Ethylhexanal: Progress, Properties, and Pathways
Historical Development
Ethylhexanal didn’t just appear on chemistry’s stage one day. It took decades of industry-driven research for companies and scientists to figure out the optimal ways to create and use this chemical. Manufacturers started working with aliphatic aldehydes like ethylhexanal during the postwar boom, chasing better plasticizers and chemical intermediates to ramp up materials for construction and consumer products. As chemical engineering grew more precise, so did methods for producing aldehydes. By the 1960s, companies deployed oxo synthesis—hydroformylation of alkenes—to churn out branched-chain compounds like ethylhexanal in reliable quantity. The process continued evolving with environmental regulations and catalyst improvements, minimizing byproducts and waste streams. The Western chemical industry still relies on this backbone to provide the 2-ethylhexanol used in PVC and plasticizer production, and that feedstock runs through ethylhexanal. Every advance in catalysis or plant design over the years links to real-world demands for safer or more efficient output, rather than chemistry for chemistry’s sake.
Product Overview
Ethylhexanal stands as a colorless liquid, often with a mild fruit-like odor. Most people outside industrial chemistry wouldn’t run into it, unless they’ve set foot in a plasticizer plant or a fragrance laboratory. This molecule—structurally an aldehyde with a branched carbon chain—primarily supports the production of 2-ethylhexanol, a valuable plasticizer precursor. Synthetic fragrances and flavorings also exploit its aroma, and its versatility has kept it on the books of large producers like BASF or Eastman. Companies don’t sell it directly to consumers; rather, it travels behind the scenes in supply chains. Its main value lies in how easily it plays a middleman—reacting further, changing into something else, or adding distinctive notes to formulations where a regular C8 aldehyde falls short.
Physical & Chemical Properties
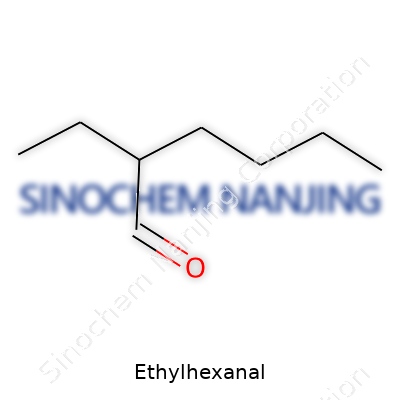
Look at ethylhexanal’s molecular makeup and some things jump out. The chemical formula comes in as C8H16O, and its molecular weight rests around 128.21 g/mol. It boils at just under 159°C with a flash point around 48°C, putting it in the category of combustible, not highly flammable. Density sits at about 0.82 g/cm³ at 20°C. This aldehyde dissolves easily in most organic solvents, yet stays stubbornly out of water, like most hydrocarbon-rich compounds. Its branched structure makes it less reactive in some pathways compared to straight-chain aldehydes. The functional group at the end of the chain—CHO—makes it a target for all kinds of addition reactions or reduction steps, setting it up as a workhorse in organic synthesis.
Technical Specifications & Labeling
Companies set out clear limits for ethylhexanal quality. Purity counts, usually listed at greater than 98%, with less than 1% water, and minimal amounts of related aldehydes or alkanes. Labels display the CAS number 123-05-7, United Nations number UN 1989, and proper shipping names, since safety regulations demand it for any significant transport. Regulatory papers call out risks—skin and eye irritation, vapor inhalation limits, and the need for secondary containment in case of leaks. The Safety Data Sheet lays out PPE needs, boiling and flash points, hazard numbers, and recommendations for ventilation, handling, and first aid. I’ve seen workplace audits take these details seriously, with warehouses double-checking secondary labels and checking for oxidizer incompatibilities before accepting shipments.
