Ethylaluminum Dichloride in Focus: Past, Present, and Paths Ahead
A Journey from Historical Roots to Modern Labs
Ethylaluminum dichloride might sound like an obscure entry in an old chemistry catalog, but its story started with some major shifts in science. In the post-war era, the world looked for better plastics and synthetic materials. Researchers soon found organoaluminum compounds, like this one, could accelerate olefin polymerization. The Ziegler-Natta revolution of the 1950s would have limped along without these kinds of catalysts. In practice, these odd-smelling, moisture-sensitive liquids soon became workhorses in the production of simple, rugged plastics that fill homes and factories. Decades later, chemists keep these bottles behind double-sealed cabinets, aware of the impact a leaky stopper can have on a whole lab. The story of this compound runs parallel to the rise of plastics—not in the public limelight, but always present in the background.
A Closer Look at the Compound
As a pure substance, ethylaluminum dichloride appears as a colorless-to-yellowish liquid, with sharp fumes and a knack for reacting with water. Every chemist who handles this material remembers the hiss and white vapor when a drop touches skin or bench—those are the aluminum oxides and ethylene jets forming in a split-second. The molecular formula, C2H5AlCl2, gives away a lot about its personality. One ethyl group and two chlorines bond to an aluminum atom that prefers forming bridges with others of its kind, leading to clusters rather than lonely molecules. People often underestimate just how fast this compound decomposes in moist air, so every bottle comes with stern warnings and sturdy, airtight caps.
Technical Details in the Real World
Chemists rarely just pick up a jug and pour. If you want pure material, the standards insist on low water content, tight controls on aluminum concentration, and careful handling under argon or nitrogen. Labels tend to spell out purity in percentages, with awareness growing that trace impurities can shift reaction yields or create dangerous byproducts. People working on industrial scale often use stainless steel lines or glass syringes to transfer this stuff, never forgetting stories of cracked glass or blocked pipelines from clumsy handling. Temperature affects both storage and reactivity—too warm, and containers build dangerous pressure; too cold, and the compound thickens up, making dosing inconsistent and tools hard to clean.
How Chemists Make Ethylaluminum Dichloride
Building this molecule isn’t as simple as shaking two flasks together. Most labs follow a direct pathway: reacting aluminum metal with ethyl chloride, sometimes with a splash of aluminum chloride as a co-catalyst. The process throws off a lot of heat and fumes, so the scale-up from test tubes to barrels required years of innovation. People working at production plants know how easily runaway reactions can spiral out of control—ventilation, cooling coils, and well-calibrated valves keep things from flying apart. Unlike some lab curiosities, this process saw relentless tweaking, with modern setups recycling gases and managing waste to minimize both environmental and operator risks.
Reacting and Reshaping: More Than One Trick
Ethylaluminum dichloride gets its reputation from its fierce reactivity. Expose it to water, and you get an explosion of gas and a mess of aluminum hydroxide. Put it with simple olefins, and it wakes up as a polymerization co-catalyst, lending power to Ziegler-Natta systems that turn gases into solid plastics. Chemists also use it to introduce ethyl groups into molecules or to cleave certain bonds that resist milder reagents. Some skilled researchers use its reactivity to build custom organometallic complexes, always with respect for how even a small miscalculation can send a reaction haywire. Studies published in reputable chemistry journals show ongoing efforts to tweak the core structure, swapping ethyl or chloride groups for others, hoping to rival its catalytic punch with less risk or greater selectivity.
Other Names in the Literature
This compound goes by more aliases than any secret agent. You’ll find “dichloroethylaluminum,” “ethylaluminium dichloride,” and “EADC” cropping up in papers from Europe, North America, and Asia. Chemical registries group them under various trade names, often based on the supplier or manufacturing route. Dictionaries of chemical synonyms sometimes stretch for pages, reflecting the compound’s global presence and long research history. In many labs, “EADC” rolls off the tongue, everyone knowing the bottle with a red hazard diamond and a complicated stopper.
