Ferrous Arsenate: A Closer Look from Past to Future
Historical Development
Ferrous arsenate tells a curious story reaching back to the 1800s. Back then, chemists mixed iron and arsenic compounds looking for new pigments and agricultural aids. It showed up in Victorian pigments or as an early attempt to manage pests on farms. Over the years, regulatory changes and modern understanding of toxicology shifted the focus away from casual use towards greater care and research. Knowing this, looking at ferrous arsenate today feels like reading two stories at once: the raw chemistry, and the lessons learned about health and safety.
Product Overview
This compound, often appearing as a powder or small crystalline solid, links iron to arsenic using oxygen atoms. Usually, the commercial product sports a light green or pale yellow hue, which hints at the iron content. Labs and certain factories keep it around for specialized use, especially where iron’s reactivity with arsenic matters. Its current footprint feels small compared to other iron compounds, mostly due to safety and regulatory concerns.
Physical & Chemical Properties
Ferrous arsenate, with the formula Fe3(AsO4)2•8H2O, forms in a hydrated state. Water content gives crystals stability but also restricts certain applications. It barely dissolves in water, making it stick around in the environment—a blessing for some uses, and a problem when thinking of waste cleanup. At higher temperatures, it can break down, letting off arsenic compounds that demand special handling. Its density sits around 3.87 g/cm³. These features, though textbook, draw a clear line: it’s a robust, heavy mineral, and not something that fades quietly into the background.
Technical Specifications & Labeling
Bags or barrels of ferrous arsenate carry sharp warnings due to the poison within. Labels flag arsenic content and urge gloves, goggles, and other safety gear. The global system for classifying chemicals stamps it as extremely hazardous if swallowed, inhaled, or handled carelessly. EU’s REACH and US OSHA guidelines push strict boundaries around exposure—few labs keep much on hand. Purity standards usually dictate iron and arsenic both stay above 98% by weight, and moisture below 15%. Trade documents show technical data, lot numbers, handling instructions, batch purity, and shelf lives measured in years, not months.
Preparation Method
Chemists bring ferrous arsenate into the world by mixing ferrous sulfate with sodium arsenate under water. A double displacement reaction makes the insoluble ferrous arsenate drop out, which they filter and dry. Every step requires attention. If left to chance, impurities like ferric forms or mixed arsenates creep in, undermining quality. Clean water, deliberate stirring, and precise temperatures help keep rogue phases at bay. Some labs tweak the pH for a finer grain, while big manufacturing plants monitor every stage using real-time analytics, hunting for that sweet spot of consistency.
Chemical Reactions & Modifications
Ferrous arsenate plays a role in redox reactions, where shifting balance between Fe(II) and Fe(III) states nudges its chemical fate. Strong acids dissolve it, letting out toxic arsenic, so working with it demands respect for chemistry and safety. When heated past 200°C, it breaks down—generating iron oxides and letting off arsenic trioxide, a known human carcinogen. Over the years, industrial chemists learned to create mixed-metal arsenates with manganese or zinc, tailoring properties for battery research or pigment production. These exchanges shape not just what the compound is, but how and where it fits.
Synonyms & Product Names
Trade catalogues or chemistry texts might call it iron(II) arsenate, ferrous arsenate hydrate, or even ferrous meta-arsenate. Old papers from before digital indexing show names like “arsenous iron salt” or “green arseniate of iron.” Modern supply lists may use CAS Number 10102-50-0 to clear up confusion, since similar names swim around in iron chemistry. Searches across international databases turn up matched synonyms, so checking the fine print on safety sheets is smart practice.
Safety & Operational Standards
No one in their right mind handles ferrous arsenate with bare hands. My first safety lecture about arsenic hammered home the brutal risk—chronic exposure can cripple the nervous system, shred kidneys, and spike cancer risk. Spills aren’t swept under the rug; trained staff in sealed gear handle cleanup, and any waste streams hit special regulated disposal routes. There’s no shortcut around regular air quality and hands-washings checks in workplaces. If it gets into water, fish and wildlife suffer. Hard rules from OSHA, EPA, and the EU’s CLP regulation make sure anybody using it invests in top-notch ventilation, emergency plans, and routine blood checks for staff. Scrimp on safety, and a trail of lawsuits and sickness will follow.
