Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate: A Closer Look at a Chemical Player
Historical Roots and Emergence in Chemistry
Back in the latter decades of the twentieth century, organic synthesis began pushing past its old boundaries. Among a wave of new compounds, Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate caught attention, especially as researchers started exploring potential pharmaceutical and agrochemical leads. It didn’t just show up out of nowhere; its design ties into a long history of tweaking amino acids and ester groups, tracing a path through medicinal chemistry where minor shifts in structure led to significant changes in biological activity. Chemists sometimes called it by its synonym “Ethyl 2-[(benzyl)(3,4-dichlorophenyl)amino]propanoate,” or referred to it just by shorthand among familiar circles in the lab. Unpacking its story means looking at the decades of incremental progress, as each tweak to the basic alanine scaffold brought new possibilities and occasional pitfalls.
Getting to Know the Compound
This molecule combines the familiar backbone of alanine, ramped up with a benzyl group and a 3,4-dichlorophenyl substitution, stitched together as an ethyl ester. Its structure hints at its dual roles—balancing hydrophobic and hydrophilic properties—potentially giving it a broader range of applications in chemical and pharmaceutical research. What stands out is not just the formula, but the carefully decided positions of chlorine atoms around the phenyl ring, which influences everything from solubility to metabolic stability. In a lot of practical benchwork, its crystalline form and molecular weight both play a part in how it gets handled and used, sometimes limiting, sometimes unlocking new uses in synthetic pathways.
Physical and Chemical Character
The compound often appears as a crystalline solid, pale in appearance, packing a density that leans toward the heavier side thanks to its dichloro substitution. Its melting point marks a distinctive spot on a researcher’s data sheet, offering clues about purity and chemical resilience. Ethyl esters like this don’t just survive rough handling; they inform how reactive or selective a molecule can be once it enters a reaction flask. Its moderate solubility in organic solvents gives practical flexibility, though it tends to resist mixing with water—something anyone setting up a reaction or extraction will take into account. Chemists, juggling dozens of vials, recognize these characteristics as deciding factors for reaction setup or product isolation.
Technical Labels and Lab Handling
On the technical front, labeling matches up with globally recognized standards, flagging its identity, purity, and known hazards. The dichloro groups call attention to its potential for environmental persistence, while the ester functionality points to reactions that need special handling, especially under acidic or basic conditions. Laboratory protocol around the world advises storing this compound in cool, dry conditions, away from incompatible chemicals like strong acids or oxidizers, which could kick off side reactions or decompose the product. All these details don't just fill up labels; they represent real risks and opportunities during a working day at the bench.
How It’s Made
Synthesis leans heavily on classical organic methods. The backbone starts with alanine, converted into its ethyl ester under acid-catalyzed conditions, often using ethanol and a touch of sulfuric acid. Subsequent amination involves reacting this ester with benzylamine and 3,4-dichloroaniline, sometimes under solvent reflux. That's not just a theoretical route—variations appear in patent literature and published research, reflecting tweaks in reagents, solvents, and temperatures. Those steps might seem routine on paper, but each batch teaches its own lessons about yield, purity, and the quirks of working with chlorinated aromatic rings. Purification, by chromatographic methods or crystallization, can stretch late into the night, demanding patience and a keen eye for the appearance of a clean product.
Chemical Reactions and Molecular Tweaks
Researchers often use Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate as a pivot point for chemical modifications. The ester group begs for hydrolysis or transesterification to form new derivatives, every reaction an invitation to push the boundaries further out. Chemists sometimes swap out the benzyl or 3,4-dichlorophenyl units, exploring activity changes in analogous compounds. These substitutions alter everything from reactivity to biological action. Sometimes, scientists pursue coupling reactions, or derivatize the amine for further synthetic adventures—each attempt reflecting the drive to eke out one more bit of useful activity or insight. Every small modification in the structure brings about notable changes in physicochemical behavior, a reality that lands with impact only after you’ve run enough rounds in the lab.
