Fluoroacetamide: Past, Present, and the Road Ahead
Historical Development
People first came across fluoroacetamide in the mid-20th century, guided by a pressing need to control pests where conventional poisons came up short. Chemists working post-war tried to outsmart adaptable rodents, and synthetic organic compounds like fluoroacetamide caught their eye. The compound landed on the market with a certain expectation: take the fight directly to tough pests without costing a fortune or leaving a chemical wasteland behind. The story of its rise traces not only to farm fields but also to a time when modern chemistry pushed boundaries, sometimes too far, before public health experts caught up to the risks. The material earned a controversial spot in the warfare of agriculture—sometimes celebrated as a needed tool, then vilified as communities and regulators came to see its darker side.
Product Overview
If you look at the physical powders carried in sealed bags, you might not notice anything remarkable about fluoroacetamide at first glance. Its real difference comes from how it works, never from some catchy label or branding. Fluoroacetamide stands out because it targets living cells at their core, interfering with natural metabolic steps. The product has always been introduced as a sharp, potent thread in pest management, straightforward in use but with a heavy responsibility attached. Its notoriety grows, not just through practical use, but by stories and reports from the field—often where children, pets, or wildlife paid the price of its hidden power.
Physical & Chemical Properties
The molecule behind the story—simple in shape, just one fluorine atom swapped where swimmers in your body would expect hydrogen—behaves with an efficiency and danger hard to ignore. It looks much like a white crystalline substance, dissolves well in water, and can hide easily in grain stores or bait stations. The chemical shifts only slightly under sunlight but clings on tight in damp, sealed environments. Chemists have found it to be surprisingly heat-stable, meaning you can’t just rely on ordinary heat to break it down if a spill occurs. Environmental persistence creates more questions than answers, especially when runoff or improper disposal enters the equation.
Technical Specifications & Labeling
Labels on fluoroacetamide stress one issue above all: respect for its risk. Packages carry warnings that border on the intimidating, emphasizing its narrow margin between efficiency and disaster. The smallest dose, if handled carelessly, can slip from a scientist’s hand to an animal’s mouth with almost no visual signal. Numbers reported on toxicity lean toward the alarming, keeping regulators on guard and often reserving its use for professionals only. Distributors highlight secure packaging, tamper-proof seals, and protocols that force users to pay attention—even before the bag is opened.
Preparation Method
Preparing fluoroacetamide rarely happens outside controlled labs where exact measurements keep disaster at bay. The process usually starts with acetamide that takes on a single fluorine atom in a targeted chemical reaction involving hydrogen fluoride sources or fluorinating agents. This transformation, once seen as a modern marvel, no longer impresses in quite the same way, as the risks have become more obvious. Chemists handle the synthesis behind fume hoods, wrapped in protective layers, sometimes calling it the laboratory equivalent of working with venomous snakes. Disasters during production have been few, but always hover as a risk in the background.
Chemical Reactions & Modifications
Once fluoroacetamide is made, its reactivity pulls from the same roots that made it a threat to living tissues. In water, it breaks down only slowly, but in living bodies, enzymes crack it apart to unleash fluoroacetate, a murderously efficient metabolic poison. Researchers have tinkered with the molecule, trying to soften its danger or target its effects with more precision, but most results extend its shelf life or adjust how quickly the body processes it, rather than making it truly safe. Little in its chemistry lends itself to neutralizing or reversing the accidental impact, so emergency response winds up stuck relying on old-fashioned hospital treatment.
Synonyms & Product Names
Across decades and continents, the same basic compound emerged in different marketplaces under names like "FCH2CONH2" or less formal trade names tied to specific brands. Each new name aimed at different regulations or audiences, but beneath the surface, specialists recognize the same core structure. Whether called by a trade moniker or a string of letters, the substance’s impact cuts through any rebranding attempt. In regulatory filings, the name "fluoroacetamide" always raises bureaucratic eyebrows given its legacy of poisonings and restrictions.
