Ethyl Diazoacetate: A Deep Dive into a Versatile Chemical
Historical Development
The story of ethyl diazoacetate stretches back to the late 19th century, tracing the footsteps of organic chemists hungry for new compounds that could open up unexplored corners of synthesis. Early reports showcased this yellow liquid’s potential as a diazo transfer agent, pulling in interest from both academic and industrial labs. Pioneers such as Theodor Curtius set the stage with foundational research on diazo compounds, laying the groundwork for methods commonly seen today. By the mid-20th century, ethyl diazoacetate had found a permanent seat in the toolkits of researchers refining cyclopropanations and inserting carbene fragments into organic frameworks. Decades of evolving synthesis and a hunger for more precise, safer handling methods have shaped the landscape, fueling new routes for manufacture with an eye toward safety and efficiency.
Product Overview
Ethyl diazoacetate appears as a pale yellow, oily liquid with a sharp, pungent aroma. Its structure packs a punch—both literally and figuratively. With the molecular formula C4H6N2O2, the presence of the diazo functional group holds immense reactivity, especially in the hands of skilled chemists. Commercially, the compound comes packaged in carefully sealed containers, often under inert atmosphere to avoid contact with light or moisture. Laboratory bottles arrive with prominent safety labels, underscoring the risk that comes with mishandling volatile diazo reagents. Instead of just living on the shelf, this reactive ester finds life in countless transformations, thanks to the energy stored in the diazo moiety.
Physical and Chemical Properties
Boiling between 97°C and 99°C at atmospheric pressure, ethyl diazoacetate doesn’t play around with volatility. Its density averages about 1.2 g/cm3, and it shows limited solubility in water, preferring organic solvents like dichloromethane or diethyl ether. Open a bottle, and you’ll quickly get a whiff—ventilation is not optional. Heat, shock, and sunlight trigger decomposition, often violently. The bright yellow hue comes from the diazo group’s unique electronic absorption, easily spotted in a well-lit room. More than once, I’ve had colleagues share cautionary tales of minor skin contact leading to persistent irritation, reinforcing why those thick nitrile gloves become second nature for anyone handling this compound. On the molecular level, the presence of a highly reactive diazo group turns an unassuming ester into a powerful chemical transformer.
Technical Specifications and Labeling
Reputable suppliers bring technical insight to the table. Purity often exceeds 97%, measured by GC-MS, and each lot ships with batch-specific analytical data. Labels warn of acute toxicity, explosive potential, serious eye and skin irritation risks, and strong recommendations for personal protection. Container volumes run the gamut, from 10 milliliter research vials to 500 milliliter bottles for larger-scale labs, but the rules remain the same: store away from heat, light, and acids. Many researchers demand specifications such as minimal water and residual solvent content, aiming for reliability and reproducibility batch after batch.
Preparation Method
Most current lab syntheses start with the transformation of glycine derivatives. Reaction with sodium nitrite under strictly acidic conditions liberates nitrogen gas and yields the diazo compound in organic solution. Early processes often grappled with controlling exotherms, sometimes resulting in dangerous runaways. Over the past few decades, both continuous flow and microreactor systems stepped up, giving chemists the ability to generate ethyl diazoacetate in safer, smaller batches with greater control. These approaches reduce the risk of explosive decomposition and minimize exposure for everyone in the lab. Still, each prep has to balance yield with operator safety, and the learning curve never truly goes away.
Chemical Reactions and Modifications
The real allure of ethyl diazoacetate comes out in its chemistry. As a prime carbene precursor, it delivers razor-sharp selectivity in metal-catalyzed cyclopropanation. Ask anyone who’s tried the Simmons–Smith reaction, and they’ll nod—ethyl diazoacetate provides a more direct, versatile path. Organic chemists also turn to it for C–H insertion, O–H insertion, and ylide formation. Catalysts built from rhodium, copper, or silver unlock entire families of transformations, many of them still fueling major advances in pharmaceutical synthesis. Modifications spring up every year. Whether switching to bulkier esters for selectivity or installing chiral auxiliaries, creative chemists keep pulling unexpected results from this familiar workhorse.
Synonyms and Product Names
This compound has worn many names, depending on the context. Ethyl diazoethanoate, EDA, and Diazomethanecarboxylic acid ethyl ester most often appear in catalogues and chemical literature. Brands streamline supply under consistent tags—no matter the label, buyers remain laser-focused on purity, stability, and supplier reliability.