Other Names and Synonyms
Chemists often refer to molecules by less intimidating names in conversation or lab notes. Ethyl N-Benzyl-N-(3,4-dichlorophenyl)alaninate, Ethyl 2-[(benzyl)(3,4-dichlorophenyl)amino]propanoate, and related shorthand names pepper research articles and lab books. Multiple identifiers come from software databases or chemical catalogs, so keeping track grows important as collaborations expand globally. Losing track of a synonym can lead to missed connections in the literature or confusion when ordering reagents—something every research group wrestles with at scale.
Safety and Good Practice
Every researcher needs to treat Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate with respect. The dichloro groups signal environmental concerns, hinting at slow breakdown and persistent residues if not managed properly. Personal protective equipment—gloves, goggles, sometimes lab coats double-layered for splash risks—keep accidents from turning disastrous. Fume hoods make a difference, especially if volatilization or unexpected decomposition threatens. Clean-up and disposal add responsibility, as chlorinated compounds shouldn’t enter standard waste streams. Those who work hands-on don’t see this as bureaucratic burden, but as habits picked up from mentors, reinforcing the culture of responsibility in chemical research.
Applications That Matter
The range of uses for this molecule rides along the interests of both synthetic chemists and biologists. Its backbone acts as a scaffold for more ambitious pharmaceutical candidates, where each atom contributes to biological function—sometimes as an enzyme inhibitor, sometimes as a lead for more refined drugs. Agrochemical research often looks at such chlorinated esters for pest control activity or plant protection studies, searching for potency balanced against off-target impacts. Others take advantage of its reactivity to screen for new types of transformations—it serves as both a target and a building block for further development. Every application draws on its finely tuned chemistry, but real world impact depends on translating bench results into practical, safe products.
Research Moves and Development Horizons
Interest in Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate keeps growing as new research pushes for more selective, potent, or environmentally friendly molecules. Drug discovery efforts focus on molecules like this with a balance of lipophilicity and metabolic stability, traits shaped by the dichloro and benzyl groups. Advances in reaction methodology intend to streamline synthesis, improve atom economy, or open routes to new analogs. Analytical chemists probe metabolic breakdown, hoping to spot problems like toxic byproducts before they impact fieldwork or patient safety. Research journals burst with studies, each staking out fresh questions in this crowded yet fertile chemical space. The future of research often depends less on developing brand new molecules, and more on understanding, refining, and responsibly exploiting the ones already in our hands.
Toxicity: What Matters in the Real World
Concerns about toxicity rest on both structural alerts and real toxicological data. Chlorinated aromatics set off alarm bells because of historic experiences with environmental damage. At the same time, the ester-linked alanine structure could signal easier metabolism and potential clearance in biological systems. Studies on acute and chronic toxicity, alongside bioaccumulation risk, guide safe handling procedures and influence regulatory scrutiny. Where data comes up short, cautious researchers err on the side of safety. Regular updates to procedures follow new insights from animal studies or accidental exposures. The history of job-related illnesses in chemistry has shaped a community quick to adapt when evidence points toward long-term risks.
Peering into the Future
What comes next for Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate often depends on the collective choices of the scientific community. Pushing forward, chemists look at greener synthesis routes, aiming to reduce hazardous waste and improve efficiency. Life sciences researchers eye new pharmacological targets, testing modifications that fine-tune activity or reduce unwanted toxicity. Environmental chemists investigate breakdown pathways, seeking assurance that what benefits one field doesn’t spoil another. With pressure mounting for sustainable chemical practices, the path forward will reward those who think beyond immediate synthesis—who listen as much to safety and environmental concerns as to the allure of laboratory discovery. For any researcher, this molecule offers more than a challenge or a tool; it serves as a reminder that each step in chemistry brings responsibilities along with opportunities.
What are the primary uses of Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate?
What This Compound Means for Modern Agriculture
Stepping out on a field after rain, the smell of fresh earth is unmistakable. Yet, for those who tend crops, there’s always the looming worry of weeds taking over. Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate, known amongst agronomists for its power as a selective herbicide, plays a real-life role in these ongoing battles with unwanted plant life. This chemical targets certain weed species without harming the main crops, making it a practical tool for producing strong yields in wheat, barley, and other grains.