Application Area
Most uses these days trace back to niche research or legacy pesticides—especially before the big regulatory clampdown of the 1970s. In rare cases, some mining or metallurgy teams still rely on ferrous arsenate to help manage arsenic in ores. It shows up in geochemistry labs studying how metals move through rocks or groundwater. Early chemists sprinkled it on crops as an insecticide; today, this practice lands squarely on the “do not repeat” list. Medical interest peaked long ago, but the toxic load pushed it out of any kind of regular pharmaceutical use. You might see it in reference pigment studies or as a teaching sample in mineralogy courses, always sealed tight and kept far from careless hands.
Research & Development
Modern researchers keep exploring ways to make arsenic in industrial waste less dangerous. Ferrous arsenate’s stubborn insolubility lets scientists lock up arsenic, making cleanup easier around former mines and factories. Studies track how it forms, how fast it breaks down, and what tweaks change its stability. Teams experiment with adding extra elements like manganese, trying to push solubility down further. Nanotech groups test ferrous arsenate-based systems in batteries or sensors, chasing new functions while sidestepping the toxicity that casts a shadow over every project. The tension runs deep: the need for reliable chemistry against an ever-present background of health risks.
Toxicity Research
Early stories about arsenic read like cautionary tales—poisoned wells, wasted fields, and public health disasters. Toxicologists today approach ferrous arsenate with sharpened tools, measuring how much leaks out of the solid under real-world conditions. Studies show iron’s grip on arsenic weakens if acid levels rise, turning sealed contamination into a community threat. People exposed to even small airborne dust levels for years may face cancer, vascular disease, or organ failure. In animals or fish, experiments show slow build-up in tissues. Once inside, arsenic interferes with cell enzymes, causing widespread damage that can’t be ignored. Field reports trace the fate of ferrous arsenate spills decades after cleanups are declared complete; stories of regulatory failures still surface from time to time. This mess keeps health researchers, regulators, and cleanup crews busy long after headlines fade.
Future Prospects
Nobody expects ferrous arsenate to stage a mainstream comeback. Too many red flags sit next to its name—tight restrictions and public pressure slam the door on widespread use. Future research aims at safer ways to immobilize arsenic, engineering more stable forms for waste locks or spill barriers. Some teams look to synthetic analogs or heavily engineered nanostructures aiming to hold arsenic even tighter, with less chance of release under changing conditions. Battery research may touch its edges for specialized roles, but smarter materials and better risk management nearly always take priority. Education and transparency stand out as the best protection—making sure anyone who reaches for ferrous arsenate knows exactly what they’re holding, what it can do, and what damage it can cause if care slips.
What is Ferrous Arsenate used for?
Why People Use Ferrous Arsenate
Ferrous arsenate doesn’t show up on grocery lists, but you might bump into it if you dig around in old agriculture manuals. For a good chunk of the twentieth century, this chemical helped farmers deal with bothersome pests. Mostly, it’s been used to control insects that threaten crops, especially those underground types like wireworms and cutworms. Folks looking for an answer to stubborn infestations relied on ferrous arsenate because it worked. Farmers liked that it stuck around in the soil and got the job done, even if it came with risks. That old tin of bug killer gathering dust in a wooden barn corner might very well be this compound.
The Drawbacks No One Ignores Anymore
Decades back, nobody thought much about environmental fallout. More people pay attention now, and for good reason. Ferrous arsenate, like many arsenic-based chemicals, can linger in soil and water. The World Health Organization points out that arsenic exposure can mean serious trouble for people and animals, including cancer and developmental problems. Even small amounts can build up, so it’s clear this isn’t a chemical for backyard gardens or playgrounds. Some older farmers still talk about cases where overuse poisoned nearby wells or left patches of soil completely useless for planting anything anyone would dare to eat.
What About Other Uses?
