Ethylene Glycol Ethyl Ether Acetate: A Comprehensive Commentary
Historical Development
Back in the early phases of industrial chemistry, organic solvents played a critical role in defining the capabilities of manufacturers. Ethylene glycol ethyl ether acetate, often known in the industry as EGEEA for short, came onto the scene when there was growing demand for solvents that combined strong solvency with balanced evaporation rates. Chemical companies in the late 20th century started looking for alternatives to more hazardous, volatile compounds, prompting intensive research. Early adoption began in the coatings industries, where traditional solvents often failed to provide the smooth finish or stability that demanding applications required. Teaching in university chemical engineering labs, I saw first-hand how students learned the shift to more tailored, less toxic compounds, with EGEEA as a case study. When regulatory scrutiny grew on emissions and workplace exposure, EGEEA offered a compromise between performance and compliance, giving product developers a solvent that met shifting safety expectations and kept pace with productivity standards.
Product Overview
Ethylene glycol ethyl ether acetate shows up in a variety of products, but it never loses its identity as a high-performance organic solvent. Known under various trade names and sometimes referred to simply as Cellosolve acetate, this clear, flammable liquid has helped formulators bridge the gap between aggressive solvency and moderate evaporation. In printing inks, paints, and certain cleaners, EGEEA brings the power to dissolve resins, dyes, and synthetic polymers. Workers in these sectors value its ability to break down hard-to-handle materials, which allows for processes that avoid waste and boost throughput. Its odor profile sets it apart from older, harsher solvents, earning it preference where working conditions and indoor air quality have an impact on worker satisfaction. Companies have stuck with EGEEA not just for what it can dissolve, but for the reliable way it leaves behind a less streaky, more predictable finish.
Physical & Chemical Properties
EGEEA possesses a molecular structure that grants it a unique set of physical and chemical attributes. As a clear liquid at room temperature, it boasts a boiling point around 156–159°C, melting point barely below zero, and a viscosity that keeps production lines running even in cooler climates. The dual presence of the ethylene glycol and acetate group empowers this chemical with balanced polarity—enough for dissolving tough organic materials but restrained enough to survive in formulations without causing premature drying or gelling. In the chemical warehouse, team members quickly learn to respect its flammability; EGEEA will ignite under the wrong storage or handling, so inventory management emphasizes sound safety standards. Its low water solubility compared to alcohols makes it a standout in paints, where water resistance has become a selling point.
Technical Specifications & Labeling
Looking at the bottles or drums coming from suppliers, EGEEA must follow strict labeling standards. Product datasheets specify purity often greater than 99%, water content below 0.1%, and mention every relevant impurity, such as related esters or unreacted glycol ether. Density sits at about 0.97 g/cm³, so every shipping manager knows to check weights as a safeguard against dilution or fraud. Regulatory compliance takes center stage during audits; hazardous labeling and GHS symbols are prominently displayed, and the Safety Data Sheet (SDS) always stays close at hand in manufacturing plants. Data integrity matters—real people depend on clear information when working around such volatile substances. Local and international laws dictate threshold limit values, transport limitations, even waste management protocols. Decades working in quality systems taught me: over-informing always wins when workplace safety and community trust are at stake.
Preparation Method
Commercial production of EGEEA isn't just about mixing and bottling. The standard route involves reacting ethylene glycol monoethyl ether (commonly called ethyl cellosolve) with acetic acid or its derivatives, often under catalysis to sharpen yields. Process engineers monitor temperature and concentrations closely; runaway reactions or overheating can spark fires or create off-grade material. Over the years, process optimizations made these syntheses more eco-friendly, using less energy or safer catalysts. At bench scale, students sometimes attempt small runs, marveling at the ease of esterification and the learning that comes from monitoring a real-time distillation. Industrial facilities invest heavily in purification and quality checks to guarantee each batch hits the physical and chemical benchmarks—failures risk spoiling end products and reputations.
Chemical Reactions & Modifications
EGEEA’s chemistry stems from its ester and ether groups, making it a playground for reaction chemists. It resists hydrolysis under neutral conditions, but strong acids or bases can force breakdown into acetic acid and glycol ethers—an outcome useful for selective degradation or recycling. In paint laboratories, technicians have experimented with blending EGEEA with other glycol ethers or acetates, sharpening evaporation curves and manipulating solvency through clever formulation. Upstream chemical manufacturers sometimes use EGEEA as a feedstock to craft specialized surfactants or intermediates. For me, watching this chemical serve as both product and raw material always drives home how a substance can stretch across value chains and push different sectors ahead.
