Ethylenimine: A Closer Look at an Industrial Powerhouse
Historical Development
Ethylenimine's story reaches back to the early 20th century, in a time when chemists leaned hard into pushing the boundaries of cyclic molecules. Hermann Staudinger is often remembered for identifying this molecule’s unique three-membered ring structure in 1909. The post-war chemical industry discovered that ethylenimine, with its strained aziridine ring, could do some heavy lifting in polymer chemistry and agriculture. The jump from laboratory curiosity to an industrial commodity didn’t happen overnight, but driven by the wild growth in plastics and specialty chemicals from the 1940s onward, ethylenimine established itself. Big chemical outfits scaled up production as demand for polyethylenimines and specialty surfactants picked up, showing just how inventive people got when they needed new solutions for the booming synthetic age.
Product Overview
This colorless, flammable liquid has a sharp, ammonia-like odor that tells you right away it’s not an everyday chemical. Ethylenimine, also called aziridine, shows up as a core building block for manufacturing polymers, pharmaceuticals, and even paper chemicals. The base chemical looks simple, but packs a lot of energy in its three-membered ring, making it reactive in ways you don’t always see with other amines. Big plants generally manufacture it in bulk, shipping it in airtight vessels with all the regulatory paperwork in order. For anyone working on resin production or cross-linking agents, ethylenimine offers unique advantages, partly because it loves to react and add to other molecules down the line.
Physical & Chemical Properties
It vaporizes quickly with a boiling point near 56°C, so handling demands solid ventilation—nobody wants accidently to catch a whiff of the intense, irritating vapor. Water dissolves it at any ratio. Under lab light, it looks just like water, but unlike water, ethylenimine polymerizes spontaneously if you leave it exposed to acid, water, or air. Molecular formula C2H5N, molar mass at 43.07 g/mol, and a density lighter than water, at roughly 0.83 g/cm³. Its unique aziridine ring holds significant ring strain, blessing the molecule with reactivity that's been a boon for chemists but a caution sign for operators. The flammability can’t be downplayed—flash point measures in single digits (°C), setting expectations for robust fire-safety standards wherever tanks or drums are opened.
Technical Specifications & Labeling
Regulations demand strict labeling for ethylenimine. Tankers and drums bear transport labels to reflect its flammable and toxic properties: UN 1185, hazard class 3 (flammable liquids), and toxicity warnings under GHS. Certificates of analysis often include purity over 99%, trace metal content, water content, and color by APHA scale. Chemists appreciate specs like boiling point, miscibility, and flash point at a glance on any documentation. Most suppliers emphasize closed handling systems as well as complete training for their employees, since even a slight exposure may carry significant health consequences.
Preparation Method
The classic approach to make ethylenimine involves dehydrating aminoethanol (monoethanolamine) with alkaline catalysts like potassium hydroxide at high temperatures. Another established route uses ethylene dichloride reacting with ammonia in a pressurized reactor, stripping away hydrochloric acid at each step. Chemists monitor these exothermic reactions closely because uncontrolled temperature spikes can start runaway polymerization. Industrial plants today optimize these processes with continuous flow reactors and automated controls to keep yields high and limit the piles of hazardous waste that older setups used to generate.
Chemical Reactions & Modifications
Ethylenimine doesn’t hesitate to branch out chemically. That highly strained ring reacts with acids, bases, and various nucleophiles, priming it for polymerization or for building more sophisticated chemical architectures. In practice, ethylenimine gets used to make polyethylenimine, a branched polymer with all sorts of uses in water treatment and adhesives. It adds to carboxylic acids and epoxides, offering up cross-linking or chain extension. The molecule also gets alkylated, acylated, or sulfonated in industry workshops, producing derivatives used in everything from paper strength agents to oil field chemicals. Not many small molecules offer such a springboard for customizing material properties.
Synonyms & Product Names
Ethylenimine often appears in documents and product listings under several names, most commonly aziridine. I’ve seen it sold under trade names when branded for specialty resins, but chemical procurement usually settles on ethylenimine (CAS 151-56-4) or aziridine as the commercial tags. Other synonyms include 1-aziridine, aminoethylene, and ethylene imine. That tricky ring structure remains the giveaway no matter the name on the barrel.
