Ethyl Isocyanoacetate: Exploring Complexity and Opportunity
Historical Development
Diving into the world of organic synthesis often brings up chemicals with obscure names yet remarkable influence, like Ethyl Isocyanoacetate. Its discovery dates back to the growing interest in isocyanide chemistry in the mid-20th century, a period marked by pushing synthetic boundaries. Early researchers sought building blocks capable of generating molecular complexity from simple starting points. The isocyano functional group, notorious for its pungent odor and high reactivity, opened doors chemists couldn't ignore. Ethyl Isocyanoacetate stood out, catching the attention of academic labs in Europe and the United States. For decades, its role has grown in tandem with advances in multicomponent reactions, helping streamline synthesis routes that would frustrate even the steeliest organic chemist with traditional methods.
Product Overview
What starts as a niche compound in the chemical catalogues now draws wide attention from medicinal chemists, material scientists, and agrochemical developers. Ethyl Isocyanoacetate offers a key to unlock unconventional reaction pathways, all thanks to its dual reactivity as both an isocyanide and an ester. The balance between these two functional groups drives applications that rely not just on one transformation but on entire sequences built around this single molecule. Chemists count on it for assembling heterocycles, fine-tuning molecular scaffolds, and propelling structure-activity relationship studies in drug discovery screens.
Physical & Chemical Properties
A clear, colorless to pale yellow liquid, Ethyl Isocyanoacetate typically comes with that distinct, pungent isocyanide smell—one you do not forget after working with it even once. Boiling at around 170°C and sporting a density in the range of 1.01 g/cm³, it's manageable by standard lab protocols, storing well under nitrogen to prevent degradation. The dual nature arises from the ethyl ester group and the isocyanide carbon, which sets up nucleophilic and electrophilic behavior, letting it participate in multicomponent syntheses or nucleophilic substitutions much more flexibly than simpler isocyanides. Its modest solubility in organic solvents like dichloromethane and diethyl ether offers some versatility for different reaction setups.
Technical Specifications & Labeling
Commercial bottles of Ethyl Isocyanoacetate bear standardized GHS and REACH labeling, signaling its hazards and the need for responsible handling. Purity typically runs above 95%, and buyers receive COAs giving full analytical breakdowns—NMR, GC-MS, IR tracing the authenticity and cleanliness of every batch. Labels warn of acute toxicity, skin and eye irritation, and potent odor, directing chemists always to work in well-ventilated areas. Packaging uses amber glass to block stray light and vented caps to handle any pressure build-up, reflecting hard-earned lessons from decades of chemical shipping.
Preparation Method
Synthesis often builds on well-trod literature, starting with ethyl cyanoacetate or ethyl bromoacetate as a base. Treating these substrates with silver cyanate or related reagents yields the intermediate needed for the isocyanide group; careful temperature control prevents runaway reactions or tar formation. Sometimes labs scale up using phase-transfer catalysis, streamlining yields in more industrial contexts. Purification leans on fractional distillation under reduced pressure, since the raw product tends to pick up colorates and side-reaction debris if the process drags or the solvent boils too hard.
Chemical Reactions & Modifications
Ethyl Isocyanoacetate shines brightest in multicomponent coupling, especially Ugi and Passerini reactions—staples for constructing peptidomimetics or bulky heterocycles in a single-pot process. The isocyano group latches onto electrophiles in carbonyl-rich mixtures, while the ethyl ester tags along, setting up subsequent hydrolysis or amidation steps. Chemists modify it further via nucleophilic substitutions or cross-coupling to append aromatic rings or aliphatic chains, tweaking electronic properties to suit follow-up steps. This built-in versatility speeds up library generation and lets teams skip weeks of separate synthetic steps.
Synonyms & Product Names
Like many specialty reagents, Ethyl Isocyanoacetate goes by several aliases in literature and sales catalogues. Common synonyms include Ethyl 2-isocyanopropanoate, Isocyanoacetic acid ethyl ester, and occasionally simply as EIA in shorthand. These names pop up regularly in patents, research papers, and supplier lists, so knowing them can save hours when trawling for data or sourcing.