You might hear about ferrous arsenate in certain pigment applications, but it’s rare now. Lab workers might use it to show a particular chemical reaction—it’s never been a common pick for mainstream industry outside of pesticides. Iron and arsenic don’t combine for strength or building; instead, this odd couple has mostly meant pest control or scientific study. For big manufacturing, there are safer and cheaper substitutes. The risks and tight government rules mean you won’t find it in household paint, dyes, or medicines. Anyone who still handles this material in a lab has to keep it under strict controls, storing it well away from anything fragile or easily contaminated.
No Quick Fixes for the Past
Some of the places where ferrous arsenate was dumped or used heavily still show traces today. I’ve seen it firsthand in my region where certain plots are marked off because crops just won’t grow—tests came back with arsenic levels above what’s safe. Cleaning contaminated fields isn’t as easy as tilling the dirt. Specialized companies step in, dig out soil, and haul it to treatment facilities. These jobs don’t come cheap, and local governments often struggle to pay for full cleanups. Sometimes, the only answer is to grow grass and keep people out. That’s a hard lesson when food and water safety is on the line.
Better Options Exist
Turning away from ferrous arsenate didn’t leave growers empty-handed. The shift moved agriculture toward integrated pest management and newer, less toxic chemicals. Safer options cost more at first, but they do less harm in the long run. For everyday folks, that’s peace of mind when you bite into a carrot or tap a well. These days, more people want answers about what’s in their food and water, and most want assurance that what worked for the past isn’t making anyone sick now.
Is Ferrous Arsenate toxic or hazardous?
The Toxicity Lurking Behind the Name
Ferrous arsenate sits in a category of substances many folks would rather not cross paths with. Arsenic already carries a notorious reputation, tied to tales of poison and pollution, and combining it with iron doesn’t soften its reality. Many people ask if ferrous arsenate is hazardous or simply misunderstood. Let’s get straight about what’s at stake.
Real Life Encounters with Arsenic Compounds
Years ago, I helped test soil in an aging orchard, and the memory stuck with me. Farmers used arsenic-containing pesticides for decades. Even as those chemicals faded from use, their residue remained. Ferrous arsenate, like other related compounds, doesn’t disappear with rainfall or time. It lingers, clinging to the dirt and making its way into plants, water, and sometimes even the food on our table.
Long-term exposure to arsenic compounds in soil and groundwater links tightly to health risks—skin problems, cancer, neurological drag, and heart issues show up more often where people breathe or eat small doses over time. Ferrous arsenate isn’t an exception simply because its name starts with “ferrous.” It poses real risks because its arsenic doesn’t flush out of bodies or environments easily.
Science on the Table: Not Just Anecdotes
Toxicologists have spent years documenting these risks. The International Agency for Research on Cancer, part of the World Health Organization, counts arsenic and its compounds, including ferrous arsenate, as carcinogenic. Breathing in dust or swallowing contaminated particles often kicks off slow-moving health problems. Children run bigger risks than adults, mostly because they play outdoors and hands find their way to mouths.
Some experiments show arsenates disrupt metabolic processes at the cellular level. They get in the way of enzymes and block the body’s natural functions. Even in lower amounts, chronic exposure makes people sick over months or years. Drinking water in contaminated areas can contain enough arsenic to breach safety thresholds set by EPA and other agencies, compounding the issue beyond just old orchards or industrial sites.
Reducing Exposure—A Community Task
Everyday safety means taking these risks seriously. Homeowners, farmers, and workers in places with industrial history should test water and soil if contamination seems possible. Protective clothing, gloves, and masks cut down the risk for folks working on old land or handling chemicals. Washing hands, sticking to safe play habits for kids, and keeping food growing away from known contamination zones helps a lot.
Larger issues call for group action. Community groups partner with local governments to remediate soil and replace tainted water sources. Some cities bring in new soil for playgrounds or install modern filtration where arsenic levels run high. These fixes aren’t cheap or quick, but they work over time—for generations, not just the next harvest season.
