Fluoranthene: A Closer Look at Its Role, Chemistry, and Outlook
Historical Development of Fluoranthene
Chemists stumbled upon fluoranthene in the thick tar left over from burning coal during the late 1800s, right at the dawn of modern organic chemistry. As folks broke down coal tar to pull out new chemicals, fluoranthene caught their eye thanks to its distinctive blue fluorescence. Back then, a lot of the science focused less on practical uses, more on the thrill of cracking secrets open. As leaders built more machines and tools relying on combustion and heavy industry, the presence of fluoranthene and its cousins — the polycyclic aromatic hydrocarbons (PAHs) — forced researchers to consider health and safety far earlier than many other industrial byproducts. Fluoranthene moved quickly from laboratory oddity to investigative target, especially in environmental tracking and public health debates.
Product Overview
Today, fluoranthene pops up as a specialty compound for labs testing air, water, oil, sediment, and other materials. Its use runs far beyond research, landing in reference standards for calibration, chemical analysis, and even pilot-scale synthesis of new functional materials. Most suppliers label it as a crystalline solid, usually in purified form, sometimes mixed with related PAHs for environmental monitoring research, or neat for manufacturing pathways demanding strict control over contaminants. Pricing and supply vary, shaped mainly by demands from the environmental testing sector and academic labs studying toxicity and mechanisms of action.
Physical and Chemical Properties
Fluoranthene looks and feels like many of its PAH relatives: flat, almost greasy needles or flakes, ranging from pale yellow to off-white. At room temperature, it barely dissolves in water. Given enough organic solvent — think benzene, toluene, or even acetone — it slips right in, making it useful for extraction and separation steps in chemical and environmental work. Its molecular formula, C16H10, reflects the alternation of fused rings, delivering high thermal stability and resistance to acids and bases, but not to strong oxidizing agents. Melting starts around 110-112°C, and at even modest heat, it gives off fumes that require attention in the lab. Its vivid blue fluorescence under ultraviolet light still surprises newcomers in analytical chemistry.
Technical Specifications & Labeling
Technical documentation for fluoranthene usually highlights purity, moisture content, packaging atmosphere, and trace impurities. Lab-grade batches tend to list purity above 97%, with high-performance liquid chromatography (HPLC) and gas chromatography/mass spectrometry (GC/MS) results provided. Labels spell out name, batch number, manufacture and expiration date, and hazard warnings according to the Globally Harmonized System (GHS). Reputable suppliers include instructions for handling, storage requirements — cool, dry, and out of direct sunlight — along with suggestions for safe disposal. Those working close to regulatory standards, especially in Europe or North America, rely on these labels to match local and international compliance requirements.
Preparation Methods
The main route to fluoranthene still takes cues from its early days, with coal tar distillation playing a starring role. Modern techniques blend distillation with chromatography to isolate the compound from other PAHs, then further purify with crystallization and filtration. For research, synthetic approaches sometimes use dehydrogenation or cyclization reactions starting from simpler aromatic hydrocarbons. Chemical engineers keep adjusting flow rates, temperatures, and packing materials based on scale, price point, or purity requirements. These steps generate crystalline fluoranthene ready for testing or further modification, though yields often depend on choices made at the very first fractionation stage.
Chemical Reactions & Modifications
Fluoranthene’s rigid structure holds up through mild reactions, but strong oxidizing agents carve out several products, including fluorenone and phthalic acids. Nitration, hydrogenation, halogenation, and sulfonation all churn out useful intermediates, which sometimes slip into the dyes, agricultural chemicals, or advanced material pipelines. Environmental chemists track how sunlight, bacteria, or other natural forces break down fluoranthene in soil or water. Each pathway hints at new methods for destroying or transforming the compound in hazardous waste streams. Some synthetic chemists look for ways to install functional side-chains or rings, aiming to improve solubility, reactivity, or biological activity for pilot studies.
Synonyms & Product Names
Lab catalogs list fluoranthene under its systematic name, along with alternatives such as "1,2-Benzofluorene," "o-Fluoranthene," and the core name "Benzo(jk)fluorene." Researchers running searches in large databases or libraries see a dizzying range of spellings and registry numbers, reflecting updates to chemical indexing systems through the years. Despite the confusion around names, molecular formula and structure usually settle any arguments for cataloging and procurement.