Preparation Method
Most commercial ethylhexanal comes from oxo synthesis, a process that involves hydroformylation of 1-heptene using a cobalt or rhodium catalyst, mixed with hydrogen and carbon monoxide at elevated temperatures and pressures. Over time, the industry shifted away from early cobalt-based technology to newer rhodium-phosphine systems, which bring both higher selectivity and energy savings. This step yields both linear and branched aldehydes, so downstream separation steps play a key role—distillation, washing, and fractionation. Some academic labs have dabbled in more exotic methods, like catalytic dehydrogenation of corresponding alcohols or selective oxidation of alkanes, but on a commercial level, oxo synthesis dominates. The plant-scale process reflects the shift toward green chemistry: Lower catalyst loadings, less effluent, and minimized worker exposure.
Chemical Reactions & Modifications
Ethylhexanal offers plenty of opportunity for further chemical transformation. One major outcome comes from hydrogenating it—adding hydrogen across the carbonyl group—to get 2-ethylhexanol. That alcohol then goes into phthalate plasticizers. In another route, condensation with active methylene compounds or amines yields intermediates for agrochemicals or specialty organics. The aldehyde group provides a point for oxidation—leading to carboxylic acids that find specialty uses in surfactants or lubricants. Companies sometimes play with these reactions at bench scale to tweak aroma molecules for perfumery, exploiting the branched chain to provide unique olfactory effects. Researchers keep exploring mild oxidation or greener reduction technologies, lower temperature processing, and biocatalytic pathways, but conventional hydrogenation remains firmly entrenched.
Synonyms & Product Names
Ethylhexanal comes packed with alternative names, a result of different naming systems in chemistry. Some documents call it 2-ethylhexanal, giving the branch position. I’ve seen n-heptylcarboxaldehyde, 2-ethylcapronaldehyde, and even abbreviated versions like 2-EHA. Catalogs sometimes group it with medium-chain aldehydes for fragrance or flavor use, or as a parent feedstock in list entries for 2-ethylhexanol. Shipment paperwork, especially for international trade, picks up all the variations—forcing companies to check reference numbers closely before accepting deliveries.
Safety & Operational Standards
Safety officers at production plants look at ethylhexanal as a moderate irritant with flammability risks, not an acutely toxic compound. Proper protocols flag it as harmful if inhaled, and as a cause of skin and eye irritation. I’ve worked with safety managers who won’t allow workers to handle it without gloves, goggles, and proper respiratory protection. Standard ventilation, explosion-proof storage, spill kits, and closed-systems for transfer all show up on the job. OSHA and EU workplace exposure guidelines specify threshold limit values; regular air monitoring in facilities checks for accidental release or vapor buildup. Fire departments expect companies storing aldehydes to keep extinguishers and neutralizing agents on hand, just in case of a thermal event or uncontrolled reaction. Everyone involved has seen the consequences of lax safety, so facilities don’t take shortcuts with checking connections or maintaining pressure relief systems.
Application Area
The biggest consumer of ethylhexanal sits in the plasticizer supply chain. Every time a flexible PVC wire or vinyl flooring goes to market, there’s a good chance some ethylhexanal entered the process during manufacture of 2-ethylhexanol. Some fragrance and flavor companies use it very sparingly—usually as a trace component to add green, aldehydic notes to synthetic perfumes or certain fruit flavor profiles in commercial foods. A few specialty chemical sectors use it to introduce branching or length into tail groups for surfactants or as building blocks for agrochemical products. Its reach rarely spreads into consumer hands, but it supports industries that produce safety flooring, synthetic leathers, inks, adhesives, and sometimes even custom pharmaceuticals.
Research & Development
Research teams keep hunting for better catalysts and more energy-efficient synthesis routes. Academic literature from the last decade focuses on minimizing waste in oxo processes, seeking more selective catalysts, or using renewable starting materials. Companies back pilot projects to employ biocatalysts or metalloenzymes, especially as pressure mounts to source greener chemicals. Analytical labs develop improved methods for detection—gas chromatography-mass spectrometry figures heavily. Environmental scientists work on cleaner destruction or recovery technologies, since aldehyde emissions attract regulatory scrutiny. I’ve met chemists who developed scale-down versions of the hydroformylation process for educational use, making it easier for students to connect industrial output to foundational organic chemistry principles. All this ongoing work ultimately aims for safer workplaces, cheaper production, less environmental impact, and smarter downstream chemistry.