Safety & Operational Standards
The biggest challenge for anyone working with fluoroacetamide runs deeper than gloves and goggles. Mishandling a pinch can spell catastrophe, and this isn’t just theoretical. Over the years, accidents turned rural towns upside down, killed livestock, and left law enforcement scrambling for answers. Authorities require airtight storage containers, lock-and-key protocols, and strict records of every milligram that changes hands. Disposal policies demand neutralization agents—not just a trip down the drain—because too many cases showed what happens otherwise. Even transport falls under close scrutiny, with strict limits on routes and carrier types.
Application Area
Farmers and public health teams once used fluoroacetamide with gusto, sometimes achieving quick reductions in pest populations. The appeal fell apart as stories of off-target casualties mounted: pets, wildlife, and even children became unintended victims. Selective bans now limit its place to extreme circumstances, or countries where alternative controls have yet to gain a foothold. Researchers weighing its use look over their shoulders at public opinion as much as pest populations. Any program proposing its deployment faces tough questions about monitoring and emergency action plans.
Research & Development
In laboratories, interest in fluoroacetamide remains, but less as a field solution and more as a chemical tool for understanding metabolism and neurotoxicity. Scientists probing the ways cells process energy lean on the compound’s unique mode of action to map out metabolic pathways. Efforts to tweak the molecule continue, sometimes seeking antidotes, sometimes exploring "suicide inhibition" structures for more targeted research outcomes. Industry spends less time defending the task of replacing fluoroacetamide for pest control, and more time supporting alternatives that carry less legal and ethical baggage.
Toxicity Research
Few chemicals spark more debate in toxicology circles than fluoroacetamide. Human and animal studies, often forced by accident rather than design, tell hard stories: sudden illness, organ shutdown, nervous system collapse. The compound works by snarling up the Krebs cycle, an essential biological engine in every oxygen-breathing creature. Doctors rarely get a second chance to intervene, which fuels calls for its retirement. Long-term environmental studies show this molecule lingers in soils and water, passing up the food chain in the most unwelcome ways, forcing future generations to deal with cleanup.
Future Prospects
The future for fluoroacetamide seems set on a forked path. Chemical historians and regulatory watchdogs cite its track record as a warning, not a model. Countries with strong regulatory systems have phased out large-scale agricultural use, turning to less toxic, more targeted agents. Innovation focuses on biological controls, genetic solutions, and chemicals with escape hatches built in—compounds designed to fade fast after doing their job. Calls for global bans ring out at regulatory conferences, but in some corners of the world, the compound lingers on shelves, a last-ditch effort where governments juggle hunger and pestilence against public health. Demands for tighter testing standards and wider transparency reflect a growing consensus: chemistry’s power works best in daylight, not in the shadows of forgotten storerooms.
What is fluoroacetamide used for?
The Role of Fluoroacetamide in Pest Control
Fluoroacetamide makes headlines in agriculture and pest management for a simple reason: it kills pests, quickly and effectively. It has carved out its place thanks to its toxicity to small mammals, insects, and rodents. In the past, farmers and pest controllers reached for fluoroacetamide to slash numbers of rats, mice, and other invasive species threatening both crops and stored food. Its lethal nature stops pests before they can cause widespread damage, saving livelihoods and entire harvests.
I grew up around family farms. Rats tearing into stored rice and corn meant true losses—not just for the fields, but for families who counted on those crops for income. When rat poisons didn’t work, stories made the rounds about someone trying out “the really dangerous stuff” like fluoroacetamide. Calling it potent isn’t exaggerating. Even a small dose brings on fatal effects. Its reliability made it a tool of last resort in settings where populations of vermin refused to budge with other measures.
The Human Risk Behind the Chemical
Sitting at the kitchen table, hearing stories about “one drop” and strict warnings not to touch or breathe anything near fluoroacetamide, the serious dangers became clear. Its toxicity doesn’t stop with rats. In the wrong hands, or handled carelessly, this is a substance that can kill people and pets just as quickly. Poisoning cases—often by accident—have happened wherever uncontrolled use has crept in, leaving families dealing with tragedy instead of relief. Children are especially vulnerable because, to the untrained eye, little pellets or powders don’t look harmful.
