Ferric Citrate Dihydrate: A Detailed Look
Historical Development
Ferric citrate has its roots in the intersection of nineteenth-century chemistry and medicine. The compound began showing up in medical records and chemistry patents as scientists tried to manage conditions tied to iron metabolism and phosphate retention. Pharmaceutical history books often bring up citrate salts as key turning points, showing how ideas about kidney health and anemia evolved alongside advances in inorganic chemistry. Back then, resources were modest, yet researchers tested this salt across patient groups, adjusting manufacturing to provide purer forms. As production scaled up, manufacturers moved from small-scale crystallization to more precise, quality-focused processes, setting the stage for today’s pharmaceutical-grade ferric citrate dihydrate. This evolution came from real clinical demand—patients with chronic kidney disease needed better options, and there was growing awareness about the consequences of phosphate buildup. So, the path from curiosity to necessity pushed ferric citrate into the mainstream, and industry experts saw plenty of opportunity for further research.
Product Overview
Ferric citrate dihydrate shows up in pharmacy shelves as a fine, rust-colored powder. In a world full of complicated drug labels, this one ships with a clear purpose: it binds phosphate in the digestive tract for those who need to manage kidney-related issues, and serves as an iron supplement. The dual use is no accident; developers recognized the compound’s affinity for phosphate and reliability in releasing iron. Beyond medicine, some industries employ it in water treatment and pigment production. Regulatory bodies have signed off on certain uses, and hospitals keep it on hand for renal patients. Iron supplements have always had a narrow lane between helpful and harmful—ferric citrate fits because it is relatively stable, and offers controlled release in the stomach, providing some peace of mind when treating delicate patient populations.
Physical & Chemical Properties
Anyone taking a close look will note its brick-red appearance, which hints at the heavy iron content. It dissolves in water with some coaxing, due to its hydrates. This powdered form can clump together unless handled properly. Under a microscope, ferric citrate dihydrate reveals angular crystals, and chemists point out the ferric ion’s tendency to form stable complexes with citrate. Formula-wise, it balances Fe3+ with the organic acid, and the bound water molecules ensure it stores well without drastic changes during normal humidity swings. Those familiar with chelating agents will recognize why stability matters: degradation or loss of hydration alters both potency and safety, so storage rules remain strict in pharmacies and warehouses.
Technical Specifications & Labeling
Manufacturer certifications must meet a long checklist. United States Pharmacopeia publishes clear thresholds for purity, allowable impurities, moisture content, and assay ranges, all enforced with regular batch testing. Label information spells out the concentration of elemental iron, recommended storage conditions (dry, cool), and batch traceability. Hospitals and retail pharmacies look for clear expiration dating and allergen statements. Labels also mark intended use: mineral supplement or phosphate binder. Years in warehouses taught many in the business the importance of batch integrity to avoid cross-contamination; manufacturers include tamper-evident seals and detailed handling instructions, which help downstream users maintain compliance and patient safety.
Preparation Method
Chemists walk through a familiar route using ferric salts and citric acid. A controlled reaction in aqueous conditions accomplishes the initial combination; the pH is kept acidic but not extreme to keep ferric ions in solution. As reaction proceeds, additional water ensures the hydrate forms correctly. Slow evaporation or cooling allows crystals to form, then repeated washing removes excess acids. Drying at moderate heat completes the dihydrate state. Processing teams monitor color and solubility, since deviation signals mistakes in reaction proportion or temperature control. Plant operators have plenty of stories about the headaches caused by moisture swings and raw material changes. It’s easy for a batch to shift toward a monohydrate or anhydrous form if care slips, so hands-on expertise remains the best insurance against bad product.
Chemical Reactions & Modifications
Ferric citrate participates in reactions typical of metal-chelate complexes. In the presence of strong acids or bases, citrate can break from the ferric ion, making the compound less useful as a binder or supplement. Exposure to reducing agents shifts iron from the ferric (Fe3+) to the ferrous (Fe2+) state, which changes both color and reactivity. This property sees use in redox titrations and industrial pigment applications. Flexible synthetic chemists modify the ligand environment for research purposes—attaching other carboxylate groups or mixing with additional metal ions can tweak solubility or stability. Researchers continually explore new modifications to create more selective phosphate binders or to enhance oral absorption in anemic patients. Small changes in molecular structure deliver a surprising range of new properties, so there’s always another experiment underway.