Toxicity Research
Toxicology won’t let ethylhexanal off the hook entirely. Animal studies flag moderate acute toxicity and mild irritation at low doses. Regulatory bodies don’t classify it as a carcinogen or reproductive toxin, but prolonged or repeated exposure causes concern—dermal contact leads to irritation, inhalation sometimes provokes coughing, headaches, or drowsiness. Testing labs monitor for metabolites in blood and urine after accidental exposures. Environmental fate studies show moderate persistence, with breakdown products that need tracking in wastewater and air emissions. Occupational safety agencies keep strict workplace exposure limits, reinforcing good handling practice, and environmental impact reviews before approval for new or expanded facility permits.
Future Prospects
Ethylhexanal has plenty of life left in major chemical sectors, but its future links to the push for safer production and sustainable sourcing. Research keeps targeting lower-impact synthesis—biocatalysis, renewable feedstocks, and high-yield, low-emission processes. Countries tighten environmental grip around chemical manufacturing, pushing companies to account for even minor byproducts and trace emissions. Small-scale specialty producers may pivot to greener chemistry inspired by academic progress, aiming for certifications that appeal to eco-conscious buyers. The need for reliable plasticizer intermediates won’t disappear anytime soon, and as long as flexible PVC features in construction or car manufacturing, ethylhexanal stays locked into global supply chains. Companies that have invested in process improvement and hazard reduction will find themselves better poised for coming shifts in regulation and consumer expectations around chemical stewardship.
What is Ethylhexanal used for?
The Role of Ethylhexanal in Manufacturing
Ethylhexanal stands as one of those behind-the-scenes chemicals that hardly shows up in daily conversation, but life would look different without its presence. I first ran across ethylhexanal a decade ago at a midwestern chemical plant, where tanks of the stuff arrived on railcars. The safety briefings drilled its strong odor and volatility into my mind, but most attention focused on what workers did with it. They used ethylhexanal to produce special kinds of plasticizers—the stuff that makes plastics bend and flex instead of snapping like glass. These plasticizers show up in products from floor tiles to garden hoses. Without flexible plastics, industries would spend more money on materials that don't hold up under bending or twisting. Flexible plastic products tend to last longer and perform better in real-world settings, keeping replacement costs low for businesses and consumers alike.
How Ethylhexanal Shapes the Fragrance World
Beyond plasticizers, ethylhexanal plays a lesser-known but important role in fragrance creation. It's a building block for specialty aroma chemicals. In the hands of a skilled perfumer, ethylhexanal becomes part of a formula that helps scents move from the bottle into the air—and linger long enough for people to notice. While a single molecule may not carry a distinct scent, combining these allows for everything from cheap air fresheners to luxury perfumes. People tend to dismiss the importance of chemistry in scent, but without these raw materials, the magic found in a fresh-smelling laundry room or a well-loved cologne wouldn't exist. In my experience, this tiny detail makes an outsized difference in how a product is perceived.
Challenges and Health Concerns
There’s always a flipside with industrial chemicals, and ethylhexanal is no exception. I’ve spoken to technicians and safety officers who handle this compound daily. Most discussions focus on its potential for skin and eye irritation and the key requirement for strong ventilation. Accidental releases create headaches and discomfort, and high doses can be harmful if inhaled or touched. Keeping track of these risks takes solid training, good protective equipment, and personal discipline. Companies using ethylhexanal can't afford to cut corners. Regular monitoring of workplace air, personal protective gear, and clear emergency procedures—all these keep workers out of harm’s way. The chemical industry is already subject to robust rules in Europe and North America, but not all corners of the globe hold themselves to these standards. There are still stories of poorly ventilated workshops or missing safety training, and that always ends up costing someone their health.