Checking science journals and poison control records confirms the fears. Reports from hospitals show symptoms can start within hours—nausea, dizziness, followed by seizures and cardiac failure. Professional handbooks stress that there is no specific antidote for exposure. Lives often hang on early detection and rapid, aggressive medical care. Simply put, the same qualities making fluoroacetamide a feared pest killer make it a real health hazard beyond the field.
Regulations and Safer Paths Forward
Authorities haven’t stood by blindly. Australia, China, and the European Union have put tight restrictions, or outright bans, on fluoroacetamide for decades. These decisions didn’t come out of nowhere. Health agencies review the facts: hospitalizations, unintentional deaths, environmental harm, and potential for misuse. Policies follow. Storage, transport, sales, and use have to run through strict regulation.
At the same time, scientists and engineers search for better ways to tackle pests—ones that don’t load up the environment or endanger children. There’s a growing market for rodenticides broken down quickly in soil, or new traps using baits the target species find irresistible but that won’t build up in water or food supplies. Biological controls, like encouraging natural predators or using sterilization, increasingly get a closer look. These options may require patience and good planning, sometimes more labor up front, but leave fewer scars on communities and ecosystems for years down the line.
Looking Ahead
Experience shows that no easy fix solves pest problems forever. Relying on any single toxin—especially one as potent as fluoroacetamide—carries real risks. Communities can protect themselves and their food by demanding safer alternatives, following science-backed advice, and supporting responsible regulation. Staying alert and careful isn’t just good sense, it’s good stewardship, for land and for the people sharing it.
Is fluoroacetamide toxic to humans or animals?
The Reality Behind Fluoroacetamide
Ask anyone who’s worked in pest control or agriculture, and they’ll likely have a story about fluoroacetamide. It’s a chemical that’s stayed in the shadows, but its effects don’t go unnoticed by anyone who’s been around it. It’s used to get rid of rodents and pests that threaten crops or stored grain, but the cost can be steep—not just for unwanted animals, but for farm dogs, cats, livestock, and sometimes people.
Known Dangers Supported by Science
Fluoroacetamide is not subtle. Scientists have classified it as extremely toxic to both people and other mammals. Even a tiny amount can stop life in its tracks. Research points out that fluoroacetamide disrupts how cells produce energy. In practice, this means organs can’t do their job and the body starts to fail.
Hospitals often deal with poisonings from substances like this, especially in rural spots. Poison control experts warn that animals or people exposed through skin contact, inhalation, or swallowing can suffer serious symptoms. Early signs include nausea, vomiting, convulsions, and, if left unchecked, coma or death.
The U.S. National Library of Medicine lists fluoroacetamide as highly hazardous. Animal studies show that dogs, cattle, sheep, and wildlife are just as vulnerable as rats or mice. Australia banned the chemical for use in pest control decades ago after reports of farm dogs and sheep dying around treated fields. China and some other countries still use it, but accidental poisonings have kept happening. It’s not only a tool for pest management; it’s a risk for anything living nearby.
Exposure and Real-Life Impact
Anyone who’s worked at grain storage facilities or on a farm knows stories of animals getting into baited areas. A friend once lost two barn cats this way—the critters didn’t stand a chance, and the vet could do little. Often, folks using chemicals trust labels and rules, but the risk doubles when baits get picked up by birds or rodents carried away by pets. Even a garden with poisoned rodents can be a problem for local wildlife that eat carcasses.
Handling or mixing fluoroacetamide without proper gear brings trouble. Gloves, goggles, and full-body suits are standard in labs for a reason: a splash on the skin or a bit of dust breathed in is enough to set off serious poisoning.
Tracking numbers is tough because not all poisonings get reported, especially in poorer rural areas. WHO estimates thousands of deaths globally every year from rodenticide poisonings that include fluoroacetamide, mostly in countries with fewer regulations.
What Could Improve Safety?