Safety & Operational Standards
Dealing with ethylenimine isn’t for the inexperienced. It is both toxic and carcinogenic, with low permissible exposure limits (OSHA recommends keeping airborne concentrations below 5 ppm averaged over an eight-hour shift). The EPA and European REACH framework set stringent controls around workplace exposure, spill management, and transportation. Direct skin or eye contact burns badly, and inhaling vapor irritates or even damages the respiratory tract. Employees wear chemical-resistant gloves, goggles, and often full-face respirators, and work takes place in tightly controlled ventilated hoods. Emergency shutdown drills, spill containment kits, and plume exhaust monitoring form the backbone of plant safety routines. Medical surveillance follows employees exposed even to small amounts. Ethylenimine serves as a textbook example of why chemical plants invest heavily in hazard communications and rigorous safety audits.
Application Area
From my own time working with specialty polymers, I’ve seen ethylenimine play a quiet but indispensable backstage role. Manufacturers lean on polyethylenimines as cationic fixatives in papermaking, as adhesion promoters for ink, and as key ingredients in water treatment chemicals that scavenge heavy metals. Drug companies investigate derivatives as alkylating agents for chemotherapeutics, though most clinical use leans toward other aziridines due to the toxicity of ethylenimine itself. Textile finishing and oil recovery both rely on its cross-linking properties to boost durability and performance. For a molecule with a reputation for danger, ethylenimine finds its way into dozens of products you might not expect, supporting industries that focus on durability, chemical resistance, or specialty coatings.
Research & Development
Academic groups and private labs remain busy reimagining what the aziridine ring can do. Modern work explores using ethylenimine-based polymers to deliver gene therapies or to capture atmospheric carbon, driven by that backbone of high reactivity. Researchers try to craft safer derivatives or limit uncontrolled polymerization using precision catalysts. In R&D, one challenge comes from mediating reactivity to unlock new properties without introducing unacceptable toxicity. Some teams look deeply at controlling polymer architecture at the molecular level, aiming for next-gen adhesives and biocompatible coatings that can outperform older materials. With automation and finer process control, labs can now screen and optimize ethylenimine reactions without the same safety compromises that used to hold back innovation.
Toxicity Research
Toxicologists have flagged ethylenimine as a probable carcinogen—rodent studies saw tumor formation at relatively low doses. The primary worry comes from its ability to alkylate DNA and proteins, setting off a chain of mutagenic and cytotoxic events. Acute exposure leads to rapid onset symptoms: burning, nausea, headaches, and in severe exposures, pulmonary edema or neurological effects. Chronic, low-level exposure carries risks to liver, kidneys, and the immune system. Factory safety guidelines draw heavily from decades of studies at national labs, where even trace exposures earned strict hazard ratings. Most of the research aims at establishing safe exposure thresholds, improving monitoring technologies, and working out how genetics and dose-response relationships play into real-world occupational health. The goal for industry has shifted more toward substituting less hazardous materials wherever possible and investing in engineering controls that shut down exposure entirely.
Future Prospects
Industrial chemistry keeps seeking new uses for sharp-edged tools like ethylenimine, though its future looks tied to stricter safety and environmental rules. The push for green chemistry means researchers hunt for ways to capture the performance of ethylenimine-based products without the same human health risks. Some next-era materials, from precision drug carriers to advanced nanocomposites, draw on the unique reactivity of the aziridine ring. Bio-derived feedstocks, careful containment, and smart reactors may let industry keep the benefits while dialing down the risks. Regulatory pressure will probably reduce direct worker exposure year over year, prompting more investment in process automation and remote monitoring. If chemists can tame the hazards, the molecule’s combination of high reactivity and structural versatility could keep it in the toolkit for applications that demand more than what conventional amines can deliver.
What is ethylenimine used for?
What Drives Demand for Ethylenimine?
Ethylenimine, sometimes called aziridine, brings muscle to manufacturing. My first real encounter with it popped up years ago working with textile engineers, tracking how chemical treatments shape fabric performance. The fact that ethylenimine showed up in those conversations says a lot. This stuff isn’t flashy, but it's a behind-the-scenes driver for everything from paper to fuel additives.