Making Production Safer
It’s possible to make things safer for workers. Transparent data from regulatory bodies, up-to-date safety protocols, and investment in better containment save lives. Every time someone builds a new facility or upgrades an old one, there’s a chance to rethink how chemicals are handled. Automated systems now allow many operations to take place behind sealed doors, cutting back on worker exposure. On a broader scale, research keeps aiming at ways to switch to greener alternatives. While ethylhexanal has proven effective and affordable, the push for safer substitutes continues in the chemicals community. Progress doesn’t always happen quickly, but steady steps can make a real difference.
Looking Ahead
Ethylhexanal isn’t going anywhere soon. Its uses in everything from bendable plastics to perfume oils mean it will stay relevant. People in industry have figured out how to work safely with it most of the time. My experience shows it takes a quietly stubborn dedication to safety and a willingness to adapt with new science. That’s how the impacts of a single, strong-smelling chemical from a railcar stretch through manufacturing, fragrance, and even home improvement projects—affecting lives in small but meaningful ways, every single day.
What is the chemical formula of Ethylhexanal?
Looking at Ethylhexanal from a Practical Perspective
Ethylhexanal, at its core, carries the formula C8H16O. To anyone in chemistry, these letters and numbers come loaded with meaning. It’s not only about learning another formula for a quiz; it’s about unlocking a molecule’s nature. The name might not come up often at the dinner table, but it shows up in labs, factories, and in plenty of behind-the-scenes processes in industry.
Getting to Know the Structure
Chemical formulas tell us more than count; they hint at how atoms connect and interact. For ethylhexanal, C8H16O says you get eight carbons, sixteen hydrogens, and an oxygen. The molecule represents an aldehyde, and that means you’ve got a carbonyl group at the end of a carbon chain. The name gives a clue—ethylhexanal breaks down to an ethyl group attached to hexanal, so there’s a six-carbon backbone with an ethyl side chain. This structure carries real consequences for reactivity in synthesis and flavor chemistry.
Why Ethylhexanal Matters in Everyday Life
I’ve spent hours poring over chemical catalogs and working up reaction plans that include compounds like ethylhexanal. Sometimes it’s about making fragrances. Sometimes it’s part of building plastics or coatings that handle exposure to sun or rain. Sure, the molecule seems simple, but what people forget is that every product in the supermarket stems from hundreds of small pieces like this. No one wakes up thinking about hexanal derivatives, but these compounds make paints last longer, add zip to scents, and help experiments go off without a hitch.
The Bigger Picture: Health, Safety, and Environmental Considerations
Formulas are a starting point, but safety data sheets sit beside every bottle in the lab for a reason. Aldehydes can irritate the eyes, skin, or lungs. In my experience, gloves and lab ventilation are non-negotiable. Reading up on handling procedures and understanding the structure of ethylhexanal has real-world consequences. It might not grab headlines, but public health depends on how carefully people treat these compounds both in the workplace and in wider manufacturing settings.
Waste disposal raises another concern. Aldehydes like ethylhexanal break down in the environment, yet careless dumping isn’t acceptable. Communities near production sites deserve factories that prevent leaks and limit pollution. As someone who’s had to explain chemical safety rules to new team members, I know misunderstandings can lead to accidents or fines. A little attention to a formula and its implications saves a lot of trouble down the line.
How to Foster Better Understanding and Safer Practice
Ethylhexanal reminds me that chemistry isn’t just a branch on the science tree—it’s involved in everything from product design to regulatory frameworks. Teaching about these compounds in schools and at work doesn’t just prepare people to pass a test; it fosters responsibility. Manufacturers need to follow regulations, but they also need a workforce that understands why these rules matter.