Safety & Operational Standards
Ethyl Isocyanoacetate demands serious respect in the lab. Even seasoned chemists reach for double gloves, full face shields, and work exclusively in fume hoods due to the intense odor and risk of skin or eye contact. Regular ventilation checks and spill kits matter greatly, and waste disposal routes to specialized solvent incineration rather than the general hazardous chemical stream. Labs running student rotations routinely reinforce these standards, using the compound as a teachable moment on risk and preparation. Training documents stress the need to avoid inhalation, minimize splash risks, and keep the chemical tightly capped and away from oxidizers or acids.
Application Area
Active pharmaceutical ingredient (API) development leans hard on Ethyl Isocyanoacetate, owing to the efficient access it provides to nitrogen-rich heterocycles. Cancer and infectious disease research rely on such frameworks due to their bioactivity and capacity for late-stage derivatization. Polymers and specialty coatings tap into the isocyanide’s reactivity, letting scientists build materials with targeted cross-linking or self-healing properties. Agrochemical firms also use it to engineer new herbicide and insecticide backbones, pushing for enhanced environmental breakdown while retaining potency in the field.
Research & Development
Research into isocyanide-based synthesis has expanded, especially with the explosion of combinatorial chemistry and automated platforms. Ethyl Isocyanoacetate features in countless studies focused on diversifying small molecule libraries quickly—essential for finding leads in drug discovery. Lab automation and flow chemistry have raised throughput, beyond what manual benchwork could ever manage, and scientists leverage these workflows to test reaction scope and scale-up feasibility. Collaborative efforts between academia and industry further push the utility of EIA, as teams explore not just classic reactions but also photoredox and biocatalytic options, trying to tame or tune its reactivity for new fields.
Toxicity Research
Toxicological profiles show an acute risk from exposure, especially through inhalation or skin absorption. Rodent data point to low LD50 values, and animal studies underline the necessity for robust exposure controls. The sharp odor is not just unpleasant; it actively irritates mucous membranes and may sensitize over prolonged exposure. Researchers and industrial hygienists continue to probe chronic toxicity and environmental fate, since isocyanides in general show poor biodegradation yet potential for off-target bioactivity. Such factors steer labs and suppliers to continually revise MSDS documentation and to support safe handling through improved engineering controls and PPE recommendations.
Future Prospects
Ethyl Isocyanoacetate sits at the intersection of pressing needs: faster drug development, higher molecular diversity, and streamlined manufacturing. Scientists search for greener production routes, using less hazardous reagents and better waste management to reduce environmental impact. Advances in flow synthesis and in situ generation may help limit exposure, increase safety, and give process chemists more tools for scaling reactions. In drug development, computational prediction and machine learning open doors to smarter molecular design, pairing EIA’s versatile reactivity with predictive analytics to target rare disease pathways. The next decade promises significant expansion in how and where this once-esoteric molecule finds its value.
What is Ethyl Isocyanoacetate used for?
A Tool for Building Up Molecules
Ethyl isocyanoacetate might sound like an obscure chemical, but it plays a key part in how scientists build complex molecules for the modern world. This pale liquid doesn’t make headlines, but behind the scenes it helps chemists craft new antibiotics, cancer drugs, and unique materials. Walking through the halls of a university chemistry building, you’ll find researchers who keep a small bottle of this compound tucked away for those rare but critical reactions.
Used by Chemists Making New Medicines
Let’s get real. Most of us never hear about ethyl isocyanoacetate. Still, pharmaceutical labs depend on it. Chemists use it to connect carbon atoms in creative ways, making rings or branching structures needed for drug candidates. Some anti-viral compounds and antifungals couldn’t exist without its help. The main function: it’s a “building block.” That means it can be plugged into bigger chemical puzzles, helping researchers reach destinations that would be much harder by other routes.
Think about the way a carpenter keeps an odd-shaped chisel for a tricky job — ethyl isocyanoacetate fills a similar spot on the chemistry bench. One famous example involves the Ugi reaction, a process that’s almost like a shortcut for stitching together four separate pieces into one new molecule. This shortcut has powered up how quickly scientists can explore new drugs. Published studies from organic synthesis journals confirm its importance in building diverse molecules efficiently.
Beyond Medicine: Role in Advanced Materials
Not every use ties to pills or patient care. Scientists rely on ethyl isocyanoacetate in materials science too. It’s a go-to chemical for creating molecular libraries—collections of thousands of related chemical variations. That approach lays the groundwork for new adhesives, coatings, or specialty plastics. Sometimes those discoveries work their way into electronics, solar panels, or even aerospace materials.