Synonyms & Product Names
The product pops up under several names. International catalogs list it as ferric ammonium citrate, ferric citrate hydrate, or iron(III) citrate dihydrate. Pharmaceutical products get branded names based on marketing departments’ input, commonly highlighting ease of absorption or specialty for kidney patients. Clinical literature typically opts for ferric citrate, but the water content is always stated to avoid confusion with the anhydrous version. Patent filings can add regional variations: in Japan and Europe, formulas may shift slightly, prompting alternate names. Regulatory authorities tend to prefer CAS numbers for clarity in import/export, but anyone working in manufacturing needs to stay alert for synonym confusion when placing bulk orders.
Safety & Operational Standards
Factory floors and pharmacies adopt strict rules for handling. Product dust poses risks for allergies and, rarely, iron overload. Material Safety Data Sheets call for gloves, particle-filter masks, and good ventilation during bulk processing. Spills can stain floors and clothing, and iron deposits build up fast on equipment if cleaning slips. Storage areas track humidity to prevent unwanted hydration or caking. Occupational health teams update safety guidelines after accident reviews in chemical plants, and those lessons trickle down to supplier contracts and pharmacy protocols. On the clinical side, overdose reveals as gastrointestinal distress or, in vulnerable patients, iron toxicity—dosing guidelines are carved from years of post-marketing surveillance. Safety isn’t a paperwork exercise; every recall or patient complaint sparks a review of everything from warehouse pallets to packaging seals.
Application Area
Healthcare takes the lion’s share: ferric citrate dihydrate serves patients with chronic kidney disease, helping remove excess phosphate and supplement iron without complicated infusion regimens. Doctors like the oral route, which offers comfort and less monitoring. Dieticians plan dosing around meals to maximize phosphate binding, and pharmacists keep a steady supply for transplant and dialysis units. In water treatment, ferric citrate functions as a mild flocculant, promoting removal of metals and contaminants. Artists and pigment suppliers value its color stability, using it in inks and textile dyes. These varied applications stem from strong, predictable iron chelation—one compound, multiple markets, all tied back to its basic chemistry.
Research & Development
Large hospital networks share clinical trial data with drug manufacturers, tracking absorption rates, side effects, and therapeutic benefits in real time. University labs dig deeper, investigating structure-activity relationships by using analogs and iron isotopes. Patent offices track a steady flow of filings on combination therapies, delivery systems, and even new uses such as antimicrobial action. Research budgets reflect optimism, aiming for new drugs that do more than just bind phosphate—future versions may target hereditary iron disorders and autoimmune kidney diseases. The drive for innovation owes a lot to patient advocacy, as real lives are shaped by small differences in absorption or tolerability. Funding from public-private partnerships accelerates proof-of-concept trials, which keeps ferric citrate at the edge of both science and practical medicine.
Toxicity Research
Iron toxicity has kept researchers busy for decades, knowing that not everyone processes iron the same way. Acute overdose causes distinct symptoms—nausea, vomiting, black stools, and in severe situations, metabolic acidosis. In laboratory models, high doses may trigger oxidative stress and liver injury. Carefully managed clinical studies have shaped dosing recommendations, flagging patient populations with hereditary hemochromatosis or active gastrointestinal bleeding as high risk. Regulatory scientists rely on this work to update package inserts, while emergency medicine teams keep protocols current for accidental ingestion. Toxicologists study combinations with other drugs to predict drug-drug interactions, knowing patients rarely take just one medication at a time. Research continues to look for markers that spot early iron overload, aiming for prevention well before symptoms start.
Future Prospects
Looking ahead, more tailored versions of ferric citrate dihydrate promise to enter the market. Researchers are optimizing release profiles and absorption curves, working toward single-dose formulations that match individual kidney function and dietary habits. Machine learning models already sift through clinical records to find patterns suggesting which patients benefit most, turning bulk chemical production into personalized medicine. The expansion into new therapeutic areas—such as treating rare, iron-processing diseases—shows investment continues to grow. Supply chains face challenges as interest spreads into low- and middle-income countries, pushing manufacturers to develop scalable, cost-effective processes. With increasing environmental awareness, research into biodegradable or recyclable packaging for iron-based drugs joins the agenda. The balancing act has never been simple—between efficacy, safety, affordability, and innovation—but ferric citrate dihydrate keeps finding new ways to prove its value in both old and emerging fields.
What is Ferric Citrate Dihydrate used for?