Research and communication make all the difference. Teams that share updates—whether it’s a new risk assessment or a change in storage—are more likely to avoid incidents. People can’t predict every possible outcome. They can build habits that protect others and the environment, one bottle of C8H16O at a time.
Is Ethylhexanal hazardous or toxic?
Getting to Know Ethylhexanal
Ethylhexanal doesn’t appear in the news every day, but for anyone who works with chemicals, it’s not an unknown name. This compound pops up thanks to its role as a building block for plasticizers, flavors, and scents. Labs use it to help create everyday products found on shelves, ranging from cosmetics to cleaning supplies. With how often synthetic compounds show up in daily life, it makes sense to ask: Could ethylhexanal bring more risk than reward?
The Toxicity Question
Some folks worry about chemicals right away just from the name. In my experience, that’s usually a signal to dig deeper rather than panic. Ethylhexanal, based on published studies and chemical safety sheets, lands somewhere between mild and moderate when it comes to dangers. Research shows it can irritate eyes and skin. Breathe in its vapor, and your throat or nose might get sore. In high doses, animal tests show damage to the liver and kidneys, but day-to-day handling doesn’t reach those extremes. The fact is, the dose matters — the old rallying cry of toxicologists everywhere. A drop on skin won’t cause the same issues as years of heavy exposure in a factory. That’s not unique to ethylhexanal, either.
Comparing Hazards in Context
I’ve handled many solvents and flavorings over the years, and the biggest problems rarely come from brief exposure. People forget that even water can be deadly if misused. Ethylhexanal demands respect, especially in concentrated form, but it isn’t out of line with many other household or lab compounds. The European Chemicals Agency lists it as an irritant, so workers wear gloves and goggles — the same deal as with stronger cleaning products under the sink.
Protecting Health: Practical Solutions
Science says safe habits do most of the heavy lifting. Companies using ethylhexanal in manufacturing already install proper ventilation. Employees wear gloves, safety glasses, and sometimes masks depending on the job. Regulatory bodies like OSHA and the EU's REACH program keep a close watch, setting limits for workplace air and skin exposure. Training remains just as important as rules. I’ve seen coworkers skip basic measures, only to regret it once irritation hit. Simple, consistent habits — washing hands, storing chemicals correctly, using fume hoods — make a bigger difference than any fancy piece of equipment.
Public Concerns and the Road Ahead
Outside the lab, the main concerns rise when people picture these chemicals leaching into products or the environment. Most consumer use cases include ethylhexanal at trace levels, well within the safety margins regulatory agencies set. That doesn't mean oversight can slip. Companies owe it to the public to run regular safety checks on ingredients, label products clearly, and keep looking for safer alternatives if possible. The fragrance and plastics industries keep investing in green chemistry. In my view, that’s the long game — making sure innovation doesn’t trade off human or environmental health just to shave a few dollars off production costs.
Final Thoughts on Safety
Ethylhexanal isn’t a villain in the chemical world, though it deserves caution and respect. Most evidence points to manageable risk, as long as folks follow safety rules and stay informed. Trust in science, clear communication, and common sense are the best tools anyone can use, whether mixing chemicals at work or choosing products for home.
How should Ethylhexanal be stored?
The Real Risks Behind Ethylhexanal
Ethylhexanal doesn’t get much attention outside of the chemical industry, but those who’ve worked around it know it deserves respect. As someone who’s seen the effects of mishandled chemicals, I can say slick guidelines don’t always hit home unless they’re grounded in day-to-day experience. This colorless liquid, with its strong, sometimes fruity smell, crops up in labs, factories, and even niche fragrance production. Behind that aroma lies a substance known for volatility and some serious health consequences if left unchecked.
