ion formation, not yet foreseeing modern uses. These breakthroughs didn’t happen in isolation; each step responded to demands for efficient pickling, unique catalyst systems, and new ways to create stable salts or dissolve stubborn oxides. Scientific curiosity, paired with the needs of metal finishing and electroplating, pushed experts to refine synthesis and understand the true heft of this acid’s usefulness.
Product Overview
Fluoroboric acid, better known in some corners as tetrafluoroboric acid, distinguishes itself in the crowded world of mineral acids. Unlike the more common hydrochloric or sulfuric alternatives, this one teams up boron tetrafluoride with water to generate a strong liquid acid, privy to applications in manufacturing processes where typical acids just can’t hold up. People rely on it in large-scale situations—pickling stainless steels, producing specialty chemicals, and fine-tuning electrolytic reactions. The acid handles a mix of roles, stretching from pharmaceuticals to cleaning agents designed for tough industrial grime. Each of these uses comes down to its high acidity, unusual resistance to oxidation, and the ability to produce fluoroborate salts.
Physical and Chemical Properties
Pour a bottle of fluoroboric acid and the clear, colorless liquid hints at nothing out of the ordinary, but the smell—sharp and reminiscent of other strong acids—hits the nose quick. With a density around 1.2-1.24 g/mL and available in various concentrations that run up to about 50 percent by weight, this acid doesn’t play around. Boiling comes at a lower temperature than water, and the fumes remind anyone in a lab to tread carefully. On the chemical side, fluoroboric acid sets itself apart by not reacting with many metals. It produces tetrafluoroborate anions, which resist hydrolysis in solution and keep from breaking down when exposed to air, setting the stage for stable reactivity. This property fuels much of its appeal in industries that demand reliability from their reagents, especially compared to stubborn cousins like perchloric or nitric acids which can go off unexpectedly.
Technical Specifications and Labeling
Working with chemicals usually means sifting through labels, and fluoroboric acid stands out for its required hazard markings. Labels pinpoint concentrated acid percentages, show hazard pictograms, and carry warnings about contact with skin or eyes. Manufacturers signal the need for proper handling with equipment rated for corrosive materials and containers that withstand acid attack. One can spot detailed warning icons and safety instructions—not to satisfy bureaucracy, but because spills or improper mixing can end in hospital trips. Standard solutions come with data sheets explaining exact molarity, typical impurity levels, and safe storage advice to help those in charge keep accidents at bay.
Preparation Method
In practice, to get fluoroboric acid, a chemist might bring together boric acid and hydrofluoric acid in controlled conditions. The chemistry seems straightforward—mix, stir, and cool. In reality, the hazards of handling hydrofluoric acid up the stakes. Mistakes can burn flesh to the bone or kick off dangerous reactions. The mixture liberates water and forms stable tetrafluoroboric acid, captured in glass or polyethylene containers. Due to the need for care, most users today trust industrial suppliers to handle production and purification, favoring prepared solutions over home-brewed batches in research or manufacturing settings.
Chemical Reactions and Modifications
Chemists don’t just let an acid sit in a bottle. In reactions, fluoroboric acid steps up as a strong proton donor, a creator of stable tetrafluoroborate salts, and a catalyst in organic and inorganic transformations. Drop it into alkali metal solutions, and out falls a group of widely-used tetrafluoroborate salts: compounds key in pharmaceuticals, dyes, catalysts, and batteries. It resists oxidation and stands its ground, making it a choice candidate for reactions where stiffer acids would create unwanted side-products. The acid’s role in ion-pairing helps control reactions in both lab and scalable production, often aiding in cleanup steps and downstream purification. Researchers explore further tweaks, using subtle substitutions or partner reagents to tease out even more from what, at first blush, looks like just another acid.
Synonyms and Product Names
Chemists and industry folks toss around a few names for this acid. Tetrafluoroboric acid comes up often, especially in technical literature. Sometimes the shorthand HBF4 appears, marrying its chemical formula and highlighting the four fluorine atoms encircling boron. It rarely travels alone; in commercial catalogs, it pops up in varied concentrations and packaging adapted to customer need, labeled sometimes just as “fluoroboric acid” but bearing hazard codes that tip off its true strength.