Safety & Operational Standards
This compound carries risk as a known PAH — gloves, goggles, and fume hoods are standard kit during any handling. Flammable dust, risk of inhalation, and skin contact hazards all get special mention on labeling and safety data sheets. Unfiltered exposure may contribute to long-term health problems, including cancer, so waste disposal and spill cleanup should always follow legal protocols. Safety drills and up-to-date training set the tone in good labs, while storage routines demand finished containers stay away from oxidizers, open flames, and moisture. The expectation is simple: treat fluoranthene as more than just a lab curiosity; treat it with respect shaped by occupational health experience, use engineering controls, and check monitoring devices in confined spaces.
Application Areas
Monitoring teams lean hard on fluoranthene as a touchstone for environmental contamination. It pops up in studies of soil, sediment, and water near cities, oilfields, highways, and industrial facilities. Air quality monitoring in urban hotspots uses fluoranthene as an indicator of incomplete combustion, especially from engines and power plants. Some specialty ink and dye manufacturers rely on its strong aromatic structure as a chemical precursor. Polymer scientists test modified fluoranthene derivatives in organic semiconductors and optoelectronic devices because electron-rich rings drive interesting conduction behavior. Occasionally, you find smaller research teams exploring its use in solar cell materials and nonlinear optics.
Research & Development
The push in R&D zeroes in on three fronts: environmental fate, chemical transformation, and new material development. Analytical chemists keep refining detection techniques to catch trace amounts in complex mixtures. Environmental engineers search for biological or chemical pathways that can safely degrade or detoxify fluoranthene in polluted zones. Synthetic researchers keep chasing ways to twist the core rings, hoping for improved performance in electronics or photonics. Competitive pressure and regulatory shifts spark new approaches to minimize exposure, remediate spills, or reclaim contaminated sites. Investment tracks the growing need for green chemistry solutions and more effective testing protocols for PAHs as a category.
Toxicity Research
Over decades, toxicologists documented fluoranthene’s persistence and tendency to lodge in soils, sediments, and living tissue. Animal studies linked repeated exposure to increased cancer risks, tissue inflammation, and harmful effects on the immune system. Unlike some PAHs, fluoranthene tends not to accumulate as quickly in human tissue, but its breakdown products spark concern for both acute and chronic toxicity in fish, amphibians, and mammals. Public health groups keep tracking exposure in air, food, and water, flagging the need for long-term studies on subtle, low-dose effects in large populations. Calls for stricter standards keep mounting as analytical capabilities sharpen and data piles up from different regions.
Future Prospects
Fluoranthene’s reputation as both a useful analytical tool and a health concern places it on the radar for innovation and regulation alike. Governments face rising pressure to clamp down on PAH emissions, driving demand for better removal and remediation technology. In chemistry, work continues to push fluoranthene’s potential in electronics and photoresponsive materials, especially as researchers prize stable aromatic frameworks for next-generation devices. As society doubles down on pollution monitoring and green tech, demand rises for cleaner preparation, smarter disposal, and ever-finer testing. Beyond these challenges, any breakthrough in harmless breakdown or transformation would change the landscape — both for industry and communities affected by legacy pollution.
What is Fluoranthene used for?
Digging Into Fluoranthene
Ask anyone in environmental health or chemistry, and they’ll tell you fluoranthene sparks as much debate as it does scientific curiosity. Most folks would recognize it as a mouthful of a word rather than a daily hazard, but this compound often pops up where people least expect it: in diesel exhaust, cigarette smoke, and charred food. In short, fluoranthene belongs to the big family of polycyclic aromatic hydrocarbons (PAHs), molecules formed when organic stuff burns—think coal, oil, tobacco, even the steak on your summer grill.
Why Anyone Cares About Fluoranthene
From my years working in the environmental field, I’ve seen how small details like residual air pollution shape big policy and personal choices. Fluoranthene holds a place in the spotlight because it appears in soil and waterways near heavy industry. Research published in journals like Environmental Science & Technology finds that fluoranthene lingers in sediment, refusing to break down for years. The trouble arrives when runoff from roadways, tire debris, or power plant emissions drags these chemicals into places where people and wildlife eat, drink, and live.