Heat, Light, and Air: The Three Enemies
Anyone who’s ever stocked a storage room learns quickly that temperature control isn’t just a nice-to-have. Ethylhexanal, like so many flammable liquids, demands a cool spot. I once visited a facility where the only storage “guideline” was to shove everything in an old broom closet. That led to broken seals, chemical fumes, and one panicked maintenance worker. Keep this substance below 25°C (77°F) and well away from radiators, sunlight, and machinery that throws off sparks. High temps lead to evaporation and pressure build-up, which turns a bottle of ethylhexanal into something you really don’t want to open in a hurry.
A Good Seal Goes a Long Way
After years working in shipping and receiving, I learned to trust a tight, screw-top container far more than any plastic wrap or makeshift lid. Ethylhexanal reacts with air, especially when left unsealed or in half-empty bottles. That reaction not only dulls the purity but also risks the creation of irritating byproducts. Metal drums with airtight seals send the right message: keep air out, and you keep people safe. Glass containers with vapor-tight caps make sense for bench-scale work, but stick with verified chemical storage bins for anything larger.
No Room for Guesswork: Keeping People at the Center
Good practice in chemical handling comes down to respect for people and process, not fear. I’ve seen how one leaky bottle on a high shelf can mean headaches or worse for teams working nearby. Store ethylhexanal at eye-level, clearly labeled, and on a non-reactive surface. No stacking heavy items on top, no sharing space with acids or oxidizers. Cross-contamination in a cramped storeroom sets you up for real trouble. Signs help, but supervisor training and routine checks do more for day-to-day safety than a hundred warning stickers.
The Hidden Value in Routine Checks
Routine isn’t glamorous, yet it’s where accidents either start or get stopped in their tracks. Workers who handle ethylhexanal should have gloves made of nitrile or polyethylene, protective goggles, and a spill kit handy. Tidy storage helps locators spot leaks or bulges in time—before vapor escapes and the place smells like rotten fruit. We once caught a pinhole leak during a slow Friday sweep thanks to a change in odor, which in a locked storeroom marks the difference between a clean up and a hospital visit.
Learning from Every Incident
Years ago, our team dealt with a spill because someone stored a reactive chemical next to ethylhexanal by mistake. That day hammered home how much hinges on one step overlooked. Clear records, honest training, and the willingness to pause and check labels mean fewer risks for everyone. The way we store a chemical like ethylhexanal says a lot about how much we value the people doing the work. Before it ever gets to an emergency, a good storage plan keeps the peace—and lets everyone head home healthy at the end of the shift.
What are the common applications of Ethylhexanal in industry?
Taking a Closer Look at a Little-Known Chemical
Most folks never hear about ethylhexanal, but this colorless liquid has found a place in several factories and labs. I’ve tracked its route through different industries, and its uses reveal a lot about how much chemistry shapes everyday products.
Fragrance and Flavor
Perfume companies rely on ethylhexanal for its fresh, slightly citrus scent. As someone who worked a summer loading trucks at a flavor warehouse, I’ve seen small barrels of this compound head out to customers in the fragrance world. Perfumers turn to it when they want to craft a subtle blend for colognes, air fresheners, candles, or even shampoos. Makers of artificial flavors look for compounds that add depth and realism to food, and it takes just a tiny bit of ethylhexanal to boost citrus and fruit notes in processed foods and drinks.
Helping Plastics Get Soft
Plastic isn’t worth much if it snaps or bends the wrong way. Manufacturers use ethylhexanal as a building block to make certain plasticizers, especially in polyvinyl chloride (PVC) production. Plasticizers keep cables flexible, floors cushy, and car interiors able to tolerate real heat and cold. People sometimes forget how many goods depend on flexible plastics: rain boots, tubing, wire coatings, credit cards—the list runs long. Keeping these plastics soft without breaking down is part of the reason chemical firms invest in molecules like ethylhexanal.