Staying Safe: Standards and Stories from the Field
No experienced operator takes shortcuts with ethylaluminum dichloride. Splash a drop on your hand, and you feel the burn in seconds—a lesson learned only once. Most facilities require full-face shields, acid-resistant gloves, dedicated fume hoods, and emergency showers. Even the best-prepared technicians stay wary of pressure buildup in containers, always grounding vessels to avoid static sparks. The stories circulate: a careless transfer causing a spray of burning liquid, or a loose-fitting stopper sending fumes through an entire section of a plant. These hard lessons fuel constant updates to handling protocols, with oversight from occupational health agencies and insurance auditors. Each year, safety training grows more sophisticated, building on chemical knowledge, hard data, and real incidents.
Applications: Why It Matters Beyond the Lab
At first glance, the only people who seem to care about ethylaluminum dichloride are chemists and chemical engineers. Dig deeper, and you see its fingerprints in everyday life. The compound sits at the core of catalytic systems that drive the creation of polyethylene, polypropylene, and related polymers—plastics that shape packaging, automotive parts, medical devices, and household goods. It has some uses as a Lewis acid in organic synthesis, especially for specialized pharmaceuticals or fine chemicals. While the world debates the future of plastics, no one forgets the impact this reagent had in making durable, cheap materials out of simple building blocks. It helps push down waste by increasing production efficiency and purity.
Research and Development: Progress with a Cautious Eye
Decades of R&D have built up a strong base of publications, patents, and corporate know-how around this compound. The drive for greener chemistry nudges both academic groups and manufacturers toward safer alternatives or improved versions. Some teams focus on modifying the ligand environment on the aluminum, chasing less toxic, easier-to-handle compounds that keep the catalytic efficiency. Other efforts look for additives or new formulations that suppress violent reactions with air or water. Funding agencies steer research toward recyclability and environmental impact, never losing sight of how these reagents enter and leave the production chain.
Toxicity: An Ongoing Public Health Study
Long-term studies still chart the health risks of exposure. Acute contact usually leads to burns or severe irritation, but chronic impacts deserve scrutiny. Researchers collect anecdotal cases and lab animal evidence, testing for evidence of organ damage, sensitization, or carcinogenicity. No one recommends casual exposure—regulators insist on closed handling systems, appropriate disposal protocols, and ongoing air monitoring where the compound gets used. Toxicologists work with data from factory workers, laboratory incidents, and emergency room visits to shape updated policies. Even as analytical methods improve, the guidance remains clear: respect the risks and treat every drop with caution.
Looking Forward: What’s Next?
Debate swirls as chemists eye the future of ethylaluminum dichloride. Will it hold its place, or give way to less reactive, easier-to-handle alternatives? Climate concerns and sustainability targets pressure manufacturers to find replacements or update production routes with green chemistry in mind. Some innovative startups invest in modular reactors or continuous flow systems, promising both higher safety and lower waste. Big chemical players chase molecules with tailored reactivity, dialing back on dangerous side effects. Consumers will never glimpse these shifts directly, but future plastics and specialty chemicals might owe their safety or sustainability to silent changes in chemistry’s building blocks. What won’t change is the community’s commitment to balancing progress with responsibility—hard-earned wisdom that grows every year, bottle by careful bottle.
What is Ethylaluminum Dichloride used for?
Digging Into Its Role in Industry
If you step into the world of industrial chemistry, you hear about ethylaluminum dichloride fairly often. This compound plays a central part in catalytic processes, especially in the manufacturing of plastics. Growing up in a region surrounded by manufacturing plants, I noticed how the movement of raw materials shaped local economies and jobs. Ethylaluminum dichloride serves as both a tool and a challenge for these industries.
Polymerization and Making Plastics
This chemical acts as a strong co-catalyst in the Ziegler-Natta method, a core process for making polyethylene and polypropylene. Creating cleaner and more efficient plastics matters now more than ever as single-use items continue stacking up in landfills. Building polymers with tighter control gives us plastics that can last longer or break down easier, depending on what society needs.
The Ziegler-Natta process wouldn’t move ahead without this aluminum compound. It helps activate titanium-based catalysts. That step allows for the precise linking of molecules, which means you get stronger shopping bags and more reliable food containers. Engineers and scientists have to balance performance, safety, and waste. Every time a new line of containers lands on store shelves, it relies on repeating this catalytic dance millions of times in giant reactors.