You may not see its name on many labels, but chemists and engineers keep it close for a reason: in the right hands, ethylenimine helps them build bigger molecules with special properties. Its three-membered ring structure makes it highly reactive and ready to link up with acids, alcohols, and other targets. A big use comes in making polyethylenimine—a family of polymers holding together water treatment chemicals, adhesives, and paint thickeners. In papermaking, polyethylenimine helps boost paper’s “wet strength,” so your coffee filter or tea bag doesn't fall apart mid-brew.
Everyday Goods Rely on an Edge
Most people won’t ever handle pure ethylenimine. Just a whiff can burn the nose and eyes, and contact spells trouble. Still, its derivatives pop up all over. Beyond paper, companies lean on ethylenimine compounds in asphalt additives that keep city roads flexible through freezing winters and summer heatwaves. Even the humble bottle of shampoo might owe its texture-stabilizing chop to ethylenimine-based polymers, helping gels and creams stay uniform.
These real-world examples back up why the chemical industry keeps investing in the process. Research tallies from the European Chemicals Agency and the U.S. Environmental Protection Agency show a steady demand, not just for building polymers but also in pesticides. Farmers see value in the water-resistance and sticking power that ethylenimine chemistry can lend to components in crop protection products.
Weighing Safety and Sustainability
Ethylenimine’s usefulness doesn’t wipe away risks. On the contrary, it has a reputation for danger, classified as a probable human carcinogen. Direct contact in factories and labs still causes severe health scares. A workplace accident in the early 2010s stuck with me: even minor exposure led to aggressive decontamination protocols and evacuations. Stringent workplace controls, engineering safeguards, and mandatory protective gear became permanent, not optional, at sites using this compound.
The industry’s embrace of ethylenimine comes with a growing sense of caution. Factories are switching to closed systems that trap fumes and prevent leaks before human exposure ever becomes a risk. Several research groups and manufacturers explore bio-based alternatives and greener processes as well, but the challenge isn’t small. Many proposed substitutes still lag behind in performance or price.
Looking to the Future
Ethylenimine isn’t going away. As the chemical backbone for so many products, it keeps finding roles that others can’t easily fill. At the same time, rules around its use keep tightening—as seen in European Union REACH regulations and moves from U.S. agencies. Companies see incentives to improve training, invest in engineering controls, and monitor health outcomes. Smarter handling, better protections, and a drive for innovation might shrink some of ethylenimine’s risks over time, even if nothing replaces its unique power just yet.
Is ethylenimine hazardous to health?
Understanding What Ethylenimine Does
Ethylenimine shows up in places where industrial chemistry moves fast and doesn’t always slow down for comfort. Used to make things like resins, adhesives, and pharmaceuticals, it plays a backstage role most people never notice. Workers in these settings can’t afford to just hope everything stays safe. They face ethylenimine at the supply shelf and production line.
Main Health Concerns
Exposure to ethylenimine doesn’t only mean skin irritation or watery eyes. Breathing in its vapors, even for a short stretch, can lead to coughing, shortness of breath, sore throat, and headaches. Skin contact may cause redness, pain, or even blistering. What makes this compound really alarming is the data behind it. The International Agency for Research on Cancer places ethylenimine as a possible human carcinogen. Long-term or repeated contact has been linked with tumor growth in studies on animals.
In my experience talking with plant operators, real worry often centers on vapor exposure. Simple spills or leaky connections become big health worries quickly. I once saw a technician dealing with a drum that wasn’t sealed tight. Within minutes, he started complaining about his skin itching and burning. Immediate first-aid made a difference, but this example sticks with me for a reason.
Routes of Exposure and Real-World Cases
Breathing becomes the main way people get exposed. Liquid ethylenimine can also seep through gloves or get splashed onto arms, so skin risk can’t be ignored. Stories shared by safety trainers point out accidents where simple mistakes—like using the wrong protective gear—turn common jobs into real health scares.
A 2022 report from the U.S. National Institute for Occupational Safety and Health followed cases in a chemical plant, finding workers reporting a range of symptoms, from mild discomfort to serious eye and lung irritation, even after using regular protective gear. Once folks started using full-face respirators and double gloves, symptom rates dropped sharply. This reminds us that safety steps matter more than written policies.
Testing and Regulation
Regulators haven’t shrugged off the dangers. The Occupational Safety and Health Administration (OSHA) set the permissible exposure limit at a low 0.5 parts per million in air, averaged over eight hours. This number signals just how quickly trouble can start. The Environmental Protection Agency treats spills with strict cleanup codes, too.
