Ethyl Vanillin: An In-Depth Commentary
Historical Development
Ethyl vanillin didn’t just appear out of thin air. As a teenager, I remember my uncle, a food chemist, sharing stories of how flavoring agents drew interest well beyond bakers. Vanilla prices surged once global demand outpaced supply. Chemists got curious about alternatives back in the late nineteenth century, pursuing not only plant-derived but also lab-made vanilla options. Officially synthesized around 1876, ethyl vanillin stood apart because its flavor punches harder than vanillin itself. France played a central role during the formative years, driven by the growing need for reliable, strong aroma enhancers. Over the decades, its adoption spread thanks to an impressive resilience to heat and a pleasant, consistent taste. This compound didn’t just help manufacturers—it gave small bakeries and candy makers a fighting chance to offer enticing treats even in lean vanilla seasons.
Product Overview
Ethyl vanillin looks like a white or faintly yellow crystalline powder. In a bakery, you’ll notice how just a pinch turns a chocolate icing from good to genuinely mouthwatering. Its aroma isn’t shy: one whiff brings to mind confectioner’s shops or vanilla-rich desserts, yet its scent leans toward something cleaner and crisper than natural vanilla. The molecular structure closely mimics vanillin, but the distinctive ethoxy group boosts its sweetness and intensity. These days, you spot ethyl vanillin in everything from colas to perfumes, highlighting its versatility. Any cook, from commercial kitchen staff to hobby home bakers, can tell you how much less you need compared to vanilla extract for the same punch.
Physical & Chemical Properties
It all boils down to the numbers and tactile qualities. Ethyl vanillin’s melting point hovers around 76–78°C, which matters when you dial in a recipe that needs precise thermal control. Solubility in alcohol and slightly less in water lets it dissolve easily for beverage or cosmetic work. This powder keeps stable under regular storage conditions—just keep it dry and away from sunlight. The compound’s molecular formula, C9H10O3, gives a molar mass of roughly 166.17 g/mol. Ethyl vanillin resists oxidative degradation and maintains its signature scent long-term, which sets it apart from natural extracts that fade over time.
Technical Specifications & Labeling
Expect to see purity standards in the 99% range for food and pharma grades, often established by organizations like the Food Chemicals Codex and European Pharmacopoeia. Packaging labels must declare purity, batch numbers, manufacture and expiry dates, and country of origin. Regulatory agencies demand proper allergen warnings, especially since some ethyl vanillin batches derive from materials like guaiacol, possibly triggering reactions. For bulk sales, safety datasheets travel with shipments, and every container must show hazard classification, even if this compound rarely poses a risk under normal use. Quality audits from buyers examine spectral analysis and check for heavy metals, which no one wants anywhere near food.
Preparation Method
From my talks with process engineers, commercial producers favor synthesizing ethyl vanillin through ethylation of vanillin. The classic route starts with guaiacol and glyoxylic acid to make vanillin, hitting the right balance of purity and cost. Next step swaps out the methyl group for ethoxy, using ethylating agents under controlled temperatures and pressure. Workers have to monitor reaction pH, temperature spikes, and agitation levels every hour, with strict sampling during the critical synthesis windows. Once the main reaction winds down, purification follows—typically recrystallization from solvents like methanol and water. In smaller labs, some chemists play with direct oxidation of ethyl ethers of isoeugenol, but these processes are rarely cost-effective for scale production.
Chemical Reactions & Modifications
Ethyl vanillin holds promise for researchers looking to tweak flavor or fragrance. As a phenolic aldehyde with an ethoxy tail, it reacts predictably with strong acids and bases. Chemists often form Schiff bases, reduce the aldehyde function, or tinker with the aromatic ring for specialized ingredients. I once watched as a lab dissolved ethyl vanillin with sodium borohydride, converting the aldehyde to an alcohol for a less-volatile, sweeter note. Some manufacturers experiment with enzymatic modification to craft novel aroma profiles for the perfume industry. If you’ve handled this compound, you know it withstands moderate heat without breaking down, letting flavorists play with it in baked goods, syrups, or even essential oils.
Synonyms & Product Names
Ethyl vanillin goes by several names: 3-ethoxy-4-hydroxybenzaldehyde, bourbonal, and sometimes just “vanillin, ethyl” in regulatory circles. The codex Alimentarius or US FDA ingredient lists may display all these aliases, so anyone with food allergies knows precisely what they’re eating. Major brands—Sigma-Aldrich, TCI, Merck—usually label product codes and ensure CAS number 121-32-4 accompanies every bottle. These names help buyers avoid confusion and make it easier to comply with local or international regulations.
