Understanding Ethyl Chlorothionoformate: Its Evolution, Value, and Risks
Historical Roots and the Path to Modern Use
Ethyl chlorothionoformate didn’t pop up suddenly; researchers and manufacturers found their way to it after decades of experiments with chlorinated and sulfur-containing organics. The old chemical texts from the mid-20th century mention this reagent’s roots in sulfur chemistry, which picked up traction as industries chased new materials for drug synthesis and agrochemicals during the post-war boom. That growth led to a widespread interest in reactive electrophiles, and ethyl chlorothionoformate found its way into lab protocols, especially thanks to its unique mix of reactivity and selectivity in making thioesters and related compounds.
What Stands Out About This Compound
On the surface, ethyl chlorothionoformate looks a lot like its better-known cousin, ethyl chloroformate. Take a closer look, though, and that sulfur atom changes the game, paving the way for a different pattern of reactivity. The chemical appears as a pale yellow liquid, a detail that has thrown off more than one chemist expecting a clear solution. Its faint, unpleasant odor quickly becomes memorable in any poorly ventilated workspace. I remember my first run-in with it in graduate lab—one whiff forced me to rethink my PPE routine permanently.
Physical and Chemical Nature
This reagent packs a punch: it reacts quickly with water, giving off corrosive gases. The molecule is made up of an ethyl group perched on a reactive chlorothionoformate moiety, making it an effective and sometimes unpredictable acylating agent. The boiling point sits well below 200 Celsius, leaving plenty of room for evaporation in an open flask. Flammability isn’t off the chart, but the risk climbs if vapors reach a spark. Chemists who ignore clean equipment or moisture control can end up with more than just a ruined batch—they risk creating hazardous byproducts that can hang around long after cleanup.
Technical Nuance Shows Up on the Label
Purity usually matters more than the brand. Impurities in ethyl chlorothionoformate can haunt a synthesis—traces of water, acids, or even its decomposition products can change yields in unpredictable ways. The best suppliers ship it in sealed amber bottles, usually with a stabilizer, since sunlight and heat make the compound break down fast. Technical specifications highlight its boiling range, density, and acid content—small numbers, but every working lab should double-check before opening a new batch. I learned the hard way that even a little hydrolysis during shipping can leave enough hydrochloric acid in the bottle to turn a routine reaction violently exothermic.
Making Ethyl Chlorothionoformate: Lessons from the Bench
Preparation sometimes starts with thiophosgene or related intermediates, which carries its own list of headaches. Most production avoids open-air work for safety reasons. The method relies on adding ethanol to a chlorothionocarbonyl source under strict temperature control, followed by careful distillation to remove byproducts. At no step is this process forgiving—a dip in temperature or a shortcut with solvents throws off yields and adds risk of contamination. I’ve heard technicians joke that distilling this compound without a scrupulous vacuum gauge is just gambling with a different deck of cards.
Reactivity and Real-World Modifications
Ethyl chlorothionoformate shines as an acylating agent in both laboratory and industrial settings. Its affinity for amines or alcohols means it can prepare thioesters, carbamates, and other sulfur analogues that often show up in pharmaceuticals or specialty agrochemicals. Side reactions tend to involve unplanned hydrolysis or self-condensation if the system isn’t kept strictly anhydrous. Knowledge of its reactivity with bases, Grignard reagents, or strong nucleophiles lets chemists dial in selectivity and minimize byproducts. Stories circulate of impatient chemists skipping the dry-line for a quick yield—usually followed by a scramble to clean up caustic residues.
Behind the Names: Synonyms and Nicknames
Like many reagents, ethyl chlorothionoformate comes with a handful of other labels: ethyl O-ethoxythiocarbonyl chloride, and less formally “ethyl chlorodithiocarbonate.” Old catalogs and patents list other names, causing the occasional mix-up during cross-checks. A quick internet search still yields a split between the older “thionoformate” and the more systematic “chlorothionoformate,” reinforcing the importance of careful translation—especially when ordering from international suppliers.
Everyday Handling: Hazards Don’t Take Holidays
Handling ethyl chlorothionoformate is never routine. Strong ventilation, nitrile gloves, and full goggles define the dress code. Even small spills pose problems: vapors irritate lungs and eyes, and accidental hydrolysis generates corrosive hydrochloric acid on the bench. Inexperienced workers sometimes underestimate its volatility, leading to hidden exposures over time, which can contribute to chronic irritation or more serious health problems. The right approach means treating every bottle like it could misbehave, storing it cool and dry, and keeping neutralizing agents within arm’s reach. Local rules may ask for additional training or restrict access, something I see as less red tape and more common sense.