Safety and Operational Standards
Handling fluoroboric acid isn’t for the unwary. The acid’s corrosiveness can destroy skin tissue, eat through clothing, and even seep into less obvious areas to weaken glassware over time. Inhaling its vapors delivers strong irritation and, over repeated exposures, risks long-term lung effects. Factories and labs answer back with thick gloves, splash goggles, and ventilation systems that keep people’s lungs clear. Emergency washes and neutralizing agents line benches near active-use areas. Companies also keep spill plans in plain sight, aiming to avoid environmental mishaps given the threats posed to water supplies. Staff get regular drills, ensuring that anyone handling this acid respects the hazards and knows what to do in a pinch.
Application Area
Few acids can claim such a wide berth of usefulness. Fluoroboric acid takes part in the pickling of stainless steel, prepping surfaces by stripping oxides ahead of coating or soldering. Galvanic and electroplating shops depend on it to help lay down bright, uniform metal layers on jewelry, electronics, and machine parts. In organic synthesis, researchers rely on its catalytic ability to wrangle challenging transformations or stabilize reactive intermediates. The acid makes terpenoid and alkaloid chemistry manageable, and in battery research, it offers a path to robust electrolyte systems. Water treatment and etching in microelectronics round out its utility, showing a rare versatility that sets it apart from the more single-purpose mineral acids crowding most shelves.
Research and Development
Despite its solid industrial roots, fluoroboric acid attracts ongoing research, especially where sustainability or reactivity come into play. Researchers dig into safer handling protocols, greener synthesis routes, and less wasteful applications in electrochemical and pharmaceutical manufacturing. At conferences and in journals, studies focus on improving selectivity of catalytic reactions, finding alternatives to higher-hazard acids, and improving on the purification of fluoroborate salts. The acid has even surfaced in new battery chemistries aimed at longer cycle lives and faster charging. Each avenue of R&D points to maximizing value while cutting down on ecological cost and workplace hazards.
Toxicity Research
Long-term exposure and acute incidents both put fluoroboric acid in the sights of toxicologists. Animal studies and reports from workplace exposures link it to severe burns, tissue destruction, and systemic toxicity if not handled with respect. Hydrofluoric acid content, as a precursor or impurity, brings added danger due to its unique bone-binding properties and risk of cardiac effects. Surveys of affected workers, post-exposure treatments, and antidote protocols fill the research literature. Best practice points toward rapid removal from affected tissue, calcium-based neutralization, and well-drilled emergency response. Regulators look at workplace monitoring, limits for airborne exposure, and disposal methods that prevent unintended releases into the environment.
Future Prospects
The story of fluoroboric acid keeps moving forward. Pushes for cleaner technology, sharper efficiency, and improved safety standards drive new studies and regulatory attention. More companies explore alternatives or upgrades to production that cut down on acid loss and recycle by-products into useful streams. Battery researchers play with new fluoroborate systems aimed at sustainability and energy storage breakthroughs. Automation and advanced robotics promise to take humans out of direct contact, reducing accidents and opening up more intensive chemical processes. Progress in toxicity management, spill containment, and worker education keeps improving outcomes, while the stubborn usefulness of this acid ensures it remains on the shelves of labs and factories with cutting-edge ambitions.
What is Fluoroboric Acid used for?
What Fluoroboric Acid Brings to the Table
You won't find fluoroboric acid in your local hardware aisle. That's a good thing. This is one tough acid—clear, colorless, and so strong it eats through glass if you let it sit long enough. Most people never run across it, unless they work with metals, electronics, or chemicals all day. Still, its role touches a ton of products people use, sometimes without even realizing it.
How Industry Gets Serious Cleaning Done
Anyone who has scrubbed rust off an old bike knows some stains just don’t budge with soap and water. Multiply that problem by a hundred, and you’re in the shoes of a metal finisher. In factories, rust and oxides grind production to a halt. Fluoroboric acid tackles these stubborn layers. Metal surface cleaning relies on it—before plating, engineers dip metal parts in a fluoroboric acid bath. The acid breaks down the oxides fast, leaving steel, copper, silver, or brass clean and ready for a protective or decorative coating.
