Ethyl Sulfuric Acid: Insight Into a Multifaceted Industrial Chemical
Historical Development
Chemists in the early 19th century often found themselves navigating uncharted territory. The need for new reagents and intermediates drove them to experiment with strong acids and alcohols. In that era, discoveries were rarely just academic — people scrambled to improve dye chemistry, medicines, and industrial processes. Ethyl sulfuric acid came about from these very experiments, mostly through the pursuit of more efficient ways to make ether and other organic products. Over time, it showed up in textbooks as a starting material and intermediate, especially after Williamson’s synthesis, which drew attention to the compound’s reactivity and highlighted its importance in organic synthesis. Its story crosses from dusty lecture halls to modern industrial labs, showing just how much progress humans can make when they keep asking questions.
Product Overview
Ethyl sulfuric acid serves as more than just a laboratory curiosity. Chemists see it pop up when converting ethanol to other useful compounds. Companies use it where selective sulfonation is a must. Years of chemical studies reveal it bridges certain gaps between simple alcohols and more robust sulfonates. For those with their hands in a beaker or behind a spectrometer, its needle-like odor and corrosive touch leave no doubt about its strong acid character. Handling such a substance demands respect and speaks to the daily triage scientists do: balancing risk, value, and progress.
Physical & Chemical Properties
Anyone who has encountered ethyl sulfuric acid remembers its clear, oily appearance. The liquid sits at room temperature with a density slightly greater than water, running somewhere around 1.2 g/cm³. Its boiling point and solubility in water hint at its strong ionic nature. It readily mixes with water, diving straight in and releasing heat. The acidity packs a punch, with a pKa similar to sulfuric acid — not something to get careless with, since even small spills burn skin and etch glass. This sharp reactivity forms the backbone for the compound’s usefulness in making ethers, esters, and sulfonic acids.
Technical Specifications & Labeling
Technical grade ethyl sulfuric acid arrives with all the caveats expected of a strong acid: >95% purity for industrial blends, a color index that leans clear to pale yellow depending on storage, and specific gravity specs taped right onto shipping containers. Hazard symbols show skulls and corrosion icons. The fine print spells out UN numbers and proper shipping names in bold letters, with a nod toward updated GHS standards. All those warnings remind even the most seasoned handler that this chemical doesn’t forgive mistakes.
Preparation Method
Chemists make ethyl sulfuric acid by mixing ethanol with concentrated sulfuric acid. The mix kicks off an exothermic reaction, splitting water from the alcohol and forming the protonated ester in situ. Later, they might distill or filter the result. Control matters — add acid to ethanol too quickly, and you get a boiling eruption or unwanted byproducts. Tinkering with temperature and concentration gives more yield, but I’ve learned that patience and a steady hand produce the cleanest product. Large plants scale up the same approach, using jacketed reactors and acid-resistant pumps.
Chemical Reactions & Modifications
Ethyl sulfuric acid holds a reputation for versatility. React it with bases, and you get ethyl sulfate salts — important for detergents and as a stepping stone to other chemicals. If you heat it with strong dehydrating agents, diethyl ether forms in a well-controlled reaction. Its structure makes it prone to hydrolysis, breaking back down to sulfuric acid and ethanol if water lingers. Side reactions dot the process, from charring to polymerization, so labs monitor closely. Modern research explores using the compound as a linker or modifying agent in greener syntheses, trying to pin down conditions that favor efficiency and safety.
Synonyms & Product Names
Across different catalogs or textbooks, ethyl sulfuric acid travels under several aliases: ethyl hydrogen sulfate, monoethyl sulfate, and sometimes even sulfuric acid, monoethyl ester. In industry, the name matches the intended use — some look for the acid itself, others for the sodium or potassium salt. The diversity in naming can trip up even experienced chemists, so checking the CAS number and structure proves essential before ordering or handling.
Safety & Operational Standards
Safety officers and lab managers hammer home that personal protective equipment makes all the difference with ethyl sulfuric acid. Direct contact leads to burns, and inhaling its vapors irritates eyes, throat, and lungs. Emergency eyewash stations and acid-resistant gloves help, but training and diligence offer the most reliable shield. Hazard communication rules now compel manufacturers to print handling, storage, and spill control instructions right on the barrel, cutting down on the likelihood of miscommunication. Environmental rules classify it as hazardous waste — disposal routes filter through neutralization and water treatment before reaching municipal systems, reflecting the push for more sustainable chemical practices.