Switching to safer alternatives has helped in countries that stopped using fluoroacetamide. Encouraging better storage, marked signs, and education for anyone handling chemicals works better than relying only on warning labels. Pet owners and farmers need clear instructions in their language, not just fine print in English.
Rapid treatment can save lives. Hospitals and vet clinics stocked with antidotes and basic awareness of the symptoms catch poisonings before they become deadly. Sharing real stories and teaching through local networks—community meetings, farm supply stores—brings the danger closer to home, where people might otherwise ignore a distant risk.
How should fluoroacetamide be handled and stored safely?
Understanding the Risks
Fluoroacetamide has earned a serious reputation in labs and agricultural settings. This isn’t your typical household chemical. Even tiny amounts can prove lethal. Its history includes use as a rodenticide, but many countries restrict it because accidental poisoning has claimed too many human and animal lives. Handling it with a casual attitude endangers not only the person opening the container, but anyone else nearby.
Real People, Real Stakes
I remember a phone call from a former classmate who worked in pest control. He described a situation where someone spilled a few grams because gloves tore. The result: an emergency trip to the hospital, panic across the whole facility, and a drawn-out cleanup. Their team learned the hard way that a single slip-up can have ripple effects on health and safety, not just for staff but for anyone living or working nearby.
Best Practices Save Lives
Always store fluoroacetamide in a locked, clearly labeled container—never just on an open shelf. Any container should have visible warnings and hazard symbols. Choosing a dry, well-ventilated place makes a difference, both for reducing fume build-up and making accidental spills less likely.
Spills can ruin a day fast. Keep absorbent materials nearby—those that won’t react with the chemical and can be disposed of safely. Nitrile or neoprene gloves, lab aprons, and proper goggles turn a risky task into a nearly routine one. Every lab manual calls for these. From experience, shortcuts tempt people who want to save ten minutes, but that attitude risks hours dealing with emergency services or worse.
Building a Culture of Responsibility
Younger workers and students sometimes feel pressure to “prove themselves” by skipping some formalities, thinking real danger only happens somewhere else. The opposite holds true: the professionals who make it through years of safe lab work never treat something like fluoroacetamide casually. Risk management isn’t just about ticking boxes for a supervisor or auditor. It means understanding how failing to follow procedures could permanently harm a coworker, customer, or even a family.
Learning From Accidents, Not Repeating Them
A 2022 review by the World Health Organization lists fluoroacetamide among high-risk pesticides. Some countries, including the United States and China, now require tightly regulated storage, tracking, and transport. Each incident—whether reported in big cities or remote farms—adds detail to guidance for the next generation, making rules more effective.
Consider regular training not as a chore, but as a way to keep up with best practices. Practice drills and spill-response training turn theory into muscle memory. Store emergency phone numbers where people actually see them, not hidden in a binder. Build and maintain a safety checklist for handling and storing toxic chemicals, and tweak it as new insights or regulations develop.
Practical Solutions, Not Empty Promises
Locking away dangerous chemicals and using proper gear might sound inconvenient at first. Still, so does explaining to authorities why you cut corners if something goes wrong. Storing fluoroacetamide the right way shows respect for yourself, your colleagues, and the community. This isn’t just a policy—it’s the only sensible way to work with something this dangerous.
What are the symptoms of fluoroacetamide poisoning?
Understanding What Happens to the Body
Fluoroacetamide comes up rarely in daily conversation, yet its risks shouldn’t be dismissed. Used at times as a rodenticide, it poisons by disrupting how cells produce energy, which feels as alarming as it sounds. People don’t just feel “off”—they experience symptoms that start subtly and then quickly snowball.
Early Symptoms: Not Just a Stomach Ache
The first signs leave a person guessing. Nausea, vomiting, and stomach pain appear within a few hours. It’s easy to mistake these symptoms for common food poisoning—until you realize they resist quick fixes. After working in a rural hospital for a few years, I saw more than one family come in thinking they just had a bad lunch, only to learn something toxic had slipped into their meal.
