Ethyl Cyanoacetate: Tracing Its Journey, Adaptation, and Role in Science
Looking Back: Historical Development
Ethyl cyanoacetate has traced a long and winding road from its early days in organic chemistry labs to its present status as a linchpin in synthetic research. Chemists first explored this small, sharp-smelling ester to harness its potential as a versatile building block for synthesizing pharmaceuticals, dyes, and agricultural products. Through the decades, it threaded its way into core reaction schemes in both academia and industry. The push to create efficient, reproducible pathways in chemical synthesis fueled interest in compounds like ethyl cyanoacetate that offered flexibility and multiple reactive sites. My experience points to how discoveries in the old bench labs of the 20th century shaped modern organic chemistry, with ethyl cyanoacetate often popping up as a key ingredient in both publications and patents.
Product Overview: Everyday Use and Appeal
There’s something practical about a compound that blends affordability and usefulness the way ethyl cyanoacetate does. Its utility boils down to a trifecta: a reactive methylene group, an easily handled liquid form, and decent solubility in many common solvents. I remember colleagues relying on it for preparing intermediates in drug synthesis, where speed and flexibility matter most. Its faint almond-like scent in the lab sometimes pointed to a new project in the making. Today’s chemists reach for it, knowing that it forms new carbon-carbon bonds with ease, making what seemed a tough synthesis only a few steps away. Its role feels less glamorous than the final pharmaceutical product, but anyone working on routes to new molecules probably bumped into it before reaching their patent.
Physical & Chemical Properties: How It Acts in Practice
Ethyl cyanoacetate presents itself as a clear, colorless liquid with a low flash point and a sweet, almost fruity odor. Its boiling point runs around 206 °C, with a melting point well below standard room temperatures, so it never crystallizes in the bottle. The main draw comes from a CH2 group set between an ester and a nitrile, causing high acidity for that methylene hydrogen. That tiny change matters; it lets the molecule participate in Knoevenagel and Michael reactions, serving as a launching pad for many derivatives. In my hands, it seemed easy to weigh, poured smoothly, and cleaned up readily with basic lab solvents. Its lower volatility compared to other esters makes storage less tricky, but the strong odor reminds you to cap the bottle fast.
Technical Specifications & Labeling: Production and Purity Close-Ups
Producers usually aim for high-purity lots to keep up with demands in fine chemicals, with specifications calling for assays above 98 percent. Color, water content, and acidity get close scrutiny since impurities change how it behaves in sensitive reactions. Chemists look out for telltale discoloration—yellowing signals breakdown or oxidation, which can derail planned syntheses. Common labeling emphasizes hazards and regulatory language. Safety data sheets always show warnings about skin and respiratory irritation, so many labs stick to fume hoods when handling open containers. In my experience, younger researchers learn early to respect labels and double-check bottle caps.
Preparation Method: Lab Bench to Factory Floor
Early approaches to making ethyl cyanoacetate relied on simple chemistry: reacting cyanoacetic acid with ethanol through an acid-catalyzed esterification. Scaled-up batches in factories swapped glassware for reactors, fine-tuning catalyst choice and solvent ratios to boost yield and minimize waste. Over time, attention shifted to greener routes, with milder catalysts and lower energy input because sustainable chemistry moved from fad into necessity. In university courses, instructors show students the classic route as an example of how functional group transformations occur, and some advanced labs seek out alternative starting materials, especially when fossil-based chemicals face scrutiny. Seeing the shift to cleaner, more efficient processes convinced me that chemistry adapts quickly when the stakes include both cost and environmental impact.
Chemical Reactions & Modifications: The Real Action
Ethyl cyanoacetate plays a starring role in synthesis projects thanks to its lively methylene group, which works well for alkylation, condensation, and cyclization. Traditional reactions like the Knoevenagel condensation pair it with carbonyl compounds, and Michael additions let it tack on to double bonds, opening up a world of heterocyclic chemistry. Skilled chemists run with these reactions to develop sedatives, antihistamines, pesticides, and fluorescent dyes. Some reach for it to construct barbiturates or pyrimidines, and the push for new antiviral drugs relies on it as a scaffold. From my own projects, I saw how small tweaks in conditions—temperature, base strength, solvent—flip yields dramatically. That kind of hands-on chemistry keeps research lively and full of surprises.