Years ago, before advances like this, farmers spent more hours hunched over, pulling weeds by hand or paying for labor that ate into already tight margins. Now, with strategic application, there’s a genuine reduction in workload and a bump in crop health. Modern field trials show this compound clears out broadleaf weeds, especially those stubborn ones that shrug off basic methods. That means fewer headaches, and a better shot at a quality harvest.
Environmental and Health Considerations
With every chemical sprayed over a field, questions about safety rise to the surface. Farmers I talk to weigh every decision, not just for short-term convenience, but for the safety of their families, the water they drink, and the land their kids will inherit. Regulatory agencies closely watch the use of Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate, setting guidelines for application rates to limit risks of runoff and residue.
Testing by agriculture departments and independent labs looks for traces in soil and water, and these results steer decisions on continued use. When rules get ignored or weather events push chemicals off fields, everyone loses. Data suggests that, when applied as recommended, this herbicide breaks down over time, which limits its spread into surrounding ecosystems. In my own experience counseling local growers, the importance of following label instructions can’t be overstated. Mistakes here cost soil health and community trust.
Weighing Benefits Against Resistance
One major challenge crops up with repeated use. Just like bacteria can shrug off antibiotics, weeds adapt. Some farms now face populations that simply ignore standard treatment. This throws a wrench into what once seemed a sure bet for weed control. Switching up chemicals and blending old-fashioned practices—like rotating crops or planting cover species—creates a more resilient approach.
I’ve watched fields turn around after a season or two of changing tactics—there’s no “silver bullet,” but combining chemical tools with soil stewardship delivers longer-lasting results. These strategies help slow resistance and reduce chemical load, making the land and the food coming from it safer.
Better Choices for Future Harvests
Farmers and scientists keep searching for new answers. Public investment in research means safer, smarter products hit the market every few years. Some are challenging the old model, chasing mechanical weeders, or looking at biopesticides that mimic natural predators. Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate hasn’t solved every issue, but it made weeding more manageable and kept farms profitable through uncertain seasons.
Every decision in the field ripples through families and communities. It takes responsibility from every side—growers, researchers, regulators—to keep food safe and land healthy. As policies shift and new technologies develop, staying grounded in transparent science and honest conversations matters more than ever.
Is Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate safe to handle and use?
Understanding the Risks
People in labs and factories often find themselves working with chemicals that sound like they came straight out of a science fiction novel. Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate lands squarely in that territory. Even though most folks will never encounter this compound outside a research setting, questions around its safety matter a lot to the people who do.
This compound belongs to a family of organic molecules, many of which serve as building blocks in agriculture, pharmaceuticals, and materials science. Some folks might know it from its use in specialty crop protection products. These kinds of chemicals tend to come with health and environmental risks. Chlorinated aromatic compounds often cause skin and eye irritation, and in some cases, even have long-term effects like liver toxicity or negative impacts on aquatic life. The concern grows deeper if you don’t have clear documentation on how toxic the compound gets through skin absorption or inhalation.
Direct Experience With Chemical Handling
In the years spent working around labs, protective gloves, lab coats, and fume hoods became more than just rules—they were regular parts of any safe routine. Too many stories float around about minor spills leading to rashes or headaches. Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate doesn’t come with the household familiarity of acetone or ethanol. That unfamiliarity often means the margin for error closes quickly. People sometimes make the mistake of treating “rare” chemicals as less dangerous. Truth is, new or unknown chemicals deserve extra respect. If you don’t know the exposure limits, you can’t tell if breathing a little vapor will keep you up all night with a migraine.
Manufacturers and Transparency
Today, manufacturers are under pressure to provide clear, complete Safety Data Sheets (SDS). These sheets need real numbers, not vague warnings. You need to see details about acute and chronic toxicity, routes of exposure, ecological data, and first aid instructions. Safety comes down to trust and good information. If the SDS feels thin—missing physical, toxicological, or disposal data—then the risks jump up right away. Without detail, workers end up guessing. That leads to mistakes, and mistakes with chemicals like these can carry a heavy cost.
Safe Handling Practices
Every lab or workplace uses protocol: keep the product in well-ventilated areas, wear gloves and goggles, and avoid eating or drinking near the chemical. Containment is big—secure caps, minimize open handling, and use spill kits nearby. Quick, informed action contains most accidents before they turn serious. Proper chemical storage and clear labeling also make a difference.