Some manufacturers harness fluoranthene for lab work, not direct consumer products. It helps create fluorescent dyes and can serve as a building block in organic electronics and solar cells. Researchers find value in its distinct fluorescence, which makes tracking chemical reactions or pollution movement possible. While cutting-edge, its industrial use stays niche. For the average person, the main worry is unintentional exposure, not handling the compound itself.
Public Health and Pollution
Growing up near a busy railway corridor, I breathed air touched by locomotive emissions every summer. Later studies backed up what the nose suspected: urban areas harbor higher concentrations of PAHs like fluoranthene. According to the U.S. Environmental Protection Agency, this class of pollutants links to cancer in lab animals and possibly humans. It doesn’t stop there—fluoranthene can harm fish and aquatic insects, disturbing entire ecosystems when it washes into creeks after storms.
Getting a true sense of impact requires context. The World Health Organization flags air pollution as a leading cause of chronic illness worldwide. Fluoranthene might play a smaller role than road-level particles or sulfur dioxide, yet it signals just how complicated pollution control becomes. You don't need to live next to a chemical plant for these issues to land on your doorstep. Just living in a city increases background exposure to PAHs.
Practical Steps and Solutions
No single fix wipes fluoranthene from the environment, but communities get ahead with a few smart choices. Tighter emission standards on vehicles and industrial plants cut off the problem near its source. Urban planners can favor green spaces that filter stormwater, keeping pollution out of local rivers and lakes. Advocacy by local groups pushes for soil testing around playgrounds, schools, and brownfields, making sure cleanups happen where families gather.
Each step toward less pollution feels small, but over time, the results show up as cleaner air and healthier neighborhoods. Awareness counts. When people understand the origins and dangers of substances like fluoranthene, they push leaders to make changes, leading to safer communities—and that’s a difference worth working toward.
Is Fluoranthene hazardous to health?
Understanding Fluoranthene
Fluoranthene shows up in places you might not expect. Burning coal, wood, gasoline, even cigarettes releases these invisible chemicals. It's not isolated to factory smokestacks—grilling outside, driving on busy roads, or living near high-traffic areas leads to regular exposure, sometimes without a person realizing it. Fluoranthene falls under the big group of polycyclic aromatic hydrocarbons (PAHs), a family known for causing concern in the health and environmental communities.
Why Should We Worry?
Research over the years paints a pretty clear picture—fluoranthene brings risks, especially over long periods. The main health worry comes from how the body processes this substance. Fluoranthene doesn’t usually act alone; it teams up with other PAHs, and together, they can damage DNA and mess with cells. Scientific studies have linked these actions to cancer, especially with repeated or heavy exposures.
The World Health Organization and the U.S. Environmental Protection Agency have classified PAHs based on these kinds of risks. Even though fluoranthene isn’t at the top of the list, nobody in medicine shrugs it off as harmless. It’s especially important for people who work in industries with high dust, soot, or fumes from combustion to take precautions, because these workers see some of the highest exposures.
Who Faces the Greatest Risk?
Not everyone has equal risk. People living near roads, busy factories, or plants that burn fossil fuels face higher levels in their air and soil. In cities, pollution from cars and industrial activities bump up background levels. Indoor pollution also matters—smoking, heating with wood, and even candle soot bump up fluoranthene indoors.
Children, whose bodies are still growing, breathe and eat more per kilo than adults. That means they can take in more pollutants for their size. This fact made me think hard about my own lifestyle when living in a city apartment; I noticed how soot gathered on my windows every week. If this residue lands outside, it’s also settling on playgrounds, parks, and gardens where children spend time.
Getting Ahead of Exposure
Good science gives us enough reason for action. Limiting the burning of fossil fuels at home, keeping windows shut during heavy traffic, and using air filters indoors knocks down risk. Cities that push for cleaner public transportation and fewer emissions see PAH numbers drop. For workers, protective gear and strong ventilation systems in factories work better than wishful thinking.
One solution sits in making better choices about energy. Cutting back on coal, switching to cleaner heating sources, and using fewer single-use plastics help everyone. Community action can speed up these changes, and education helps people spot sources and reduce risk. I learned this firsthand through community workshops that offered simple advice—like washing fruits and vegetables grown near busy roads.