Safety & Operational Standards
Safety remains crucial in every step, from handling to end-use. Ethyl vanillin rarely irritates skin or eyes at the concentrations found in food and perfume, yet workers in factories wear gloves and eye protection. In industrial kitchens or labs, proper ventilation limits inhalation risk, even if acute toxicity is minimal. Regulatory audits demand trials to verify that cross-contamination doesn't occur with allergens or hazardous chemicals. Producers must maintain material safety data sheets and offer employee training—even seasoned handlers sometimes skip steps, which I saw back in my days at a flavor house, only for a missed glove to ruin a full batch. The European Food Safety Authority and US FDA regularly evaluate exposure limits. Currently, levels up to 7 mg per kg of food get approved for human consumption, balancing safety with flavor needs.
Application Area
Ethyl vanillin shows up in more products than most people realize. Walk through a supermarket and spot it in chocolates, ice cream, baked sweets, chewing gums, and even specialty teas. The food industry leans on ethyl vanillin when seeking a deeper vanilla note without spending a fortune on scarce vanilla beans. Outside food, perfumers love its strength and radiant, powdery scent—think luxury cosmetics or high-end candles. Pharmaceutical developers use ethyl vanillin to mask bitter medicine flavors, making tablets easier for kids and adults alike. Some niche industrial users incorporate it in specialty inks and tobacco blends, banking on the way it harmonizes sharp base notes. Specialty coffee roasters sometimes boost blends with a tiny dose, creating a unique edge in house blends that regular vanillin can’t deliver.
Research & Development
Research into ethyl vanillin evolves every year. Food scientists examine its flavor release curves across different temperatures and pH levels, hoping to better synchronize it with novel sugar substitutes. I’ve attended conferences where newer encapsulation techniques received attention, such as using microcapsules to control flavor bursts in chewing gum. The pharmaceutical sector pushes for higher purity and fewer residual solvents, while biotech firms explore enzymes to synthesize ethyl vanillin from lignin waste, chasing both sustainability and cost reduction. Developers in fragrance prioritize finding compatible fixatives and delivery systems to stretch ethyl vanillin’s profile without causing off-notes or volatility losses. University labs partner with multinational food and fragrance conglomerates, looking for smarter ways to reduce carbon footprint and improve process yields.
Toxicity Research
Anyone in regulatory or QA roles keeps a close eye on toxicity data. Decades of lab studies reveal low acute toxicity for both humans and most animals when consumed within regulatory limits. Long-term feeding studies in rats, for instance, did not indicate carcinogenic effects at approved intake levels. The most common adverse response, mild skin or eye irritation, arises with direct or undiluted contact. The American Chemical Society’s published reports show that, unlike artificial flavors developed in the early 20th century, ethyl vanillin rarely triggers allergic or mutagenic effects at everyday concentrations. Ongoing research explores metabolization, sensitive populations, and combined effects with other common flavoring agents. Regulatory agencies utilize these findings to continually refine the guidelines, aiming to stay ahead of new evidence and changing consumption patterns.
Future Prospects
The next decade feels ripe for ethyl vanillin’s growth, driven by both demand for plant-based foods and ongoing vanilla bean shortages. Synthetic biology startups, already tinkering with yeast fermentation as a novel production route, may cut costs and environmental impacts in tandem. Food security experts push for stable, scalable alternatives to natural vanilla to guard against crop failures or climate-induced shortages. Analysts expect emerging markets, especially in Asia and South America, to spark new uses for ethyl vanillin in dairy and beverage categories. Sustainability will likely steer the next phase, as large producers experiment with bio-based raw materials to deflect criticism facing petrochemical synthesis. As consumer watchdogs scrutinize ‘natural’ vs. ‘nature-identical’ labeling, transparency in sourcing and production processes will matter more than ever, with traceability and audit trails becoming a necessary part of doing ethical business.
What is Ethyl Vanillin and how is it different from regular vanillin?
Why Vanilla Flavor Matters
The scent of vanilla sneaks into childhood memories—cookies in the oven, ice cream cones on summer afternoons, and boxes of supermarket treats. Many of those flavors don’t come from vanilla beans grown in Madagascar or Mexico. Instead, they’re built from molecules created in labs. Two names show up on ingredient lists: vanillin and ethyl vanillin. They look similar on the surface, but the story behind each one leads straight into questions of taste, allergies, and food transparency.