Safety and Operational Standards
No one I know in research or industry treats ethyl diazoacetate lightly. Any lab planning to use it needs exhaust hoods, full-face shields, thick gloves, and layered protective clothing. Standard operating protocols call for immediate disposal of any spills using absorbent, inert media, far from heat and direct sunlight. Hands-on training comes before the first reaction flask even leaves the storeroom. Emergency eyewashes and showers sit close by. Fire departments respond to accidents with full hazardous-materials gear, aware that a mishap can escalate fast. Suppliers ship in UN-certified containers, and regulatory frameworks place ethyl diazoacetate squarely among substances subject to tight control.
Application Area
The chemical finds its strongest foothold in organic synthesis, especially as a carbene source for ring construction and insertion chemistry. Drug discovery teams rely on it for building strained motifs that bear biological activity or squeeze into complex natural products. Agrochemical and flavor industries dip into its reactive core for late-stage modification and scaffold-hopping. Academic research often leans on ethyl diazoacetate for methodology development, probing reaction mechanisms or expanding the playbook for asymmetric catalysis. My own time in an industrial lab highlights one point: access to this reagent speeds up project timelines, unlocking routes that simply stall with less-active precursors.
Research and Development
Front-line research focuses on safer, greener ways to use and manufacture ethyl diazoacetate. Process chemists keep searching for alternative oxidants and nitrosating agents, with hopes of cutting out nitrite’s environmental footprint. Teams developing continuous flow systems tout not just safety gains but improved yield through enhanced mixing and stricter temperature control. Teams in academia explore new ligand frameworks for greater selectivity in cyclopropanation or C–H insertion, feeding the cycle of discovery in pharmaceutical and specialty materials innovation. My own graduate work often bumped into the problem of scaling reactions from milligrams to grams, and it became clear that innovation in reaction engineering has just as much impact as breakthroughs on the molecular side.
Toxicity Research
Toxicologists document ethyl diazoacetate as an acute toxin, with skin, eye, and inhalation exposure each posing major health threats. Animal studies report respiratory distress, convulsions, and, at higher exposures, systemic toxicity. Chronic exposure data remain limited, sparking fierce debate in regulatory circles. Waste disposal protocols demand oxidative quenching, far from drains or incinerators ill-equipped to handle diazo off-gassing. Many health and safety officers advocate for periodic monitoring and routine medical check-ins for any lab staff working with high volumes. After reviewing dozens of safety reports, I learned to respect the human cost that can come from a single lapse in handling—something a 10-minute training session just can’t cover.
Future Prospects
The horizon for ethyl diazoacetate keeps expanding, led by synthetic chemists eager for new tools. As the pharmaceutical industry embraces ever-tighter regulations, ongoing work targeting better shelf-life and less hazardous precursors will only draw more attention to this compound. Green chemistry initiatives already push for benign synthesis routes, tighter solvent control, and scalable continuous flow setups. Innovation in catalyst design will likely squeeze out better yields and new transformations, keeping ethyl diazoacetate a fixture in both classic labs and cutting-edge facilities. All signs point to a busy—and safer—future, supported by the experiences and hard lessons of those who’ve worked with this powerful reagent.
What is Ethyl Diazoacetate used for?
Inside Organic Chemistry Labs
Ethyl diazoacetate doesn’t turn up in daily life, but it shapes plenty of what happens behind the scenes in organic chemistry. In research spaces, people reach for this chemical whenever they need to stitch together complex molecules. It looks like a clear, yellow liquid, and chemists appreciate the way it reacts with other substances, especially for tricky steps where they need to add a carbon atom right in a precise place. The active part of the molecule, known as the diazo group, tends to grab the spotlight. This part makes ethyl diazoacetate very reactive, and as a result, it finds regular use in making cyclopropanes, which are tiny, triangle-shaped molecules that fit into medicines and pesticides.
Pharmaceutical Synthesis
Building new drug compounds often demands the kind of special reactions ethyl diazoacetate can provide. One example is the Wolff rearrangement, which helps chemists transform starting materials into more valuable molecules, sometimes even adding pieces that mimic natural substances in the body. Industry reports and scientific journals often describe how these steps make the difference between a failed project and a novel medicine. Sometimes, the only tool for the job is a diazo compound like this. In the past, people relied on older, messier methods, but ethyl diazoacetate offers more control. Fewer unwanted leftovers means purer medicines and less environmental clean-up.