The Bottom Line on Ferrous Arsenate’s Hazards
Ferrous arsenate carries more than a complicated name—it punches above its weight in terms of risk, especially in places with a history of chemical use. Knowing what’s in the soil and water makes all the difference. Action at the household and community level can prevent small exposures from ballooning into lifelong health problems.
What is the chemical formula of Ferrous Arsenate?
Breaking Down the Chemistry
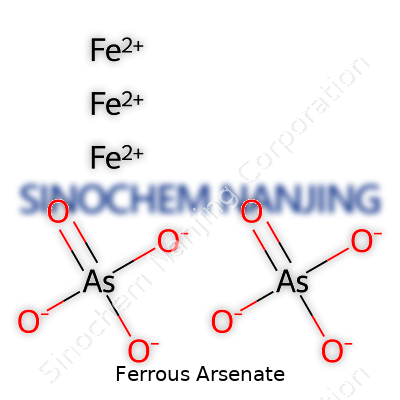
Anyone curious about chemistry probably wonders how something as specific as ferrous arsenate gets its formula. The answer comes down to iron’s behavior in its +2 state and arsenate’s nature as a complex oxyanion. In ferrous arsenate, iron (Fe) brings a +2 charge, pairing up with the arsenate anion (AsO43-). The chemical formula Fe3(AsO4)2 shows how three iron atoms link up with two arsenate groups to balance out all the charges.
Uses and Importance
Ferrous arsenate may never end up on a supermarket shelf, but it has played a role in a handful of material and environmental processes. Soil, for example, can contain this compound. Its limited solubility means it doesn’t easily dissolve into water, which keeps arsenic from leaching out and polluting groundwater in many areas—an issue millions face globally. In my days working with water treatment technologies, ferrous arsenate kept cropping up as a way to lock away arsenic safely, limiting the risk of contamination.
Arsenic Contamination and Possible Answers
Arsenic poisoning has haunted communities, especially where groundwater gets tapped for drinking in countries like Bangladesh and India. The World Health Organization has labeled arsenic one of the most dangerous contaminants. Here, ferrous arsenate’s stability under normal conditions can help: researchers build filters and barriers that encourage the formation of these kinds of compounds. Well-designed water treatment tanks sometimes introduce iron salts. As water percolates through, arsenic binds and gets sequestered as ferrous arsenate. In rural installations I’ve observed, this simple chemistry has protected families from serious illness.
The Toxic Side
Handling any arsenic-containing compound requires care. Workers dealing with ore mining or refining—especially those involved with minerals like scorodite, a natural form of iron arsenate—must control dust and personal exposure. Chronic low-level arsenic exposure causes organ damage and cancer. Putting safety first must never be negotiable for these industries. From gloves to proper venting, the right steps save lives. My own background in occupational safety underscored the risk; even with strict protocols, mistakes hurt real people.
What Can Be Done?
Better awareness, education, and technology give communities real hope. Government support for upgrading infrastructure and testing water—using field kits that detect arsenic at low levels—should become standard in high-risk zones. I’ve seen firsthand how local training programs empower residents to check and treat water independently. On the science front, researchers continue searching for even more efficient ways to capture and neutralize arsenic, aiming for compounds that resist breaking down or leaching.
Final Thoughts on Responsibility
Understanding the chemical formula of ferrous arsenate means more than memorizing numbers and letters from the textbook. It connects chemistry to real-life problems, from industrial risk to public health victories. Every time a water treatment expert, a scientist, or a local leader helps communities keep arsenic levels down, the knowledge behind Fe3(AsO4)2 proves its value outside the lab.
How should Ferrous Arsenate be stored and handled safely?
Real Risks Call for Real Precautions
Many people overlook the dangers some industrial chemicals carry. Ferrous arsenate sits among them. Touching or inhaling dust from this compound exposes workers and bystanders to health issues; folks who work around it must pay close attention to safety. I spent a lot of years in facilities where hazardous materials showed up on the shelves: my respect for storage rules grew each time the emergency alarm sounded. Ignoring protocols often ends with a trip to the hospital—or worse.