Jewelry makers, too, count on this acid to give silver and gold a mirror shine. After soldering, they throw pieces in a “pickle” solution, often based on fluoroboric acid. The acid eats away scale and grime, making precious metals gleam without damaging the underlying material. Safer for gold and silver than sulfuric alone, fluoroboric acid helps keep intricate work intact, even on tiny chains and filigree.
Electronics and Cleaning Challenges
Electronics manufacturers face a different beast: super thin copper traces. During circuit board production, unwanted copper must disappear without chewing up the board itself. Fluoroboric acid allows precise etching and cleaning, stripping away excess copper with control no amateur chemistry can match. This kind of repeatable, clean etching supports high-tech advances, where every fraction of a millimeter counts.
People don’t think about the chemicals that keep their phones or tablets humming, but this acid has quietly shaped modern electronics. Mess up the purity or handling, and entire batches go to waste, driving up costs and delays. That’s why experience counts—a seasoned technician knows how to handle it, when to use it, and how to contain any hazards.
Risks Demand Respect and Proper Training
Fluoroboric acid doesn’t play around. It causes burns on contact, and inhaling fumes gets dangerous for the lungs. Old-timers talk about safety with a kind of reverence—goggles, gloves, good ventilation. Proper containers matter. Fluoroboric acid turns careless mistakes into disasters. Even glass isn't safe storage if it sits around too long.
Strict rules control where and how to use it. Factories must train workers, provide emergency wash stations, and store the acid in chemical-resistant containers. Disposal requires neutralization, all tracked by law. Mistakes make headlines, and cleanups cost real money.
Better Ways—Or Are There?
No one wants more dangerous chemicals on the job. Researchers keep searching for greener alternatives. Some companies choose milder acids or new cleaning processes using electricity or different compounds. Transition takes time and real-world testing. Until something safer matches its performance, fluoroboric acid keeps its place in plenty of plants around the world.
With smart safety, respect for risk, and a drive for innovation, industries keep this acid working for them instead of against them. In skilled hands, it’s a valuable teammate; with shortcuts, it quickly turns into an expensive mistake.
Is Fluoroboric Acid hazardous to handle?
Getting Real with Fluoroboric Acid’s Dangers
Fluoroboric acid turns heads for a reason. Few chemicals demand such firm respect and a steady hand in industrial settings. I remember working my first job near a plating line where hazmat training ran back-to-back, all because of this single compound. Even the old-timers treated it as more than just “another acid.” There’s good reason.
Direct Health Risks on the Job
Skin contact pulls no punches. Drops on bare skin can burn quickly, eating into tissue with little warning. Burning doesn’t always happen straight away, which fools people into thinking the exposure isn’t as bad as it is. Eyes hit with the acid can face lasting damage, even blindness. Breathing in the mist or fumes irritates airways and lungs, leading to coughing fits, tightness in the chest, and sometimes chronic respiratory trouble.
Data from the U.S. National Institute for Occupational Safety & Health paints a stark picture. Emergency visits following accidental exposure often end in intensive intervention. Fluoroboric acid gets through protective barriers fast, dissolves rust and metal, and delivers toxic fluoride ions that travel right into tissue and bloodstream.
Equipment Corrosion and Facility Damage
Beyond personal safety, the stuff chews through equipment. Pumps, gaskets, and seals tasked with containing fluoroboric acid live on borrowed time. Leaks start small but can speed up wear and lead to environmental spills—big headaches for plant managers and city officials alike. Insurance never covers all the cleanup costs. Neighboring workers, and even the community, pay the price if storage tanks rupture or if disposal protocols slip.
It doesn’t help that fluoroboric acid is colorless and blends right in with water, making accidental mixing or mislabelling possible in busy workspaces. One misstep in the labeling process and a maintenance worker uncaps the wrong drum—a recipe for disaster.
Long-Term Environmental Impact
Runoff containing fluoroboric acid needs careful control. Untreated discharge seeps into municipal water, working its way into soils and crops. Regions mishandling acids like this face long-term groundwater contamination, which can force communities to invest in new filtration systems or alternative water sources. These costs climb quickly, especially in places where budgets already run thin.
Tools for Safer Handling
What stands between a safe day at work and an emergency room dash comes down to training, regular checks, and strict routines. Everyone in the handling chain needs clear instructions—easy-to-read labels, up-to-date Safety Data Sheets, and shake-ups that keep bad habits in check. My training involved mock spills followed by real walk-throughs of evacuation routes. That stuck more than any printed warning.
