Ferrous Perchlorate: Paths of Discovery, Use, and Care
Historical Development
Chemistry never stands still, and ferrous perchlorate—an iron(II) salt paired with the powerful perchlorate ion—has come into the picture through a mix of curiosity and chance. Chemists started its story by pushing iron compounds into new roles, testing their limits with strong oxidizers and acids. Researchers in the twentieth century, drawn by the rise of analytical chemistry, started producing perchlorates to explore their oxidizing abilities and their stubborn stability. Ferrous perchlorate entered the record as part of this fervor: iron and perchlorate thrown together, yielding a material that combined an ancient metal’s appeal with a modern ion’s chemistry. Every era, from classic inorganic labs to today’s advanced setups, has left fingerprints on ferrous perchlorate’s path. Historical diggings show it in old patents and scientific literature, usually surrounded by debates on safe handling and the ever-present risks of perchlorate chemistry.
Product Overview
Ferrous perchlorate isn’t found on every shelf, but its specific role draws in researchers and industry types who need iron(II) in a strong, water-soluble package. The pairing of iron and perchlorate changes up the usual iron salt chemistry: the perchlorate’s non-coordinating nature leaves iron(II) more accessible for redox activity and complex formation. While rare in daily life, this compound finds its community wherever scientists want a precise, active source of iron(II). Compared to sulfates or chlorides, ferrous perchlorate allows chemists to step outside common limitations, pushing redox work or catalysis further.
Physical & Chemical Properties
Ferrous perchlorate usually appears as a crystalline solid, often pale green thanks to the iron(II) center. Solubility in water sets it apart from less dramatic iron(II) salts. Its real character shows up under the microscope and through chemical testing—perchlorate ions stay silent in coordination, but their oxidizing power can’t be ignored if given the right trigger. Unlike organic perchlorates, it shows more stability, though heating or contact with organics always calls for caution due to explosion risks. The iron(II) ion inside tends to react with air, gently nudging the state toward iron(III), which keeps chemists on their toes during prep and storage.
Technical Specifications & Labeling
Labeling practices draw lines around ferrous perchlorate’s risks and standards, with suppliers focusing on ruling out cross-contamination and water content. Chemically, buyers and users look for purity—both perchlorate and iron need to show up at expected ratios, with trace impurities kept in check. Testing often centers on oxidation state analysis, moisture assay, and perchlorate ion confirmation. Because the compound handles sensitive redox chemistry, even small shifts in content can crash a process or change outcomes, so trust in both specs and supplier honesty makes all the difference in research and application.
Preparation Method
Handling strong acids and reactive metals puts a spotlight on the prep process. Typically, chemists build ferrous perchlorate by reacting iron or iron(II) salts with perchloric acid, often under a flow of inert gas. The procedure calls for slow addition, temperature management, and clean-up of by-products—each action reduces the risk of iron(III) contamination and runaway oxidation. Once the reaction winds down, crystallization and careful drying finish the process. Mistiming or sloppy technique here doesn’t just reduce yield; in the presence of organic matter or heat, mishaps have led to real accidents. Those who know, work these syntheses behind shields and with good ventilation, keeping iron(II) unspoiled and perchlorate away from sparks.
Chemical Reactions & Modifications
Ferrous perchlorate jumps into the action in redox experiments. The iron(II) center gives up electrons to a range of oxidants, shifting easily to iron(III) and feeding into broader reaction cycles. Coordination chemistry books show off the compound’s tendency to form complexes with nitrogen or oxygen donors, with the perchlorate ion usually hanging back as a spectator in the company of bulky ligands. Some labs hunt for ways to swap perchlorate for other anions to fine-tune reactivity, but for many processes, the inert nature of perchlorate provides just the background needed to focus on iron’s behavior alone. In mixed reactions, ferrous perchlorate sometimes pushes other salts or reagents in new directions, especially in organic synthesis where selective reduction or oxidation matters.
Synonyms & Product Names
Ferrous perchlorate goes by several names in older literature and global trade—iron(II) perchlorate, ferrous(II) perchlorate, or perchloric acid iron(II) salt all pop up in papers and catalogues. The chemical formula Fe(ClO4)2 keeps things clear, though confusion with ferric perchlorate, its iron(III) cousin, leads to mix-ups for those not checking oxidation states. Naming rests mainly on the oxidation number, so sticking to “ferrous” rather than “ferric” saves headaches in the lab and in trade.