Synonyms & Product Names: What’s on the Label
No matter how often a chemical appears in literature, confusion over names crops up at every turn. Ethyl cyanoacetate also turns up as cyanoacetic acid ethyl ester and a few legacy trade names hanging on in catalogs. Abbreviations like ECA may crop up on bottle labels, especially in older labs. I’ve found myself double-checking synonyms in catalogues to make sure I grabbed the correct bottle, since a small slip turns an experiment sideways. Consistency in naming not only prevents mix-ups, but also underpins everything from ordering supplies to searching global patent records, so researchers tend to keep synonym lists close at hand.
Safety & Operational Standards: Staying Smart
Lab life brings with it a host of safety considerations, and ethyl cyanoacetate sits squarely among compounds needing careful handling. Even short exposure to its vapor can irritate skin, eyes, and lungs, especially in closed spaces or without good air flow. Proper gloves and goggles count as standard gear, and spill kits should be kept at the ready in a busy organic chemistry lab. Instructors drill safety procedures into students because shortcuts end in costly mistakes. I saw firsthand how rapid cleanup and good ventilation kept minor spills from escalating. Regulatory bodies set up exposure limits and outline safe disposal, making chemical stewardship less a legal box-tick and more a matter of personal and lab-wide responsibility.
Application Area: Real-World Uses
Ethyl cyanoacetate has kept its niche in synthesis, but the range of end products it helps create continues to widen. Drug makers rely on it to put together molecules for treating epilepsy, insomnia, and infections—the benchtop reactions ripple out to pharmacy shelves worldwide. Agrochemical firms use it as a precursor for herbicides and growth regulators, helping deliver tools for food production that keep up with population growth. Dye and pigment companies also draw from its versatile chemistry to bring new colors to fabrics and inks. I’ve seen graduate students set up week-long syntheses turning ethyl cyanoacetate into fluorescent molecules that end up in imaging tools or even forensic science. Its adaptability has made it a reliable anchor in industries that need chemical innovation.
Research & Development: Pushing Boundaries
Workshops and research journals keep turning out new ways to use ethyl cyanoacetate, often with a focus on catalysis or environmental sustainability. Researchers look to tighten up reaction yields, cut down on hazardous waste, and coax the molecule to fit into more elaborate architectures. Recent projects home in on asymmetric synthesis, aiming to produce single-enantiomer pharmaceuticals that work better and come with fewer side effects. I’ve spoken with chemists trying to tether cyanoacetate to biodegradable polymers, eyeing greener materials for future electronics. This drive to keep refining techniques extends to universities and corporate labs alike, feeding a steady stream of new data to patents and publications.
Toxicity Research: Probing Risks and Limits
Toxicology work helps set the boundaries for safe use of ethyl cyanoacetate. Acute exposure triggers skin and respiratory irritation; with higher doses, animal studies report muscle tremors, ataxia, and even convulsions. Chronic tests on rodents show liver and kidney effects, though human data remain scattered outside occupational case reports. Risk assessments blend animal results with real-world exposure scenarios to inform worksite ventilation and set ceiling concentrations wherever it’s handled. In my own academic work, proper ventilation and personal protective equipment stood out as the difference between safe research and regrettable mistakes. Bad habits in handling this compound catch up fast, which is why every responsible supervisor drives home safe practices.
Future Prospects: On the Horizon
With sustainability climbing industry agendas, ethyl cyanoacetate’s future ties closely to cleaner, smarter reaction processes. As more chemists explore non-fossil starting materials and low-impact catalysts, this old staple will find itself rethought in light of green chemistry. Pharmaceutical and agrochemical pipelines show no signs of letting go of compounds rooted in cyanoacetate chemistry, especially since their cost and accessibility offer an edge over expensive designer reagents. The growing demand for fine chemicals built from efficient, reliable precursors helps keep ethyl cyanoacetate on the shopping list for any lab looking to innovate. From what I’ve seen at conferences and workshops, conversations have shifted from simple yield boosts to lifecycle analysis and long-term impact, so practical know-how with this molecule will keep paying off in the years ahead.
What is Ethyl Cyanoacetate used for?
From Laboratory Benches to Household Goods
Ethyl cyanoacetate probably doesn’t sound familiar unless you’ve dabbled in a lab or read the ingredients on a tube of super glue. It’s a clear liquid, not much to look at, but it's behind several things that make a difference in our lives. Chemists like myself know it as a small molecule with a big range of uses—from pharmaceuticals and agricultural products to materials you find around the house.