Role in Academic Research
Graduate students and postdocs in university labs learn to respect the power and hazards of ethyl diazoacetate. They wear layers of safety gear since it can explode with rough handling. Everyone remembers the training around diazo chemistry, because one mistake can end up as a lesson for the whole department. Stories float around the lab about syringes clogging or bottles venting unexpectedly. Mistakes teach people to double-check their set-ups, keep reaction scales small, and run reactions in blast shields or fume hoods. The chemical’s danger means that it’s not usually a teaching tool for beginners. But for advanced students, mastering a reaction involving ethyl diazoacetate opens doors to cutting-edge projects and can even launch careers in pharmaceutical research.
Challenges and Safer Alternatives
Safety remains the biggest challenge with ethyl diazoacetate. The very property that makes it useful—strong reactivity—can make it unpredictable. It demands patience and attention to detail. Over the past decade, researchers have been searching for less hazardous substitutes or safer protocols. New reactor setups, such as flow chemistry, allow for smaller amounts of the chemical to move through tubes, reducing the risk of explosive build-up. Still, nothing has yet matched the versatility for every reaction where chemists want to use ethyl diazoacetate.
The Road Ahead
If demand for new therapies and advanced materials keeps rising, more people will want to work with tricky but useful reagents. Industry partnerships with university research groups may help refine safety guidelines even further. From my own experience seeing chemists debate whether to scale up a reaction or keep it limited to small vials, it’s clear that careful respect for ethyl diazoacetate’s power must always guide its use. Progress in chemistry usually comes with trade-offs between safety, new discoveries, and practical application. No single molecule demonstrates that balance quite like this one.
What are the storage and handling precautions for Ethyl Diazoacetate?
Understanding the Risks
Ethyl diazoacetate catches the eye in a typical organic chemistry lab. The yellow liquid may sound friendly, often popping up in textbooks for cyclopropanation and insertion reactions, but it demands real respect. I’ve worked with it in my own research, and it left a lasting impression—literally, since even a small whiff when uncapped sends a sharp, unpleasant odor straight up your nose. This chemical, with its diazo group, stands out for being a potential explosion risk, even at room temperature.
Storage: No Corners to Cut
A few careless mistakes—leaving the bottle out after use, stacking incompatible chemicals on the same shelf—can turn an ordinary workday into a dangerous mess. Ethyl diazoacetate sits on my shelf inside a tightly sealed amber glass vial, stashed far away from light and heat sources. The fridge or a dedicated explosion-proof freezer works best for this. My PI drilled into us the rule of never using domestic fridges: even the faintest spark from a compressor can ignite vapors from volatile chemicals.
Avoid storing ethyl diazoacetate near acids, alkalis, or metal powders. I’ve heard a story where a careless mix-up left it next to a strong acid, and the resulting gas could have caused a bigger problem if an alert tech hadn’t caught it quickly. Silica gel in the storage box soaks up stray moisture, since water contamination can set off slow decomposition. My colleagues always marked the storage area with bold labels, not only for compliance but to remind everyone that a single slip-up with this stuff risks more than just a ruined experiment.
Handling: Treat Every Step Like It’s Hazardous
Personal protection turns from suggestion into necessity with ethyl diazoacetate. I learned to use full nitrile gloves, goggles, and a buttoned lab coat, with the fume hood sash kept as low as possible. It’s not just about protecting your hands; the vapor from this chemical stings the nose, and longer exposures threaten the lungs. Splash risk drove us to use weighed-out micro amounts with pipettes and syringes instead of pouring, which makes spills much less likely. No one wants to clean up a diazo spill off the bench or the floor—the risk of an unexpected bang or toxic vapor lingers long after the initial incident.
Disposal and Emergency Measures
Every bottle carries its own limited shelf life. Old bottles can turn especially hazardous; we learned to check for crystals or colored residues around the lid—warning signs the contents may have started to decompose. Fresh solutions get made just before use, never stockpiled. If any spill occurs, evacuating and letting the professionals handle it always trumps quick DIY fixes. Spill kits meant for diazo compounds and immediate reporting to safety teams saved a friend from what could have been a major accident. Local fire codes and university guidelines all spell out very specific disposal methods, usually involving slow, controlled neutralization under supervision.