Taking Next Steps
Fluoranthene reminds us that daily habits often bring hidden risks, but steps exist for everyone to push back. Listening to scientists, improving air standards, and staying aware of pollution in homes and workplaces brings health risks under control. People who have a say in policy can push for faster transition away from dirty energy and stricter industrial controls.
We all have a role in protecting health—at home, at work, and in communities. A little knowledge, some action, and talking about these issues moves things in the right direction for everyone.
What are the safety guidelines for handling Fluoranthene?
Understanding Fluoranthene Risks
Fluoranthene, a polycyclic aromatic hydrocarbon, shows up in creosote, exhaust fumes, and sometimes even grilled meats. Sometimes lab workers, site managers, or anyone involved in cleanup efforts will encounter this chemical out in the field or in the workplace. Health agencies like the Centers for Disease Control and Prevention (CDC) and the Occupational Safety and Health Administration (OSHA) have been clear: fluoranthene can bring health risks if it’s not handled with respect. Skin contact, inhalation, or accidental ingestion can irritate the eyes and respiratory tract, and—over long periods—may contribute to more serious health issues.
I remember a chemistry class where the professor drilled into our heads the necessity of solid lab safety habits. We worked with several compounds, and these early lessons stuck with me when I started my first job in environmental consulting. Chemical safety comes down to vigilance and common sense. With fluoranthene, it’s important people focus on direct protective measures rather than trust that the odds will stay in their favor.
Best Practices for Personal Protection
Before handling fluoranthene, start with personal protective equipment (PPE). Nitrile gloves provide a strong barrier against skin contact, and safety goggles protect the eyes from splashes or dust. Lab coats or chemical-resistant aprons keep particles away from regular clothing. Respirators, fitted for the right filtering capacity, matter most in environments where vapors or dust could linger. Facilities usually require these during spill response or larger cleanup jobs.
Always check that ventilation in the workspace is running. Fume hoods or local exhaust systems should be working every time you begin a task. Never try shortcuts or work in closed rooms—simple steps like this can make the difference between a normal day and a health scare.
Storage and Spill Management
Fluoranthene needs cool, well-ventilated, and dry storage. Use sealed containers with clear hazardous material labels. Lock these containers in cabinets away from sunlight or sources of heat. Keep compatible chemicals separate, since mixing can mean trouble. When transferring fluoranthene, use secondary containment like trays to catch spills. This stops the chemical from spreading if a bottle tips over or cracks.
Spill kits should always be within arm’s reach where any hazardous chemical gets used. An ideal spill kit for fluoranthene includes absorbent pads, dedicated chemical waste bags, and cleaning agents that break down or trap the compound. Quick cleanup is vital so that vapors don’t build up and skin exposure doesn’t happen. The team should know the emergency plan before they need it—drills and refresher training go a long way to keeping muscle memory sharp. This isn’t just about checking boxes; people’s health may depend on these habits.
Medical Response and Proper Waste Disposal
Contact with fluoranthene, especially in the eyes or on the skin, requires immediate rinsing under flowing water. If someone feels dizzy, breathes in vapors, or shows any contact symptoms, get them into fresh air and seek medical help. Keeping emergency eyewash stations and showers unobstructed might sound obvious, but it’s often ignored until the worst happens.
Disposing of fluoranthene waste means using clearly marked hazardous material bins. Never throw it in normal trash or pour it down the drain. Facilities should work only with certified hazardous waste handlers. This step keeps the community and environment safe, something I came to appreciate most after I saw how easy mistakes can spread outside a work site. Safety in chemical work rests as much on these habits as it does on science or regulations. Real protection comes from preparation and the willingness to stick to the basics day in and day out.
How should Fluoranthene be stored?
What Makes Fluoranthene Tricky
Fluoranthene sounds like the kind of chemical most people never run into, but industries use it in research, manufacturing, and testing labs all the time. It doesn't take much digging to learn that this compound doesn’t just pose a minor inconvenience—it can do real harm if ignored. Found in fossil fuels, cigarette smoke, and byproducts of incomplete combustion, fluoranthene isn’t friendly stuff. Being careful isn’t just about ticking off a few boxes on the safety checklist. Poor storage could spell trouble for workers, the environment, and the budget.