Fine Chemical Synthesis
Most chemistry happens in chains—turn one thing into another, then build something completely new. Ethylhexanal can act as a stepping stone in this chain-reaction world. Labs use it to make other specialty chemicals, from rubber accelerators to herbicides and antioxidants. That breadth reflects the value of having a reliable building block with a strong chemical structure. Synthetic routes often choose ethylhexanal so they can branch off into diverse markets, be it pharmaceuticals or agricultural sprays.
Industrial Solvents and Coatings
Some jobs in construction and automotive work require solvents that evaporate at just the right pace. Paints and coatings sometimes include ethylhexanal or its derivatives—these compounds help balance drying time and surface finish. In my early days painting houses, I noticed certain commercial paints left a smoother, more robust film thanks to these specialized additives. Keeping a paint smooth in both hot and cool weather, or helping coatings stick to metal in automotive plants, depends on careful chemical choices.
Looking at Safety and the Future
Handling chemicals like ethylhexanal calls for respect and solid safety routines. The industry saw tighter standards in recent decades, especially because this compound can irritate skin or the lungs in high concentrations. Growing up around a chemical plant, I remember the strict training and safety drills that kept people informed and prepared. Companies track exposure limits closely, push for better air filtration, and train workers about personal protective gear. They’re also searching for greener substitutes or ways to make manufacturing less wasteful, especially as pressure mounts to shrink the sector’s environmental footprint.
Why It Matters
Ethylhexanal might not be famous, but it's a part of life: in the perfume you wear, the power cable you plug in, and the processed snack you eat during a break. Industry keeps finding new ways to use basic building blocks like this one, quietly bridging the gap between raw chemistry and daily convenience. Tracking these uses helps shine a light on the unseen work that shapes the world around us.
| Names | |
| Preferred IUPAC name | 2-Ethylhexanal |
| Other names |
2-Ethylhexanal α-Ethylhexanal Ethyl caproaldehyde |
| Pronunciation | /ˌɛθ.ɪlˈhɛk.sə.næl/ |
| Identifiers | |
| CAS Number | 123-05-7 |
| Beilstein Reference | 1721576 |
| ChEBI | CHEBI:79013 |
| ChEMBL | CHEMBL4210720 |
| ChemSpider | 10084 |
| DrugBank | DB14096 |
| ECHA InfoCard | 14d55748-1bf2-4b1f-8b91-19ecf8f9b194 |
| EC Number | 203-979-7 |
| Gmelin Reference | GN196590 |
| KEGG | C20829 |
| MeSH | D000086246 |
| PubChem CID | 12508 |
| RTECS number | KI5075000 |
| UNII | 7T16V9476N |
| UN number | UN 1992 |
| CompTox Dashboard (EPA) | DTXSID7021244 |
| Properties | |
| Chemical formula | C8H16O |
| Molar mass | 142.24 g/mol |
| Appearance | Colorless liquid |
| Odor | strong, fatty, green, citrus |
| Density | 0.827 g/mL at 25 °C (lit.) |
| Solubility in water | slightly soluble |
| log P | 2.8 |
| Vapor pressure | 0.32 mmHg (25°C) |
| Acidity (pKa) | 16.6 |
| Basicity (pKb) | '11.72' |
| Magnetic susceptibility (χ) | -7.41×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4190 |
| Viscosity | 1.33 mPa·s (20 °C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 383.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -274.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4065 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | ``` GHS07 ``` |
| Signal word | Warning |
| Hazard statements | H226, H315, H317, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P303+P361+P353, P305+P351+P338, P337+P313, P370+P378, P403+P235, P501 |
| Flash point | 61 °C |
| Autoignition temperature | 215 °C |
| Explosive limits | Explosive limits of Ethylhexanal: 1.0% - 5.9% |
| Lethal dose or concentration | LD50 (oral, rat): 2,400 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Ethylhexanal: "1,900 mg/kg (rat, oral) |
| NIOSH | KI1950000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 50 mg/m³ |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
2-Ethylhexanol 2-Ethylhexanoic acid n-Hexanal Octanal Heptanal |