Real-World Solutions for Safer Labs
Labs run best with redundancy and communication. Someone always checks the dated log on reactive bottles. Training new students on ethyl diazoacetate happens early, never left to chance. Color-coded storage, strict limits on purchase size, and regular audits keep everyone honest and aware. Investing in better ventilation and blast shields feels like a no-brainer after hearing enough stories of near misses.
Respect for ethyl diazoacetate makes good science possible without trading away safety. In my experience, nothing matches the value of peer support, good training, and clear procedures. Remembering the stories that circulate after every safety incident reminds us all why we follow these precautions—no experiment justifies the risk of ignoring best practices in handling a tricky reagent like this one.
Is Ethyl Diazoacetate hazardous or explosive?
Chemicals in the Real World
Ethyl diazoacetate sounds like something reserved for the back corners of a university lab, but it’s become a staple in synthetic organic chemistry. New chemists coming into the field often hear warnings about its bright yellow color, odd smell, and potentially hazardous properties. Knowing how this chemical acts in the real world—outside of textbooks—can make all the difference.
Dangers Hidden in Small Bottles
Anyone who’s ever worked with ethyl diazoacetate knows stories float around labs about containers popping under pressure or bottles shattering after being left in the sunlight. The truth isn’t all rumor. Ethyl diazoacetate belongs to the family of diazo compounds, known for containing nitrogen double bonds that sometimes snap apart with very little provocation. That gas release can translate directly to explosive behavior. Even small bottles, if stored incorrectly or shaken too much, may develop higher pressure. On top of that, it’s flammable, and the vapors pose inhalation risks.
There’s a reason manufacturers slap hazard symbols all over these bottles. A simple review of the Material Safety Data Sheet will tell you ethyl diazoacetate is both toxic and potentially explosive. The biggest risk isn’t from a single drop landing on a glove; it’s from what happens if a larger amount gets exposed to open flame, heat, or rough handling. The energy stored in the diazo group can get released in a flash, which sometimes means disastrous lab accidents.
Lessons from the Lab Bench
Speaking with colleagues and training newcomers, I’ve stressed the impact of treating certain reagents more like live ammunition than simple tools. Watching old videos of chemical demonstrations, you see the controlled detonations of diazo compounds and their decomposition pathways. The possibility of uncontrolled release makes every professional careful.
Back in undergraduate synthesis, I watched an experienced postdoc use a blast shield every time diazo compounds entered the setup. Even years into my own experience, that memory sits close. One miscalculation, or a forgotten step in the safety checklist, and a harmless-looking experiment could escalate fast. Accidental releases won’t only harm the person handling the bottle but risk others in the room.
Why Proper Handling Matters
In 2018, the United States Chemical Safety Board reported multiple incidents tied to mishandled energetic chemicals. Often, these substances defeat even experienced hands because safety measures slip—protective eyewear gets replaced with prescription glasses, or bottles migrate out of the fridge. For ethyl diazoacetate, using a fume hood and carefully documented transfer procedures is standard. Ignoring these steps isn't just risky—it’s negligent.
Engineers and safety officers encourage training, clear labeling, and frequent safety drills. These aren’t just for formalities. With compounds like ethyl diazoacetate, a five-minute rundown on proper waste disposal or the difference between saving and discarding off-spec solutions can save a life. Glass vials must never fill to the brim, since temperature changes can induce pressure spikes without warning.
Moving Toward Safer Chemistry
Research keeps evolving. Scientists look for less hazardous alternatives or methods allowing for safer slow generation of diazo intermediates. Some universities even publish their near-miss stories, sharing both scares and lessons. These open conversations drive improvements in bottle design, storage methods, and even insurance requirements for hazardous lab work.
Ethyl diazoacetate demands respect, focus, and a willingness to stop a process if something feels off. The chemical’s value in synthesis stays high, but so does the need for awareness. Chemistry, after all, rarely rewards shortcuts, especially with reagents this energetic.
What is the chemical structure and formula of Ethyl Diazoacetate?
Understanding Ethyl Diazoacetate from the Ground Up
Ethyl diazoacetate might sound like a compound for specialists, but it connects to plenty of discoveries in organic chemistry. For anyone tackling complex reactions or making new molecules, knowing its structure unlocks a bigger toolbox for creativity in synthesis. Let’s break this down and show why this simple-looking molecule punches above its weight.