Why Storage Matters for Health and Safety
Not all chemicals deserve the same level of fear, but fluoranthene calls for respect. I remember handling it back in my lab days. The worry wasn’t only direct contact—it was the risk of fumes, dust, or leaky containers. The U.S. National Institutes of Health and OSHA both list this compound as a possible carcinogen, and that label should make anyone rethink how tightly they screw the lid back on. If fluoranthene gets loose, airborne particles threaten staff—mostly by inhalation or skin exposure. Over time, this can trigger headaches, breathing issues, or long-term effects still not completely mapped out.
Keeping fluoranthene far from food, medicine, or simple office supplies is just basic common sense. It deserves a place on the chemical storage shelf that promises no chance for cross-contamination, accidental mixing, or casual wandering from room to room. One overlooked spill or misplacement feels like an invitation for more serious trouble.
Best Practices: Real Solutions, Not Just Guidelines
Experience in a busy lab taught me a few golden rules. First, always use sealed, labeled containers—preferably made of glass or high-grade plastic that will never react with fluoranthene. Locking cabinets away from sunlight, heat sources, and moisture help slow any chemical breakdown. Darkness and a cool, stable temperature preserve the stability and cut down any chance for fires, since fluoranthene catches flame when the heat rises enough.
A separate, fireproof cabinet never hurts. Chemical storage guides from CDC and EPA agree with this: treat all semi-volatile, combustible chemicals with a wide margin of caution. Storing it with acids, bases, or oxidizers cranks up the risk; so smart workers keep all incompatible items apart. Clearly marking storage locations isn’t a “nice to have”—it tells emergency teams and new staff exactly what’s inside, even if the main label falls off or gets scratched.
Addressing Waste and Environmental Risks
Disposing of leftover fluoranthene takes just as much thought as storing it. Forgetting about old bottles, or letting them build up, can poison water and soil around waste disposal points. Local regulations often demand hazardous waste pickup—not a half-hearted flush down the drain. My own lab once faced a shutdown because stored chemicals had seeped through aging floor tiles. Salvage contractors charged a small fortune to clean it up, and the paperwork that followed felt endless.
Preventing that kind of disaster stands as the real reason for storing fluoranthene right. It saves money, protects staff, and shields neighborhoods from contamination all in one careful step.
Simple Habits, Big Impact
Training everyone—not just new hires—keeps those lessons fresh. Regular checks on containers and stockrooms don’t cost much, but they spot worn seals and missing labels before they cause harm. For anyone working with fluoranthene, safe storage means more than just following rules; it reflects a deeper responsibility to coworkers and the environment.
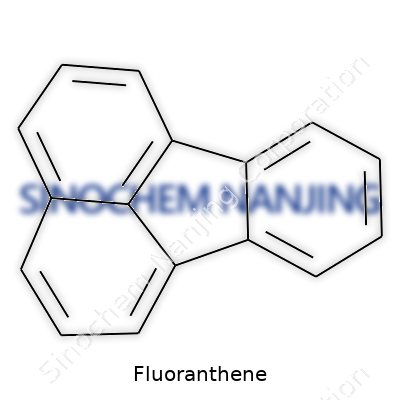
What is the chemical structure of Fluoranthene?
A Look at the Skeleton of Fluoranthene
I remember learning about polycyclic aromatic hydrocarbons in college chemistry, digging into why some of these ring-laden molecules grab so much attention. One that always popped up in soil and smoke samples was fluoranthene. Its structure isn't flashy, but it tells a complex story about environmental chemistry and the byproducts of fossil fuel use.
Fluoranthene’s Structure: More Than Just Rings
Fluoranthene carries the formula C16H10. It belongs to the polycyclic aromatic hydrocarbon (PAH) family. Looking at it on paper, you’ll spot four fused aromatic rings. Three of them form a straight path, and a fourth one clings to the side. Some chemists call its shape “bent” or “nonlinear,” which sets it apart from much neater, fully lined-up rings like those in anthracene.
If you’re curious about where to place the atoms, fluoranthene’s unique feature involves a five-membered ring stuck right in the middle, spliced between two benzene rings and linked to a third. This bent shape not only looks special but also tweaks how fluoranthene behaves in real life—how it reacts, where it goes in nature, and the roles it plays in pollution.
