Methyl Nitrite: A Full Look at an Unassuming Chemical
Historical Development
Methyl nitrite first grabbed chemists’ interest back in the 19th century. Early records show it showed up in experimental labs during the rapid expansion of organic chemistry. Anton Mitscherlich explored its structure in the 1830s, eventually linking it to alcohols and nitrous acid. Over the next decades, as laboratories improved synthesis techniques, methyl nitrite provided a tool for understanding how nitroso and nitrite groups interact with organic molecules. It didn’t lead to grand discoveries overnight, but it taught researchers quite a bit about the behavior of esters and became a reference point for similar compounds.
Product Overview
Methyl nitrite, with the formula CH3ONO, appears as a colorless gas with a faint, fruity smell. It dissolves in water to a small degree but mixes better with most organic solvents. Industries and labs mainly use it as a reagent or an intermediate, never quite becoming a star product but always filling a useful niche. Over the years, its small presence in textbooks and research catalogs has kept it relevant, especially during studies focused on alkyl nitrites’ reactive properties.
Physical & Chemical Properties
Physically, methyl nitrite packs a volatile punch, boiling at around −12 ℃, and melting well below freezing. Its vapor is heavier than air, and it burns with a characteristic greenish flame. The molecule reacts rapidly with oxidizing chemicals and should never be around open flames. Water doesn’t break it down as quickly as bases do, but it travels swiftly through air as a vapor, making proper handling essential. Chemically, its bonds carry extra electron density on the nitrogen and oxygen atoms. This split charge helps explain its behavior in synthesis and why it participates so eagerly in both reduction and oxidation reactions.
Technical Specifications & Labeling
Pure methyl nitrite arrives as a compressed gas or within solution. Labels typically flag its flammability, toxicity, and need for vented storage. Routine purity standards hover above 98%, with gas cylinders or ampoules marked with clear hazard symbols. Shipping it between research sites falls under strict national guidelines; a shipping error with methyl nitrite could trigger a fire or a toxic exposure. Industry insiders recognize the bright blue or green warning labels and pay extra attention during weighing or dispensing.
Preparation Method
Most small-scale preparations rely on dropping sodium nitrite into cold methanol with an acid such as sulfuric or hydrochloric. The nitrosation kicks up bubbles of methyl nitrite gas, which need collection by chilled traps or displacement over saltwater. The process looks simple at first glance, but experience teaches that keeping everything cold makes all the difference. Too much heat and the yield drops—or the risks climb sharply. In my own experience with bench chemistry, well-insulated reaction vessels and slow addition of acid improve both safety and product quality. Larger setups swap in temperature-controlled reactors and automated venting, guided by well-calibrated sensors.
Chemical Reactions & Modifications
Researchers often turn to methyl nitrite for diazotization and nitrosation. Its nitroso group transfers easily, opening doors to synthesize dyes, pharmaceuticals, and specialty polymers. Sometimes, folks use it to create unstable intermediates that break down quickly or shape catalysts for oxidation reactions. It won’t replace bulkier alkyl nitrites but fills gaps where reactivity and volatility outrank sheer yield. Newer studies focus on swapping its methyl group for bulkier alkyls or adjusting conditions to steer product formation, reflecting how versatile a simple ester can become in an expert’s hands.
Synonyms & Product Names
Methyl nitrite shows up under many aliases. The most common names include “nitrous acid, methyl ester,” “methyl ester of nitrous acid,” and “methyl nitrosooxy.” European and American chemical catalogs might label it numerically—common registry numbers include CAS 624-91-9. Sometimes instructions call it just “MeONO.” Old synthesis journals may refer to it as “spirit of nitrous ether,” recalling early confusion with diethyl ether and related vapors. Clarity in naming isn’t just a paperwork exercise; mislabeling in chemical stores can have serious consequences, especially in busy research environments.
Safety & Operational Standards
Handling methyl nitrite without the right precautions can lead to disaster. Its vapors catch fire easily, so spark-proof tools, flame arrestors, and ventilated hoods are non-negotiable. Inhalation causes headache, dizziness, and promotes the formation of methemoglobin, meaning even a small leak may drop blood oxygen in exposed workers. Storage needs steady, low temperatures and gas-tight containers with clear emergency protocols for leaks. Training drives the message home: you don’t cut corners when working with volatile nitrites. Teams prepare for the worst—chemical-resistant gloves, face shields, and respiratory masks stay at hand in every prep room.