Ethylaluminum Dichloride as a Chemical Builder
Beyond plastics, ethylaluminum dichloride helps with organic synthesis—building blocks for pharmaceuticals, fragrances, and fuel additives. My time working as a technician in a chemical lab showed me how a single change in process can ripple outward. A tweak using compounds like this one often means a cleaner reaction, a bigger yield, or fewer byproducts.
Instead of describing it as just a supporting player, it’s better to see ethylaluminum dichloride as a workhorse that quietly powers modern chemistry. Its reactivity draws on the strength of aluminum and the complexity of its molecular structure. In the right hands, those traits open doors for new medicines and performance chemicals. Mistakes or accidents, though, carry risks for people and the environment.
Challenges and Safer Handling
Those who work with hazardous reagents like ethylaluminum dichloride walk a tightrope. Contact with water creates flammable hydrogen gas. Exposure can damage skin, eyes, and lungs. During my time near industrial facilities, I saw the drills and emergency protocols in place day after day. You can’t cut corners. Companies train teams rigorously and outfit them with protective gear. Emergency response plans exist not only for their own safety but also for entire neighborhoods.
Moving forward, the chemistry field faces pressure to find substitutes that perform well but come with less risk. Research continues into catalysts that are easier to store and handle. Investments in automation and monitoring tech let plants manage dangerous materials carefully. In my experience, the dialogue between industry, researchers, and communities must stay open—because only through cooperation do safer and cleaner solutions emerge.
What safety precautions should be taken when handling Ethylaluminum Dichloride?
Understanding the Risks
Ethylaluminum dichloride sets off alarm bells for anyone familiar with reactive chemicals. This substance doesn't mess around. Contact with water creates a violent reaction that spits out flammable gases and heat. Skin, eyes, and lungs feel the pain in an instant after exposure. Shipping labels slap on the 'corrosive' and 'flammable' warnings with good reason.
Personal Protective Gear Isn't Optional
Handling ethylaluminum dichloride always means gearing up. From personal experience in chemical labs, the basic gloves and goggles don’t cut it. Go with heavy-duty nitrile gloves. Cover arms with a long-sleeved chemical-resistant lab coat. A face shield ranks much higher than safety glasses alone — splashes move fast, and so do reactions. For folks dealing with large containers, adding a chemical-resistant apron and boots has saved some from nasty burns. Respirators work as a backup if fumes become too strong, especially in poorly ventilated spots.
Control the Workspace
Open this chemical in a fume hood. Relying on just open windows or room fans invites fumes right into your lungs and puts coworkers at risk. I’ve seen labs gutted after a careless spill; vapor builds up quickly and one spark means disaster. Keep all sources of ignition away — no open flames, no static-inducing electronics in reach.
Dry as a Bone: It Matters
Dampness spells trouble. Even grease from a fingerprint can start a chain reaction. Always use dry, clean utensils. The storeroom needs a dehumidifier running. Desiccators and sealed cabinets do the heavy lifting when the day ends. Longevity and safety both depend on a dry bottle — moisture eats through containers and has caused more than a few surprise fires. Never use glass containers with ground-glass joints; bonded joints can freeze and shatter unpredictably.
Emergency Planning Can’t Wait
Lab newcomers scoff at drills, but seasoned staff know spills test nerves. Spill kits absorb hazardous liquids and neutralize them fast. Buckets of dry sand, ready to cover leaked material, make a big difference. Drains and sinks nearby? Block them before a drop lands. Ethylaluminum dichloride turns water into a reaction zone instantly.
Never downplay the burn hazard. Eye-wash stations and quick-drench showers belong within a few steps. Clear labels turn seconds into safety saves. Alarms and fire extinguishers should be tested regularly; dry powder extinguishers outperform CO2 or water units, which make things worse.
No Lone Rangers
Someone should always know you're working with this compound. Solo handling courts disaster; even the most careful handler can slip. Teamwork doesn't just keep projects moving — it keeps people alive. Clear communication about when a bottle hits a bench, how much is used, where it's stored, and when a spill happens beats silent efficiency every time.