Treating Chronic Kidney Disease
Ferric citrate dihydrate finds its place in the world largely because of people who live with chronic kidney disease (CKD). Many years ago, I watched a close friend of mine struggle through the endless routines of dialysis. Alongside everything else, he had to track his phosphate levels like a hawk. Healthy kidneys keep phosphate in check, but CKD throws that balance out fast.
The body needs phosphorus, but too much can harden blood vessels and make the heart work harder. Ferric citrate dihydrate works here by binding phosphate in the gut. The combination leaves less to slip into the blood and cause problems. Patients have long relied on phosphate binders like calcium acetate or sevelamer, but for some, ferric citrate dihydrate steps in when those don’t do enough—or bring side effects. Several studies, supported by reputable journals like Kidney International, show ferric citrate not only lowers phosphate but helps manage iron levels. Doctors see value in hitting two birds with one stone: controlling phosphorus and topping up dwindling iron stores, both common challenges for people with CKD.
Supporting Iron Deficiency
Iron deficiency can knock the wind out of a person. Anemia, fatigue, pale skin, all can stem from simple iron loss or non-absorption. It makes sense: red blood cells need iron for carrying oxygen. I’ve spent time volunteering at clinics where patients struggle to keep their energy up, and iron is often the culprit. Ferric citrate dihydrate comes into play for folks who can’t take standard supplements, usually because their gut can’t handle it, or absorption falls short.
Its mechanism doesn’t rely on big technology or elaborate procedures. The iron in ferric citrate can absorb in the digestive tract and gradually replenish the body’s stores. That means less dependence on infusions or risky workarounds for many patients. Clinical trials, including those reviewed by the New England Journal of Medicine, confirm improvements in iron levels for patients with CKD who stick to a ferric citrate plan.
Beyond the Hospital Setting
Most folks think only of medications and treatments, yet ferric citrate dihydrate also shows up in food fortification. Some breakfast cereals and nutritional drinks quietly include it to support people who need their iron built up, especially in populations where meat or green veggies aren’t easy to get.
I grew up in a community where iron deficiency in children was a fact of life, and iron-fortified foods made a concrete difference. Ferric citrate offered an effective dose without the metallic taste that often made kids refuse their medicine. That might sound minor, but anyone who’s ever tried coaxing a five-year-old to swallow something chalky will know—it’s not.
What Needs More Attention
While ferric citrate dihydrate shows clear benefits, it isn’t for everyone. People with too much iron or specific allergies should steer clear. Overuse risks iron overload, a real problem for those with hereditary conditions like hemochromatosis. Medical professionals urge regular monitoring, blood tests, and open discussions about side effects.
Healthcare staff could do more to educate patients on phosphate control and iron balance. Pharmacies could offer clearer information, not buried in packet inserts. Research still moves ahead to understand long-term use cases and risks. Safety comes from both community awareness and strong clinical guidance. I’ve seen the difference straightforward communication can make, especially for older adults managing multiple medications at once.
Finding a Balance
Ferric citrate dihydrate carries real weight in patient care, especially for people dealing with kidney disease and persistent iron shortages. It offers more than medicine; for many, it brings normalcy back to daily life—enough energy to pick up grandkids, to go for a walk, to stay connected with family. That’s something worth improving and safeguarding with both research and compassion.
How should I take Ferric Citrate Dihydrate?
Learning What Ferric Citrate Dihydrate Does
Ferric citrate dihydrate usually ends up on a prescription list for people with chronic kidney disease. The main reason doctors choose it: it helps control phosphorus levels in the blood. For anyone with failing kidneys, keeping phosphorus under control is a big deal. Too much of it puts stress on the bones and heart and drags down energy levels. The pill combines iron with something called citrate. This combo binds with phosphorus and carries it out of your body, mostly through stool.
Lots of folks receive a container of big brown tablets and feel unsure what the next step should look like. I’ve seen patients hesitate in the pharmacy, nervous about side effects or worried about how a new pill will change their routine. Having watched family struggle with kidney issues, I know that sticking to even a simple medication plan gets tough fast. Life’s interruptions add up: meals don’t always come at the same time, and your stomach isn’t always ready for the same food.
How to Take Ferric Citrate Dihydrate Safely
This medicine works best alongside a meal. The idea is pretty simple: the food in your stomach helps the tablet find and grab extra phosphorus. Trying to guess if you can take it alone on an empty stomach won’t give the same results. Consistency matters most; pick regular meal times and make taking your pill part of those moments. If breakfast, lunch, and dinner fall at different hours on a busy day, set alarms. Busy nurses, working parents, or anyone juggling appointments know alarms and pill boxes aren’t just for grandparents.