Safety & Operational Standards
Real experience with ferrous perchlorate means treating it with respect—both perchlorates and iron(II) demand strict handling. Perchlorate-based salts offer the risk of serious explosion if contaminated with organics, dust, or struck by sparks. Iron(II), while less dramatic, oxidizes easily to iron(III), sometimes releasing heat and changing reaction outcomes. Standard operating procedures drawn up in responsible laboratories involve gloves, eye protection, fume hoods, and serious storage guidelines: avoid heat, mixing with flammables, and uncontrolled open containers. Spills require special disposal methods. Institutions with chemical hygiene plans put ferrous perchlorate under restricted use, often logging its movement and use to prevent misuse or unnoticed build-up. Emergency planning, including safe quenching and neutralization, runs alongside daily operations.
Application Area
Ferrous perchlorate picks up attention mainly in specialist laboratories—redox chemistry, analytical procedures, catalysis, and prepping coordination compounds take up most of its industrial footprint. Certain fields, like photochemistry and organic synthesis, depend on pure iron(II) sources, and perchlorates let researchers avoid the interfering side reactions seen with other counterions. Environmental scientists sometimes use iron(II) perchlorate as a reference in soil or water studies analyzing perchlorate fate and stability. Pure research into electronic structures, magnetism in transition metal complexes, and high-energy chemistry also finds a partner in this salt, though in small, careful batches rather than tons.
Research & Development
Much of the recent research around ferrous perchlorate digs into its redox abilities, complex-building skills, and influence as a starting material. Labs chase better, safer synthetic paths that reduce waste and lower accidental oxidation. Analytical chemists probe the subtle effects perchlorate has on electron transfer rates, while materials scientists look for new complexes or hybrid materials with iron(II) centers for magnetic or catalytic use. Detailed studies routinely check stability under varying conditions, seeking understanding on decomposition points, potential pollutants, or ways to sensibly recover iron or perchlorate after use. Such work means lots of trial, error, and learning—students and experienced researchers alike shape the evolving story of this compound.
Toxicity Research
Nobody ignores risks with any perchlorate. Ferrous perchlorate demands extra care because both perchlorate and iron can end up harming people and the environment. Perchlorates disrupt thyroid function if they get into the water supply, a fact supported by studies monitoring exposed communities. Iron(II), in large enough doses, produces its own toxicity—though most cases concern more common iron compounds. Toxicity testing has shown the need for closed systems, containment, and good lab hygiene, especially since accidental exposure comes less from vapors and more from spills or direct contact. Regulatory groups and environmental researchers keep monitoring perchlorate’s impact, shaping safety codes and suggesting improved alternatives in sensitive applications.
Future Prospects
Ferrous perchlorate’s next chapters hinge on how labs and industries weigh its benefits against the growing focus on safety and environmental stewardship. As more is learned about perchlorate’s environmental fate and persistence, regulations around its use continue to tighten in many regions. Researchers keep chasing stable, less hazardous iron(II) sources, but in very controlled settings, ferrous perchlorate still holds unique value for certain reactions and analysis. Advances in containment, real-time sensors, and automated synthesis help reduce risks and open chances for new chemistry involving the iron(II) center. The drive for greener materials and less persistent pollutants could also spur innovation in recovery and recycling methods for perchlorate-containing wastes, reshaping the compound's role in the long term. Experience tells those working with it to stay alert to both its promise and its perils, shaping thoughtful stewardship in labs and industries alike.
What is Ferrous Perchlorate used for?
What Ferrous Perchlorate Brings to the Table
Ferrous perchlorate stands out because it holds iron in the +2 oxidation state, paired with a strong oxidizing anion, perchlorate. In industrial labs, this compound barely makes a ripple for everyday folks, but to specialists, it’s a small tool with outsized consequences. It doesn’t build cars or show up in household cleaners, but it sits in a niche where chemists push boundaries or look for answers no other salt can offer.
Lighting Up Analytical Chemistry
Looking back, I remember my first year in a research lab, surrounded by ampules of compounds with names I could barely pronounce. Some were there for routine tasks, but ferrous perchlorate had a sharper job: it came out during complex analysis of transition metals. Analysts often seek out sharp contrast or particular redox reactivity, and this compound fits when you want a reliable ferrous source that brings no extra baggage from anionic contaminants.