Waste Matters More Than Most Think
Ethylaluminum dichloride waste can't go down the drain or even into the usual hazardous container. Neutralize slowly, with practiced hands, inside a fume hood by adding small amounts to a cold, dry, properly chosen solvent. Trained waste handlers know the drill — outside of that, outsourcing disposal saves more than stress.
Real-World Solutions
Ongoing training makes all the difference. Bringing in real stories of near-misses or injuries hammers home why strict protocol sticks. Supervisors push for regular checks on personal gear. Audits surprise everyone and often reveal weak spots in the process. Safety posters fade to background, but culture sticks when everyone feels responsible for each step. Mistakes hurt most when they're repeated, so sharing lessons openly, not just memorizing rules, has kept more than one workplace incident out of the news.
How should Ethylaluminum Dichloride be stored?
Understanding the Risk
Walk into any chemical plant that handles organometallic compounds and a few storage drums stick out. Ethylaluminum Dichloride belongs on that list, not because it’s common, but because it reacts violently with water. A single drop of moisture can trigger flaming reactions. Most folks aren’t ready for a chemical that spits fire and toxic fumes if humidity sneaks in. Stories about this compound often go untold outside laboratories, but incidents remind us how easily situations slip out of control.
Never Underestimate Water’s Presence
Every safety trainer I’ve met stresses this: even sealed drums sweat. Air finds a way. Slight leaks, an unseen pinhole, or condensation inside will set off a chain reaction with Ethylaluminum Dichloride. Handling the chemical outside a glovebox? One splash spells trouble for skin and lungs. The right container for this chemical matters. Steel won’t work – it causes violent decomposition. People use tightly sealed glass or Teflon-lined vessels. Checking seals, double gasketing, and adding desiccant packs becomes routine life in the warehouse.
Temperature Matters More Than Labels Suggest
Most workers focus on flammables, acutely aware of flashpoints and heat sources, but Ethylaluminum Dichloride creates its own heat even in storage. If temperature creeps up above room level, pressure in containers jumps and raises the chance for leaks or violent rupture. Warehouses built with evaporative coolers or air conditioning help, even if it takes investment. Leaving drums out in direct sun isn’t an error anyone should make more than once. One summer day can scorch a warehouse and force an emergency.
Ventilation and Segregation Save Lives
Many chemical facilities place incompatible materials far apart. Shelves labelled “keep away from oxidizers” don’t just make life easier for inspectors – they prevent fire. Ethylaluminum Dichloride won’t share space with acids, water-reactive compounds, or room humidifiers. Some warehouses even keep it surrounded by inert gas blankets using nitrogen, just to cut oxygen out if something goes wrong.
Good ventilation keeps fumes from pooling. Engineers tell me about designing hoods or vents that kick in automatically if sensors pick up rising levels of hydrochloric acid vapors. It’s not an extra, it’s as vital as a fire alarm at home. If fumes reach a worker before alarms go off, that lag can cost serious health.
Training and Preparation Trump Everything
Every safe storage rule means little if workers don't know or trust the process. The best chemical storage setups don’t skip steps – regular inspections, fresh training, replacing worn seals, and updating emergency plans if anything changes. PPE like goggles and gloves comes standard, but training teaches why those gloves matter, not just which ones to choose.
Accidents involving chemicals like Ethylaluminum Dichloride rarely happen from sabotage. Most stem from overlooked procedures, someone in a rush, or failing to replace failing containers. Each event costs money, trust, and sometimes lives. Safe storage starts long before a drum arrives, with written procedures, engineered systems, and real buy-in from people on the ground.
Looking Ahead
Storing a highly reactive chemical doesn’t always mean expensive tech. Some of the strongest storage systems build on simple rules done right every time: dry containers, cool and stable temperatures, real separation from water sources, and trained eyes. A bit of caution and rigor saves worry, keeps products pure, and, above all, keeps people unharmed. That earns trust from regulators and creates peace of mind hard to buy any other way.
What is the chemical formula of Ethylaluminum Dichloride?