Getting to the Core Structure
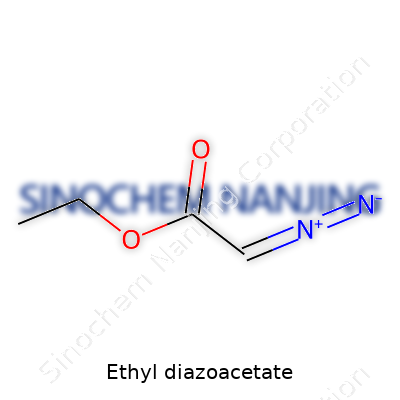
Ethyl diazoacetate’s chemical formula is C4H6N2O2. Picture two main pieces linked together: a diazo group (–N2), and an ethyl ester group (–COOCH2CH3). Looking at its line structure, you see CHN2 (the diazo bit) sitting next to a carbonyl group, which holds onto the ethoxy chain. The full IUPAC name, ethyl 2-diazonoacetate, reflects the placement of the reactive diazo group right next to the carbonyl carbon.
Chemists often draw it like this:N2=CH–COOCH2CH3
Even if the formula looks simple, the arrangement of these groups is what sets reactivity in motion.
Importance Rooted in Reactivity
In practical lab settings, ethyl diazoacetate acts as a gateway for carbene chemistry. This molecule stands as one of the classic sources for diazo compounds since the late 19th century. With the diazo bit attached to the right spot, chemists can generate carbenes—those short-lived intermediates that let you build rings, make new bonds, or insert into existing ones. For students and professionals alike, this means discovering opportunities for new reactions, green chemistry, or access to different molecular backbones. People who work in pharmaceutical or agrochemical research encounter it in ring-expansion reactions, cyclopropanations, and as a starting point for more complex chemicals.
Handling and Hazards: Why Experience Matters
It’s hard to talk about ethyl diazoacetate without mentioning safety. The diazo group can decompose with minimal provocation, producing nitrogen gas. While that’s useful in controlled setups, mistakes in handling lead to pressure build-up and risks. From my early days working with diazo compounds, the lesson always stuck: wear goggles, keep volume small, and use shielding. Respecting these conventions keeps the process safe and reproducible.
Lab manuals and safety datasheets back it up. The European Chemicals Agency (ECHA) lists ethyl diazoacetate as both toxic and potentially explosive. Training and strict supervision matter more than any shortcut with such reagents.
Paths Forward: Safer Synthesis and Applications
Over the past decade, more chemists focus on steady, slow-release diazo sources to address the safety problem. Encapsulated or polymer-bound diazoacetate provides a friendlier alternative, reducing risks. Microfluidic reactors and flow chemistry setups give another route, letting small amounts react at a time. These advances mean the field keeps growing without adding unnecessary danger.
Ethyl diazoacetate shows how one molecule can shape research and technology. It keeps people on their toes and rewards those who treat it with respect. The formula may be short, but its reach in discovery is broad.
What are the common applications of Ethyl Diazoacetate in organic synthesis?
Breaking Out of the Lab Textbook
Ethyl diazoacetate shows up on the bench as a yellowish liquid with a not-so-subtle reputation. Unlike many reagents, this compound doesn't just end up sitting around waiting for a moment to shine; it brings action to modern labs, especially in the hands of both graduate students and experienced chemists looking for smoother synthetic routes.
Why Alkylation and Cyclopropanation Matter
One of the first places I learned to respect ethyl diazoacetate was in cyclopropanation reactions. It acts as a source for carbene generation. In practice, this means a chemist gets to take a simple alkene, add a metal catalyst like copper or rhodium, and turn that flat molecule into a three-membered ring, fast. Cyclopropane rings have carved out their own place in both pharmaceutical research and natural product synthesis. They show up in everything from antivirals to insecticides, thanks to their unique strain and reactivity.
Ethyl diazoacetate's role doesn't just end in small-scale tests; it's been the workhorse for building rings that otherwise would need expensive or unpredictable reagents.
Into the Toolbox: C–H Insertion Reactions
Ethyl diazoacetate stands out for C–H insertion reactions as well. C–H bonds are everywhere, but making one carbon hop onto another where there’s no obvious leaving group takes guts and a little danger. Ethyl diazoacetate, under the right metal catalyst, lets chemists skip protecting groups, as well as extra manipulations. That saves both time and solvents, which is always appreciated when scaling up reactions or chasing yields.