Steps Toward Safer Work
No one should have to work in the dark about safety. Pushing companies to fund more thorough toxicology research can build a clearer picture of risk. Requests for updated SDSs force suppliers to dig deeper into their data. Regular training refreshers help everyone stay alert and spot new risks before they become problems. Fostering a culture where people speak up about unclear instructions or missing information saves both money and lives.
Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate may never enter the mainstream, but its risks are no less real than the classic acids and bases. Good information, training, and solid routines keep people in control and out of harm’s way.
What is the recommended storage condition for this product?
Storing Product Safely: What Matters Most
If you’ve ever opened a package and found something off—maybe a strange smell, a faded label, or unexpected clumping—you know how important proper storage can be. It’s more than just keeping things tidy; it’s about making sure a product works, tastes, or performs as expected. Keeping an eye on storage conditions gives any product, whether food, medicine, or household material, a better shot at lasting longer and staying safe.
Everyday Truths from the Pantry and Beyond
I’ve spent plenty of time checking expiration dates in my own kitchen, tossing out spices that lost their zing or medicine stashed in a bathroom cabinet a bit too warm and steamy. Turns out, those notices on labels exist for a reason. Heat, moisture, and even a bit of sunlight can change the way things work or taste faster than you might think. In food, those changes can attract mold or bacteria. In medicine, unstable storage often leads to less reliable results—sometimes with real risks for health.
Several studies back up the importance of this. For example, research published by the World Health Organization points out that as temperatures climb above 25°C, some pharmaceuticals break down far more quickly. Food standards agencies around the world warn about letting perishables linger outside chilled spaces—meat left at room temperature can develop dangerous bacteria in just a couple hours.
Simple Steps for Reliable Storage
Most products benefit from a similar set of rules: keep it cool, dry, and sealed. For perishable food, the fridge or freezer creates a reliable safety net. Medicines work best in a dry cupboard away from heat, not in a steamy bathroom. Cleaning products and chemicals often demand a locked, well-ventilated space away from curious kids or pets. Even batteries prefer cool, dry drawers over warm, cluttered garages. These habits seem basic, but they prevent a lot of waste and avoidable risk.
Whenever companies update packaging or develop new storage instructions, they usually turn to real-world testing. Some manufacturers use accelerated aging studies, running products through high-heat environments to guess how they’ll last in a normal home. Quality teams track changes in consistency, flavor, or chemical structure, ensuring anything on a shelf is as trustworthy at the end of its life as at the start. Their work helps build those “store in a cool, dry place” recommendations that often get shrugged off but can save a lot of hassle.
Better Storage Practices Start at Home
People sometimes underestimate how much their own environment affects storage. A product kept in an air-conditioned room lasts longer than one left on the porch in summer. Tracking changes in temperature and humidity makes a bigger difference than just buying bigger packages. Even upgrading to airtight containers can stretch out the life of pantry staples and prevent pests. It’s about taking little steps that make daily routines safer and more efficient.
Following storage guidance isn’t about perfection; it’s about building habits that help everyone get the best from what they buy. By respecting those small instructions on labels, it’s possible to save money, reduce waste, and protect health. The wisdom behind all those “keep sealed” or “avoid sunlight” notes has always come from real lessons and years of trial and error—worth keeping in mind every time a new product shows up on the shelf.
Are there any known side effects or toxicities associated with this chemical?
Why Understanding Chemical Safety Matters
Every day, chemicals show up in cleaning products, cosmetics, and the foods we eat. Some claims about these chemicals sound scary, but sorting through the facts gives peace of mind—and sometimes helps us push for smarter safety rules. If you've ever read the back of a shampoo bottle or glanced at an ingredients list, you know how overwhelming the scientific names can get. It matters to ask what’s actually in there, and more importantly, if it can do harm.
What Side Effects Really Mean in Real Life
My first run-in with chemical side effects came from a simple cleaning spray. I used it to shine up my kitchen, only to end up with red, itchy hands. Turns out that chemical—one you might spot in any hardware store—can trigger skin allergies. That taught me two things: reactions don’t always show up in lab animals, and label warnings aren’t just for show.