In my experience working in a small biotech lab, we hit roadblocks in making just the right molecule for a diagnostic tool. Commercial suppliers offered only a few options, and almost every promising path circled back to reactions needing ethyl isocyanoacetate. It became clear: this chemical does not show up in finished products, but the world of innovation depends on it.
Handling With Respect and Safety
Every field demands safe handling of tricky chemicals, and ethyl isocyanoacetate is no exception. This compound gives off a strong, pungent odor and needs careful storage. Lab safety data sheets stress using gloves and working in fume hoods—this isn’t something to keep in a high school lab desk drawer. Researchers have reported skin and eye irritation from improper handling. Higher standards across universities and industry help minimize those health risks, and regulatory agencies like OSHA in the US provide strict guidelines for use.
Waste also matters. In my old lab, the safety officer made it clear: waste streams containing any isocyanides go into special containers and get handled by licensed professionals. Those rules protect workers and prevent harmful chemicals from escaping into groundwater.
Room for Advancement
Some green chemistry groups focus on making this compound in more sustainable ways, given the current processes require toxic reagents. Better methods could lighten the environmental impact, cutting hazardous waste and limiting exposure. The push for safer, greener chemistry keeps gaining ground, with journals publishing progress every year.
Ethyl isocyanoacetate might sound technical, but the world leans on this tool in more ways than most realize. Supporting innovation, safer workplaces, and cleaner production methods will keep this silent helper valuable for the next wave of science and technology.
What is the chemical formula of Ethyl Isocyanoacetate?
The Chemical Formula and What It Means
Ethyl isocyanoacetate is a small molecule, and its chemical formula is C5H7NO2. Seeing a formula on paper never really shows just how useful or problematic a compound is. With this one, there’s a blend of simplicity and complexity. Ethyl isocyanoacetate brings together an ethyl group, an isocyano group, and an ester function. This combination is far from random—it makes the molecule pretty versatile for chemists designing new compounds.
Where You Encounter It
Lab folks often run into ethyl isocyanoacetate during the synthesis of heterocyclic compounds. In the pharmaceutical industry, researchers keep it handy when looking to build rings found in potential drugs. I spent time in research where achievement wasn’t just about racking up reaction yields—it was making new connections between basic chemicals and real products like drug candidates. Ethyl isocyanoacetate played its part in several key steps.
Why the Isocyano Group Matters
Isocyano groups tun into powerful tools in organic reactions. Especially in multicomponent reactions, like Ugi or Passerini. These reactions don’t just save time; they save a lot of raw material, which matters for anyone watching cost or environmental footprints. Instead of long, laborious stepwise syntheses, a team can toss in ethyl isocyanoacetate and punch out complex molecules faster. I’ve seen these reactions slash project times for graduate students who often face tight deadlines. One simple choice of reagent can change the experience in the lab, and this is a textbook example.
Safety and Handling
Not every flashy chemical comes without baggage. Ethyl isocyanoacetate raises eyebrows for its potential toxicity and its powerful, sometimes unmanageable smell. Working with it, gloves and good ventilation are non-negotiable. The isocyano group isn’t just reactive in test tubes—it can be irritating to skin and lungs. Teachers and lab managers should stress respect for these rules. Useful chemicals shouldn’t shift all focus to what they can make; they require responsible handling. This comes directly from long afternoons in fume hoods, double-checking seals and breathing masks for leaks, as nobody wants to take shortcuts with chemicals like this one.
Where the Field Goes From Here
Ethyl isocyanoacetate keeps a mostly niche spot in organic chemistry, but methods for making and using it keep evolving. Manufacturers look for ways to produce it cleaner and safer. Some labs try to swap out hazardous substances for less risky ones without giving up reaction diversity. That balance, between innovation and caution, tends to drive progress. Sharing best practices, supporting safety culture, and backing improvements in production helps reduce chemical risks in workplaces.
Responsible Use and Broader Impact
Awareness spreads fast in the community—stories of accidents, as well as breakthroughs, travel much faster today than a decade ago. As someone who often answers student questions, I stress understanding the details behind every formula. Chemicals like ethyl isocyanoacetate aren’t only about reactions on a whiteboard; they tie into safety, environmental choices, and ethical decisions. Making better choices with molecules benefits more than the next drug—it supports healthier labs and, down the line, healthier communities.