Chemical Identity: C2H5AlCl2
Ethylaluminum dichloride brings together aluminum, two chlorine atoms, and one ethyl group, forming the formula C2H5AlCl2. It’s more than a string of letters and numbers out of a chemistry textbook. I’ve seen how its reactivity and structure play a central role in real-world processes, especially in catalysis for polymer production.
A Builder for Plastics
Years back, during a visit to an industrial lab, I watched how small bottles of ethylaluminum dichloride served as gatekeepers for the creation of polyethylene and polypropylene. The chemists called it a co-catalyst, but the impact showed up everywhere — from stronger packaging materials to lighter car parts. C2H5AlCl2 reacts with compounds like titanium chloride to help build long chains of plastics. Its role illustrates why the formula means so much in heavy manufacturing, which shapes everything from shopping bags to medical supplies.
Risk and Responsibility Come With the Formula
Talking about this compound, safety matters just as much as chemical structure. Ethylaluminum dichloride reacts instantly with water, producing sparks, fumes, and fires if mishandled. In some labs, I’ve seen safety gear checked twice before the bottle leaves its shelf. Without strict controls, spills could threaten both people and equipment. Occupational health studies back up that point, showing injuries from accidental exposure. The formula is powerful, but control and respect for its hazards must match its utility.
Ethics in Handling and Waste
Discussions about chemicals like ethylaluminum dichloride always come back to what happens after use. Aluminum-based catalysts can contaminate soil or water if waste isn’t managed. Some companies use closed-loop systems, recycling both solvent and catalyst residues. Others partner with certified hazardous waste processors. Making those choices isn’t just technical—it crosses into ethics, responsibility to workers, and public trust in the chemical industry.
Innovation With a Foundation in Chemistry
C2H5AlCl2 sits at the intersection of chemistry and technological ambition. Researchers explore tweaks to this formula, replacing ethyl groups or shifting the aluminum’s surroundings, all to tune the end properties of plastics or speed up reactions. Some startups have even looked at greener alternatives, aiming for safer catalysts with similar punch but less risk. My conversations with young chemists show a drive to keep learning—knowing that a formula like ethylaluminum dichloride isn’t the end of discovery, but a stage in a bigger journey.
Trust, Facts, and the Public Good
Ethylaluminum dichloride reminds me that chemical formulas only tell part of a story. Industries depend on compounds like C2H5AlCl2 for progress, but society expects stewardship. Recognizing the facts—how powerful, dangerous, and useful a formula can be—paves the way for making smarter, safer choices.
Is Ethylaluminum Dichloride hazardous to health or the environment?
Understanding Ethylaluminum Dichloride
Ethylaluminum dichloride doesn’t turn heads the way mercury or asbestos do, but its dangers are no less real. This colorless liquid shows up mostly in chemical plants, especially where plastics and rubbers get made. In labs and factories, people count on it for its strength as a catalyst—meaning it helps speed up reactions, sometimes in big batches. Handling this stuff comes with rules, goggles, and a healthy dose of respect.
Health Risks on the Job
I’ve walked through industrial sites where safety is drilled into staff from day one. Chemicals like ethylaluminum dichloride force everyone to pay attention. Splash a little on skin and burns appear fast. A whiff won’t just make you cough; this vapor can fry the inside of your nose and lungs. Reactions can get frighteningly severe, needing more than just a rinse with water. Stories from plant workers highlight the risk: one moment of carelessness, and someone’s hands or eyes might never be the same.
Accidents speak louder than any warning label. The U.S. Occupational Safety and Health Administration (OSHA) classifies ethylaluminum dichloride as hazardous, warning of everything from chemical burns to permanent lung damage. Hospitals nearby heavy chemical plants have policies shaped by accidents with this compound. In real terms, a drop in the wrong place—an open cut, or worse, near the eyes—can mean months of treatment. Even diluted, the threat remains.
Environmental Impact: Spills and Worries
Factories keep their tanks sealed tight because the environment takes a beating from spills. Ethylaluminum dichloride reacts fiercely with water, so a leak turns rain puddles toxic fast, releasing ethylene and hydrochloric acid as it breaks down. These byproducts don’t just vanish. They travel through air and water, causing danger for local plants and animals in ways that don’t get fixed overnight.