Personal protective gear stays essential—acid-resistant gloves, face shields, and chemical aprons, even if it’s only for a quick transfer or sample draw. Spill kits belong within arm’s reach, not locked away where nobody can find them in a pinch. On the engineering side, double-containment piping and automated leak sensors cut down risk and catch small failures early.
Safer processes lead to fewer health emergencies and stronger compliance with regulation. Improving handling methods draws on the experience from past accidents—sometimes tragic ones. Nothing beats preparation, both for the workers who clock in every day and the community living down the road.
What is the chemical formula of Fluoroboric Acid?
HF and BF3 Combine: The Science Behind HBF4
Fluoroboric acid, known by its chemical formula HBF4, sounds more complex than most people run into day-to-day. My first real encounter happened in a university chemistry lab, my professor handling it with care and stressing the importance of clear safety rules. He pointed at the flask, explained its reactivity, and launched into a story about a spill from earlier in his career. Hazards with acids don’t just cause problems in the lab. In manufacturing and electronics, uncontrolled exposure to reactive agents like HBF4 translates to both safety and environmental issues if handled poorly.
HBF4 forms when boron trifluoride (BF3) meets hydrofluoric acid (HF). What stands out for people working with chemicals: both ingredients carry risks of their own. Hydrofluoric acid attacks flesh and bone, while boron trifluoride brings severe respiratory hazards. Now, think about combining the two—better be ready with thick gloves and a fume hood. Every good chemistry curriculum highlights these dangers.
Why It Earns Respect in Industry
In plating and metal cleaning shops, I’ve seen staff use fluoroboric acid preparations for tough jobs. Copper electroplating runs smoother with HBF4-based baths. The solution keeps metal ions happy, stopping them from clumping up or sticking stubbornly to the wrong places during the process. For those in the know, having reliable chemistry means fewer surprises and cleaner finishes, even on oddly shaped parts. This reliability saves hours, and hours translate directly into earnings.
Glass etching presents another dimension. Creating frosted glass or precise decorative patterns depends on acids that can chew through silica. HBF4 brings powerful etching without the heavy toxic cloud associated with some other etchants. This lets artisans and engineers produce sharp designs with fewer work hazards, at least compared to harsher alternatives.
Environmental and Health Challenges Won’t Disappear
Acid fumes don’t just vanish—they pose localized risks to both workers and communities downwind. I remember working alongside a facility manager haunted by an incident involving faulty ventilation. It became clear that regulations, routine monitoring, and proper disposal practices are the backbone of safe chemical use. The environmental impact of accidental HBF4 releases highlights the need for stringent control systems. Waste must pass through neutralization before leaving a lab or plant. Careless handling results in fluorine and boron compounds ending up in local waterways—a nightmare for aquatic creatures and anyone drinking downstream.
Effective Risk Management and Safer Solutions
Training and awareness make the biggest difference. Any lab or workshop serious about safety invests in hands-on instruction for new staff. Personal experience shows that written protocols only go so far: face-to-face guidance, real equipment, and clear accountability give the lessons sticking power. PPE policies should never be fudged, even for “quick” tasks. Inspections and routine maintenance of hoods, scrubbers, and emergency showers matter just as much.
Alternatives and safer substitutes continue to gain attention. Some companies seek ways to reduce their HBF4 footprint while preserving process reliability. Research into non-acidic cleaning or plating baths adds promise, though switching over proves slow for established production lines. Regulatory pressure and economic incentives might nudge change faster as best practices keep shifting.
HBF4: Vital Yet Demanding Respect
Knowing the chemical formula and understanding the properties behind HBF4 matter both for professionals and curious minds. The more people understand the risks, uses, and better handling methods, the safer everyone will be. What you don’t know about HBF4 can hurt you. Good training, environmental care, and continual improvement shoulder the real backbone of safe chemical science and industry practice.
How should Fluoroboric Acid be stored?