Application Area
Methyl nitrite has value in the fine chemical world, where it shows up in organic synthesis and chemical research. Most synthetic chemists learn about its role in producing certain diazonium salts or facilitating mild nitrosation. Though not as common in medicine or agriculture, it plays a niche role in theoretical studies on radical reactions and the modeling of atmospheric compounds. Some industrial labs use it to tweak reaction pathways where only a volatile, short-lived nitrite will do the job. While consumers never encounter it directly, its impact echoes in dyes, specialty reagents, and select polymer manufacturing lines.
Research & Development
Ongoing studies explore how methyl nitrite can fine-tune reactivity in organic synthesis, especially when looking for greener chemistry processes. Chemists experiment with analogues by adjusting the surrounding groups, then measure how small tweaks affect reaction selectivity. Some environmental scientists look at methyl nitrite’s fate in the upper atmosphere, since related nitrites accumulate in polluted air. As the chemical toolkit grows, folks reach for methyl nitrite to test out new catalysts or initiate more complex reaction cascades. I’ve seen scientists lean on its reliable nitroso transfers to probe stability boundaries in new materials.
Toxicity Research
More than one safety case study underlines methyl nitrite’s toxicity. Inhalation in poorly ventilated rooms knocks out coordination and slows cognitive skills—signs point to the chemical’s tendency to disrupt oxygen delivery in blood. Animal studies track its rapid absorption and swift breakdown, showing risks tied to cumulative exposure. It breaks down into formaldehyde and other reactive fragments, though at low levels indoor and environmental persistence stays short-lived. Nevertheless, regulatory bodies still flag it for strict workplace controls, keeping airborne levels well below threshold limits. Accident reports stress the need for clear emergency procedures, swift evacuation plans, and real-time gas monitors when using or transporting this compound.
Future Prospects
New types of catalysis and green synthesis invite fresh interest in methyl nitrite. Some teams explore its use in energy storage, carbon capture chemistry, and molecular imaging. Its volatility makes it tough to control at scale, but nanotechnology might offer new routes for safe packaging and timed release. There’s growing pressure for alternatives with fewer health risks, but methyl nitrite’s unique reactivity means it will stick around as a research staple for years to come. Its journey forward rests on tighter regulations, more robust equipment, and a collective push toward safer lab practices, so no promising research comes with preventable setbacks.
What is Methyl Nitrite used for?
Behind the Lab Door: Methyl Nitrite’s Unique Place
Every now and then, a chemical pops up in the news or on a safety sheet, and folks start to wonder what exactly it does. Methyl nitrite fits that bill. Plenty of people have never heard of it. This compound holds a unique spot in the world of chemistry, especially for those working in research labs and specialty production facilities.
Role in Research and Analysis
Researchers turn to methyl nitrite for specific reactions. The compound plays a role in making other chemicals, mostly through methylation and nitrosation reactions—a mouthful, but simply put, it helps add certain groups to molecules. Some drug discovery projects and studies on new materials count on it because it delivers reactions that would be hard to pull off with other substances. Sometimes, a scientist tries out methyl nitrite in a process looking for a better way to make active pharmaceutical ingredients or pesticides.
Applications in Organic Synthesis
The biggest draw for working chemists comes from methyl nitrite’s role as a reagent. It reacts predictably and can help introduce nitroso groups into organic molecules. That might sound technical, but picture it as building blocks—if you need to shape molecules in a certain way, you need the right kind of building block at the right time or the project falls apart. Take the field of pharmaceuticals: getting one step wrong sometimes means restarting the entire process. Chemists stick with methyl nitrite for its reliability in certain steps where no simple replacement exists.
Industrial Importance and Hazards
It’s true that methyl nitrite doesn’t appear in your average consumer product. Its main life plays out quietly in controlled settings. Safety is a serious concern. This compound gives off a distinct odor and can be toxic in high concentrations. Lab workers use strong ventilation, rigorous training, and protective gear to prevent accidents. Studies and experience show that exposure can cause lung and nasal passage irritation and headaches. Chemical manufacturing facilities design equipment specifically to limit leaks or spills, and staff work in small teams for easy monitoring during each step involving this chemical.