Why Pay Attention to This Structure?
It’s easy to overlook why such details matter, but in my lab experience, the exact layout of carbon atoms often helped predict how persistent a contaminant would be. Fluoranthene doesn’t dissolve well in water and sticks stubbornly to soils and sediments. Its non-linear design blocks easy breakdown by many bacteria, so it lingers in the ground and stacks up in riverbeds.
This sort of persistence tells researchers and policymakers plenty about how fluoranthene acts after escaping car exhaust pipes or seeping out of creosote-contaminated soil. The same intertwined carbon rings that dodge rapid breakdown also let it interact with organic matter and even move up food chains. That’s why you find fluoranthene in grilled meat, city dust, and fish tissue.
What to Do With This Knowledge?
Taking lessons from fluoranthene’s chemical structure means adjusting how we approach pollution. City planners, for example, can use this information to guide where stormwater flows, trapping dirty runoff before it hits waterways. In cleanups, chemists look for microbes or chemical agents that can break into stubborn fused rings. Some bacterial communities can even learn to nibble at those fused carbons more effectively—sometimes by adding nutrients to soils or adjusting pH levels in water.
I’ve seen firsthand how soil doused with creosote gets treated in bioremediation projects. Teams watch for changes not only in overall contamination but in levels of specific PAHs like fluoranthene. Technology has gotten better at measuring trace levels, but it’s the understanding of molecular structure that shapes everything from risk assessments to site cleanups.
Fluoranthene’s Story in Environmental Health
Researchers keep an eye on fluoranthene not just because it’s common, but because its chemical backbone offers hurdles for natural breakdown. Agencies like the Environmental Protection Agency monitor PAH levels in food and water due in part to these chemical quirks. The fused-ring format means health effects can range from skin irritation to possible cancer risk if exposure continues at high levels.
It’s not enough to sweep up the dust or clean the river’s surface. Real progress needs a combination of better science, technology, and everyday awareness, all shaped by the “skeleton” of problem chemicals like fluoranthene.
| Names | |
| Preferred IUPAC name | Dibenzo[def,mno]fluorene |
| Other names |
Benzo[j]fluorene 3,4-Benzofluorene Fluoracene |
| Pronunciation | /fluˈɔːr.ən.θiːn/ |
| Identifiers | |
| CAS Number | 206-44-0 |
| Beilstein Reference | 1903450 |
| ChEBI | CHEBI:28691 |
| ChEMBL | CHEMBL1406 |
| ChemSpider | 986 |
| DrugBank | DB08230 |
| ECHA InfoCard | tons-211-293-9 |
| EC Number | 205-912-4 |
| Gmelin Reference | 82151 |
| KEGG | C08217 |
| MeSH | D005473 |
| PubChem CID | 9157 |
| RTECS number | LL9275000 |
| UNII | S7X6G6FB8H |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C16H10 |
| Molar mass | 202.25 g/mol |
| Appearance | White to pale yellow-green crystals |
| Odor | Odorless |
| Density | 1.25 g/cm³ |
| Solubility in water | Insoluble |
| log P | 4.88 |
| Vapor pressure | 1 mmHg (at 177 °C) |
| Acidity (pKa) | pKa ~ -1.1 |
| Basicity (pKb) | 13.93 |
| Magnetic susceptibility (χ) | -10.9×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.640 |
| Viscosity | 2.5 mPa·s (at 25 °C) |
| Dipole moment | 1.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 267.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 77.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −6108 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V10BX02 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08, GHS09 |
| Pictograms | GHS07,GHS08,GHS09 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335, H351, H411 |
| Precautionary statements | P201, P202, P261, P273, P280, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 149 °C |
| Autoignition temperature | 550 °C |
| Explosive limits | Explosive limits: 0.9–5.9% |
| Lethal dose or concentration | LD50 oral rat 5,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 5,000 mg/kg (oral, rat) |
| NIOSH | NT 31532 |
| PEL (Permissible) | 0.2 mg/m³ |
| REL (Recommended) | 0.04 mg/m³ |
| Related compounds | |
| Related compounds |
Benzo[a]pyrene Phenanthrene Pyrene Naphthalene |