Super Glue’s Secret Weapon
It’s hard to forget gluing your fingers together as a kid—or as an adult—using super glue. That instant bond comes from cyanoacrylate adhesives, and ethyl cyanoacetate plays a central role in making them. This chemical forms the backbone of the famous quick-bonding reaction. Anyone fixing broken mugs, shoe soles, or even medical professionals closing wounds benefit from its quick-curing properties.
Medicine’s Hidden Hand
Ethyl cyanoacetate doesn’t grab headlines in the world of health, but it acts as a starting point for several drug compounds. Pharmaceutical research teams use it to build molecules for anti-cancer, anti-viral, and anti-convulsant medicines. Its structure helps craft heterocyclic compounds, the core of a lot of medicines that keep people out of the hospital or help them recover quicker. Drug makers prize it for its versatility and reliability in making new therapies possible.
Agriculture and Everyday Products
Stepping outside the lab, ethyl cyanoacetate finds a role in pesticide production. It serves as a building block for chemicals that target harmful insects, protecting food crops that end up on store shelves. For the people working in fields or managing orchards, safer and more effective pesticides cut down on crop loss and raise yields, helping food security and rural livelihoods.
Risks and Responsible Use
With chemical production, the conversation must include safety and environmental protection. Ethyl cyanoacetate, like many chemicals, calls for careful handling: it can cause skin and eye irritation. Factories need proper ventilation, staff use gloves and goggles, and spills require immediate clean-up. Down the line, companies face regulations on how they dispose of it. Some countries enforce tough standards to make sure runoff or fumes don’t hurt workers, folks living nearby, or wildlife.
Balancing Innovation and Responsibility
Relying on chemicals like ethyl cyanoacetate brings a responsibility to innovate safely. Researchers keep searching for greener processes—making adhesives and medicines with fewer toxins or using renewable sources. It reminds me of conversations with chemist friends who push for new ways to get the same result with less environmental impact. Approaches like continuous flow manufacturing can lower waste and energy use, offering a future where chemistry keeps benefiting people without leaving a heavy environmental footprint.
Community Role in Safe Chemistry
People and policymakers can steer the future by demanding transparency. Clear labeling on household products, frequent inspections at production sites, and publicly accessible reports hold companies accountable. It pays to remember that the everyday items in our homes often have a long, complex story behind them, from raw materials to finished products. Awareness helps protect our health and the world we share.
What are the storage conditions for Ethyl Cyanoacetate?
The Real Risks of Ethyl Cyanoacetate
In my early chemical work, handling ethyl cyanoacetate rarely drew much attention until someone left a half-open bottle on a warm windowsill. The sharp, irritating smell let us know right away this stuff reacts to its surroundings. Ethyl cyanoacetate does not act like water; unmanaged, it evaporates and can even give off toxic hydrogen cyanide. So, the location and method of storage influence both safety and quality over time.
Temperature and Ventilation Decide a Lot
Keep ethyl cyanoacetate somewhere cool. High temperatures speed up decomposition and evaporation, raising toxic vapor risk. Industry standards push for room temperature or lower—around 15-25°C. Even at these temperatures, a slow breakdown can occur, so chilling it below 15°C, without freezing, extends its shelf life. Ordinary fridges can work, though a designated chemical refrigerator is a better option.
Good airflow helps manage vapors. In my experience, cramped shelves and closed, stuffy cabinets trap odor and make spills more likely to go unnoticed. A well-ventilated chemical cabinet, ideally with a spot for volatile organics, limits risks if a bottle leaks.
Light, Moisture, and Material Compatibility
Sunlight shouldn’t hit those glass bottles directly. Light sets off certain decomposition reactions, and ethyl cyanoacetate can yellow or degrade. Even if you can’t see much change, trust that reactions are happening that may affect purity or producing hazardous fumes. Store in amber bottles or a dark cupboard.
Dryness counts. Ethyl cyanoacetate reacts to water, generating cyanoacetic acid and ethanol. Humid conditions in old labs led to surprises—thicker, sometimes cloudy liquid and a drop in expected reactivity. A good, tight cap keeps moisture out, and a couple of silica gel packs in the cabinet won’t hurt.
Some folks use plastic containers, but solvents gradually attack many plastics. Glass stands up better against leakage or reaction. Corrosion-resistant shelving or secondary containment trays help catch spills. After a busy semester, I once found a corroded metal shelf with a sticky, slightly blue stain—proof metal contact invites unwanted side reactions.