What safety precautions are necessary when handling Ethyl Isocyanoacetate?
The Unforgiving Nature of Ethyl Isocyanoacetate
Few chemicals command respect in a lab quite like ethyl isocyanoacetate. With this reagent, you get something that quickly bites back if treated with anything less than caution. Even years after my first brush with the substance, the faint, piercing smell sticks out in my memory—and it serves as a sharp reminder to never take shortcuts.
Personal Protective Gear Protects More Than Skin
A splash to the skin or eyes with ethyl isocyanoacetate brings more than a little irritation. Rapid redness, burning, or worse—blistering—comes fast. Gloves made from nitrile or butyl rubber keep hands out of danger. Lab coats and eye protection block stray sprays and vapors. I always make sure my goggles fit snugly before even cracking the seal on the bottle. Lash-free eyes beat a chemical flush every time.
Ventilation Guards Your Lungs and Neighbors
Tiny droplets of ethyl isocyanoacetate vaporize much faster than a casual labmate realizes, and once in the air, the stinging odor warns of each breath. A fume hood isn’t just good practice here—it’s essential. There’s no real substitute, as a standard lab mask leaves too much risk uncovered. I’ve seen coworkers forget, only to spend the rest of the day nursing headaches or burning throats.
Don’t Leave Storage to Luck
Ethyl isocyanoacetate causes trouble even at room temperature. Keep it sealed tight in a chemical-safe cabinet, far from sunlight and away from acids or other incompatibles. My lab stacks the bottles in small trays, just in case a glass crack springs a leak. Strong ventilation around storage helps prevent a slow build-up of fumes. It’s easy to get lazy and just tuck it anywhere, but that only creates long-term headaches or real emergencies down the road.
Clean-Up Isn’t a Guessing Game
A minor spill doesn’t sound daunting until you’re gagging from fumes three feet away. Any cleanup starts with proper gear and goes slow—never try just paper towels or a rag. You want absorbent pads suited for organic chemicals, and always plenty of baking soda or a neutralizing agent close by. Disposal follows local hazardous waste rules, with no exception. Regular trash cans have no business handling these leftovers.
Training Is Not a Shortcut
For newcomers, training matters more than anything. Every set of hands handling ethyl isocyanoacetate gets practice runs, not just a walkthrough on paper. I remember a time someone skipped refresher sessions—one rushed move, and an emergency shower ran for half the afternoon. Skills build through actual drills, not just instructions posted on a wall.
Solutions That Really Work
Use clear, visible labels on every bottle. Keep emergency equipment like eyewash stations and showers less than a stone’s throw from every point of work. Encourage a culture where double-checking isn’t nagging—too much is at risk for sloppiness or pride. Digital chemical tracking systems let everyone know what’s on hand and whether inexperienced users get blocked from working alone. These approaches foster both accountability and safety—no one gets hurt, and science marches forward without regret.
Why This Matters for Every Lab
Ignoring protocol with ethyl isocyanoacetate ends poorly, whether for new researchers or lifelong chemists. The tragedy of preventable accidents comes from forgotten steps, skipped gear, or stored bottles gathering dust. It’s not about just ticking boxes—it’s about ensuring everyone gets home unscathed, every day. Real respect in science often shows in how we treat our most dangerous tools—and in that regard, no detail proves too small.
How should Ethyl Isocyanoacetate be stored?
Understanding the Risks with Practical Eyes
Ethyl isocyanoacetate does more than just sit on a shelf. This chemical, used mostly in organic synthesis, causes real problems if it ends up in the wrong place, gets too warm, or finds its way into open air. Years around high-strung reagents have drilled one lesson home: letting your guard down even once can ruin a whole lab—and hurt people. Vapors from ethyl isocyanoacetate can cause breathing trouble or skin reactions. A single spill can fill a bench area with a sharp, irritating odor, making a safe retreat the only smart move until the mess gets controlled. That’s why storage isn’t just about a tight lid or a dark cupboard.