Understanding Ferrous Arsenate’s Health Hazards
Ferrous arsenate carries risks because it contains arsenic, notorious for its toxicity. Even a small amount taken into the body harms the nervous system, lungs, skin, and kidneys. Extended exposure builds up toxic effects over time, especially if the chemical gets into the air or water supply. I’ve known colleagues who let their guard down, thinking a little exposure “wouldn’t hurt.” They changed their minds after symptoms went from mild headaches to long-lasting breathing trouble. Serious chemicals require persistent caution.
Smart Storage Practices Make the Difference
Keeping ferrous arsenate out of reach begins in the storage room. Only store this chemical in sealed, labeled containers. Never use makeshift buckets or unlabeled jars. For extra protection, choose containers made to resist corrosion and keep moisture outside. Damp air doesn’t just ruin material inside—it can speed up dangerous leaks. Place containers in a cool, dry spot without direct sunlight. Chemicals respond to heat and light in ways most folks underestimate. Every site I’ve worked in used lockable cabinets or designated rooms with strong ventilation for this reason. Too many avoidable spills happen from improper shelving or poor airflow.
Personal Protection Matters Every Time
No shortcut exists here. Workers who handle ferrous arsenate need reliable gloves, safety goggles, and full-coverage clothing. Reusable gear should get cleaned right after each use, away from break rooms and high-traffic areas. I learned many lessons after accidental splashes and unseen dust landed on my face shield. If people cut corners, residue lingers, and trouble follows. Fit-tested respirators also play a crucial role, especially if any dust threatens to settle around breathing zones. Some companies still treat protective equipment as an afterthought. Real-life emergencies proved the opposite—these steps keep families from receiving calls nobody wants.
Training Saves More Than Compliance
Strong storage policy needs a team that understands what’s at stake. Regular training makes sure everyone on site knows how to identify risks, use gear, clean up safely, and respond to emergencies. Posters help, as do drills that rehearse leaks or spills. Real situations test how well people pay attention to lessons. In every plant I worked, staff meetings drove home the message: “If you think something looks off, speak up.” That courage prevented more than one disaster.
Clear Labels and Waste Disposal Prevent Disaster
Every drum or small container deserves a legible label that spells out the danger. Sloppily written tags create confusion in busy storerooms. All wastes, rags, or leftovers must go in designated, marked bins—not into general trash, not down any drain. Licensed hazardous waste disposal services exist for a reason. Years ago, a careless disposal caused an evacuation and a visit from the regulators. That’s a mess nobody wants to create or clean up.
Safe Storage Stands as Everyone’s Job
Cutting corners with ferrous arsenate never pays off. Shared vigilance, solid training, and strict routines decide how safe a workplace stays. Never assume today will be fine just because yesterday passed without trouble. People’s health depends on the habits developed around the most dangerous materials in the room.
What are the physical properties of Ferrous Arsenate?
Getting to Know the Basics
Ferrous arsenate doesn’t show up in the headlines, but its physical properties tell a story that few substances can match. With a chemical formula of Fe3(AsO4)2, this mineral forms a pale green powder or crystalline solid that’s almost insoluble in water. You try to dissolve it, it just settles to the bottom. I’ve seen this firsthand in chemistry classes — mix it into your average beaker and you’ll wait a long time before anything shifts. This reluctance to dissolve is one reason it’s played a role in agricultural formulations aimed at keeping certain compounds locked in place.
Stability and Structure
The powder comes off as stable, not quick to react. Storing a sample, you won’t notice it giving off gas or changing color. Under bright light or heat, ferrous arsenate holds up, up to a point. It has no real odor, which makes it easier to handle than some metal arsenates that fill the air with a whiff of garlic. Its structure, based on orthorhombic crystals, means it forms well-defined grains, sharp-edged if you look close with a magnifier. This matters if you’re working with filtration, because blocky crystals flow differently compared to soft, fluffy powders.