Misuse and Social Risks
Every so often, reports surface about recreational misuse. Like “poppers,” a group of chemicals sometimes abused for their short-term effects, methyl nitrite draws risky crowds. These cases aren’t common, but they lead to poisonings. Hospitals in Europe and North America have tracked incidents linked to inhaling this substance, highlighting just how quickly things can go wrong. News articles and poison control centers stress that misuse doesn’t just bring about a headache—overdose can result in life-threatening drops in blood pressure and respiratory trauma.
Better Paths Forward
Some folks argue that chemicals like this should be better regulated, with closer tracking even for lab use. Others point out that better education about the dangers of inhalant substances might head off tragedies well before they happen. What works best, based on public health research, looks like a mix: strong safety policies plus outreach to schools and at-risk groups. Strong labeling, public service campaigns, and industry partnerships help protect people outside the lab as well as workers inside it.
Is Methyl Nitrite safe to handle?
Understanding the Real Risks
Many people working in labs, or just curious about chemicals, run into the name methyl nitrite and wonder about its safety. It’s a compound known for its bright yellow color and its use in chemical syntheses. What many folks might not realize at first glance—methyl nitrite packs some serious risks both as a compound and during storage or handling.
Fast Evaporation, Faster Trouble
Methyl nitrite boils at room temperature, turning from liquid to gas in a flash. Even with a brief opening of a bottle, the space can flood with toxic vapor. I learned this firsthand while shadowing a chemist in graduate school. One misstep meant vapors spreading through our workspace, setting off alarms and sending us outside. Inhaling even small amounts can cause headaches, dizziness, or worse—blue-tinged lips and skin signal a problem with how the body carries oxygen.
Toxicity Isn’t the Only Risk
Breathing it isn’t the only concern. Methyl nitrite acts as a strong oxidizer and poses a fire hazard. A bottle toppled in a fume hood once, and sparks from nearby equipment nearly started a flash fire. A compound like this doesn’t ask permission—it takes every opportunity to cause harm.
In addition, methyl nitrite can react violently with everyday materials. It doesn’t get along with common metals, acids, or bases. Contact with copper or iron can set off unwanted, dangerous reactions. If the lab isn’t set up with the right ventilation and explosion-proof storage, the risk grows.
Regulation and Recommendations
Occupational safety standards treat methyl nitrite with extra caution. Regulations from agencies such as OSHA, and safety data from the CDC, clearly flag this compound. The Centers for Disease Control notes that along with the risk of inhalation, methyl nitrite damages red blood cells, impairing how our bodies use oxygen.
The EU and U.S. both limit its commercial use. In industry, technicians rely on full fume hoods, flame-resistant lab coats, and heavy-duty gloves tailored for chemical resistance. The smallest mistake—wrong gloves, poorly maintained ventilation—means real danger.
A Lesson from Lab Experience
In my own time handling volatile compounds, overconfidence almost always led to problems. It only takes one shortcut—propping open a hood, skipping glove checks, or using a regular waste container—for an emergency to unfold. Emergency services are trained to treat methyl nitrite releases as high-priority calls.
Where Do We Go from Here?
Education can’t stop at lab safety posters. Hands-on practice and a culture of accountability matter most. Young chemists learn protocols from mentors who model caution, not from warning labels alone. Safety gear needs regular checks. Proper labeling and chemical logs flag trouble before it starts. Disposal isn’t just a matter of dumping old bottles—specialized hazardous waste procedures help prevent injuries.
This isn’t a chemical for casual or curious handling. Industries and research teams treat it with respect built on hard-earned lessons. Methyl nitrite isn’t safe to handle without training, equipment, and a strong commitment to following strict guidelines every single time.
What are the storage requirements for Methyl Nitrite?