Swallow tablets whole with water. Chewing or crushing changes how your body absorbs the medication—something most people overlook. If you have trouble swallowing pills, let your doctor know. Sometimes, adjustments to your prescription make all the difference.
What to Expect and Watch Out For
No one likes surprises with side effects. The most common ones: your stool could look dark or even black. That throws people off at first, but it isn’t anything to stress about. Upset stomach, diarrhea, or mild constipation sometimes tag along. If stomach issues stick around for days, the prescribing doctor should hear about it. Nausea that completely ruins meals, vomiting, or sharp abdominal pain signals you need more help.
It pays to keep an eye on iron levels, too. Ferric citrate contains iron, and on rare occasions people absorb too much, especially if other supplements get mixed in. Regular blood work helps doctors track any creeping changes so adjustments come before anything serious kicks in.
Building Habits and Talking With Your Healthcare Team
New medication can bring a thousand questions, especially for someone juggling a handful of pills every morning. Pharmacists answer tons of questions about timing and side effects. Nurses often give advice about tricks that really work–pairing doses with brushing teeth or keeping the pills on the kitchen table where nobody forgets.
If a dose gets skipped, don’t double up. Just take the next scheduled pill with the next meal. Skipping too many doses, on the other hand, turns into a real problem over time.
Every person’s experience with ferric citrate dihydrate looks a little different. The best advice: keep close to your doctor and pharmacists. Be honest about how you feel and what’s actually happening at home. Trouble with pills isn’t just about willpower; finding the right way to fit them into your real life makes all the difference in staying healthy.
What are the possible side effects of Ferric Citrate Dihydrate?
Listening to Patients Facing Iron Overload or Kidney Disease
Many doctors use ferric citrate dihydrate to manage high phosphate levels in people with chronic kidney disease. Some rely on it as an iron supplement, too. But medicine doesn't always work smoothly for everyone, and every treatment brings its own set of tradeoffs. Through years of talking to folks in dialysis clinics and seeing how daily pill routines land differently for each person, I’ve seen that understanding the day-to-day realities of a drug matters as much as the numbers on a lab result.
Gut Reactions Hit First
Nausea and diarrhea pop up often after the first few doses. It’s less of a rare reaction and more of a common complaint. Some people have enough discomfort that meal planning and social plans take a hit. One friend described having to plan bathroom access every morning. Constipation can show up, too, even though it seems odd when diarrhea is also on the list. It’s a reminder that gut reactions to new substances swing both ways for different bodies. Over time, some folks adjust, but others just keep struggling.
What Happens to Stool?
Many get a surprise when their stool starts to look dark or almost black. This isn’t necessarily dangerous, but without a heads-up it can send someone straight to Google—or worse, assuming the worst. Talking about bowel habits isn’t exactly dinner conversation, yet keeping tabs on color and texture makes a difference in tracking your health.
Impact on Blood Levels
As an iron-rich compound, ferric citrate dihydrate shifts iron stores in the body. That sounds good if you're iron-deficient. But some patients end up with too much iron, and that’s a real problem. Too much iron damages organs over time, especially in folks already battling long-term kidney disease. Blood tests help watch for this, but busy clinics sometimes miss the warning signs until symptoms—like painful joints or skin changes—start to show.
Phosphorus and Calcium Balance
Ferric citrate dihydrate works by soaking up extra phosphorus in the gut, which helps people on dialysis avoid bone and heart problems. But it can tip the body into low phosphorus, too. Muscle weakness, confusion, and pain can sneak up when levels drop. Low calcium can tag along, worsening cramps and even causing heart rhythm problems. Folks with underlying bone disease face a tougher time here.
Allergic Reactions Are Rare, But Worth Knowing
A small number of patients experience hives, swelling, or trouble breathing after starting ferric citrate dihydrate. Even though these are rare, they must be treated seriously. Doctors need to educate patients about the early warning signs and what to do if they show up, instead of relying on medication inserts that get tossed aside.
What Helps Keep People Safe?
Ongoing blood work and open conversations with a care team do the most to spot trouble before it grows. People need space to share side effects that feel embarrassing or minor—like gut changes—and know that they can tweak doses or switch medications. Supporting dietary adjustments helps, too. Lowering processed phosphorus in the daily diet can reduce the need for high doses of binders.
Navigating medication isn’t only about what’s on the label. It’s about knowing how real people live with side effects, knowing what to look for, and not shying away from honest conversation. Better patient education and more time with healthcare professionals can help avoid problems from going unnoticed.