Labels, Segregation, and Security
Label every bottle clearly—not just the substance name, but date of receipt or opening. Over time, bottles accumulate dust and the labels fade, so a yearly audit is practical. My own system involved color-coded labels for reactivity; ethyl cyanoacetate pairs better with organics and non-oxidizing chemicals. Keep it far away from acids, bases, and oxidizers.
Security isn’t just about theft. Ethyl cyanoacetate sits on some watch lists because of its use in illegal synthesis. Lock it away after hours. Support staff and new lab workers benefit from explicit instructions and updated safety sheets posted nearby.
Dealing With Spills and Disposal
Spills need immediate attention, not tomorrow’s shift. Absorb with sand, ventilate, and dispose according to local hazardous waste guidelines. Leftovers from undergrad labs sit too long in forgotten corners. I’ve seen graduate students argue about what’s in the unmarked bottle; these debates don’t belong around dangerous chemicals.
Better Storage Means Less Risk
Safe storage starts with attitude—treating every bottle as if a leak could end up in the hospital. Regular checks and thoughtful placement keep everyone safe, save money in lost inventory, and cut down on those chemical headaches from poorly stored compounds.
Is Ethyl Cyanoacetate hazardous or toxic?
Looking Beyond the Lab Bench
People run into chemicals every day, but not many outside of industries or research labs recognize names like ethyl cyanoacetate. Most folks just want to know, will this hurt me? This liquid, with a sharp smell, pops up in fragrance-making, medicines, adhesives, and sometimes even in undergraduate chemistry experiments. The issue with ethyl cyanoacetate isn’t just the name—it’s the mix of risks that make people wonder about its safety.
What Science Says About Toxicity
I’ve spent plenty of bleary-eyed hours in university labs and chemical storerooms, so I can tell you: labels marked “toxic” or “hazard” grab your attention. Ethyl cyanoacetate deserves a place on that list. If someone gets it on their skin, it can irritate and even blister. Breathing in too much vapor starts to burn nose and throat, and eye contact brings stinging tears. Cases exist where high exposure injured the lungs or central nervous system. It’s no cyanide, but it’s not apple juice, either.
Data from the European Chemicals Agency and the U.S. National Institute for Occupational Safety and Health agree: direct contact can damage tissue, and too much breathing of fumes may leave someone with chest pain, headaches, or worse if they are sensitive. Animal studies link high exposure to impacts on the liver and kidneys—organs you don’t want to mess with lightly.
What Makes It Useful—and Risky
This chemical holds value in making things stick, combining parts in drug molecules, and even giving perfumes a whiff of fresh fruit. Chemists like it for its reactivity. Yet, every advantage comes with a caution sign. It can go up in flames with the right mix of air and spark. Spills cause a sharp odor that signals irritation long before you see the liquid. Years ago, a small bottle cracked in my university’s storage: within minutes, classmates nearby covered their faces and left the room.
Staying Safe: Knowledge Beats Fear
Taking precaution doesn’t mean swearing off all chemicals. Education means more than memorizing dangers; it means learning proper storage, using gloves and goggles, and having chemical hoods vent away fumes. Employers and school labs need to keep material safety data handy and encourage everyone to recognize the warning signs—skin tingling, coughing, watery eyes—so nobody shrugs off a problem.
Disposing of unused stock also matters. Dumping it down the drain risks harming water treatment systems. Chemical waste collectors exist for a reason. National and local rules usually require special handling, with labeling and secure containers, to cut down on accidental exposure.
Better Alternatives and Future Steps
The push for “greener” chemistry has helped scientists hunt for replacements—new compounds with similar performance but fewer health worries. Increasing public awareness about chemicals like ethyl cyanoacetate might convince companies and labs to look at alternatives, or at least tighten up training and emergency preparedness.
People overlook the everyday impacts of chemicals because they seem distant from daily routines. But risks come quietly, building up over time or turning serious after one careless moment. Treating ethyl cyanoacetate with respect, investing in safety gear, and keeping information transparent stack the odds on the side of safety.
What is the chemical formula of Ethyl Cyanoacetate?