Why Methyl Nitrite Demands Respect
Methyl nitrite brings a serious punch in both laboratory and industrial spaces. It’s a volatile, highly flammable gas often used as a methylating agent or a reference point for measuring reaction kinetics. From long hours dealing with chemicals in academic labs to industry visits, I’ve seen experienced professionals treat methyl nitrite storage as a matter of habit and training, not as an afterthought. It doesn’t forgive mistakes. Exposure to heat or sparks, and the risk of build-up under pressure, can end up causing fires, explosions, or life-altering accidents.
The Practical Side of Safe Storage
Flammable gases like methyl nitrite require cold storage. Most seasoned chemists keep it in well-ventilated outdoor gas cages or in explosion-proof refrigerators. Storing this chemical near sources of ignition—like standard wall sockets, heaters, or open flames—means inviting trouble. Repeated training drives one point home: even the tiniest spark or static electric discharge from synthetic lab coats can flash the entire storage.
Every cylinder or container must remain secured upright to prevent falling and damage. Pressure relief valves need attention too. I’ve seen teams log checks twice a day during the colder seasons, because rapid temperature changes lead to unexpected pressure fluctuations, especially in older storage setups.
Why Proper Labeling and Isolation Matter
Labeling isn’t just red tape. In shared environments, mislabeled bottles become silent hazards. A well-maintained chemical inventory and clear, bright warnings prevent other workers from mistaking methyl nitrite containers for nonhazardous compounds or reagents. Isolating methyl nitrite from incompatible chemicals like oxidizers, organic acids, or strong bases builds a strong safety net—far more critical than for many less reactive gases.
From my own stints at bench work, I can say that secondary containment trays deliver peace of mind. Trays catch accidental leaks and prevent gases from spreading. I remember a time when containment trays averted what could have been a real disaster after an unnoticed valve failure late one Friday afternoon.
Health Impacts and Emergency Planning
Exposure to methyl nitrite brings more than fire risk. Inhalation can cause intense headaches, breathing issues, and methemoglobinemia—an oxygen transport disorder. Personal experience and medical records both highlight repeated small exposures that lead to symptoms escalating quickly—especially when ventilation falls short. Gas detectors and continuous air monitoring serve as crucial watchdogs. If one detector goes off, quick evacuation sometimes turns out to be the line between a lesson learned and a trip to the ER.
Written emergency protocols save lives. Employees need drills—not just reading material—on handling leaks or exposures. The best-run labs and plants arrange regular surprise drills, just to keep everyone sharp.
Room for Better Systems
More advanced storage solutions bring in real innovation. Automated venting and electronic monitoring systems catch problems faster than people, especially in big facilities. Employees often urge their management to invest in newer explosion-proof enclosures or smart vented cabinets. Tight budgets can make upgrades slow, but one close call can change company priorities overnight.
Methyl nitrite storage works best with a culture of open communication and strong reporting. Even the smallest valve leak or labeling flaw gets reported early and solved before it grows bigger. Old lab habits—stashing “just a little” outside the secured storage—should get called out and corrected by everyone in the room. Generations of chemists and plant operators have learned these lessons, often the hard way, and there’s no substitute for listening to that lived experience.
What are the potential health hazards of Methyl Nitrite?
Breathing Trouble and Dizziness
Walking into a lab or facility working with methyl nitrite hits you with a sharp, sweet odor. It’s not just unpleasant—your body can react to small exposures. Methyl nitrite messes with how oxygen moves around your blood, which puts anyone at risk for headaches, dizziness, and sometimes confusion. I’ve heard stories from researchers who only needed a few breaths of poorly ventilated air before feeling lightheaded and shaky. If someone breathes enough of this stuff, they’re heading for big trouble—fainting or even seizures show up when the brain and organs cry out for oxygen.
Liver, Kidneys, and the Blood
The liver and kidneys work overtime trying to handle chemicals like methyl nitrite. This molecule changes hemoglobin into methemoglobin. Normal hemoglobin carries oxygen just fine, but methemoglobin doesn’t let your blood do its main job. If levels climb high, the risk of serious damage gets real—especially for folks with health problems or those who already handle other toxic substances. Chronic exposure can push organs closer to failure, and there’s not always a red flag until things suddenly get scary.