It serves well in redox titrations, where knowing exactly what’s reacting matters more than anything. Fe(II) ions convert quickly, which lets scientists pin down reaction steps or pinpoint unknowns with confidence. This accuracy ripples out to fields like pharmaceuticals, where one small measurement shifts millions of dollars in drug quality and safety.
Chemical Synthesis: Not Just Another Reagent
Researchers love ferrous perchlorate for building more complex molecules, especially when crafting coordination compounds or new materials. If you’ve got an experiment that wants a perchlorate counterion—say, to control crystal growth or to limit interference—this salt shows up with just what you need. The purity and defined oxidation state make a difference at a level where impurities can ruin days or weeks of work.
It played a key part during my graduate research, as our group worked on synthesizing new types of organometallic complexes. Only a few iron sources gave us the reproducibility we craved, and ferrous perchlorate made the shortlist. Consistency, even at this level, breeds trust in data and can help unlock new discoveries.
Questions on Safety and Accessibility
Some folks raise eyebrows when they spot “perchlorate.” There’s a reason: perchlorates can be dangerous, depending on use and waste handling. Industry pros know the story—rigorous protocols, tightly sealed storage, meticulous records. It reminds me of how many steps we take in labs to make sure innovation doesn’t come at the expense of human health.
Perchlorates in water supplies have prompted environmental watchdogs to push for tighter regulations. Labs turning to ferrous perchlorate need to plan waste management up front, so contaminants don’t leave the fume hood and hit the wider world. Small choices here—opting for neutralization and containment—scale up into public health wins.
Better Alternatives, Safer Practices
As interest in sustainable chemistry grows, some researchers hunt for alternatives that sidestep the perchlorate risks and deliver similar reactivity. Ferrous sulfate and ferrous chloride step in where specific perchlorate chemistry isn’t needed, sparing both the lab and environment unnecessary exposure. Manufacturers improve safety data sheets and storage packaging all the time to help users stay one step ahead. The more transparent the risks and best practices, the more trust the scientific community earns.
The Bigger Picture
Ferrous perchlorate may be obscure to most, but it has a real place in the world of chemical research. Specialists depend on it for clarity and reliability, both in the classroom and the lab. Its use, like that of other powerful chemicals, carries responsibility—toward both science and society.
What is the chemical formula of Ferrous Perchlorate?
The Formula Unwrapped
Ferrous perchlorate carries the formula Fe(ClO4)2. This combination brings together iron in its +2 oxidation state and the perchlorate ion, ClO4-. In lab language, it means you’re pairing up one Fe2+ ion and two perchlorate ions. Nothing fancy, just simple chemistry at work. If you’ve ever taken apart a compound in high school, it draws on the routine method of balancing charges: iron sits at +2, perchlorate comes with a hefty -1, and a little arithmetic sorts the stoichiometry.
Where Real Use Shows Up
In my early college years, I once watched a classmate handle perchlorates with such reckless confidence that a safety advisor stepped in and shut the whole group down. Working with iron(II) perchlorate isn’t a casual job. This salt stands out for its strong oxidizing qualities. Even seasoned chemists eye perchlorates with caution since they react with organic materials and can go off if given half a chance. In an academic or industrial context, you rarely find ferrous perchlorate just sitting around. Most of the time, it steps into the ring as a reagent for complex syntheses, especially for research chasing new materials or investigating redox properties.
Data from various safety studies highlight that perchlorate salts, including the ferrous type, can pose challenges for storage and transportation. The oxidizer label requires dedicated training, especially for the young techs who might not yet take chemical hazards seriously. No matter how much experience you clock in a lab, the risks tied to perchlorates push for extra layers of safety gear and ventilation. The Environmental Protection Agency addresses perchlorate contamination, shining a huge spotlight on the compound’s environmental persistence and the way it messes with thyroid function after exposure.