Keys to Good Storage—Beyond a Locked Cabinet
Every chemist I know trusts their nose to spot trouble, but a nose shouldn’t be the first alert. Ethyl isocyanoacetate stays stable if it sits in a cool, dry spot, kept apart from acids, oxidizers, and moisture. Temperature shifts in storerooms speed up decomposition or reactions. I’ve seen bottles start to sweat and bulge just from sitting in sunlight through a supply room window. So, constant temperature—ideally lower than 25°C—makes a real difference.
Moist air interacts with the reagent and makes it fall apart or react before you’ve even cracked the cap for real work. I’ve switched chemicals from leaky flasks to air-tight amber bottles and watched shelf-life triple. Using desiccators or silica gel inside the storage cupboard helps a lot, especially in humid regions where moisture sneaks in unnoticed.
Protection Means Thinking Ahead
I once found out the hard way about incompatibilities when a container of this stuff, stored too close to an oxidizer, started to leak and corrode the shelf. Since then, I always keep it isolated from acids, bases, and common oxidizing agents. Segregation may sound fussy, but it prevents 3 a.m. emergency calls.
Glass works best for containment, with a solid seal—those screw caps with PTFE liners go a long way. Open shelving never passes muster. Secondary containment, such as a plastic tray, catches leaks if the bottle fails, sparing the headache of cleaning up spilled hazardous material. Fire-proof safety cabinets, well-ventilated and clearly labeled, pay for themselves in peace of mind.
Health and Legal Responsibility
If something happens and you can’t produce records showing safe storage, authorities step in faster than you might think. Organizations like OSHA and the European Chemicals Agency don’t play around. Labels fade or peel, so frequent checks matter, not just for compliance but so coworkers know what’s inside any plain-looking bottle. Safety Data Sheets, up-to-date and posted at eye-level, back up the storage controls and make it easier when training someone new.
Personal safety comes first. Staff need gloves, goggles, and a working fume hood when handling the reagent. Respiratory protection isn’t optional if spills happen, and every proper lab keeps a spill kit ready. Having a clear, posted emergency plan for chemical exposures isn’t just policy—it stops panic and limits harm.
Thinking Long-Term
No matter how careful workers stay, chemicals break down and risks shift with each passing month. Inventory checks, regular disposal of expired stock, and reviews of storage setups coast under the radar until something goes wrong. A proactive approach keeps people and labs in business. My time in shared academic labs taught me that one person’s shortcut can trigger a group’s disaster.
Simple habits—dated inventory, tidy logs, dry shelves, and careful oversight—count more than any single piece of equipment. Ethyl isocyanoacetate isn’t the most famous chemical on a bench, but respect for its potential danger, reinforced with honest safety culture, is what separates busy, productive workplaces from tomorrow’s headline.
Is Ethyl Isocyanoacetate available for laboratory or industrial purchase?
Availability Concerns in the Chemical Market
Finding the right chemicals for research or manufacturing isn’t just about budget and demand. Ethyl isocyanoacetate draws special attention from both ends of the market. On one side, researchers look for high purity; on the other, safety regulations put up extra hurdles. Searching supplier catalogs, I noticed that access to ethyl isocyanoacetate isn’t simple. Only a handful of chemical distributors, often based in Europe or Asia, list it for sale. Even then, requests usually hit a barrier—special order status, non-stock items, or requests for additional credentials.
For chemists, tools like Sigma-Aldrich, TCI Chemicals, or Alfa Aesar often serve as benchmarks. Yet, this particular reagent pops up rarely. Listings show it as “on request” or “available for institutions only,” hinting at its limited supply and elevated risk profile. I’ve heard from colleagues who needed it for peptide synthesis or specialty transformations, and many hit the same wall—the paperwork, safety data, and procurement hurdles slow research progress.
Safety, Regulation, and Responsible Use
Isocyano compounds demand extra vigilance because of their reactivity and potential toxicity. Ethyl isocyanoacetate, in particular, falls under strict regulatory oversight. Authorities often classify it as a suspicious precursor, linking it to controlled substance manufacture due to its nucleophilic isocyanide group. Regulatory checks cover toxicology, environmental handling, and proper disposal. European REACH protocols and U.S. agencies like the DEA or EPA keep a close watch, so smaller labs may lack the licenses or dedicated storage needed to secure shipments legally.