Can I take Ferric Citrate Dihydrate with other medications?
Understanding Ferric Citrate Dihydrate’s Role
Ferric citrate dihydrate often finds its place in treatment plans for people managing chronic kidney disease, especially when keeping phosphate levels in check proves tricky. Doctors also look at it for patients with certain types of anemia because it can help bump up low iron stores. With more folks living longer and collecting more prescriptions as they go, questions always pop up about mixing drugs safely. That’s not just a minor concern. Add Ferric citrate to a packed pillbox, and the story gets complicated.
Mixing Ferric Citrate Dihydrate Into Daily Routines
Juggling pills every day comes with its own set of surprises. Blood pressure meds, diabetes pills, phosphate binders, or vitamins often crowd the medicine cabinet if kidneys are acting up. Ferric citrate does more than add iron — it binds phosphate in the gut, keeping dangerous levels from building up. But this same ability also lets it trap other medicines, stopping them from working as well as they should.
A lot of people, including some of my own pharmacy clients, have mixed iron products with common antibiotics or thyroid meds and noticed the later pills just didn’t seem to work right. Iron catches drugs like doxycycline and levothyroxine, turning them into leftovers for the toilet rather than letting them get to the bloodstream. The science backs this up: a study in Nephrology Dialysis Transplantation showed levels of certain drugs like doxycycline and ciprofloxacin drop if someone takes them too close to Ferric citrate.
Why This Matters
Missing out on antibiotics or thyroid pills doesn’t sound good. Skipping a dose or getting partial medicine lessens the chance of staying healthy. If phosphate stubbornly sticks around because of missed binders, that can cause bone and heart trouble down the line. Doctors know about these problems, but the rush of everyday life leads plenty of folks to accidentally mix up their pills anyway.
In my own circle, I’ve seen relatives struggle with complex schedules. Tracking which pill at what hour sometimes feels like a cruel math problem with bad penalties for mistakes. It gets harder for older adults, especially with memory fog in the picture.
Finding a Safer Routine
Spacing out medicines can solve most of the trouble. Experts usually tell people to take antibiotics or thyroid pills at least two hours before or after Ferric citrate. This gap boosts the chances each drug will make it to the bloodstream without hitchhiking on the iron. A pharmacist can help set a schedule that won’t turn the day into one giant pill alarm.
Keeping a list of all current medicines and sharing it with each provider helps everyone stay on the same page. Digital pill reminders or even a sticky note by the coffee pot go a long way too. Family support matters, so if someone else checks in during the day, mistakes drop.
What To Ask At The Pharmacy
Don’t hesitate to speak up about everything in your cupboard. Some over-the-counter antacids, calcium pills, and other binders compete with Ferric citrate or get tangled up in similar ways. If in doubt, pharmacists love fixing medicine puzzles—they’ve seen just about every mix-up before.
Laying out your medication schedule honestly beats toughing it out in silence. Safety rests on teamwork: you, your providers, and anyone who helps with your care. Catching drug conflicts early keeps your health plan on track instead of derailed.
Is Ferric Citrate Dihydrate safe during pregnancy or breastfeeding?
Why People Ask About Ferric Citrate Dihydrate During Pregnancy
Doctors prescribe ferric citrate dihydrate for people with chronic kidney disease who need to keep phosphate levels in check. Many folks also rely on it for treating iron deficiency, which is common during pregnancy. Iron keeps oxygen flowing to both mom and baby, and low iron leaves you feeling wiped out. So, once a healthcare provider suggests ferric citrate, one big question pops up: “Is it safe to take while pregnant or breastfeeding?”
What the Science Says About Its Safety
Researchers have looked into ferric citrate mostly for kidney patients, but data in pregnant women trails behind. The FDA classifies medications based on risk in pregnancy. Ferric citrate hasn’t landed in a clear safety group, mostly due to a lack of robust, well-controlled studies in expectant moms. The few studies in animals at high doses kicked up some red flags (such as low birth weights or developmental quirks), but animal results don’t always tell us what happens in people.
On the other hand, iron itself is more than just safe—it’s essential. Expecting mothers need about double the iron compared to other adults, so supplements often come into play. The difference is that most prenatal vitamins use other forms of iron, like ferrous sulfate or ferrous gluconate, because they have more data supporting safety during pregnancy. With ferric citrate, less data leaves more unanswered questions. That gap worries both doctors and expectant mothers alike.