What Happens When People Cut Corners
Fluoroboric acid makes anyone in the lab perk up and pay attention. It’s a substance known both for its strong acidity and how quickly it chews through materials that look tough on paper. I once saw a careless colleague store it in a makeshift bottle “just for a few hours.” The result? The shelf got etched, fumes drifted into the hallway, and we spent a morning explaining ourselves to the safety officer. Incidents like this keep me strict about storing dangerous chemicals, and fluoroboric acid sits high on my list of things never to underestimate.
Understanding What’s at Stake
Fluoroboric acid breaks down most plastics and reacts with bases and metals in a snap. Besides burns, its vapors can eat at your lungs and eyes. That harsh nature means basic plastic containers don’t work—HDPE or polypropylene sound good for some acids but lose the battle here. Glass holds out better but gives way too easily when moisture and high concentration come into play.
This isn’t just theory. In 2022, an industrial accident in Asia led to dozens being hospitalized after fluoroboric acid leaked from an old, plastic-lined tank. Investigators found traces of corrosion spreading through the storage room. Their report recommended a shift to safer, purpose-built containers and clear, simple storage instructions. If you look at safety statistics, most issues come down to shortcuts or outdated gear, not lack of knowledge.
Proper Containers and Why They Work
Always pick containers lined with PTFE (polytetrafluoroethylene). Some might know it as Teflon. This lining stands up to strong acids and helps prevent the slow creep of leaks. If you work in a lab, check for these markings every time. Avoid any old containers, even if they look clean. Surfaces can hold on to residues, and those unglamorous details add up to risky situations.
Strong ventilation matters, too. Many people think a closed cap solves the problem. Experience tells a different story—vapor buildup finds its way out, spreading to anyone working near the area.
Temperature and Labeling—Simple Steps, Big Results
Store fluoroboric acid in a cool, dry place away from direct sun. I personally double-check storage room temperatures weekly. High heat accelerates the acid’s corrosive nature and can warp even the best PTFE linings. Dedicated storage cabinets really help. OSHA recommends acid cabinets marked with big, easy-to-read warning labels. Trust me, simple labeling saves headaches and helps new team members spot risks faster.
The ideal practice includes keeping incompatible chemicals at a safe distance. Acetic acids, metals, and bases stay well apart. In shared spaces, I post diagrams to remind staff of safe separation. These steps guard against accidental mixing, which can cause explosions or violent releases of gases.
Keeping People First
Routine safety checks keep everyone accountable. Spills get cleaned up with special absorbents, never with water. Emergency showers and eyewash stations should work and stand out. Building a culture where people speak up early—if a container looks off, if a valve sticks just a bit—keeps small problems from turning into disasters.
There’s no trick or fancy system here. Storing fluoroboric acid safely relies on sturdy materials, good habits, and not brushing off warning signs. Plenty of lessons pile up from seeing small mistakes invite big problems. Clear steps and a healthy respect for the damage this acid can cause give both employers and workers peace of mind.
What are the safety precautions when working with Fluoroboric Acid?
Understanding the Risks
Fluoroboric acid does not play by friendly rules. Whether you’re running an electroplating bath or cleaning up lab glassware, a single splash can create real trouble. This acid eats through skin and eyes, but it does its worst damage deeper, by letting fluoride ions travel beneath the surface. A lab tech at a previous job once told me stores didn’t even keep it on “normal shelves.” He once spilled just a few drops on the outside of a glove, and the tingle turned into searing pain minutes later.
Getting careless in safety routines can leave lasting injuries, especially on hands and unprotected faces. Even in a controlled academic lab, I’ve watched someone fail to double-check a transfer tube, leaking acid vapor into the fume hood, which set off instant coughing fits down the line.
Personal Protective Equipment is Non-Negotiable
Bare hands and arms should never be exposed. Nitrile gloves break down quickly, so I prefer heavy neoprene gloves every time. Eye protection has to cover from the sides—regular goggles won’t cut it. Face shields block random splashes. Chemical-resistant aprons and sleeves are not optional, especially if mixing up more than a small volume. Every facility I’ve ever worked in with fluoroboric acid has kept fresh eyewash and emergency showers within easy reach, and not once did anyone joke about skipping a safety drill.
Ventilation and Storage
Those vapors sting before you realize you’re exposed. Fume hoods with solid airflow keep the workspace safe. Fumbling around with acid bottles outside a hood risks breathing fumes that can scar lung tissue. Proper ventilation stopped one colleague from ending up in the hospital after a beaker cracked—smoke never reached her face.