Why the Formula Still Matters
Some of the latest research circles around iron-based catalysts and batteries. Chemists hunting for cleaner, longer-lasting battery solutions check every angle, including lesser-known salts like ferrous perchlorate. The simple formula, Fe(ClO4)2, pops up in journals, hinting at its role in energy technology or advanced redox reactions. Every time I glance over a new synthesis that pivots to perchlorates, I recall the importance of clear labeling. Labs that mislabel even a single bottle might wind up with explosive consequences. In some older chemistry labs, you still spot faded labels—an accident waiting for the wrong set of conditions.
Facing the Hazards Head-on
Safe handling of any perchlorate salt springs from upfront education and solid lab infrastructure. Schools and companies should invest in regular safety drills. Mix-ups with simple formulas sometimes trigger near-misses, especially when shared workspace gets crowded. Proper waste management steps in as the unsung hero. Rules laid down by major scientific bodies, along with strict adherence to environmental disposal guidelines, keep communities safe. Ferrous perchlorate deserves a place in the chemistry playbook for its usefulness but demands respect for its hazards. I’ve seen far more projects interrupted by poor chemical housekeeping than by funding cuts. Keeping the humble Fe(ClO4)2 on your radar sharpens awareness for everyone who cracks open a fresh bottle in the lab.
Is Ferrous Perchlorate hazardous or toxic?
Getting Real about Risks
Ferrous perchlorate doesn’t usually stick around in everyday conversation, but if you work in a lab or a chemical plant, the name means something very specific: danger in a bottle. This isn’t a compound you’ll find under your kitchen sink; the chemical world knows perchlorates, in general, have a reputation that is anything but friendly to health or the environment. With ferrous perchlorate, you get a combination that brings the usual risks—plus a few extras nobody wants to breathe, touch, or drink.
Why Perchlorates Raise Eyebrows
I grew up with a chemist in my family. He told me early on that the perchlorate ion itself has a tendency to explode under the right conditions and almost always plays dirty with human health. The Environmental Protection Agency doesn’t mince words: perchlorates get called out for messing with iodine uptake in the thyroid. Even small traces in water prompt health advisories, and agencies across the globe ban or strictly limit perchlorate exposure.
Ferrous perchlorate combines the instability of perchlorates with iron. Ferrous salts on their own can bring health risks—think about rust poisoning if ingested in significant quantities—but perchlorate ramps up the stakes. Inhalation or even skin contact with ferrous perchlorate dust or solution can bring out the worst symptoms: breathing problems, skin burns, and eye irritation. Swallowing this salt is a fast route to metal poisoning, thyroid disruption, and with larger amounts, potentially fatal internal injuries. That isn’t a scare tactic—that’s straight from hazardous material studies and firsthand response reports.
Blasts, Burns, and Environmental Fallout
On the industrial side, ferrous perchlorate isn’t invited to the party without good reason. It shows up mainly where strong oxidizing agents are needed, like rocket propellants or exotic lab experiments. But the same reactivity that makes perchlorate useful also creates headaches: improper storage sets up powerful fires or explosions. One spill, a static spark or heat source nearby, and things get out of control. Those aren’t rare events—industrial accident databases list perchlorate-triggered blasts with alarming regularity. Cleanup isn’t simple, either. Perchlorates don’t break down easily, so they continue to threaten groundwater and soil if not properly contained and managed.
Safeguarding Workers and Waterways
I’ve seen chemical safety done right and wrong. In a lab where perchlorates are handled with respect, people stick with personal protective gear, chemical fume hoods, and sealed containers. Training isn’t an option—it’s a must, and the goal is no accidental exposure, ever. Waste disposal needs to stay sharp and aware: sending perchlorates down a regular drain is forbidden for good reason. Compliance inspections catch surprises more often than most would guess. Reliable detection and monitoring systems help keep the material out of drinking water, but smaller operations sometimes skip critical steps. Here, government regulation and penalties play an important role.
People sometimes ask if all the worrying is overblown. The evidence says otherwise. Strong guidelines exist for reasons grounded in decades of research and real-life disasters. I believe the more people understand about why ferrous perchlorate gets labeled hazardous and toxic, the better decisions can be made on every level—from national regulators to folks running small research projects. Respect for the risks isn’t fear—it’s a practical response.
How should Ferrous Perchlorate be stored?