Breastfeeding and Ferric Citrate
So far, the research wheel hasn’t spun much faster for breastfeeding. We know most minerals a mom takes can pass into breast milk, but scientists haven’t nailed down how much ferric citrate ends up there or whether it affects infants. The American Academy of Pediatrics doesn’t single out ferric citrate as risky, but no active endorsement exists either. Once again, reliable information is thin.
My Take: Questions I’ve Heard From Parents
People care about every substance brought into their body when growing or feeding a child. In clinics, I’ve heard worried parents ask, “Will this hurt my baby?” or “What happens if I skip my iron pills?” It’s a natural concern—no one wants to take risks. Most obstetricians will lean toward safer, better-studied options for iron supplementation, given the lack of clarity with ferric citrate. If someone has a tough time with other forms of iron, and ferric citrate seems to work, only a healthcare provider can weigh the risks versus benefits.
How To Make Smarter Choices
A big part of safety during pregnancy and breastfeeding rests with open, honest conversations with your healthcare team. If a doctor suggests ferric citrate dihydrate, ask for the reason and bring up any research you’ve found. Don’t stop or switch iron supplements on your own. Let nutrition assessments and blood tests help guide treatment. Whenever options exist, choosing something with a longer safety record in pregnancy and lactation feels comforting.
Real progress on these questions comes only through future research. Until then, listen to your doctor, keep communication lines open, and stick to supplements with better-established track records, unless ferric citrate turns out to be the safest or only choice for you. Your baby’s health and your own well-being both deserve this careful attention.
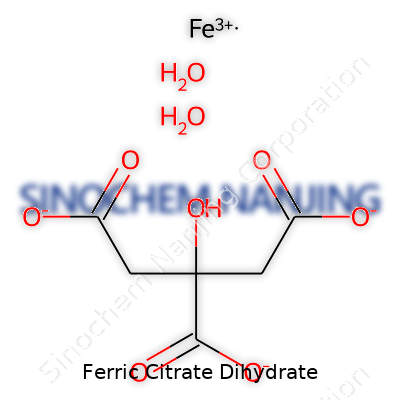
| Names | |
| Preferred IUPAC name | Iron(3+) 2-hydroxypropane-1,2,3-tricarboxylate dihydrate |
| Other names |
Ferric Ammonium Citrate Iron(III) Citrate Dihydrate Citrate of Iron Ferric Citrate, hydrate |
| Pronunciation | /ˈfɛrɪk ˈsɪtreɪt daɪˈhaɪdreɪt/ |
| Identifiers | |
| CAS Number | [35947-98-7] |
| Beilstein Reference | 3318736 |
| ChEBI | CHEBI:133751 |
| ChEMBL | CHEMBL2108700 |
| ChemSpider | 21476655 |
| DrugBank | DB12967 |
| ECHA InfoCard | 03cb8b6e-efa6-4e65-8ad5-15d1def3fdc7 |
| EC Number | 40-070-2 |
| Gmelin Reference | 84259 |
| KEGG | C18736 |
| MeSH | D006976 |
| PubChem CID | 71586965 |
| RTECS number | GE7520000 |
| UNII | Y2PVT7S4MR |
| UN number | UN3077 |
| CompTox Dashboard (EPA) | DTXSID2051523 |
| Properties | |
| Chemical formula | C6H5FeO7·2H2O |
| Molar mass | 244.94 g/mol |
| Appearance | A reddish brown to brownish yellow powder |
| Odor | Odorless |
| Density | 1.8 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | -2.1 |
| Acidity (pKa) | 3.1 |
| Basicity (pKb) | 10.7 |
| Magnetic susceptibility (χ) | '~470 × 10⁻⁶ cm³/mol' |
| Dipole moment | 12.77 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 501.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -2346.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AE10 |
| Hazards | |
| Main hazards | May cause respiratory irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | Hazard statements: May cause respiratory irritation. |
| Precautionary statements | Precautionary statements: P261, P280, P301+P312, P304+P340, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Lethal dose or concentration | LD50 Oral Rat: > 2,000 mg/kg |
| LD50 (median dose) | 2000 mg/kg (rat, oral) |
| PEL (Permissible) | Not established |
| REL (Recommended) | 6.0 mg/kg bw |
| Related compounds | |
| Related compounds |
Ferric citrate Ferric ammonium citrate Ferrous citrate Ferric chloride Ferrous sulfate Citrate (citric acid) Iron(III) sulfate |