Ordering from outside your jurisdiction isn’t straightforward, either. Importers confront customs paperwork, and sometimes, parcels face seizure. A single mistake in manifesting the chemical name or hazard class can ruin a whole project’s timeline. I’ve known postdocs who watched experiments stall for months waiting for a single vial to clear customs, even after preparing detailed risk assessments and standard operating procedures.
Why It Matters for Science and Industry
Difficulties in obtaining ethyl isocyanoacetate affect more than lab schedules. Medicinal chemists, academic researchers, and process engineers often use isocyanide reagents for Ugi reactions and other multicomponent syntheses that open doors to new drugs. Discovery slows down if scientists can't test these routes. Delays ripple through drug pipelines, feeding into higher costs and longer times for life-saving therapies reaching patients.
On the industrial side, specialty chemical producers juggle security and product quality. They can’t just buy reagents off unvetted websites; global incidents of counterfeit or contaminated chemicals have led to strict audits. Legitimate suppliers need thorough vetting, safety protocols, and clear records of each transaction, which can drive up cost and limit access for smaller players.
Suggestions for Improved Access and Oversight
Researchers, suppliers, and regulators could work together to strike a better balance. Transparent sourcing, verified distributor databases, and shared resources between institutions could help qualified professionals access these tools without skirting on safety. Communication lines open between chemical suppliers and end-users give room for negotiation on lead times, documentation, and storage conditions.
Investing in chemical inventory-sharing programs across universities or creating certified “chemical banks” lets vetted users draw upon essential materials for short-term research, sidestepping repeated orders or excessive stockpiling. These changes need trust, communication, and ongoing risk management. They also support the progress of science while respecting public safety concerns—a hard line to walk, but worth the effort.
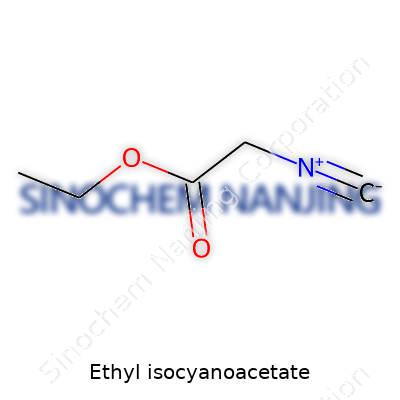
| Names | |
| Preferred IUPAC name | ethyl 2-isocyanidoacetate |
| Other names |
Ethyl 2-isocyanoacetate 2-Isocyanoacetic acid ethyl ester Ethyl isocyanomethylformate NSC 63207 |
| Pronunciation | /ˈiːθɪl aɪˌsoʊsaɪəˈnoʊæsɪteɪt/ |
| Identifiers | |
| CAS Number | 2999-45-7 |
| Beilstein Reference | 906164 |
| ChEBI | CHEBI:132965 |
| ChEMBL | CHEMBL196656 |
| ChemSpider | 132821 |
| DrugBank | DB08715 |
| ECHA InfoCard | 100.034.367 |
| EC Number | 205-956-3 |
| Gmelin Reference | 9257 |
| KEGG | C19233 |
| MeSH | D017841 |
| PubChem CID | 12511 |
| RTECS number | KK7175000 |
| UNII | 7I6I3OP9X6 |
| UN number | UN2283 |
| Properties | |
| Chemical formula | C5H7NO2 |
| Molar mass | 115.12 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | fruity |
| Density | 1.05 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 0.29 |
| Vapor pressure | 0.4 mmHg (20 °C) |
| Acidity (pKa) | 12.7 |
| Basicity (pKb) | 8.86 |
| Magnetic susceptibility (χ) | -35.87 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.423 |
| Viscosity | 1.254 cP (25°C) |
| Dipole moment | 2.91 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 344.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -152.5 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and serious eye irritation, may cause respiratory irritation, toxic to aquatic life. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS06 |
| Signal word | Warning |
| Hazard statements | H302, H311, H315, H317, H319, H332, H334, H335 |
| Precautionary statements | P210, P261, P271, P280, P301+P312, P304+P340, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-1 |
| Flash point | 87°C |
| Autoignition temperature | 455°C |
| Lethal dose or concentration | LD50 (oral, rat): 640 mg/kg |
| LD50 (median dose) | LD50 (median dose) is 640 mg/kg (rat, oral) |
| NIOSH | NA8750000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.07 ppm |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Methyl isocyanoacetate Isocyanoacetic acid Isocyanoacetamide |