A Substance Not to Shrug Off
Anyone who’s spent time in a lab or hung around industrial chemists knows that ferrous perchlorate brings a load of responsibility. This isn’t kitchen-table chemistry. Fe(ClO4)2 does more than just stain countertops. It craves stability but rarely settles for it. Many folks hear “oxidizer” and think bleach or pool shock; ferrous perchlorate punches above its weight and grabs oxygen with both hands, ready to escalate a simple mistake into chaos.
What Makes Storage Tricky?
Moisture, heat, friction, and stray organics press all the wrong buttons with perchlorates. These materials have powered propellants and pyrotechnics for decades because a little energy goes a long way. Ferric and ferrous forms amp that up. A damp bottle or a leaky cap will not only corrode your workbench — it may start heating up materials you never meant to react in the first place. Chemists who’ve cleaned up after badly stored perchlorates remember it. Some never took shortcuts again after seeing the aftereffects of a minor spill.
On top of the chemical risks, perchlorates tie into environmental regulation. The EPA tracks commercial users due to risks of water contamination and thyroid impacts. Stories of perchlorate in municipal drinking water haven’t helped its reputation. Storage becomes not just about safety but about meeting regulatory baseline and community trust.
How the Real World Handles Ferrous Perchlorate
Nobody wants to be the person who ignores a warning and triggers an incident. The smart move follows a clear path: store this material in airtight glass or compatible plastic, far from flammables or reducing agents. Steel shelving works, but why tempt fate with bare metal if rubber-lined cabinets exist? Never share a space with acids, combustibles, or anything that might spark. Some labs go as far as isolating a whole fridge for unstable oxidizers. Pay attention to humidity — once you see clumping or color changes, discard it by the book.
Labeling sounds like paperwork, but it pays off. Every jar or drum should spell out not just what’s inside but its hazard class and date received. I’ve seen too many mystery bottles turn into minor emergencies when some intern, unsure of what they’re holding, inadvertently sets up a dangerous situation. Even old-timers keep up with their documentation because the alternative gets expensive and, frankly, scary.
Practical Steps for Safer Storage
Packed knowledge beats shelf technique. Training never ends in a good lab or warehouse. Staff run drills for spill management and keep up to speed with the latest NFPA and OSHA guidelines. Regulators aren’t the only people who want that — everyone involved benefits when nobody’s scrambling to figure out how a reaction is going to behave in an emergency.
Routine inspection forms a safety backbone. Look for leaks, corrosion, dusty caps, or compromised seals. Dispose of old material using approved waste vendors, not down the drain or in general trash. Make sure fire extinguishers in the storage area match the hazard (water won’t cut it here).
Room for Improvement
Manufacturers and end users invest in second-generation packaging that limits exposure to air and moisture. Some facilities set up environmental monitoring for temperature and humidity. This isn’t just ticking boxes — it prevents real-life incidents that make headlines. If perchlorates remain in a work environment, expect investment in better ventilation, segregated spaces, and constant improvement.
Ferrous perchlorate behaves as an unforgiving material, so handling it demands more than textbook answers or short-term memory. Safe storage depends on experience and disciplined oversight. These are the habits that let everyone go home at the end of the day.
What are the safety precautions for handling Ferrous Perchlorate?
Why This Chemical Commands Respect
Anyone who’s worked in a lab or plant setting knows ferrous perchlorate means serious business. This chemical shows up in energetic material applications, research, and synthesis work for good reason. With a substance like this, one mistake can lead to more than just a ruined experiment—it can send someone to the emergency room or even worse. I’ve had a fair share of tense moments around strong oxidizers, and complacency is a luxury nobody gets with compounds like this.
What Makes Ferrous Perchlorate Dangerous
Ferrous perchlorate acts as a powerful oxidizer. That quality makes it useful, but also raises the stakes. If it comes into contact with organic matter or even just a greasy rag, there’s a risk of ignition or explosion. The dust itself is hazardous; even small spills can cause troubles in the right (or wrong) conditions. I remember a cleanup drill we had after a minor oxidizer spill, and nobody slept well for a week.
Practical Steps For Staying Safe
Not every lab cuts corners, but cutting corners with this one often comes back to bite. Here are some lessons learned and rules that should stick in your mind:
- Personal protective equipment isn’t just for show. Gloves rated for chemical use, splash goggles, and lab coats save skin, eyes, and clothing every single time. Face shields add reassurance when weighing or transferring the material. In my experience, nitrile gloves stand up better than latex to perchlorates, but check them for pinholes.