Ethyl Cyanoacetate: Unpacking the Formula
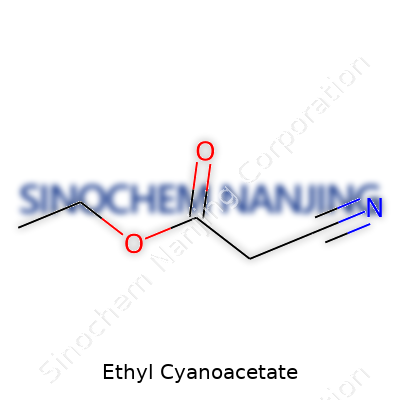
Ethyl cyanoacetate has the chemical formula C5H7NO2. In everyday chemistry, this formula isn't just a pattern of numbers and letters. It actually reveals how this molecule works, where it can fit into larger chemical puzzles, and why people across industries care about it.
Inside the Structure
Take a moment to picture the skeleton of ethyl cyanoacetate. The “ethyl” part signals the presence of two carbon atoms and five hydrogens (C2H5). “Cyanoacetate” means you're dealing with a nitrile group (-CN) stuck onto an acetate core. The full formula, C5H7NO2, covers every atom—carbon, hydrogen, nitrogen, and oxygen—in this compact structure.
Looking at the connections, the molecule includes a nitrile group bonded directly to a carbon, an ester function, and a chain that further opens options for reactions. This isn’t just textbook talk. The way chemists use ethyl cyanoacetate in real life—whether blending pharmaceuticals, paints, or adhesives—all comes back to this exact arrangement of atoms.
Why Exact Formulas Matter in Practice
Accuracy saves time, money, and resources in any lab or factory. I’ve seen chemists, myself included, double-check a bottle’s label and spot a missing atom in the formula—leading to a product callback or a halt in production. One wrong digit could disrupt a whole batch. Students also memorize these formulas, not just to pass exams, but to make real, physical products. C5H7NO2 isn’t just something to write on a board; it’s tied to a tangible molecule with distinctive properties.
A wrong formula means a wrong starting point. For example, a scientist might aim for a certain pharmaceutical and wind up with a compound that doesn’t perform as expected. The right structure, described correctly, sets the stage for safe manufacturing and effective results.
The Role in Industry
Ethyl cyanoacetate's formula allows it to become a building block for other chemicals. In my experience, it has been used in synthesizing medicines, creating jump-starts for glue production, and helping turn out coatings that last. Its cyano group enables big transformations—nitrile groups regularly serve as stepping stones in organic synthesis.
Safety teams and regulators need the formula for another reason: risk management. The functional groups in ethyl cyanoacetate make it flammable and potentially hazardous in large amounts. Plants tracking C5H7NO2 don’t just want compositional clarity; they need it for compliance. The correct formula keeps people safe and businesses legal.
Looking for Solutions in Chemical Education
Getting students and new employees comfortable with structure and formula goes beyond rote memorization. Hands-on practice, such as drawing molecules, running simple syntheses, or breaking down formula derivations, helps everyone—rookie or veteran—lock the right numbers in memory. I’ve seen mentorship and real-world case studies turn confusion about chemicals into practical skill. Digital tools that confirm formula correctness—scanners, molecule drawing apps—have started to become mainstays in labs, boosting reliability and building confidence.
The Facts Stick
Ethyl cyanoacetate comes down to C5H7NO2, a formula that shapes so much of what it does in labs and the market. Knowing those details isn’t trivia. It makes safer workplaces, sharper research, and more reliable products. In the ever-moving world of chemistry, facts grounded in practice always make the difference.
How do you safely handle Ethyl Cyanoacetate in the laboratory?
Understanding Ethyl Cyanoacetate’s Risks
Many folks who spend long hours in chemistry labs come across ethyl cyanoacetate. Standard use involves reactions making dyes, medicines, and agricultural compounds. Something about this liquid—its sharp smell, its tendency to irritate—means you pay close attention before opening the bottle. Studies point out it can irritate skin, eyes, and airways. Even a handful of drops splashed or spilled spells a bad day, so one look at the label and most researchers want every sensible precaution on their side.
Old-School Precautions Still Work Best
Preparation means more than grabbing gloves and calling it done. Nitrile gloves hold up better than basic latex, since ethyl cyanoacetate soaks right through weaker stuff. In my own grad days, I watched more than one eager undergrad skip thick gloves, only to regret it after a quick tingle and a harsh chemical smell made them drop what they were doing. Eyes need defenders too; wraparound goggles keep drifting vapors out. For folks who fuss with bottles daily, a good fume hood—fans on, window set low—makes breathing easier and heads off headaches.