Synonyms & Product Names
Across regions and various suppliers, EGEEA answers to several names. Cellosolve acetate, Glycol ether ethyl acetate, and Ethyl glycol acetate often appear in purchase orders, each reflecting certain linguistic conventions or branding legacies. Companies standardize naming internally, but workers need to watch for confusion that might cause mix-ups. In early days of my career consulting in cross-border projects, the same substance would arrive labeled differently in neighboring plants, leading to moments of unwanted improvisation. Proper identification isn’t just an academic detail—it avoids misapplication and keeps safety protocols squarely on track.
Safety & Operational Standards
Occupational safety for EGEEA demands diligence. Its volatile nature and moderate toxicity mean companies install fume hoods, monitor exposure levels, and make sure that anyone handling this substance wears gloves and goggles. Fire marshals regularly inspect solvent storage, looking for leaks, incompatibilities, and damaged labels. Proper ventilation systems get designed not only for legal requirements, but also out of respect for workers who deserve a space free of lingering chemical smells or health hazards. Over time, fines for non-compliance and the cost of accidents have taught management what can go wrong when shortcuts get taken. In operations, spill drills and real training outperform posters and one-time briefings. I always believed in rotating team leaders through hazard communication refreshers and gear inspections—errors fade when the rules become part of habit.
Application Area
EGEEA’s broad use finds it in high-value areas like automotive coatings, electronic cleaning fluids, polyurethane, and printing inks. For paint shops needing slow, thorough evaporation, this chemical replaces faster solvents that once caused uneven films or rashes on fresh surfaces. Printing businesses rely on its compatibility with pigment dispersions, cutting down on nozzle clogs and unwanted downtime. In specialty cleaners, EGEEA goes after oils, greases, and hardened polymers left behind in manufacturing lines. During a project with an industrial coatings plant, I noticed that small tweaks to EGEEA concentration meant better drying rates and harder finishes, which led to fewer product returns. Users in the electronics world count on its mild solvency to clean parts without stripping sensitive layers or inviting static buildup.
Research & Development
R&D teams continue to refine EGEEA’s applications, searching for greener processes or new uses in fields like battery manufacturing or advanced composites. Analytical chemists study new purification steps, aiming to reduce leftover contaminants and extend shelf life. Environmental chemists examine pathways to bio-based production, hoping to cut the carbon footprint left by traditional petrochemical sources. Researchers at universities compete for grants to unravel the reaction dynamics in complex blends—data from these efforts often filters down to small- and medium-sized companies looking for an edge. Real technical progress happens when academic theory meshes with practical constraints on plant floors, a combination I’ve witnessed in both major corporations and scrappy startups.
Toxicity Research
Concerns over safety keep toxicologists and regulators busy. EGEEA’s moderate level of toxicity puts it under study in labs dedicated to industrial hygiene and environmental safety. Animal studies and workplace exposure research feed into critical updates of permissible exposure limits. Detailed monitoring of air and water near manufacturing sites has uncovered patterns in bioaccumulation and chronic effect risks, prompting some manufacturers to switch partially or entirely to safer alternatives. My own students often struggled with the real-world tradeoffs between urgent performance needs and downstream health issues. Transparency about research findings, open reporting, and taking feedback from those on the factory floor builds a culture that respects both innovation and people’s well-being.
Future Prospects
Looking toward the future, EGEEA will face stiffer competition from biobased and less toxic solvents. But the strong technical profile of EGEEA ensures it remains important for industries where its performance edge matters—especially in paints, inks, and specialty cleaning. Process optimization, better waste handling, and closed-loop recovery systems raise the bar for sustainability in its use. Digital monitoring and automated dosing may shift exposure risks out of routine handling and toward a more measured, data-driven approach. Regulatory changes often lag behind, but industry leaders who invest in lower-emission, safer processes often find their investment returned through avoided compliance headaches and better workplace morale. In the decades ahead, I expect EGEEA to stick around, even if its role narrows to special applications that need just what this molecule brings to the table.
What is Ethylene Glycol Ethyl Ether Acetate used for?
But What Does Ethylene Glycol Ethyl Ether Acetate Actually Do?