Case history from Europe and the U.S. shows that fish kills and scorched earth around old plants often trace back to poor handling of this chemical. Watchdogs like the Environmental Protection Agency (EPA) have set limits and demanded accountability, but slip-ups still happen. When regulators fine companies for illegal dumping, it’s often because of the cascading damage caused by chemicals that don’t just fade away. Farmers near chemical zones sometimes report stunted crops or sick livestock; sometimes it’s the water, sometimes just the soil itself.
Better Practices: Protecting People and Nature
I’ve seen change driven by hard lessons. Employee safety training saves lives, but it goes farther. The biggest gains come from simple engineering steps—like sealed systems, filtered air, and strict storage methods. Double-walled tanks and emergency neutralizers in the workspace can absorb spills before they escape. Modern plants now use remote sensors and automatic shutoffs to catch leaks early, which helps both workers and neighbors.
On the environmental side, companies can either cut corners or clean up. Upgrading wastewater treatment and developing alternatives less reactive than ethylaluminum dichloride will make a difference. Independent audits hold businesses to account, and public reporting forces transparency. As communities get louder about clean air and water, businesses start to realize their reputation depends on genuine change. Regulators, scientists, and citizens all keep each other honest—and every story of a spill or a saved worker proves the struggle is worth it.
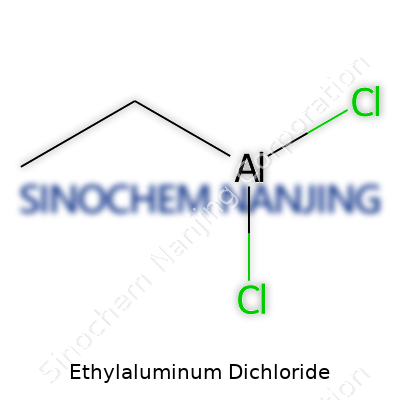
| Names | |
| Preferred IUPAC name | Ethyl(dichloro)alumane |
| Other names |
EADC Dichloroethyaluminium Aluminum, ethyldichloro- Ethyl aluminium dichloride |
| Pronunciation | /ˌiːθɪl.əˈluːmɪnəm daɪˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 563-43-9 |
| 3D model (JSmol) | ``` Ethylaluminum Dichloride|C[Al](Cl)Cl ``` |
| Beilstein Reference | 3539868 |
| ChEBI | CHEBI:38968 |
| ChEMBL | CHEMBL3400896 |
| ChemSpider | 23866011 |
| DrugBank | DB11348 |
| ECHA InfoCard | 100.012.414 |
| EC Number | 208-696-4 |
| Gmelin Reference | 6352 |
| KEGG | C07097 |
| MeSH | D000942 |
| PubChem CID | 8667 |
| RTECS number | AT4375000 |
| UNII | K7OL27H76A |
| UN number | UN1183 |
| CompTox Dashboard (EPA) | EPA CompTox Dashboard for Ethylaluminum Dichloride: **DTXSID4020710** |
| Properties | |
| Chemical formula | C2H5AlCl2 |
| Molar mass | 119.02 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | strong, pungent |
| Density | 1.2 g/cm3 |
| Solubility in water | Reacts violently |
| log P | -0.24 |
| Vapor pressure | 10 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 6.5 |
| Basicity (pKb) | -0.68 |
| Magnetic susceptibility (χ) | −72.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.433 |
| Viscosity | 1.58 mPa·s (25 °C) |
| Dipole moment | 2.53 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -434 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H225, H260, H314 |
| Precautionary statements | P210, P220, P222, P231+P232, P260, P261, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P335+P334, P337+P313, P363, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Autoignition temperature | 205 °C (401 °F; 478 K) |
| Lethal dose or concentration | LD50 oral rat 250 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 162 mg/kg |
| NIOSH | AL0175000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Ethylaluminum Dichloride: Not established |
| REL (Recommended) | 2 mg/m³ |
| IDLH (Immediate danger) | 2 ppm |
| Related compounds | |
| Related compounds |
Aluminum chloride Diethylaluminium chloride Trimethylaluminium Triethylaluminium Dimethylaluminium chloride |