Improving Safety: Where Responsibility Lies
Handling ethylenimine safely demands a mix of industrial hygiene, personal responsibility, and company investment. Standard gloves and aprons only go so far—thicker, chemical-resistant suits work better. Air monitoring needs attention, especially in older plants where equipment loses its edge.
Clear training drives down accidents. I’ve seen teams run mock drills, acting out spill or exposure scenarios. These exercises help everyone move fast under pressure and remind workers that overconfidence can backfire. Management faces constant pressure to fix leaks and replace outdated gear, and for good reason: health doesn’t get a second chance.
Better Paths Forward
Solving the risk puzzle takes honest reporting and adaptation. Regular health checks for workers help spot early symptoms and show where protective steps break down. Substitution with safer chemicals, when possible, should always be discussed—if the industry gets creative, safer options might take ethylenimine’s place down the road.
Until then, knowing the risks, respecting the regulations, and investing in solid workplace culture stays just as important as technical fixes. In my experience, most workers step up when they realize their own vigilance can be the dividing line between a safe shift and a trip to the emergency room.
What is the chemical formula of ethylenimine?
Cracking Open Ethylenimine's Basics
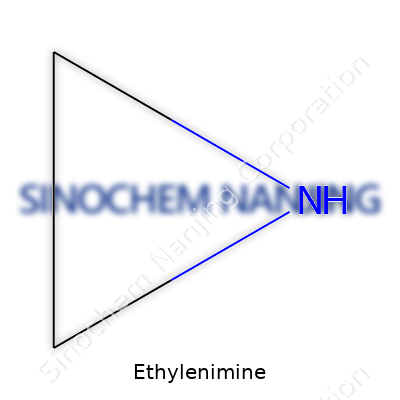
Ethylenimine doesn’t pop up in most people’s day-to-day conversations, but its presence in chemical industries is impossible to ignore. The formula for ethylenimine is C2H5N. It feels simple at a glance: two carbons, five hydrogens, and one nitrogen come together to make a molecule that packs a punch in its applications and quirks. My experience working alongside chemists showed me just how much impact a small molecule can create. Ethylenimine often carries a tall stack of responsibilities in making products that we rely on without realizing it—things like resins, adhesives, and pharmaceuticals.
Why Ethylenimine’s Formula Matters
Chemical formulas are really more than memory exercises from high school. Each letter and number tells a story, and C2H5N’s tale revolves around reactivity and practical use. The three-membered ring structure—called an aziridine—gives ethylenimine an edge. This ring is strained, so you get a volatile mixture that wants to react with other substances. That reactivity unlocks creative solutions in industry, as it serves as a building block for everything from anti-tumor drugs to tough coatings.
Lack of care with ethylenimine’s handling, though, leads straight to trouble. I recall hearing from a safety officer about an old incident: just a few drops spilled in a poorly ventilated room led to severe irritation for everyone nearby. The volatile, ringed structure that’s so prized for chemical synthesis is exactly what makes it dangerous for workers. The National Institute for Occupational Safety and Health (NIOSH) even lists ethylenimine as a substance to handle with special precautions. Long exposure can have nasty effects, damaging skin, eyes, and sometimes triggering longer-term health problems.
Safety Steps and Solutions
Companies using ethylenimine owe their teams real protections. Simple steps—like high-quality gloves, reliable respirators, and working inside closed systems—make a world of difference. During my time shadowing chemical engineers, I saw the benefits of straightforward training. When workers understood the boundaries that ethylenimine’s formula demanded, accidents fell off fast. That hands-on attitude, supported by good signage and routine safety checks, did more than a stack of unread manuals ever could.
Finding replacements also helps. Some labs now explore less volatile ring compounds that mimic ethylenimine’s behavior without its risks. Green chemistry pushes for alternatives wider still, focusing on ways to skip hazardous reactants altogether. That search isn’t easy—some uses call for ethylenimine’s particular mix of toughness and flexibility. But with modern chemical research, stubborn problems eventually get new answers.