Ethylene Glycol Ethyl Ether Acetate, or EGEEA for short, often ends up living in the shadows compared to big headline-grabbers like acetone or ethanol. Spend a few years near paint or electronics plants, and you’ll hear plenty about it. The main reason? This liquid acts as a powerful solvent, able to break down resins, varnishes, inks, and dyes in ways some more familiar chemicals just can’t manage.
Walk into a print shop or paint manufacturing facility, and you catch its faint odor near mixing drums and drying lines. That’s EGEEA at work, blending pigments and polymers so they stick together without clumping or separating. Anyone who’s accidentally left paint half-mixed knows what happens next: streaks, lumps, or bubbling. Producers rely on EGEEA to keep things smooth right out of the can.
Keeping Up with Electronics Manufacturing
For all the talk about new phones and smart gadgets, few people realize just how much chemistry goes into circuit boards and display screens. EGEEA pops up here, too. It helps manufacturers lay down thin, even coatings of photoresist materials—the types needed to etch ultra-fine lines and connections onto silicon wafers. One deviation, and performance tanks. Uniform coatings mean faster chips and brighter screens, both of which consumers demand year after year.
Whenever tech companies announce another leap forward, there’s usually some push at the factory level to tighten margins or boost yields. EGEEA provides real leverage against failed batches by supporting greater control in film deposition and etching. Missed coatings or contamination can tank a production run fast, costing serious money and possibly damaging reputations.
Health and Safety: A Real Concern
Experience in the field raises practical questions about exposure and safety. Chemical handling crews know to take precautions—well-ventilated spaces, gloves, goggles. Chronic exposure to EGEEA brings health risks like eye irritation, headaches, and even organ damage with high enough doses. Not everyone heeds the warnings until a scare forces change. This ties back to honest education and vigilance, often driven by people with direct experience rather than by inscrutable labels on storage drums.
Regulations keep tightening in developed countries, but loopholes and shortcuts may creep in where enforcement lags. Workers who know what EGEEA can do for their jobs should also know the right way to handle it. In some smaller or underfunded operations, corners get cut out of necessity or neglect, creating hidden dangers for both crews and the surrounding community.
Toward Cleaner, Safer Alternatives
Sustainability has climbed the list of corporate priorities. Chemistry departments now look at ways to replace or reduce reliance on high-impact solvents without sacrificing performance. For a while, going “greener” meant higher costs or weaker products. Lately, progress in alternative solvents and advanced filtration helped shrink those gaps.
Still, regulation only goes so far. Companies and communities do better when workers receive hands-on safety training, clear labeling, and access to the right protective gear. As industries evolve and care for the environment gains urgency, the future of EGEEA looks tied not only to performance, but to the value placed on health and stewardship. Real safety starts with recognizing the risks, learning from those who work closest to the material, and investing in better choices for all.
What are the safety precautions when handling Ethylene Glycol Ethyl Ether Acetate?
Understanding Why Safety Matters
Anyone who's stepped into a paint shop, electronics plant, or printing facility has probably run into solvents like Ethylene Glycol Ethyl Ether Acetate. What sounds like a mouthful on a label is actually a clear, sweet-smelling liquid that helps dissolve all sorts of stubborn things. But beneath that mild-mannered appearance, this solvent carries a few hidden dangers. Skin irritation, headaches, breathing issues, even damage to organs—those are real risks if you don’t take safety seriously.
Personal Experience with Industrial Solvents
My first summer job handled countless drums of industrial chemicals. I never forgot the lesson an old-timer taught me after a guy collapsed from fumes. He said, “Trust your nose and trust your mask.” Simple as it sounds, a lot of safety boils down to common sense and sticking to good habits, even if it means taking a minute longer on a job.
Protect Your Body and Lungs
Direct contact with Ethylene Glycol Ethyl Ether Acetate eats away at skin and can lead to rashes or more serious health effects. A lab coat, nitrile gloves, splash goggles, and sturdy closed shoes will block most of it. Vented chemical hoods, fume extractors, or at least solid cross-ventilation will keep the air fresher. Swapping out a clogged filter or opening a window makes a bigger difference than you’d think. Don’t be the person who takes off their mask “just for a second.”
Handling Spills and Storage Like a Pro
Accidents happen—always have absorbent pads and a dedicated spill kit nearby. This stuff evaporates fast and forms flammable vapors, so mop up spills immediately and bag up waste in flame-proof containers. Never toss rags or gloves in common bins, and don’t leave any open containers sitting around a shop. Nearby sources of sparks, flames, or even plugged-in power tools turn a minor mistake into a major emergency in a heartbeat.