Plenty of well-known compounds have clear side effects. Take formaldehyde—used in everything from pressed wood products to hair straighteners. It can cause burning eyes, breathing trouble, or even raise cancer risk after long-term contact. Asbestos, once common in insulation, brought lung disease to thousands. Just because something’s been around for decades doesn’t make it harmless.
Short-Term vs. Long-Term Toxicity
Chemicals can strike fast, like ammonia fumes that sting the nose and throat. Others—think of certain pesticides—work slowly, building up over years and raising risks for chronic disease. Many stories never hit headlines. Sometimes, people only notice after years on the job, or when public health researchers connect the dots.
It’s tough to know exactly how a chemical acts in every person. Age, health, even genetics play their part. Some folks shrug off mild irritation, while others break out in hives or face breathing problems. That’s why labs test across a wide range of subjects, and health agencies post recommended exposure limits—not hard-and-fast rules, but useful guidance for avoiding trouble.
Digging Up the Facts: How Experts Check for Safety
Learning about side effects means looking at a pile of research: animal tests, cell studies, reports from workers, and accidental exposures. Mistakes—like the lead-in-paint saga—often drive new studies and better rules. The U.S. Environmental Protection Agency (EPA), Food and Drug Administration (FDA), and other regulators update advice as new risks turn up.
Plenty of reliable information sits in public health resources and peer-reviewed journals. Checking those beats falling for scary online rumors. Some organizations, like the National Institutes of Health (NIH), keep databases where regular folks can see plain-language summaries of chemical risks, reporting allergic reactions, links to cancer, or environmental hazards.
What Makes a Safer World
People can ask for better transparency from companies and keep an eye out for robust independent testing. Workers benefit from strong protective guidelines, regular health screenings, and open reporting when issues come up. On a personal level, wearing gloves, using proper ventilation, and reading warning labels pay off—and so does choosing less risky alternatives where available. Ultimately, public demand and careful research push manufacturers to raise the bar on chemical safety.
How should Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate be disposed of after use?
Looking Beyond the Lab Bench
Ethyl N-Benzyl-N-(3,4-Dichlorophenyl)-Dl-Alaninate rarely gets public attention, but anyone who’s handled this compound in research or industry knows things get complicated when it’s time to toss leftovers. Not every chemical can take a trip down the drain—think about the contamination and what could end up in the soil or rivers. Someone once described the smell as “pungent” the first time I worked with a dichlorinated aromatic compound; it lingered everywhere. It didn’t take long to realize these molecules stick around longer in the environment than most people realize.
Real Hazards, Real Lives
Compounds like this stick out for their persistent nature. Those two chlorines change the whole picture—not just from a chemistry angle, but for health and safety as well. Even in my own experience, a spilled drop on a bench top turned into a lesson about proper gloves and fume hoods. That wasn’t just protocol; it mattered for everyone using the space after me. The U.S. Environmental Protection Agency identifies many dichlorinated and aromatic compounds as harmful to aquatic life even at low doses. These facts make responsible disposal non-negotiable.
Not Just a Local Problem
Dumping waste in general trash or into a sink doesn’t just put the building’s plumbing at risk. Wastewater plants can’t break down everything. This stuff survives. It works through pipes, sneaks out with the water, and shows up where we’d rather it didn’t. Removal costs for persistent organic pollutants eat into public budgets every year, according to the United Nations Environment Programme. Local actions hit bigger systems. That’s a lesson few people appreciate until they see the downstream results.
Common Sense Meets Regulation
Anyone who’s worked with research chemicals, especially organochlorine derivatives, learns early what can happen if disposal is sloppy. I once saw a fume hood shut down for days after an improper dump—the whole building got a lesson. Federal rules back these habits: the Resource Conservation and Recovery Act in the U.S. lists many similar compounds as hazardous waste. Laws expect chemical users to hand off this kind of waste to licensed contractors who know how to safely destroy or neutralize them, usually by incineration at high temperature. No home fixes or DIY drain solutions. If the waste comes from a university lab or an industrial site, a hazardous waste bag stays the safest call.