Looking Toward More Responsible Chemistry
Knowing the chemical formula for ethylenimine—C2H5N—doesn’t end with a trivia fact. That knowledge points to the molecule’s strengths, risks, and possibilities. It forces anyone working with it to respect the reality: small formulas can carry big weight, and careful handling keeps its benefits squarely on our side of the ledger. Smart engineering, safer practices, and new chemistry keep this compound useful yet less menacing, letting industry grow without closing its eyes to safety or the health of the people making it all work.
How should ethylenimine be stored safely?
Understanding the Real Risks
Ethylenimine packs a punch as a chemical. It’s not a household name, but in the industrial world, folks know how volatile and unpredictable it can turn. Breathing its vapors or touching the liquid risks burns and serious lung damage. Since it’s flammable and explosive under the right set of conditions, the hazards go far beyond skin-deep. I’ve seen firsthand what happens when institutions skip good storage habits—lost dollars, injured workers, and sometimes a call to the fire department that nobody wants.
Proper Containers Matter
Controlling ethylenimine always starts with the right container. This chemical eats its way through weak plastics and will react with copper or copper alloys. Steel drums, lined with Teflon or glass, block reactions that weaken safety. A tight seal on every drum or bottle stands between an ordinary workday and a nasty spill. My time in a lab taught me to never trust a container just because it says “chemical safe.” Like most seasoned technicians, I check seals twice before moving anything.
Temperature and Ventilation—A Balancing Act
Ethylenimine gives off vapors at room temperature that shouldn’t linger in the air. Good ventilation pulls those vapors away, and an exhaust hood stops headaches before they even start. This chemical holds a flash point below freezing, so storing it somewhere cool isn't a suggestion. A dedicated flammable storage cabinet, fire-rated, with self-closing doors forms a strong defense. Years of maintenance work convinced me it’s easier to deal with a fussy HVAC system than explain a vapor explosion to safety inspectors.
Labeling and Security
Accurate labels help everyone stay on the same page. Each container needs a clear sign with the chemical name, concentration, and hazard warnings. Labels wear off—so checking them feels like part of any weekly routine. I’ve seen accidents happen just because someone grabbed the wrong bottle. Keeping ethylenimine locked up, with access limited to trained folks, cuts down on mix-ups and theft.
Regular Inspection and Spill Plans
Old habits count for a lot in chemical storage. Double-checking for leaks, cracks, or corrosion turns into muscle memory with experience. Spills happen, no matter how careful people think they are. A real-world plan spreads across more than a dusty binder. Emergency kits—acid-resistant gloves, full-face shields, sorbent pads—should never sit out of arm’s reach. I remember a spill from a cracked bottle that nearly went sideways. Quick action made all the difference because everything was ready and everyone had trained for the moment.
Training: The Last Line of Defense
Anyone who handles or stores ethylenimine deserves proper training about what they’re working with. People need to spot trouble long before it starts. I’ve watched teams improve over the years, not by fancy tech, but by learning from each close call. Hands-on instruction beats any slideshow for this stuff. Experts warn about complacency, especially in places juggling dozens of chemicals. Reinforcing those lessons with refresher courses keeps old mistakes from repeating.
What precautions are needed when handling ethylenimine?
Understanding What You’re Dealing With
Ethylenimine looks harmless at first glance — a colorless liquid, not a fuming acid or a bright, scorching powder. That’s part of what makes it so risky. Exposure brings serious health hazards, including severe irritation to skin, eyes, and lungs, along with long-term concerns linked to cancer. I remember first learning about it in a university lab, where everyone seemed more nervous handling ethylenimine than almost anything else in the storeroom. There’s a good reason for that. According to the Centers for Disease Control, even a splash or stray vapor can cause skin burns, blurred vision, and respiratory distress. And because it vaporizes easily, the risk doesn’t always come with a warning smell.
Importance of Physical Barriers
Nobody should work with ethylenimine in an open space. Fume hoods, glove boxes, or local exhaust systems form a crucial line of defense. Airflow measurements matter — it’s not enough to rely on the hood just being on; check its face velocity with an anemometer. Chemical-resistant gloves, safety goggles with side shields, and full-length protective clothing help keep exposure off your skin. Nitrile gloves typically block most solvents, but ethylenimine eats through some materials like latex, so it pays to double-check the material safety data sheet instead of guessing.