Working With Asymmetry
Ask anyone who cares about drug development, and they’ll point to the value of chirality. Ethyl diazoacetate slides neatly into asymmetric synthesis. Chemists have paired it with sophisticated chiral catalysts that tip a reaction toward one enantiomer over the other. During an internship, I watched this compound used in a rhodium-catalyzed reaction to give a single, desired stereochemistry, cutting out the slog of separating mixtures later.
Peer-reviewed sources routinely share how this route has led to progress in new antibiotics and cardiovascular drugs with fewer side reactions than alternatives. The proof can be seen both in patents and in the published yields.
Problems That Don’t Disappear
Ethyl diazoacetate won’t win any green chemistry awards on its own. It’s got a reputation for volatility and can give off nitrogen gas with a bang if handled carelessly. I’ve seen more than one researcher duck when a reaction vessel vented unexpectedly. Its reactivity demands serious respect, which means beginners need real training and the right fume hood.
Better Options on the Horizon?
There’s a growing push for safer, scalable, and more environmentally friendly carbene transfer agents. Flow chemistry offers one possible solution, because small volumes and controlled conditions help reduce the risks with diazo compounds. Researchers are also exploring stable precursors that generate ethyl diazoacetate only as needed in microreactors, which limits the amount on hand at any given time.
Looking forward, some teams focus on new catalysts — using iron instead of rhodium, for example — which pushes these reactions toward both safety and cost-effectiveness.
The Real Value in Synthesis
Despite the caveats, ethyl diazoacetate hasn’t lost its edge. My own experience matches the consensus in the literature: this molecule is a tool for serious, reliable, and creative chemistry, so long as it’s treated with the respect it’s earned over decades.
| Names | |
| Preferred IUPAC name | Ethyl 2-diazanoacetate |
| Other names |
Diazoacetic acid ethyl ester EDA Ethyl alpha-diazoacetate Ethyl diazoethanoate |
| Pronunciation | /ˌiːθɪl daɪˌæzoʊəˈsiːteɪt/ |
| Identifiers | |
| CAS Number | 623-73-4 |
| 3D model (JSmol) | `Ethyl diazoacetate JSmol string`: ``` CCOC(=O)CN=[N+]=[N-] ``` |
| Beilstein Reference | 1208732 |
| ChEBI | CHEBI:52276 |
| ChEMBL | CHEMBL34285 |
| ChemSpider | 7179 |
| DrugBank | DB03716 |
| ECHA InfoCard | 100.013.771 |
| EC Number | 209-550-4 |
| Gmelin Reference | Gmelin 79238 |
| KEGG | C18701 |
| MeSH | D003375 |
| PubChem CID | 61110 |
| RTECS number | KJ9175000 |
| UNII | G87Q02U2G6 |
| UN number | UN1992 |
| CompTox Dashboard (EPA) | DTXSID9051805 |
| Properties | |
| Chemical formula | C4H6N2O2 |
| Molar mass | 116.09 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | ester-like |
| Density | 1.204 g/mL at 25 °C (lit.) |
| Solubility in water | Soluble |
| log P | 0.05 |
| Vapor pressure | 0.59 mmHg (20°C) |
| Acidity (pKa) | 11.5 |
| Basicity (pKb) | pKb = 6.24 |
| Magnetic susceptibility (χ) | -28.3×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.418 |
| Viscosity | 13 cP (20 °C) |
| Dipole moment | 2.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -53.98 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -991.0 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS02, GHS06 |
| Signal word | Warning |
| Hazard statements | H225, H301, H311, H331, H370 |
| Precautionary statements | P210, P261, P280, P301+P312, P304+P340, P305+P351+P338, P337+P313, P370+P378 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | -18 °C |
| Autoignition temperature | > 140°C (284°F) |
| Lethal dose or concentration | LD50 (oral, rat): 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): 130 mg/kg (rat, oral) |
| NIOSH | SKT |
| PEL (Permissible) | Not established |
| REL (Recommended) | 200–500 mg |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Diazomethane Methyl diazoacetate Diethyl azodicarboxylate Diazoacetic acid Phenyl diazoacetate |