Fire Risk and Explosions
Methyl nitrite catches fire easily and doesn’t wait for a spark. Stories float around about storage mishaps where a tiny mistake or leaky valve ended with an entire room or truck erupting. Facilities must respect strict rules—grounding containers, keeping out open flames, and avoiding static shock. I’ve seen firsthand how fire marshals treat methyl nitrite like a ticking clock. After a fire breaks out, toxic fumes become an instant emergency for surrounding communities and first responders. Breathing in those byproducts is one hazard; emergency rooms have to handle anyone exposed with serious care.
Skin and Eye Contact
Direct contact with methyl nitrite stings right away. Splash it on your skin and you’ll notice redness or maybe burns, and the eyes take a real beating. One squirt can cause swelling, pain, blurred vision or even blindness. Hospitals near chemical labs stay ready for this, keeping rinse solutions on hand. Workers covering themselves with gloves, goggles, and long sleeves don’t do it for show—they’ve learned those shortcuts lead straight to pain and hospital visits.
Community Exposure Concerns
Some neighborhoods live close to production sites or transport routes. I’ve talked to people worried about leaks or accidents, and those fears aren’t misplaced. Wind carries vapor farther than most realize, and emergency warnings sometimes arrive too late. Kids, seniors, and anyone with asthma or heart problems face extra risk. Real-time monitoring stations, evacuation plans, and prompt training for first responders make a real difference. Companies have a duty to use the safest containers, invest in leak detection, and follow up with transparent updates. Regular emergency drills with the local community keep everyone better protected.
Practical Safety Steps
Keeping health risks down isn’t rocket science, but it takes investment and a thorough approach. Ventilation systems need constant checks and repairs. Proper training for anyone handling chemicals pays off fast, even if that means repeating the basics year after year. Personal protective gear should fit right—nothing beats good gloves, face shields, and smart work habits. Strong labeling and storage rules, along with up-to-date emergency protocols, keep accidents from multiplying. Coordinating with local clinics or hospitals, and sharing honest data with everyone nearby, builds trust and safety from the ground up.
How should Methyl Nitrite spills be managed?
Understanding the Trouble with Methyl Nitrite
Methyl nitrite sits among those chemicals that don’t play nice. It’s toxic, highly flammable, and produces nitrogen oxides that choke the lungs when released. I’ve spent part of my career in research labs and, even in controlled settings, people treated this stuff with real caution. Picture cleaning up gasoline, only a lot touchier and with stakes running much higher. One false move, and the danger ramps up instantly.
Why Speed and Smarts Count During Spills
Once methyl nitrite hits the air, the clock starts ticking. Vapors catch fire from any spark. Skin exposure eats away at tissue, and breathing the fumes leaves folks dizzy or worse. That’s why response crews can’t hang back or cut corners. Fire doesn’t wait and neither do toxic clouds. This is not just a checklist moment; real lives hang in the balance, and so does the wider environment.
What Needs to Happen Right Away
Alarms go off and the area gets cleared. The right personal protective equipment matters—a dust mask isn’t much help here; emergency crews grab full-face respirators or self-contained breathing apparatus. No one wants to gamble with their lungs. For those who learned the hard way, proper chemical-resistant suits are a must. Many seasoned responders keep decontamination showers on standby, because splashes turn serious in seconds.
Ventilation becomes the next priority, but only if it helps sweep vapors out safely without spreading them further. Mechanical fans can stir up trouble, so responders often rely on containment. Sealing drains and blocking flow holds the spill in place. Even in small lab spills, I’ve seen how a simple tray or rolled towel can save hours of cleanup later—no high-tech solution required.
Cleanup Tactics That Actually Work
Absorbents such as activated carbon pick up the mess without sparking reactions. Scoop-and-bag works for waste, but tools and buckets stay plastic or coated metal; bare iron or steel offer an invitation for fire. The cleanup teams have to keep bystanders away even after the mess looks gone, since fumes hang low and sneak out for hours.
Every bit of collected material shifts into sealable, labeled containers. Disposal isn’t a quick trip to the dumpster—methyl nitrite contamination travels to a licensed hazardous waste facility. Once, a friend saw local responders skip this step, only for the waste truck to start smoldering at the dump. Lessons like those stick around.
Prevention and Community Safety
Training local teams goes far beyond reading manuals. Regular drills carve muscle memory into the brains of workers and first responders. I recall an old supervisor who ran mock drills every month, sometimes at odd hours, just to jam a sense of urgency into people who might get too comfortable.