Personal Health Stays on the Line
Business owners and lab supervisors can’t just stick a sign on the door and hope people play by the rules. I’ve seen labs where shortcuts led to serious accidents: people grabbing the nearest gloves, skipping respirators, or stacking bottles in the wrong place. The Occupational Safety and Health Administration (OSHA) sets a permissible exposure limit of only 0.5 ppm for ethylenimine. That’s a pretty loud warning that even tiny exposures add up fast. Using a cartridge respirator or, even better, a supplied-air mask can make the difference between a minor scare and a trip to the emergency room.
Storage and Handling Practices
Ethylenimine breaks down under light and heat — sometimes violently. Glass containers won’t always cut it. Steel or Teflon-coated vessels stand up better, and everything stays in secondary containment trays in case of spills. I always make sure to double-check tight seals, as vapors escaping rarely give much notice. Keep it away from acids, oxidizers, and any container that once had water in it, since mixing creates toxic fumes.
Emergency Responses Save Lives
No scientist or worker likes to think about spills, but action beats panic every time. Anyone working around ethylenimine should know where the nearest eyewash and safety shower are, test them monthly, and go through spill drills during onboarding. Quick action after a splash — a thorough rinse under running water for at least 15 minutes — cuts injury risk dramatically. Emergency services need clear instructions on what to expect and how to decontaminate both people and spaces.
Training and Personal Vigilance
Most risks come down to people, not equipment. Everyone handling ethylenimine should receive regular training, not just a once-a-year slide show. Even experienced chemists need refreshers; old habits can bring mistakes. Open communication about near misses, providing immediate access to updated safety data, and keeping team members alert to the dangers all combine to create a safer environment.
Solutions Rely on Culture, Not Just Tools
No technology alone keeps people safe from chemicals like ethylenimine. Clear procedures, constant vigilance, and a culture that favors safety over speed carry the day. I’ve seen workplaces where people take five minutes to do things right — and no one gets hurt. That’s the kind of environment worth aiming for with something this hazardous.
| Names | |
| Preferred IUPAC name | Aziridine |
| Other names |
Aziridine Aziridane Ethyleneimine Aziridinium hydroxide 1-Aziridine |
| Pronunciation | /ɛˌθaɪliˈniːmɪn/ |
| Identifiers | |
| CAS Number | 151-56-4 |
| Beilstein Reference | 505923 |
| ChEBI | CHEBI:42585 |
| ChEMBL | CHEMBL1231266 |
| ChemSpider | 54938 |
| DrugBank | DB04147 |
| ECHA InfoCard | 100.001.078 |
| EC Number | 203-481-7 |
| Gmelin Reference | 985 |
| KEGG | C06510 |
| MeSH | D005029 |
| PubChem CID | 6115 |
| RTECS number | KH4300000 |
| UNII | 4T6U1Y8755 |
| UN number | UN1185 |
| CompTox Dashboard (EPA) | DTXSID1020696 |
| Properties | |
| Chemical formula | C2H5N |
| Molar mass | 42.08 g/mol |
| Appearance | Colorless liquid with an ammonia-like odor |
| Odor | Ammoniacal |
| Density | 0.832 g/cm³ |
| Solubility in water | miscible |
| log P | -0.38 |
| Vapor pressure | 11.3 kPa (at 20 °C) |
| Acidity (pKa) | 8.0 |
| Basicity (pKb) | 7.9 |
| Magnetic susceptibility (χ) | -13.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.017 |
| Viscosity | 1.3 mPa·s (20 °C) |
| Dipole moment | 1.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 198.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 67.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -794.6 kJ/mol |
| Pharmacology | |
| ATC code | J01XX05 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS05,GHS08 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H314, H317, H331, H350 |
| Precautionary statements | P280, P260, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P308+P311, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | −30 °C (−22 °F; 243 K) |
| Autoignition temperature | 410 °C |
| Explosive limits | 4.2–36% |
| Lethal dose or concentration | LDL0 Human oral 20 mg/kg |
| LD50 (median dose) | LD50 (median dose): 20 mg/kg (oral, rat) |
| NIOSH | K045 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Ethylenimine: "2 ppm (3 mg/m3) as an 8-hour TWA (OSHA) |
| REL (Recommended) | 0.018 mg/m3 |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Aziridine Dimethylenetriamine Ethylenediamine |