Application Area
Industrial chemists lean on ethyl sulfuric acid as a reagent for alkylation, esterification, and sulfonation. The textile and dye sector still uses its derivatives in color-fixing reactions. In the lab, it finds a place in organic synthesis, helping build more complex molecules by transferring ethyl or sulfate groups. Water treatment plants sometimes use its salts for scaling control, and pharmaceutical researchers keep it close at hand for intermediate preparation. The utility stretches across manufacturing, development, and environmental technology.
Research & Development
Research groups explore alternative production routes, sometimes employing biocatalysis or solvent-free systems to lessen the environmental footprint. New methods seek to squeeze more product from each batch, waste less, and avoid the hazards of decades past. Universities and startups collaborate to identify catalysts that trim costs or permit continuous-flow synthesis, reducing operator risk. Funding bodies now tilt more resources toward sustainable chemistry, giving students and professionals space to try out greener, safer, and more efficient routes.
Toxicity Research
Animal studies and in vitro tests reveal that ethyl sulfuric acid damages mucous membranes, skin, and internal organs if absorbed in large doses. Acute exposures bring coughing and skin blistering, and chronic exposure could spark lung or liver issues. Regulatory agencies set strict airborne exposure limits and require detailed risk management plans in workplaces. Toxicologists track metabolic breakdown and look for links to long-term health impacts, motivated by a duty to protect both workers and surrounding communities. Layers of safety legislation and environmental monitoring show science’s commitment to preventing harm before it starts.
Future Prospects
Looking ahead, the chemical world doesn’t sit still. Ethyl sulfuric acid’s role will likely keep shifting with advances in catalysis, green chemistry, and industrial scaling. Pushes for cleaner manufacturing demand safer, more efficient synthesis methods — investors and regulatory bodies both place a premium on minimizing waste and eliminating uncontrolled emissions. Smart automation and stricter standards promise lower risk but demand ongoing vigilance in operator training and plant design. Continued research, especially on toxicity and ecological effects, will drive changes in handling, application, and disposal practices. Sustainability concerns steer more chemists toward life-cycle analysis and cleaner infrastructure, keeping ethyl sulfuric acid relevant — but under ever sharper scrutiny.
What is Ethyl Sulfuric Acid used for?
What’s Ethyl Sulfuric Acid Doing Out There?
People don’t stumble onto ethyl sulfuric acid at the neighborhood hardware store. It’s a compound with a pretty small audience, but it’s got its place in some big processes. Built from ethanol and sulfuric acid, this chemical shows up mostly behind the scenes where folks rarely look—industrial labs, specialty factories, and chemical processes. The magic happens in reactions, not in household cleaners or personal care products.
Chemistry’s Building Block
From my experience around university labs, ethyl sulfuric acid isn’t a standalone hero. Chemists often turn to it when they need to introduce an ethyl group into another molecule. In the story of organic synthesis, the stuff serves almost like a transfer agent—taking a piece of itself and joining it to another, making brand new compounds. This gets important for drug development, plastics, and chemical research. Without these basic building blocks, we don’t get aspirin, nylon, or countless other products. So when people ask why this stuff matters, the answer is that it helps make things happen that we all benefit from, even if we never see the original bottle.
Spotlight on Industry Uses
Production plants lean on ethyl sulfuric acid in ways most people never imagine. It’s not about direct end-use—nobody’s brushing their teeth with the stuff or adding it to their food. Instead, the acid supports the groundwork for dyes, pesticides, and pharmaceuticals. I’ve seen how specialists use it to create ethyl esters, which eventually land in solvents, flavors, and fragrances. These processes sometimes look like magic tricks, with the acid carving out new chemical paths.