Every storage area gets tagged with clear, sturdy labels. I once saw an intern reach for a bottle with a faded tag, thinking it was boric acid. Only a last-minute supervisor check saved him. I never forget how much safer things get when every shelf is clearly marked and acids get separate, locking cabinets.
Accident Response Matters
Local emergency plans aren’t just for show. If fluoroboric acid gets on skin, immediate rinsing with copious water is the top move—at least fifteen minutes, no shortcuts. Calcium gluconate gel lives in my lab’s first-aid kit, alongside safety data sheets posted right by the entrance. Medical help needs calling as soon as possible, because damage under the skin spreads fast. I’ve seen permanent nerve loss in fingers from slow responses.
Spills need neutralizing with calcium carbonate or similar agents, then scooping up the residue into special hazardous waste containers. Regular janitorial staff shouldn’t walk into these spills—trained hands with sealed suits step in.
Building Safer Habits
Fluoroboric acid safety goes beyond rules written on a poster. Real safety comes from an attitude built during daily routines. Team members check each other, and everyone calls out small errors before they become tragedies. These chemicals rarely forgive mistakes. The best labs don’t trust luck—they trust each other and clear, practiced actions. That’s what keeps people safe, every single shift.
| Names | |
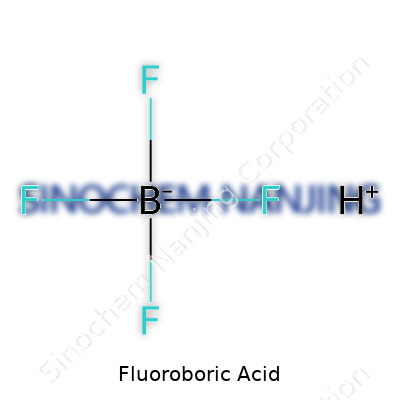
| Preferred IUPAC name | tetrafluoroboric acid |
| Other names |
Boric acid, hydrofluoro- Hydrofluoroboric acid Fluoboric acid HBF4 |
| Pronunciation | /ˌflʊə.rəˈbɒr.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 16872-11-0 |
| Beilstein Reference | 3587152 |
| ChEBI | CHEBI:29329 |
| ChEMBL | CHEMBL1231866 |
| ChemSpider | 156057 |
| DrugBank | DB14516 |
| ECHA InfoCard | 100.028.930 |
| EC Number | 231-634-8 |
| Gmelin Reference | 894 |
| KEGG | C06321 |
| MeSH | D005465 |
| PubChem CID | 24857 |
| RTECS number | ED4300000 |
| UNII | 7N1Q0ROI1V |
| UN number | UN1775 |
| CompTox Dashboard (EPA) | DTXSID4015394 |
| Properties | |
| Chemical formula | HBF4 |
| Molar mass | 87.81 g/mol |
| Appearance | Colorless liquid |
| Odor | Pungent |
| Density | 1.19 g/cm³ |
| Solubility in water | Very soluble |
| log P | -0.44 |
| Vapor pressure | 24 mmHg (20°C) |
| Acidity (pKa) | -0.4 |
| Basicity (pKb) | -0.6 |
| Magnetic susceptibility (χ) | −54.5 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.390 |
| Viscosity | 2 cP |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.5 J/mol·K |
| Std enthalpy of formation (ΔfH⦵298) | −377.0 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1039 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Corrosive, causes severe burns, harmful if inhaled or swallowed, reacts violently with water and bases, toxic fumes |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H290, H314 |
| Precautionary statements | P234, P260, P264, P270, P271, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-Acidity |
| Autoignition temperature | 100 °C (212 °F; 373 K) |
| Lethal dose or concentration | LD50 (oral, rat): 100 mg/kg |
| LD50 (median dose) | LD50 (median dose): 100 mg/kg (oral, rat) |
| NIOSH | UN1775 |
| PEL (Permissible) | 2 mg/m3 |
| REL (Recommended) | 2.5 mg/m³ |
| IDLH (Immediate danger) | 25 mg/m³ |
| Related compounds | |
| Related compounds |
Boric acid Hydrofluoric acid Tetrafluoroborate Sodium tetrafluoroborate |