- Only open containers inside a certified fume hood. The dust travels, and nobody wants to inhale oxidizer. Good lab ventilation puts a barrier between your lungs and trouble. If your hood doesn’t pass the airflow test, don’t trust it.
- Storage rules matter. Ferrous perchlorate should never live near flammable or organic stuff. I keep it in a solid, airtight glass container, far away from glacial acetic acid and lower shelves, so it doesn’t take a long fall. Moisture tight packaging keeps out water, cutting down the risk of violent reaction.
- Avoid static and friction. Everything slows down around this material. Grounding yourself before handling, using plastic or antistatic utensils, and working on a clean, dry, static-safe surface make a difference. I’ve seen static clips produce peace of mind worth the extra minute.
- Never work alone. If something goes wrong, another set of hands can call help, flush eyes, or operate a fire extinguisher. This isn’t paranoia; it’s teamwork that could save your life.
- Prepare spill kits in advance. Vermiculite, sand, and special oxidizer-neutralizing absorbents earn shelf space in any good prep room. Water won’t help and can actually set off a reaction—learn the right first-aid methods for each chemical you use.
- Dispose of waste properly. I don’t toss oxidizer-contaminated gloves or containers into the regular trash. Special chemical waste bins exist for a reason, and your local rules might make the difference between a safe site and a shut-down lab.
Making Safety Routine, Not Optional
I’ve learned that regular training takes the guesswork out of emergencies. Review the safety data sheets, conduct fire drills, and run spill cleanups just like you’d practice a fire drill at home. Most labs are only as safe as their habits, so keep good habits sharp.
Ferrous perchlorate doesn’t forgive mistakes, but it responds to respect. Solid practices, clear procedures, and an honest understanding of the risks matter more than fancy equipment or bravado. A few basic steps, done every time, bring everyone home at the end of the day.
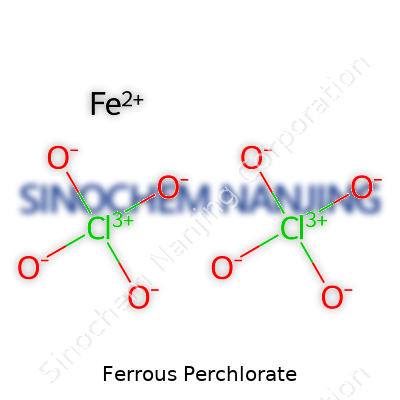
| Names | |
| Preferred IUPAC name | Iron(II) perchlorate |
| Other names |
Iron(II) perchlorate Ferrous perchlorate Iron diperchlorate |
| Pronunciation | /ˈfɛr.əs pərˈklɔː.reɪt/ |
| Identifiers | |
| CAS Number | 16923-51-8 |
| Beilstein Reference | 4268463 |
| ChEBI | CHEBI:139432 |
| ChEMBL | CHEMBL3315957 |
| ChemSpider | 16213553 |
| DrugBank | DB14638 |
| ECHA InfoCard | ECHA InfoCard: "100.031.403 |
| EC Number | 221-508-6 |
| Gmelin Reference | 37229 |
| KEGG | C18615 |
| MeSH | D019278 |
| PubChem CID | 16210852 |
| RTECS number | SC9100000 |
| UNII | 36UOW8P607 |
| UN number | UN1473 |
| Properties | |
| Chemical formula | Fe(ClO4)2 |
| Molar mass | 286.84 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.43 g/cm3 |
| Solubility in water | Soluble |
| log P | -4.14 |
| Vapor pressure | Negligible |
| Acidity (pKa) | -2.0 |
| Basicity (pKb) | -7.0 |
| Magnetic susceptibility (χ) | +7430.0e-6 cm³/mol |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 239.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –1029 kJ/mol |
| Pharmacology | |
| ATC code | B03AA01 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS03,GHS06 |
| Signal word | Danger |
| Hazard statements | H271: May cause fire or explosion; strong oxidizer. |
| Precautionary statements | P210, P220, P221, P260, P280, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3-0-4-OX |
| NIOSH | NA8200000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m3 |
| Related compounds | |
| Related compounds |
Iron(II) sulfate Iron(II) chloride Iron(II) nitrate Iron(II) acetate Iron(III) perchlorate |