Health and Environmental Risks
Every chemical comes with concerns, and this one’s no exception. Spilling ethyl sulfuric acid or mishandling it creates immediate danger. The burn risk is real. In research work, extra care always goes into storage and protective gear. When it comes to larger scale operations, careful monitoring and emergency procedures make a difference. Companies that manage this substance under strict rules help keep workers safe and prevent wider contamination.
Responsible Use Matters
Regulators in Europe and the US keep their eyes on chemicals like ethyl sulfuric acid. Any company making, moving, or using it must follow rules that cover storage, handling, and disposal. There’s no room for shortcuts. From my experience in compliance training, everyone gets a reminder: one bad practice and the whole system feels it. That’s why people working with this acid need solid training and real-world safety resources—posters on the wall, spill kits in the corner, and management that takes things seriously.
Better Solutions on the Horizon
Alternative chemicals and green chemistry trends push makers to find safer substitutes, or at least tighten up the process. Researchers have tested greener reagents that do the same job without as much risk. Some labs work on capturing and recycling corrosive byproducts, which means less waste and fewer spills. These ideas make good sense, especially as regulations get stricter and the public cares more about what’s happening beyond the factory gates.
Wrapping Up
Ethyl sulfuric acid doesn’t belong on most people’s radar, but its fingerprints show up whenever certain chemicals shape our lives. Understanding how it gets used—and how to stay safe around it—helps everyone along the supply chain sleep a little better at night.
What are the safety precautions when handling Ethyl Sulfuric Acid?
Why Respect for Ethyl Sulfuric Acid Matters
Stepping into a lab or industrial plant, you quickly learn who treats chemicals with casual hands and who respects them. Experience taught me the difference comes down to consequences. Ethyl sulfuric acid can strip the finish right off a metal bench, and it will do the same to skin if given a chance. It takes a few seconds for a minor spill to become a major problem. No job or research project justifies skipping precautions with something so corrosive and reactive.
Personal Protective Gear
Back in college, I worked an internship where lab safety was more than a checklist; it could make or break your day. The first rule for handling ethyl sulfuric acid: suit up. Goggles keep splashes away from eyes. I once watched a student grab regular glasses—two weeks later, he wore safety goggles everywhere. Nitrile gloves resist most acids, and a thick lab coat shields arms and torso. Sturdy closed shoes never seem stylish until you drop a beaker. An acid-resistant apron gives extra backup, especially when pouring or transferring larger amounts.
Ventilation and Storage
A fume hood is a friend you want on your side. Ethyl sulfuric acid gives off vapors that can sting your nose and throat or send you outside gasping for air. I always check airflow with a tissue before any transfer—one quick test, but it gives peace of mind. Storage matters just as much. The stuff eats through containers if left unchecked. Only glass or compatible plastics work safely for longer periods. Every storeroom worth its salt keeps acids away from bases or organics, since an accident can spiral fast. Segregated shelves and labels help keep everyone honest, especially on late shifts.
Emergency Readiness
Accidents don’t give warnings. The best-run labs I know keep eyewash stations cleared and checked. I start every shift by making sure I know how fast I could reach the safety shower. Vinegar neutralizes some spills, but with volatile acids, large amounts of water and neutralizing powders stay ready. Experienced crews run spill drills so no one stands around guessing. Quick action means rinsing eyes for 15 minutes, not a few seconds. Every minute counts.
Handling and Disposal Practices
Transferring ethyl sulfuric acid asks for both steady hands and sober attention. Slow pouring, small volumes, and keeping containers closed when not in use protects everyone. I label every container even when it feels redundant—one mistake could send someone to the hospital. For disposal, never dump leftovers down the drain. Local rules require neutralizing with a base and collecting waste in marked containers. I keep spill kits nearby, not buried in a closet, to take care of drips and splashes on the spot.
Respect Earned Through Experience
Years of careful work build habits that protect people and property. Looking out for new coworkers, calling out shortcuts, making the right gear available, and trusting your senses—all of it matters. Ethyl sulfuric acid rewards respect with trouble-free use and punishes lapses. Safety grows from knowledge but runs on habits.
What is the chemical formula of Ethyl Sulfuric Acid?