Proper storage helps too. Small bottles, double containment, and well-labeled containers cut down on mistakes. Digital tracking—something many forward-thinking organizations now use—shows where chemicals sit and flags expiration dates before things go wrong. In neighborhoods near chemical sites, real-time alert systems can make a difference: warning families faster than a broadcast news statement ever could.
The Role of Oversight and Honest Reporting
Spill management isn’t just about mopping up. It takes real transparency—reporting what happened, tracking long-term effects, and inviting third-party audits. Good oversight means communities don’t have to gamble on luck or vague promises. In my experience, the strongest safeguards come from teams who call it like they see it, report everything, and push for better rules, not just bare-minimum compliance.
Putting real-world know-how, fast action, solid equipment, and honest communication at the center of spill management builds trust and saves lives. Every mishap can be a lesson and a step toward safer handling in the future.
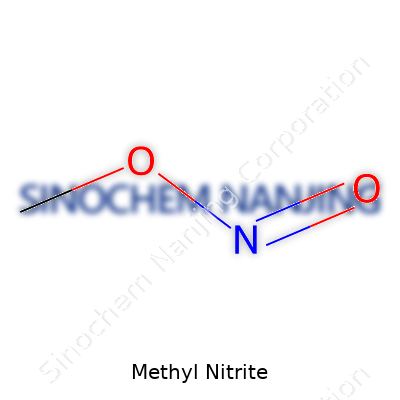
| Names | |
| Preferred IUPAC name | Methoxy nitrite |
| Other names |
Methyl nitrite Nitrous acid, methyl ester Methoxy nitrite MeN(O)O MON Nitrous ether UN 1061 |
| Pronunciation | /ˈmɛθ.ɪl ˈnaɪ.traɪt/ |
| Identifiers | |
| CAS Number | 593-53-3 |
| 3D model (JSmol) | `JSmol.loadInline("data/mol/Methyl_Nitrite.mol")` |
| Beilstein Reference | 1209227 |
| ChEBI | CHEBI:75080 |
| ChEMBL | CHEMBL135598 |
| ChemSpider | 6968 |
| DrugBank | DB09261 |
| ECHA InfoCard | 100.005.059 |
| EC Number | 200-772-9 |
| Gmelin Reference | 821 |
| KEGG | C14390 |
| MeSH | D008777 |
| PubChem CID | 6347 |
| RTECS number | NT1225000 |
| UNII | G3U12IV95E |
| UN number | UN1061 |
| CompTox Dashboard (EPA) | DJ15YM75TY |
| Properties | |
| Chemical formula | CH3ONO |
| Molar mass | 61.04 g/mol |
| Appearance | Yellowish liquid or gas with a fruity odor |
| Odor | fragrant odor |
| Density | D 1.067 g/cm³ (20 °C) |
| Solubility in water | slightly soluble |
| log P | 0.77 |
| Vapor pressure | 4000 mmHg (20 °C) |
| Acidity (pKa) | 26.0 |
| Basicity (pKb) | 11.08 |
| Magnetic susceptibility (χ) | -25.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.269 |
| Viscosity | 0.227 cP (at 25 °C) |
| Dipole moment | 3.46 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 176.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -75.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -930 kJ/mol |
| Pharmacology | |
| ATC code | N01AX08 |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Precautionary statements | P210, P220, P261, P271, P280, P304+P340, P308+P311, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-4-2 |
| Flash point | −40 °C |
| Autoignition temperature | 615°F (324°C) |
| Explosive limits | 4.2% - 20% (in air) |
| Lethal dose or concentration | LDLO-rat-alkaline solution-45 mg/kg |
| LD50 (median dose) | LD50 (median dose): mouse (inhalation) 2230 mg/m³/2h |
| NIOSH | NIOSH: QV6300000 |
| PEL (Permissible) | PEL for Methyl Nitrite: Not established |
| REL (Recommended) | 0.0003 ppm |
| IDLH (Immediate danger) | > 60 ppm |
| Related compounds | |
| Related compounds |
Nitrous oxide Nitric oxide Nitrite Dimethyl nitrite |