Know Your Emergency Responses
If you take a splash to the skin, the first move should always be a long rinse with running water. Any chemical in the eyes means a direct sprint to the eyewash station or sink. If someone passes out or can’t breathe, carry them into fresh air and call for medical help. Posting up a clear emergency list and showing new hires how to use basic first aid can turn a scary moment into a manageable one.
The Call for Consistent Training
Yearly refreshers, good signage, and clear safety data sheets make up the roots of a safe workplace. OSHA and EPA both rank Ethylene Glycol Ethyl Ether Acetate as a chemical that demands respect. Tracking your use, keeping logs, and asking suppliers for the latest safety tips all help build a culture where nobody lets down their guard. Even after years in the field, I have seen how a well-prepared team can handle surprises far better than just hoping for the best.
Looking Ahead: Smart Changes for Better Safety
Modern factories look into less toxic alternatives once they can. Closed-loop systems, better detection alarms, and sealed containers cut down on risks for everyone. Still, nothing replaces people looking out for each other, taking time to clean up properly, and talking honestly about any “close call” so others can learn. Real safety never gets old, and it never comes standard—you build it step by step, job by job.
What is the chemical formula and structure of Ethylene Glycol Ethyl Ether Acetate?
Getting Clear on the Chemical Formula
Ethylene Glycol Ethyl Ether Acetate goes by the formula C8H16O4. In the world of solvents, this one combines flexibility with a straightforward structure. On paper, it breaks down to eight carbon atoms, sixteen hydrogens, and four oxygens. It’s made up of acetic acid mixed with the ether of ethylene glycol and ethanol. In short, chemists crafted it by swapping hydrogen in acetic acid with an ethylene glycol ethyl ether group. This approach builds a molecule that behaves with a good mix of volatility and solubility, making it useful in different settings.
Peering Into Its Structure
The backbone starts as an ethylene glycol skeleton. Picture two carbon atoms, each connected with an oxygen atom, with one side extended by an ethyl group. The other end carries an acetate group — which is just a two-carbon chain topped with an oxygen double bond and a single-bonded oxygen hanging off to complete an ester linkage. The result is a molecule that can interact gently with both water and organic compounds.
Why Structure and Function Matter
Years in a university chemistry lab taught me that a solvent’s power lies not just in the atoms but in how they fit together. Ethylene glycol ethyl ether acetate excels at dissolving polar and non-polar compounds because of these oxygen atoms connecting polar and nonpolar ends. That’s why it’s found in coatings, inks, and cleaners. Industries lean on it for cleaning up sticky resins and dropping pigment into solutions for a smooth finish. Factories keep things moving thanks to molecules like this one doing work nobody sees — easing grease off parts, blending into paints, speeding up drying time without dangerous fumes pouring out like some older solvents.
Risks and Practical Handling
Handling this chemical at work means respecting its strength. Exposure to vapors can irritate skin or eyes, so safety glasses and gloves belong on every bench. In the news, stories about occupational exposure remind us that careful ventilation prevents accidents. At home, people rarely deal with chemicals this powerful unless painting, but even then, ventilation keeps headaches and risks low. These lessons stick with anyone who has worked with volatile organic compounds — a little caution goes a long way.
Environmental Impact and Safer Alternatives
Environmental health needs real-world solutions. This chemical breaks down in the environment and doesn’t linger for decades, unlike some tougher compounds. Still, pouring it down the drain won’t do rivers and soil any favors. Factories running round the clock need good waste controls, using carbon filters or thermal treatment. Groups pushing for “greener” chemistry look for drop-in alternatives with fewer risks — such as solvents made from plants or even water-based mixes — when business models allow. These swaps aren’t about fads; they keep workspaces safer for everyone down the line.
Why Chemistry Never Happens in a Vacuum
Behind every formula like C8H16O4 stand real people — workers, neighbors, families. Checks on exposure, improvements in products, and thoughtful disposal come from keeping an eye on both well-being and results. As researchers and regulators continue improving chemical management, both performance and responsibility stay in sharper focus. Knowing molecules and their structures isn’t just for passing exams — it guides safer choices every shift, every shipment.
How should Ethylene Glycol Ethyl Ether Acetate be stored?