As the toxin gets deeper into the system, other warning lights start blinking. Shortness of breath, fatigue, and odd heart rhythms make things even more serious. The body fights to keep its organs supplied with energy, but fluoroacetamide blocks that effort. You can see the shift in someone’s color and hear the difference in their breathing.
Trouble Moves Fast: Nervous System Symptoms
The impact doesn’t stay in the gut or lungs. Most people think of poison just as “stomach issues,” but fluoroacetamide’s bite reaches the nervous system, too. Headache, dizziness, muscle twitching, and even seizures can show up, turning confusion to real danger. These changes usually scare family members, who come in worried their loved one just “isn’t acting right.”
Mental changes matter a lot. The person might seem restless or confused, their speech slurred. With time, the poison can push someone into a coma. Once a patient reaches this point, help needs to arrive quickly; the risk to the brain and heart jumps.
What Makes This Poison So Dangerous?
Unlike more familiar accidents, treatment for fluoroacetamide poisoning doesn’t follow a script. Hospitals don’t keep a magic antidote in the back room. In most cases, the team relies on activated charcoal, fluids, and support for breathing or heart rhythm. Doctors try to buy time for the person’s metabolism to break down and clear the poison.
What sits in my mind are the moments that could change the outcome—spotting the telltale mix of stomach pain and confusion early, not assuming every sick person just caught the latest bug. Toxicology labs sometimes confirm the diagnosis, but nurses, farmers, or family members are usually the first to guess what’s really wrong. Education makes a difference. Every hour lost can push someone closer to irreversible damage.
Staying Prepared and Protecting Communities
Every accidental or intentional poisoning story underscores something—chemicals don’t belong unsecured around children, foods, or community water supplies. The World Health Organization lists fluoroacetamide as hazardous, and countries have started to drop its use for a reason. People need to recognize symptoms early and act fast—call emergency services, share information on possible exposures, and bring samples of food or suspected products to help doctors connect the dots.
In the end, prompt action comes down to awareness. Healthcare workers, families, and teachers deserve training on these symptoms. Recognizing nausea, confusion, and trouble breathing all together could be what saves a life. In my view, prevention and sharp observation give fluoroacetamide much less power to harm.
Are there any regulations or restrictions on the purchase or use of fluoroacetamide?
A Dangerous Chemical in the Spotlight
Fluoroacetamide doesn’t often make headlines. Yet those who handle pesticides, toxicology, or agricultural chemicals know it’s no everyday substance. This rodenticide—strong enough to earn a spot on several “do not touch” lists—triggers big concerns about poisoning risks. Even a small mistake could endanger humans, animals, and the environment. With danger running so high, staying on top of laws that govern fluoroacetamide isn’t just a bureaucratic exercise. The rules around selling, buying, or using this stuff exist for a reason.
Worldwide Restrictions Take Different Shapes
Countries don’t all draw the same legal lines on dangerous chemicals. In the United States, the Environmental Protection Agency (EPA) blocks fluoroacetamide from legal pesticide markets. No one can register or sell it here. Penalties for trying to import or trade in it can include serious fines or even jail time—laws with real teeth, not just empty threats. The story is similar across much of Europe. The European Union has marked fluoroacetamide as “prohibited,” placing it under strict rules meant to limit exposure and protect public health.
Some places—China stands out—have faced high-profile episodes of illegal use tied to mass poisonings. The Chinese government moved to pull the chemical from agricultural shelves, but black-market circulation still pops up now and then. It’s easy to see that enforcement gaps can have tragic results. Japan, Australia, and other countries ban fluoroacetamide for most uses, giving out permits only when absolutely necessary, and usually only for research inside university labs or controlled environments. If someone orders fluoroacetamide without the right paperwork or a clear scientific reason, they’ll get a firm “no.”
Why Tight Control Matters
Stories about poisonings aren’t just headlines—they bring real suffering. The symptoms that follow accidental exposure to fluoroacetamide can hit fast, and they don’t offer much time to react: convulsions, breathing problems, rapid heart rate. I’ve talked with folks working in emergency medicine, and they agree—there’s hardly anything more frightening than treating a poisoning case when antidotes are rare or hard to access.