Decoding the Structure
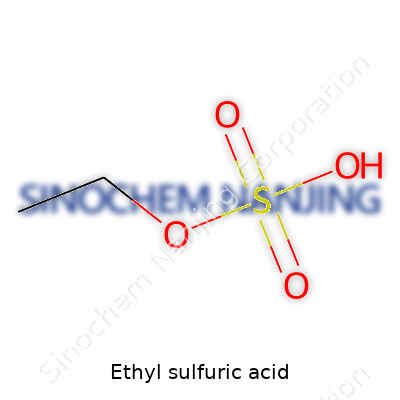
Ethyl sulfuric acid holds the chemical formula C2H5HSO4 or, less commonly, C2H6SO4. It consists of an ethyl group attached to a sulfuric acid molecule, forming a compound often described as the half-ester of sulfuric acid. You could think of it as sulfuric acid where one hydrogen atom has been replaced with an ethyl group (C2H5). Its creation demonstrates the fascinating ways chemical building blocks come together. In high school, I learned to appreciate formulas by visualizing how atoms swap, link, or anchor molecules to create new materials—this approach stuck with me as one of the more memorable parts of chemistry class.
Ethyl sulfuric acid often gets mistaken for similar-sounding compounds. Ethanol mixed with sulfuric acid, for example, delivers something quite different. Chemical accuracy matters: slip-ups in formulas can trigger confusion or even danger, especially in labs and industrial settings. It’s a reminder of chemistry’s precision, where even a missing hydrogen atom turns a molecule’s properties on its head.
Importance in Research and Industry
While ethyl sulfuric acid doesn’t show up in everyday products, its role in the chemical industry still matters. This compound often comes up as an intermediate in organic synthesis, especially in processes involving the creation of ethyl ethers and other organosulfur compounds. Years ago, working in an organic chemistry lab, we handled related esters. Proper labeling and formula knowledge meant the difference between a successful experiment and a hazardous mess. Chemical accidents, even at the smallest scale, tend to leave a lasting impression.
Health and safety agencies, such as the U.S. Occupational Safety and Health Administration (OSHA), highlight the risks pyrotechnic materials or unsuspecting mixtures can pose if precise formulas aren’t respected. Ethyl sulfuric acid, being corrosive and reactive, demands skilled handling. Studies published in journals like the Journal of Chemical Education stress the value of clear, correct structural documentation. For professionals, keeping the formula straight keeps potentially harmful errors at bay.
Addressing Knowledge Gaps
Many chemistry students and even some professionals confuse ethyl sulfuric acid with ethyl sulfate (its conjugate base). Getting it wrong can lead to unintended results, especially in analytical chemistry or pharmaceutical fields. Education rooted in hands-on activities often helps anchor concepts. As a student, building molecules out of plastic models gave me a concrete sense of how structures affect formulas and outcomes.
Academic institutions could lean more into visual learning, offering workshops with molecular construction kits and digital modeling tools. Seminars led by veteran chemists bridge textbook learning with lived experience, which tends to stick better than rote memorization. Outreach programs and online webinars open channels for sharing this knowledge further, especially for communities that lack access to advanced laboratory facilities.
A Path Toward Safer Practice
Clear communication means fewer misunderstandings and safer workplaces. Safety data sheets, real-time training, and accessible references help prevent slips with reactive compounds like ethyl sulfuric acid. Creating a culture where asking questions isn’t just accepted but encouraged can make a world of difference. Having spent time in several different research environments, I’ve seen firsthand how a willingness to double-check and cross-reference goes a long way toward both innovation and safety.
How should Ethyl Sulfuric Acid be stored?
Understanding the Hazards
Anyone who has worked in a lab or chemical plant knows EtHSO4 isn’t something to take lightly. It’s corrosive, reacts strongly with water, and its fumes can sting your throat. Inhaling or skin contact can cause immediate harm. Even someone with plenty of experience can get tripped up if they lose focus for a moment. The risks highlight the need for real caution in how this material gets stored.
Choosing Containers and Storage Conditions
Only certain materials can stand up to the acid’s aggressive nature. I’ve seen polyethylene and high-density polypropylene containers hold up well over years of use. Metal, glass, and cheap plastics tend to corrode or crack. Leaks don’t just waste product—they turn safe work into damage control. A double-layer container, with a sealed lid, cuts the risk of accidents even if the outer shell fails. You want containers marked clearly, with hazard labels that jump out—no scribbled marker or faded stickers. Colleagues sometimes ignore this part, but a clear label means nobody makes a fatal mistake on a busy morning.