Where They Come From
Vanillin comes from vanilla beans, but most vanillin people taste comes from wood pulp or chemicals. Natural vanilla is expensive, hard to grow, and gets harvested by hand. I once spoke with a pastry chef who explained that real vanilla extract sits on a shelf, saved for special cakes, while synthetic vanillin does the daily load of flavoring breakfast cereal or soft drinks. Most folks barely notice because vanillin’s sharp, sweet scent mimics actual vanilla reasonably closely.
Ethyl vanillin steps things up. Chemists swapped part of the vanillin molecule, making a cousin that shows up about three times stronger on the human nose. This extra strength lets food makers use less of it, keeping costs low and flavors bold even in processed foods where subtler aromas would disappear in the mix.
How Do They Taste?
Drop ethyl vanillin onto your tongue, it punches through with a sweeter, cleaner vanilla blast. The difference becomes clearer in foods with heavy processing—boxed brownies, mass-produced chocolate. Bakers and candy makers lean on ethyl vanillin for hitting flavor targets that regular vanillin sometimes misses. I remember testing a batch of cookies with both: one tray wafted warm, mellow vanilla, the other smelled more like a vanilla milkshake from a soda fountain.
Some people, especially those sensitive to smells or tastes, can spot a hint of artificial edge with ethyl vanillin. Others find it pleasant and even crave it because it’s tied to childhood favorites. The reality is nobody’s taste exactly matches anyone else’s, but the food industry adapts recipes to what gets the biggest smile in a taste test.
Safety and Transparency
Both vanillin and ethyl vanillin carry safety checks from food safety authorities including the FDA. Food-grade ethyl vanillin comes from chemical synthesis, usually using raw materials derived from petrochemicals or lignin (the stuff left over from making paper). People with allergies to vanilla or who react to certain synthetic additives sometimes need to read ingredient lists closely. Most sensitivities come from the natural vanilla bean or other ingredients in the mix, not from vanillin or ethyl vanillin itself, but labeling headaches happen often.
Ethyl vanillin sometimes creates confusion because it gets listed as “artificial flavoring” or buried under broader terms like “flavor.” That leaves customers wondering exactly what they’re eating, especially if they’re trying to avoid unnecessary additives. After reading countless labels at the grocery store, I find myself reaching for products that call out “vanilla extract” if I want something closer to the flavor of the actual bean, but I don’t hesitate when reaching for a favorite boxed brownie every now and then.
What Could Change?
Transparency around flavor ingredients still lags behind demand. More people care about what goes into processed foods. Simple labeling, open communication from food makers, and more education about the actual differences between vanillin and ethyl vanillin can make shopping easier. Food producers could take cues from small bakeries—let customers know how a product gets its flavor, whether from beans, synthetics, or both, so everyone can make informed choices at the checkout.
Is Ethyl Vanillin safe for consumption?
Tasting the Flavor of Vanilla—But Not Quite
Vanilla flavor gets a boost from ethyl vanillin in everything from chocolate bars to bakery treats. This chemical cousin to natural vanillin packs a stronger punch and brings a familiar taste to food, often at a fraction of the cost of real vanilla beans. Food manufacturers love it because it keeps flavors consistent and vibrant in products that line supermarket shelves. If you’ve eaten sweets recently, chances are you’ve had a taste of ethyl vanillin.
So, Is It Safe for People to Eat?
Looking into food safety, I learned that the U.S. Food and Drug Administration (FDA) lists ethyl vanillin as Generally Recognized As Safe (GRAS). This decision isn’t made lightly. Before a food additive gets GRAS status, it goes under review by scientific experts. In Europe, the European Food Safety Authority (EFSA) also gives ethyl vanillin a green light for use in foods at specified levels. The Joint FAO/WHO Expert Committee on Food Additives reviewed ethyl vanillin’s effects as well, setting clear limits so it doesn’t show up in worrying amounts in the average person’s diet.
No Perfectly Natural—But Not Toxic
Ethyl vanillin is synthetic, which sometimes makes people pause. In my own kitchen, I lean toward foods with simple, recognizable ingredients, but that isn’t always possible. Using the available research helps cut through worry. Most studies focus on toxicity, long-term effects, and potential allergic reactions. Feeding tests on rats and mice, often for weeks or months, try to catch problems like organ damage or cancer risk. According to data gathered over the years, ethyl vanillin doesn’t build up in the body and passes out quickly in urine. At the low doses used in foods, even for sensitive groups like children or older adults, scientists don’t see health problems tied to this flavoring.