Better Habits Start Early
It’s easy to think tossing a small batch here and there means little. The reality stacks up. In my old lab, every bottle carried a tag tracking what left the space and who handled it. This practice worked—accidents shrank, and nobody worried about handling surprise residues. Establishing written procedures and enforcing training does more than check regulatory boxes. Anyone who joins in can ask questions, flag problems, and suggest improvements, because they know what goes on.
Avoid the Mess Later
Local hazardous waste services usually offer advice and collection programs. If nobody around knows what the next step should be, a call to an environmental health officer helps. Disposing of complex chemicals demands more than following one rule; it’s about listening to people with experience and building smarter habits. The safest disposal methods protect everyone—now and for a long time down the line.
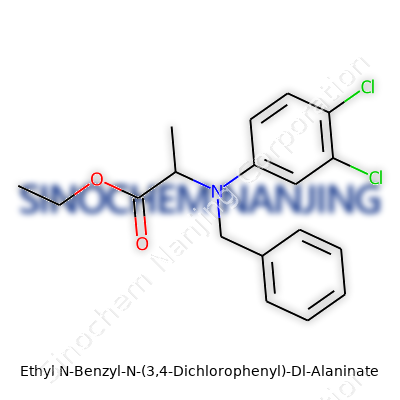
| Names | |
| Preferred IUPAC name | ethyl 2-(benzyl(3,4-dichlorophenyl)amino)propanoate |
| Other names |
Ethyl N-benzyl-N-(3,4-dichlorophenyl)alaninate Ethyl N-benzyl-N-(3,4-dichlorophenyl)-DL-alaninate Ethyl N-benzyl-N-(3,4-dichlorophenyl)alanine ester |
| Pronunciation | /ˈiːθɪl ɛn ˈbɛn.zɪl ɛn ˈθriː ˈfɔːr daɪˈklɔːr.oʊˌfiː.nɪl diː ɛl əˈlæn.ɪ.neɪt/ |
| Identifiers | |
| CAS Number | 629664-81-9 |
| 3D model (JSmol) | `/prod/jmol/data/cid/13002032.smi` |
| Beilstein Reference | 2963787 |
| ChEBI | CHEBI:131133 |
| ChEMBL | CHEMBL3704687 |
| ChemSpider | 21559648 |
| DrugBank | DB08910 |
| ECHA InfoCard | 03b3f431-05fc-48e8-aaa9-efafcf5aabb4 |
| EC Number | 610-144-8 |
| Gmelin Reference | Gmelin 83380 |
| KEGG | C18622 |
| MeSH | Dichlorphen |
| PubChem CID | 127263875 |
| RTECS number | DJ1228000 |
| UNII | KT8YJZ3Y01 |
| UN number | UN3276 |
| CompTox Dashboard (EPA) | DTXSID3039249 |
| Properties | |
| Chemical formula | C18H19Cl2NO2 |
| Molar mass | 395.28 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.3 g/cm3 |
| Solubility in water | Insoluble in water |
| log P | 3.96 |
| Vapor pressure | 5.2E-5 mmHg at 25°C |
| Acidity (pKa) | 14.98 |
| Basicity (pKb) | 7.2 |
| Magnetic susceptibility (χ) | -53.98 × 10^-6 cm³/mol |
| Refractive index (nD) | 1.583 |
| Dipole moment | 4.85 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 528.9 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | N05CM02 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin and eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H410 |
| Precautionary statements | P260, P261, P264, P270, P271, P272, P273, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P330, P332+P313, P337+P313, P362+P364, P403+P233, P405, P501 |
| Flash point | 102°C |
| Autoignition temperature | 425 °C |
| Lethal dose or concentration | LD50 oral rat 344 mg/kg |
| LD50 (median dose) | LD50 (median dose): 300 mg/kg (rat, oral) |
| PEL (Permissible) | Not established |
| REL (Recommended) | Not established |
| Related compounds | |
| Related compounds |
Benzyl N-(3,4-Dichlorophenyl)glycinate N-Benzyl-N-(3,4-dichlorophenyl)-DL-alanine Ethyl N-benzylglycinate N-Benzyl-N-phenyl-DL-alanine ethyl ester Ethyl N-benzyl-N-phenylglycinate |