Understanding the Stakes
Ethylene glycol ethyl ether acetate runs a high profile in industrial circles, especially in paints and coatings. Getting storage right keeps workers safe, protects property, and ensures this solvent stays effective. I remember a maintenance call at a plant where a single leaking drum turned into a costly clean-up—nobody wants that. Unsafe storage of this solvent can cause fire hazards, sudden leaks, and even dangerous vapors in the air.
What Experience Teaches
The first thing I learned on the job: treat volatile liquids with respect. Colleagues I trust always say that proper storage means thinking ahead. A chemical like ethylene glycol ethyl ether acetate has a low flash point—below 50°C—so it won’t take much for vapors to cause a flash fire. One plant manager told me a static spark near an open container once triggered an alarm and shut down operations for hours. Small errors add up fast with solvents like this.
Practical Storage Conditions
A dedicated, dry, and cool area far from direct sunlight always works better than improvising with random shelving. Avoiding heat sources and open flame isn’t negotiable; even electrical outlets and fridges need special approvals for hazardous materials. Metal drums tightly sealed with no dents or cracks stand up better to long-term storage than plastic containers. I once saw employees storing open drums in a corner, and the fumes meant everyone was coughing. It only took one supervisor walking in to demand every lid secured.
Good ventilation also earns its keep. A simple exhaust fan or open windows won’t cut it. Think about chemical-rated ventilation hoods that move vapors outside, and sensors that track any leaks. Containment trays or spill pallets stop leaks from spreading. Nobody expects a drum to start leaking, but the first puddle that runs across the floor convinces you to believe it happens. Most experts suggest keeping spill kits close by and making sure everyone knows where they sit.
Safety Precautions and Labeling
Every time an unlabeled barrel appears, confusion starts. Clear labels listing the chemical name, hazard warnings, and date of receipt removes guesswork. I’ve seen audits knock points off for missing labels, but more importantly, proper communication lets response teams work quickly after a spill.
Some companies invest in storage cabinets rated for flammable materials. Steel cabinets with self-closing doors resist fire, buy time for evacuation, and give the emergency teams an edge. Even smaller businesses can pick up these cabinets as a smart investment. Safeguarding your team matters more than pinching pennies. Installing proper sprinklers above storage spaces adds another shield, especially in older buildings.
Checking Regulations and Training
Rules for storage vary by city, state, and country. OSHA, EPA, and local fire marshals all have checklists and guidelines. Overlooking a single detail can turn into an expensive fine or risk. In my own experience, hands-on drills, rather than dry lectures, get workers to notice leaking drums or overheated storerooms before anything goes wrong. Employees who practice handling spills, not just reading slides, know what to do when things go sideways.
Smart Storage Saves Trouble
Storing ethylene glycol ethyl ether acetate with care means taking each risk seriously and learning from others’ mistakes. Setting up storage by the book, keeping maintenance tight, and training people for real-world problems pays for itself. In the long run, smart storage protects lives, saves money, and keeps businesses running without costly interruptions.
Is Ethylene Glycol Ethyl Ether Acetate hazardous to health or the environment?
Understanding the Risks to Health
Ethylene glycol ethyl ether acetate usually comes up in industrial workplaces. It’s a solvent often used in paints, inks, coatings, and cleaners. Now and then, I’ve seen friends in surface coating plants talk about headaches and dizziness after handling solvents like this. Science backs up those observations – breathing in vapors or getting the stuff on your skin isn’t just uncomfortable, but also harmful.
Researchers have linked exposure to this chemical to symptoms like nausea, fatigue, and skin irritation. High doses hit the nervous system and can throw off your balance or judgment. The material absorbs through skin, raising concerns that gloves or aprons aren’t just optional; they’re essential. Short-term slips are bad enough. Over longer periods, studies have connected these glycol ethers to problems with blood counts and even reproductive effects. That information matters most for people who spend workdays in spray booths, print shops, or chemical plants.
The Environmental Protection Agency tracks this family of solvents because of these risks. Regulation keeps exposure down, setting limits for employees and mandating ventilation in workplaces. A safety data sheet for this chemical reads like a warning label: avoid inhaling vapors, wear splash-proof protection, and clean up spills right away. As somebody who’s seen workers take shortcuts, I recognize why rules have teeth.
Impact on the Environment
Chemicals never stay put. Wastewater and runoff carry solvents from factories to rivers and soil. Ethylene glycol ethyl ether acetate breaks down in air, but it sticks around in water longer. Fish and aquatic life don’t always recover when exposed to these compounds. I’ve watched clean-up crews work double shifts after solvent spills at industrial parks; a few hours of inattention put months of recovery on their agenda.