What About People With Allergies or Sensitivities?
Some people wonder about allergy risks. True allergies to ethyl vanillin show up rarely in medical literature. Skin irritation might happen if it touches the body at high concentrations, but not through eating everyday foods. That said, everyone reacts a bit differently, and people with multiple chemical sensitivities might feel uneasy after consuming many processed foods—even if science can’t always explain why. Anecdotally, I have seen people mention headaches or stomach upset online after eating lots of artificially-flavored products; these stories deserve compassion, but don’t show up strongly in published research.
Staying Informed—And Keeping Control
The big concern with flavor additives often comes from eating them in huge amounts or combined with many other chemicals over a long time. Experts recommend keeping an eye on ingredient lists and aiming for a balanced diet with plenty of fresh, whole foods. If a food tastes too perfect or artificial, it likely relies on synthetic flavors to keep taste consistent batch by batch. Over time, encouraging clear labeling helps people spot ethyl vanillin and make informed decisions.
How to Approach Solutions
A good step is to support research that keeps checking for long-term or subtle effects of food additives. Food regulators could push manufacturers to use the minimum amount needed for flavor—less is usually better—and to be transparent about what’s in a product. Cooking at home more often gives control over flavorings and cuts down on surprise ingredients. Staying thoughtful about diet and following developments from trusted science and health agencies helps manage the inevitable presence of these additives in modern life.
What are the common uses of Ethyl Vanillin in food and beverages?
What Makes Ethyl Vanillin a Go-To Flavor Choice
Ethyl vanillin brings a rich, sweet scent that stands out in countless products. It tastes stronger and bolder than regular vanillin, the compound from vanilla beans. This not only makes it popular with food manufacturers but also puts it at the center of flavor science. Have you ever smelled a chocolate bar or poured syrup over your breakfast and picked up a deep vanilla note? Ethyl vanillin probably helped build that experience.
Sweet Treats and Everyday Desserts
Candy, chocolate, and baked goods are unmistakable showcases for ethyl vanillin. Bakeries and chocolatiers reach for it because it gives sweets a punch without the cost of real vanilla bean extract. It can power up the flavor in fudge, ice cream, and cookies. Modern candy manufacturers use it to deliver a reliable, bold vanilla flavor batch after batch, and because it’s more potent than traditional vanillin, smaller amounts go further.
This compound plays a big role in how "vanilla" ice cream, frosting, and cakes taste in large commercial batches. You get a consistent product at a price that makes sense for both producer and customer. I’ve seen it show up in ingredient lists for holiday treats and baked goods all year round in the supermarket.
Chocolate Gets a Makeover
Chocolate makers rely on ethyl vanillin to mask bitter notes from cocoa. A little bit goes a long way to round off the hard edges of mass-produced chocolate. As someone who’s tried both single-origin chocolate and off-the-shelf candy, I notice the flavor in the latter often feels a bit more “vanilla-heavy.” That’s no accident—ethyl vanillin stays stable at high temperatures during chocolate production, so the taste comes through even after melting, cooling, and shelf life.
Expanding Into Beverages
Beyond snacks, ethyl vanillin finds a home in flavored drinks. Think of pre-mixed milkshakes, energy drinks, or even spiced liquors. The warm vanilla character works well in cola and cream soda. Mixologists and beverage companies use it to add depth and comfort to drinks that need a signature twist. It comes through in non-alcoholic and alcoholic beverages alike, giving a familiar note that stands out on crowded shelves.
Everyday Prepared Foods
Flavored breakfast cereals, instant oatmeal, and even some processed dairy products pull flavor and aroma together with a boost from ethyl vanillin. It covers off-notes in preserved foods and pleasantly rounds out other flavors. On early mornings or busy days, many folks eat these foods without thinking about why they taste so good—ethyl vanillin handles a lot of behind-the-scenes work.
Health, Safety, and Transparency
Ethyl vanillin stands up to scrutiny. Food safety agencies—including the FDA and the European Food Safety Authority—have cleared it for use in set limits, based on years of safety research. Some shoppers prefer whole or "natural" ingredients, so brands now call out artificial flavors more clearly. Knowing what’s in your food and understanding why these ingredients are used helps everyone make better choices at the store.