Temperature and Humidity
I’ve noticed EtHSO4 stays most stable at cool, dry temperatures, away from direct sunlight. A climate-controlled storeroom—think 15–25°C and about 40% humidity—makes a real difference. One summer, a colleague’s shipment sat near a loading dock, exposed to heat and humidity. By the time they moved it, the plastic softened and the label came off. Even small lapses like that build up over time.
Segregation from Incompatible Materials
Some lessons stick with you forever. Early on, I saw storage rooms where acids and bases were lined up side by side. All it takes is a toppled bottle or a small spill for things to get out of hand. EtHSO4 mixed with bases like sodium hydroxide or bleach triggers heat and potentially harmful fumes. Keeping acids locked away from organics, bases, or oxidizers keeps accidents from snowballing into bigger emergencies. Separate shelves, well-marked areas, and clear signage have proven their worth many times in practice.
Simple Safety Steps and Spill Response
Even strong storage methods won’t stop every mishap. I’ve always made sure spill kits sit near the storage site: neutralizing agents, proper gloves, goggles, and absorbent pads. In smaller labs, a dedicated spill tray below the containers keeps leaks from running out onto the floor. Ventilation matters as well; good airflow reduces the risk from fumes, especially if storage extends into shared spaces. Training matters more than pamphlets—employees remember a demonstration or drill long after paperwork gets filed away.
Regular Inspection and Documentation
Trust in the system shouldn’t mean skipping hands-on checks. I’ve found monthly inspections catch cracks, leaks, or missing labels early. Digital inventory logs, tied directly to inspections, stop forgotten stock from sitting until disaster hits. If something seems wrong—unexpected container weight, cloudy liquid, or damaged seals—the safest move is to flag it and act right away.
Building a Culture of Respect for Chemicals
You can see right away who takes chemical safety seriously. Neat, well-labeled shelves, clear walkways, a place for everything—signs that people treat these hazards with real respect. Experienced teams pass down habits: double-checking lids, reporting problems, never working alone during transfers. Storing ethyl sulfuric acid safely doesn’t mean just following rules. It means paying attention, learning from slip-ups, and supporting colleagues in doing things right the first time.
Is Ethyl Sulfuric Acid hazardous to health or the environment?
The Risk Behind the Compound
Ethyl sulfuric acid doesn’t ring bells for most people, but it crops up in all sorts of labs and chemical processes. It acts as an intermediate in making dyes, surfactants, and sometimes serves as a reagent in research. At a glance, a name like that signals a serious substance. The structure brings together a strong acid character with an ethyl group, adding volatility and possible reactivity.
Health Hazards: More Than Just a Harsh Acid
Brushing past the chemistry, daily contact with ethyl sulfuric acid spells trouble. Its corrosive nature burns skin and eyes almost instantly. In labs, this usually means thick gloves and tight goggles, not just for show, but as a critical layer between acid and tissue. Once, during my own graduate studies, a flask of diluted ethyl sulfuric acid tipped over near my workspace. Even from the diluted splash, my arm stung through my lab coat, and redness lingered for days. That gave a lasting lesson: in the world of corrosive chemicals, even one slip causes lasting damage.
Respiratory exposure introduces more hazards. Breathing in fumes can inflame the nose, throat, and lungs, leading to coughing or shortness of breath. Those working with volatile acids in poorly ventilated rooms may feel the sting with just a few mishandled drops. Chronic exposure remains largely unstudied, but nobody bets on long-term acid inhalation producing good results. The Centers for Disease Control and Prevention (CDC) lists effects from acids of similar strength, pointing to tissue corrosion, chemical burns, and potential long-term lung injury.
Accidental ingestion rarely occurs, but it transforms into a medical emergency if it does happen. Severe burns to the mouth, throat, and gut demand rapid medical attention, leaving no time for hesitation.