Some studies show this solvent can linger in groundwater, tough to remove once it seeps below the surface. Drinking water standards exist for a reason. It’s not just about workers or wildlife – families who draw water from wells could be affected if pollution spreads beyond fences. The history of chemical spills across the world proves leaks often go undetected until problems get big enough to notice.
What Works to Lower the Dangers
Focusing on alternatives makes sense. Newer water-based paints or less hazardous solvents replace glycols like this in plenty of shops. Those swaps matter in communities near factories; less toxic chemicals can mean fewer worries about leaks or air pollution. Where substitution isn’t practical, strong oversight counts more. Air filters, personal protective equipment, and frequent safety training aren’t just rules on paper. They protect the people who come home tired from their shifts, not just the bottom line.
Public records help everyone keep track. Agencies post data about spills, emissions, and violations for a reason. If a plant isn’t following the law, neighbors deserve to know. Whistleblowers – people who courageously speak up – often send the first warning signals when shortcuts put health or the environment at risk.
Communities can push for tougher rules, and businesses that value health look for lower-risk options. The lesson is simple: chemicals like ethylene glycol ethyl ether acetate carry real hazards. Taking health seriously means substitution, protection, and a willingness to speak up before trouble starts.
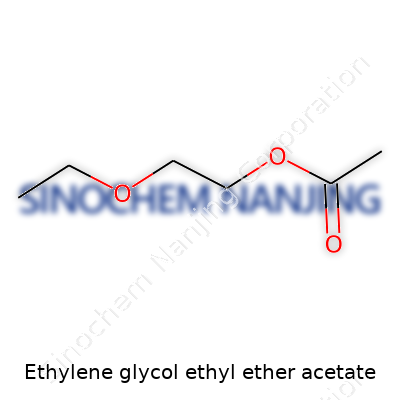
| Names | |
| Preferred IUPAC name | 2-ethoxyethyl acetate |
| Other names |
2-Ethoxyethyl acetate Ethoxyethyl acetate EEA Acetic acid 2-ethoxyethyl ester |
| Pronunciation | /ˈɛθ.ɪˌliːn ɡlaɪˌkɒl ˈɛθ.ɪl ˈiː.θər əˈsɛɪ.tət/ |
| Identifiers | |
| CAS Number | 111-15-9 |
| Beilstein Reference | Beilstein 1782480 |
| ChEBI | CHEBI:80840 |
| ChEMBL | CHEMBL57260 |
| ChemSpider | 5026 |
| DrugBank | DB14019 |
| ECHA InfoCard | 100.019.160 |
| EC Number | Ethylene Glycol Ethyl Ether Acetate" EC Number is "203-940-1 |
| Gmelin Reference | 8224 |
| KEGG | C19601 |
| MeSH | D017325 |
| PubChem CID | 10719 |
| RTECS number | KO2450000 |
| UNII | C3B4YRX2F5 |
| UN number | UN1171 |
| CompTox Dashboard (EPA) | DTXSID2021236 |
| Properties | |
| Chemical formula | C8H16O3 |
| Molar mass | 160.20 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Mild, pleasant |
| Density | 0.97 g/cm³ |
| Solubility in water | Miscible |
| log P | 0.32 |
| Vapor pressure | 0.32 mmHg (20°C) |
| Acidity (pKa) | 13.1 |
| Basicity (pKb) | pKb: 15.5 |
| Magnetic susceptibility (χ) | -62.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.406 |
| Viscosity | 1.14 mPa·s (at 25°C) |
| Dipole moment | 2.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 370.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -589.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3898.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H319 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | The flash point of Ethylene Glycol Ethyl Ether Acetate is **49°C (120°F)**. |
| Autoignition temperature | 185°C |
| Explosive limits | 1.2–10.0% |
| Lethal dose or concentration | LD50 oral rat 2,292 mg/kg |
| LD50 (median dose) | 2,700 mg/kg (rat, oral) |
| NIOSH | NIOSH: KB6125000 |
| PEL (Permissible) | PEL: 100 ppm (540 mg/m³) |
| REL (Recommended) | 25 ppm (135 mg/m3) TWA |
| Related compounds | |
| Related compounds |
Ethylene glycol Ethylene glycol ethers Ethylene glycol monoethyl ether Ethylene glycol monoethyl ether acetate Ethylene glycol monomethyl ether Ethylene glycol monoethyl ether acrylate |