Ethyl vanillin won’t take the place of real vanilla for every recipe, but it gives manufacturers room to get creative, reduce costs, and meet the appetite for bold, comforting flavors on a big scale.
What is the recommended dosage of Ethyl Vanillin in recipes?
Understanding Ethyl Vanillin’s Punch
Ethyl vanillin isn’t just another flavoring tucked away in the baking aisle. It packs a much bigger punch than regular vanillin—almost three to four times as strong by weight. Its creamy, deep vanilla scent falls somewhere between classic vanilla and a light floral note. This compound found its place in bakeries, chocolates, drinks, and ice cream, promising a hit of sweet warmth to anything it touches.
How Much is Too Much?
Walking into recipe development, many folks see ethyl vanillin as a straight swap for vanilla extract. That quickly leads to disaster. Food scientists agree that a pinch too much can turn a gorgeous batch of cookies into something medicinal and sharp. Years of playing with different flavors in home kitchens and on the job showed me: less is almost always more with this stuff.
Most culinary experts and food safety regulators, including groups like the FDA and European Food Safety Authority, settle on about 5 to 30 milligrams of ethyl vanillin per kilogram of finished product as a reasonable range for baked goods, candies, or ice cream. Home bakers can translate this to roughly one-fifth to one-half teaspoon per entire standard cake recipe, sometimes even less. Trying to match the vanilla taste in a classic chocolate chip cookie? A quarter teaspoon will easily do—use more and you risk an overpowering chemical aftertaste.
Why Dosing Matters: A Practical View
Flavors become personal memories. Maybe that vanilla ice cream after a ball game, or a thick slice of birthday cake—too much ethyl vanillin, and you lose those layers under something harsh. Overdosing prints a single, loud note on your tastebuds, one that lingers long after the treat is gone.
From a health and safety angle, ethyl vanillin carries a solid track record when used in moderation. Large manufacturing kitchens rely on digital scales, sometimes even measuring to one-tenth of a gram, to keep consistency and avoid going over the mark. I’ve watched seasoned bakers laugh about their ‘vanillin disasters’ from their early days, learning through experience that following strict weights and not just eyeing the teaspoon can make or break a whole batch of fudge or frosting.
Solutions: How to Get It Right
Approach ethyl vanillin with the same respect you give salt or yeast. Start on the low side: add the minimum, taste, then build up only if you need a touch more. For hobby bakers, a cheaper milligram scale keeps hand slips in check. Professional kitchens already see the value in precision scales and solid recipes vetted through trial and error.
Many flavor houses and recipe books share clear dosage tables—don’t toss out those charts. They’re built from both lab research and decades of kitchen experience. If you ever find the taste too flat, try mixing ethyl vanillin with real vanilla extract for a rounder, more familiar profile. Pure ethyl vanillin rarely needs to be pushed to the upper limits of any dosage chart.
Knowing the Limits Builds Better Food
Learning the recommended dose of ethyl vanillin doesn’t just improve taste—it saves ingredients, time, and headaches. Once you’ve dialed in that ratio, the finished product truly lets the vanilla notes shine, without drifting into anything synthetic or bitter. Every time, the smell from the oven brings a different kind of comfort, knowing you got the balance right.
Does Ethyl Vanillin have any allergens or side effects?
What Exactly is Ethyl Vanillin?
Ethyl vanillin gives food and fragrances a powerful vanilla flavor, even stronger than its better-known cousin, regular vanillin. This synthetic flavoring gets used in ice cream, chocolate, baked goods, and perfumes. The price stays lower than natural vanilla, which makes it a common choice for manufacturers.
Allergens in the Mix?
Many folks feel a little nervous seeing unfamiliar chemicals in their groceries and cosmetics. Ethyl vanillin comes from a scientific process, not a vanilla bean, so questions about safety pop up often. Most health agencies, including the U.S. Food and Drug Administration (FDA) and European Food Safety Authority (EFSA), have found no evidence that ethyl vanillin causes allergies in healthy people. This matches up with my own experience in the food industry where very few reports link ethyl vanillin to allergic reactions. Allergy databases rarely mention it, especially compared to common triggers like milk, nuts, or gluten.
Still, no one can promise a risk-free experience for every person. Anyone who has ever had a mysterious rash or sneezing spell from a food knows that even the most harmless-sounding additives can surprise us. Some sensitive people might react to almost anything. True ethyl vanillin allergies look extremely rare, but absolute certainty is tough to guarantee.
Are There Any Side Effects?