Wildlife gets hit just as hard. Because of how it spreads in nature, a single careless application can trigger a domino effect, wiping out not just pests but also birds, pets, and even carnivores who eat poisoned rodents. Environmental groups have published grim statistics showing the aftermath in areas with weak enforcement.
Better Solutions and Tighter Systems
I see real benefits in tougher licensing and tracking systems. Adding reporting requirements for anyone authorized to handle highly toxic compounds doesn’t just safeguard people—it helps protect whole communities. Technology can help here. Databases and digital barcodes, when layered onto shipments, let governments figure out quickly if something lands in the wrong hands. Mandating in-person training for licensed professionals moves beyond simple paperwork and ensures that handlers really know what they’re doing.
Education forms another crucial layer. The more farmers, lab workers, customs agents, and even teachers know about the danger posed by old-school poisons, the fewer accidents occur. Getting the word out about safer rodent control alternatives makes a difference. This doesn’t just keep people alive—it also protects farm ecosystems and food supplies from long-term harm.
Regulating fluoroacetamide isn’t about bureaucracy for its own sake. It’s about plugging the gaps that let tragedies unfold, making sure dangerous substances stay off the street and inside secure labs where they belong.
| Names | |
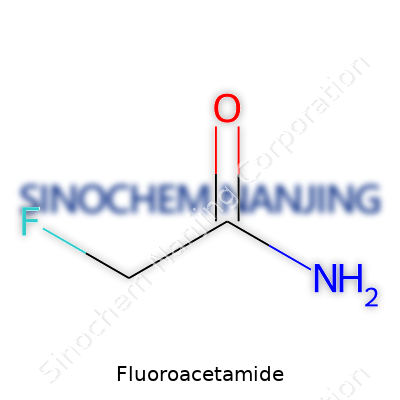
| Preferred IUPAC name | 2-fluoroacetamide |
| Other names |
Fluoacetamide Fluoracetamid FCH2CONH2 2-Fluoroacetamide |
| Pronunciation | /fluːˌɔːroʊəˈsɪtəmaɪd/ |
| Identifiers | |
| CAS Number | 640-19-7 |
| Beilstein Reference | 1717132 |
| ChEBI | CHEBI:5144 |
| ChEMBL | CHEMBL1460 |
| ChemSpider | 54614 |
| DrugBank | DB08230 |
| ECHA InfoCard | 100.004.078 |
| EC Number | 200-698-1 |
| Gmelin Reference | 9759 |
| KEGG | C14553 |
| MeSH | D005439 |
| PubChem CID | 9550 |
| RTECS number | LL9275000 |
| UNII | BKY455T20B |
| UN number | UN No. 2642 |
| Properties | |
| Chemical formula | C2H4FNO |
| Molar mass | 77.061 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.574 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.39 |
| Vapor pressure | 0.00273 mmHg at 25°C |
| Acidity (pKa) | 13.6 |
| Basicity (pKb) | 8.7 |
| Magnetic susceptibility (χ) | -41.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.372 |
| Dipole moment | 2.5466 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 160.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -535.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -559.7 kJ mol⁻¹ |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or absorbed through skin; may cause damage to organs; highly hazardous to humans and animals. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301: Toxic if swallowed. |
| Precautionary statements | Precautionary statements: P260, P264, P270, P280, P301+P310, P302+P352, P304+P340, P308+P311, P330, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-Poison |
| Lethal dose or concentration | LD50 oral rat 10 mg/kg |
| LD50 (median dose) | LD50 (median dose): 10 mg/kg (oral, rat) |
| NIOSH | UN-1694 |
| PEL (Permissible) | 2.5 mg/m³ |
| REL (Recommended) | 0.005 mg/m³ |
| IDLH (Immediate danger) | 100 mg/m³ |
| Related compounds | |
| Related compounds |
fluoroacetate fluoroacetic acid monofluoroacetic acid sodium fluoroacetate |