Spills: The Part No One Loves
Every chemist thinks a spill won’t happen during their shift, up until it does. Cleanup only works if you stay calm and reach for a spill kit before touching anything bare-handed. In my experience, sand or a proper absorbent does more than paper towels, which usually just spread the mess. It’s worth noting that this liquid gives off fumes, so getting those fans moving early matters. Waste goes straight into a labeled, sealed container—no shortcuts.
Storage: Not Just a Shelf Game
Leaving ethyl cyanoacetate around on a benchtop ranks high among poor choices I’ve seen. Storage calls for tightly sealed bottles, placed in a cabinet designed to keep out moisture and sunlight. Some labs go so far as to use flame-proof cabinets, since anything flammable poses a fire risk around that stuff. Keeping the storage area logged and limiting access cuts down on accidents, too. My own mentor once taped clear instructions to every cabinet, spelling out dangers and emergency contacts, which made a bigger difference than the fanciest lock.
Breathe Easy: Air Safety Counts
Even small labs can set up air monitors or draft alarms in their hoods, and it’s worth the effort. Levels above safe limits creep up without much warning—the human nose only catches a strong hit. I learned this lesson during a summer internship, where a hood’s alarm once caught a hidden leak before anyone’s eyes itched or throats burned. Staying proactive with ventilation beats trying to fix things after the fact.
Education and Emergency Planning
Training keeps lab folks from guessing, especially when a split-second decision matters. Knowing where eyewash stations or safety showers stand helps people act fast. Every season, our lab gathers for short drills—no one likes them, but everyone remembers what to do when alarms ring. Posting big, readable emergency numbers above storage cabinets seems basic, yet still saves time under pressure.
Staying Safe Means Staying Ready
Treating ethyl cyanoacetate with respect, using gear that actually protects, and reviewing safety routines each semester build habits that stick. Mistakes teach better than strict warnings. A culture of speaking up and making safety visible—clean benches, clear signs, well-stocked gear—keeps both newcomers and veterans out of trouble.
| Names | |
| Preferred IUPAC name | Ethyl 2-cyanoacetate |
| Other names |
Cyanoacetic acid ethyl ester Ethyl 2-cyanoacetate Ethyloxymethylidenecyanide Ethyl cyanoethanoate |
| Pronunciation | /ˈiːθɪl saɪˌænoʊˈæsɪteɪt/ |
| Identifiers | |
| CAS Number | 105-56-6 |
| Beilstein Reference | Beilstein Reference: 02-02-02-00163 |
| ChEBI | CHEBI:12053 |
| ChEMBL | CHEMBL418061 |
| ChemSpider | 5016 |
| DrugBank | DB13925 |
| ECHA InfoCard | 03b18b6c-1c9d-42a4-bca0-94ca47559a96 |
| EC Number | 203-297-6 |
| Gmelin Reference | Gmelin Reference: **101590** |
| KEGG | C14182 |
| MeSH | Ethyl Cyanoacetate |
| PubChem CID | 7845 |
| RTECS number | KK9275000 |
| UNII | LDX2Q39410 |
| UN number | UN3342 |
| Properties | |
| Chemical formula | C5H7NO2 |
| Molar mass | 111.10 g/mol |
| Appearance | Colorless to yellow transparent liquid |
| Odor | Fruity odor |
| Density | 1.095 g/mL at 25 °C (lit.) |
| Solubility in water | soluble |
| log P | 0.01 |
| Vapor pressure | 0.05 mmHg (20°C) |
| Acidity (pKa) | 9.0 |
| Basicity (pKb) | pKb = 7.94 |
| Magnetic susceptibility (χ) | -40.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.418 |
| Viscosity | 2.02 mPa·s (25 °C) |
| Dipole moment | 4.43 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 191.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -455.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1923 kJ/mol |
| Hazards | |
| GHS labelling | **GHS02, GHS07** |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H317 |
| Precautionary statements | P210, P261, P280, P301+P312, P304+P340, P305+P351+P338, P330, P337+P313, P403+P233, P370+P378 |
| NFPA 704 (fire diamond) | 2-2-2-W |
| Flash point | 79 °C (174 °F) |
| Autoignition temperature | 372 °C |
| Lethal dose or concentration | LD50 oral rat 2820 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 2,820 mg/kg |
| NIOSH | AI5950000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | IDLH: 200 ppm |
| Related compounds | |
| Related compounds |
Cyanoacetic acid Methyl cyanoacetate Isopropyl cyanoacetate Malononitrile Diethyl malonate Ethyl acetoacetate |