Weight and Texture in the Real World
If you’ve ever scooped up a jar of ferrous arsenate, you’d feel it — heavier than chalk dust, but far from dense like iron filings. The bulk density floats between 2.0 and 2.5 grams per cubic centimeter, which gives engineers clear rules for storage and handling. The powder feels slightly gritty. Pour it from bag to beaker, it doesn’t cling with static. It settles fast, which I find useful if you need to measure or separate solutions in a hurry.
Color and Visual Clues
The pale green color isn’t just cosmetic. It tells you about the iron sitting in the +2 valence state, confirmed by X-ray and electron microscopy over the years. Spot anything off — brownish tints, murky hues — and you’re seeing impurity or oxidation. Even a beginner can use color as a simple check, especially in remote labs or places with limited gear.
Why All This Matters
Knowing how ferrous arsenate acts in the physical world isn’t just for the lab. Farmers used to slip it into pest control, because it didn’t dissolve away in rain, and didn’t blow as easily as lighter powders. In waste water treatment, the property of near insolubility turned it into a tool for trapping poisons in a form that is harder to spread. Still, I’ve seen concerns over arsenic leaching grow louder. Soil and water scientists point out that even small releases can linger and harm life. Strong policies and safer alternatives now take priority, but for older cleanup sites, understanding these properties can make or break a remediation plan.
Looking for Solutions
Switching to less toxic compounds isn’t always simple. Scientists have worked on encapsulation or creating new crystal forms that trap arsenic even better, cutting the risk of exposure. Regulators now demand regular checks, not just of bulk piles, but of dust in the air and runoff in water. With better detection and regular training, toxic mishaps should drop — if rules get followed. My experience tells me that clear specs and hands-on training beat theory every time. So when handling ferrous arsenate, folks lean on real measurements, no shortcuts, and respect for what those pale green grains can carry.
| Names | |
| Preferred IUPAC name | Iron(2+) arsenate |
| Other names |
Iron(II) arsenate Ferrous arsenite Iron arsenate(2-) |
| Pronunciation | /ˈfɛr.əs ɑːr.sə.neɪt/ |
| Identifiers | |
| CAS Number | 10028-22-5 |
| Beilstein Reference | 87838 |
| ChEBI | CHEBI:86155 |
| ChEMBL | CHEMBL1201571 |
| ChemSpider | 113505 |
| DrugBank | DB14671 |
| ECHA InfoCard | 03b06c6e-b831-4115-bda3-6d80cb7c800d |
| EC Number | 234-109-2 |
| Gmelin Reference | 68244 |
| KEGG | C19700 |
| MeSH | D005247 |
| PubChem CID | 163278 |
| RTECS number | BQ9625000 |
| UNII | 6Q8K7G6Z69 |
| UN number | UN1586 |
| Properties | |
| Chemical formula | Fe3(AsO4)2 |
| Molar mass | 241.72 g/mol |
| Appearance | Light green powder |
| Odor | Odorless |
| Density | 2.87 g/cm3 |
| Solubility in water | Insoluble |
| log P | -36.65 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 11.27 |
| Magnetic susceptibility (χ) | +2320.0e-6 |
| Dipole moment | Zero Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 109.9 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | -938.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -206.7 kJ/mol |
| Pharmacology | |
| ATC code | B03AB04 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause cancer; very toxic to aquatic life |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS07, GHS09 |
| Signal word | Danger |
| Hazard statements | H301: Toxic if swallowed. H332: Harmful if inhaled. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P264, P270, P273, P280, P301+P317, P330, P302+P361+P353, P319, P305+P351+P338, P337+P317, P308+P311, P501 |
| NFPA 704 (fire diamond) | 3-1-0 |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 100 mg/kg |
| LD50 (median dose) | > 112 mg/kg (rat, oral) |
| NIOSH | XN8225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Ferrous Arsenate: "0.01 mg/m³ (as As) |
| REL (Recommended) | 0.05 mg As/m³ |
| IDLH (Immediate danger) | 15 mg As/m3 |
| Related compounds | |
| Related compounds |
Iron(II) oxide Iron(III) oxide Iron(II) phosphate Iron(III) arsenate Sodium arsenate |