Regulatory safety reviews focus on how much of an ingredient shows up in food. Ethyl vanillin typically appears in small amounts. Research so far does not show big risks at these levels. The Joint FAO/WHO Expert Committee on Food Additives calls it safe in small doses. I’ve tasted hundreds of products over the years with ethyl vanillin listed on the back, and after a stomachache or two, it’s never landed me or anyone I know in the ER.
Larger doses create a different story. Studies in rats show that massive amounts—far beyond anything found in real food—can lead to irritation or mild toxicity. The idea here: more is not better. No flavoring belongs by the spoonful. If ethyl vanillin were substituted for natural vanilla in every meal and snack, the cumulative exposure would climb. Choosing a variety of foods always helps keep any single ingredient in check.
Watching for Contamination
Beyond allergies and direct effects, another angle deserves attention. Poor manufacturing or sloppy handling might introduce trace impurities or contaminants. Most reputable manufacturers follow good practices, but the rise in global trade means not all sources can be vetted easily. Consumers with chemical sensitivities sometimes react to what rides along with the ethyl vanillin, not the molecule itself.
What Can People Do?
Anyone worried about new reactions would do well to keep an eye on food and cosmetic labels. For folks with a history of allergies or sensitive skin, jotting down suspected triggers after a reaction helps narrow things down. Medical specialists can run tests if a pattern arises. Asking brands exactly what’s in their flavor blends—and where ingredients come from—can keep things transparent.
On the bigger picture, pushing for tighter labeling helps everyone. Ethyl vanillin might not trigger allergies for most, but fuller disclosure means people can make informed choices. That control goes a long way toward peace of mind.
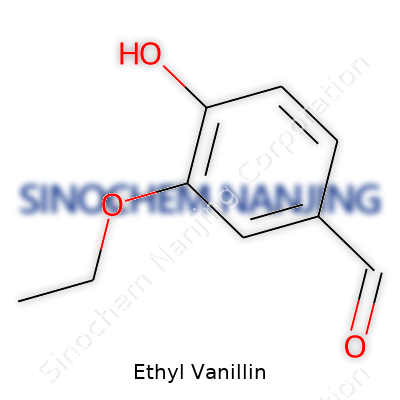
| Names | |
| Preferred IUPAC name | 3-ethoxy-4-hydroxybenzaldehyde |
| Other names |
Bourbonal Ethylprotocatechuic aldehyde 3-Ethoxy-4-hydroxybenzaldehyde Evanillin |
| Pronunciation | /ˌiːθɪl væˈnɪlɪn/ |
| Identifiers | |
| CAS Number | 121-32-4 |
| Beilstein Reference | 3599536 |
| ChEBI | CHEBI:42244 |
| ChEMBL | CHEMBL15910 |
| ChemSpider | 5795 |
| DrugBank | DB11268 |
| ECHA InfoCard | 100.012.838 |
| EC Number | 203-300-1 |
| Gmelin Reference | 736615 |
| KEGG | C16449 |
| MeSH | D014727 |
| PubChem CID | 8467 |
| RTECS number | GF4800000 |
| UNII | 7OHS5J9E5S |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C9H10O3 |
| Molar mass | 166.17 g/mol |
| Appearance | White to pale yellow crystalline powder |
| Odor | vanilla; creamy; sweet |
| Density | 1.185 g/cm³ |
| Solubility in water | Soluble in water (1 g/100 mL) |
| log P | 1.64 |
| Vapor pressure | 0.0013 hPa (25°C) |
| Acidity (pKa) | 7.40 |
| Basicity (pKb) | 13.57 |
| Magnetic susceptibility (χ) | -64.0e-6 cm³/mol |
| Refractive index (nD) | 1.553 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.74 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 252.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -537.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −4373 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | A07BC30 |
| Hazards | |
| Main hazards | May cause eye, skin, and respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | May cause an allergic skin reaction. Causes serious eye irritation. |
| NFPA 704 (fire diamond) | 2-1-0-W |
| Flash point | 143°C |
| Autoignition temperature | 500 °C (932 °F; 773 K) |
| Lethal dose or concentration | LD50 (oral, rat): 2,990 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1590 mg/kg (oral, rat) |
| NIOSH | GV0450000 |
| PEL (Permissible) | 10 mg/m³ |
| REL (Recommended) | 6.5 mg/kg |
| Related compounds | |
| Related compounds |
Vanillin Methyl vanillin Propyl vanillin 4-Hydroxybenzaldehyde Heliotropin |