Environmental Impact: Acid Meets Ecosystem
Down the drain or in a landfill, strong acids like ethyl sulfuric acid don’t quietly disappear. Waterways and soil react to spills, especially since the acid rapidly splits apart and lowers the pH. Fish and other aquatic life don’t tolerate low pH, and even temporary acidification wipes out insect larvae and eggs. Rivers affected by acid spills may take years to recover, depending on the volume and dilution available downstream. The US Environmental Protection Agency (EPA) treats spills of strong acids as serious incidents, requiring cleanup teams and careful neutralization to avoid more damage.
Ethyl sulfuric acid interacts with other chemicals, sometimes forming irritating or even toxic fumes. Pouring it into the wrong waste container in a busy lab can send up clouds that drive everyone out. Municipal water treatment plants watch for pH drops from these sources, since acid entry disrupts water purification and harms infrastructure by corroding pipes.
Steps Toward Safer Handling and Disposal
Every workplace using ethyl sulfuric acid ought to teach handling protocols until they become second nature. Regular safety drills, clear material safety data sheets on hand, and emergency eye wash stations nearby go a long way. Chemical fume hoods matter just as much for small volumes as for bulk containers. Training new staff to recognize the smell and sting helps, but working culture makes the biggest difference. A lab that eats lunch at its benches takes risks nobody needs, whereas a shop with strict clean areas keeps everyone safer.
Disposal demands neutralization—splashing ethyl sulfuric acid down the sink as-is invites trouble. Neutralizing with baking soda or lime before disposal in controlled quantities prevents pH shocks to the environment. Anyone running a building with chemical drainage should schedule regular pH checks and train janitors and custodians to spot chemical footprints before they spread. Local environmental agencies hold regular hazardous waste drives, collecting chemicals that don’t belong in domestic or municipal systems.
Why It Matters Now
More people take up chemistry as a hobby, and small businesses delve into niche manufacturing every year. Without respect for these reactive acids, real accidents happen fast. Just as with strong bases or solvents, clear rules and ongoing watchfulness make the difference between a useful tool and a health hazard.
| Names | |
| Preferred IUPAC name | Ethyl hydrogen sulfate |
| Other names |
Ethyl hydrogen sulfate Sulfuric acid, ethyl ester Ethylsulfuric acid |
| Pronunciation | /ˈiːθɪl sʌlˈfjʊrɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 110-63-4 |
| Beilstein Reference | 1100442 |
| ChEBI | CHEBI:537124 |
| ChEMBL | CHEMBL1232070 |
| ChemSpider | 160444 |
| DrugBank | DB01828 |
| ECHA InfoCard | 100.030.113 |
| EC Number | 214-684-0 |
| Gmelin Reference | 889 |
| KEGG | C01597 |
| MeSH | D013391 |
| PubChem CID | 11106 |
| RTECS number | KL8400000 |
| UNII | T84070A6X5 |
| UN number | UN2796 |
| CompTox Dashboard (EPA) | DTXSID9015506 |
| Properties | |
| Chemical formula | C2H6SO4 |
| Molar mass | 126.16 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 1.28 g/cm3 |
| Solubility in water | soluble |
| log P | -1.34 |
| Vapor pressure | 0.06 mmHg (20°C) |
| Acidity (pKa) | 1.92 |
| Basicity (pKb) | 1.43 |
| Magnetic susceptibility (χ) | -4.6e-6 |
| Refractive index (nD) | 1.454 |
| Viscosity | 2.6 cP |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 212.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -837.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -858.7 kJ/mol |
| Hazards | |
| Main hazards | Corrosive, causes severe skin burns and eye damage, harmful if swallowed, inhaled, or absorbed through skin. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | Flash point: 110°C (230°F) |
| Lethal dose or concentration | LD50 (oral, rat): 1550 mg/kg |
| LD50 (median dose) | 0.5 g/kg |
| NIOSH | WJ8925000 |
| PEL (Permissible) | 1 mg/m³ |
| REL (Recommended) | Stainless Steel 316, PE, PP |
| Related compounds | |
| Related compounds |
Sulfuric acid Sodium ethyl sulfate Ethyl sulfate Dimethyl sulfate Diethyl sulfate |