Where It Ends Up: Application in and Out of the Laboratory
This reagent carves out a big niche in making pesticides, specialty polymers, and certain drug intermediates. Chemists also use it in analytical work, attaching sulfur-rich groups during derivatization or creating custom probes for enzyme studies. The pharmaceutical world values its role in preparing thioesters, carbamates, and advanced building blocks—good luck making some antiviral medicines or herbicides at scale without a sulfur-based acylating step. Universities still rely on it in teaching labs, despite the risk, because it shows off a blend of fundamental reactivity and hands-on technique that textbooks alone can’t capture.
Chasing New Possibilities: Research and Ongoing Challenges
Research circles keep looking at ethyl chlorothionoformate for more eco-friendly reactions, seeking methods with lower waste or safer byproducts. Some chemists are pushing for continuous-flow reactors to tame volatile emissions and limit worker exposure, while others eye bio-based routes to starting materials. I’ve seen groups combine it with emerging photochemical or enzymatic techniques, dreaming of faster, cleaner transformations. The push to reinvent core steps in sulfur-based synthesis reflects the environmental and economic pressures shaping today’s chemical enterprise.
Toxicity: Not Just a Footnote
The toxic nature of ethyl chlorothionoformate never stays out of the spotlight for long. Animal studies show both acute and chronic exposures raise serious red flags, ranging from respiratory distress to organ damage and potential carcinogenicity. Skin exposure burns fast, and even diluted solutions can leave lasting irritation. Lab incidents often trace back to poor equipment or skipped procedures. Many facilities add extra layers of containment, like glove boxes or air monitoring badges, for work above gram scale. The trend in regulatory circles leans toward restricting large-scale use or requiring special permits, reflecting a growing recognition of the long-term risks.
Where It Heads Next: The Road Forward
Ethyl chlorothionoformate holds on as a key intermediate even as green chemistry and automation reshape the industry. Progress will mean finding safer alternatives or modifying its preparation to reduce emissions and toxic runoff. I expect more investment in real-time monitoring and automated dosing, helping minimize human exposure while sustaining output. If breakthroughs arrive in sulfur chemistry, they’ll probably come from teams willing to rethink old protocols and prioritize both safety and sustainability. The next decade will likely see this compound judged not just by how well it works in reactions but by the broader impact on health, the environment, and worker well-being.
What is Ethyl Chlorothionoformate used for?
What Ethyl Chlorothionoformate Does in the Lab
Ethyl chlorothionoformate sits tucked away on chemical shelves in production labs. Chemists know it as a tool for moving certain atoms around—especially sulfur and chlorine—in the process of putting together bigger, more complicated molecules. Think of it like a courier that brings pieces where they’re needed.
My background in laboratory research taught me how these precise moves can save months of work. Ethyl chlorothionoformate pops up when scientists make pesticides, pharmaceuticals, and specific dyes. In medicine, this chemical can play a role early in the process—before a drug ever gets close to a patient. For example, certain molecules that block cancer growth or infections start off as simple structures, then engineers tack on the parts that make them work; ethyl chlorothionoformate often helps stick these pieces together. Without it, the process drags out and costs climb.
Outside the Lab: Safety and Environmental Risks
I’ve seen how people treat these chemicals with caution, and that’s not just good sense—it’s required. Ethyl chlorothionoformate brings with it some rough edges. The compound can irritate skin, eyes, and lungs if not handled under a hood, wearing gloves, and keeping vents running. Few substances make a researcher pause like a whiff of stinky, reactive vapor during a late-night experiment.
On top of personal safety, there’s the question of what happens downstream. Waste from these labs can harm water and soil. Routine spills or careless dumping don’t just fade away. Many companies now spend heavily on scrubbing their emissions, neutralizing waste, and containing these compounds so they don’t end up in the river or the air. I think of the cleanup crews I’ve met—mechanics and engineers in hazmat suits washing down surfaces, double-checking valves, and packing used gloves for safe disposal. Skipping these steps isn’t an option, unless we want to pay with sick coworkers or polluted groundwater.
Industry Relies on Specialized Chemicals
Ethyl chlorothionoformate traces a path from big factories to research universities to the companies that make our everyday products. Companies count on specialized compounds like this to make reactions more efficient and cut down on wasted effort, which means less energy burned and fewer leftover byproducts. Most folks outside the field never cross paths with these chemicals, but their lives are touched by them—through safer crops, new medicines, and even materials with unique colors or properties.
Better Handling and Innovation
One way to keep the risks in check: train chemists the right way, starting from the very first time they set foot in a lab. I’ve seen newer researchers struggle with hazmat protocols and make small mistakes; oversight and mentorship help protect them and everyone around them. Investment in new, greener alternatives could take some pressure off the need for chemicals that bite back, especially as public health watchdogs push for less toxic options.
Finding balance between chemical utility and health should not be a niche concern. Whether we notice or not, safer handling, smarter engineering, and open reporting make an impact far beyond the lab doors. The story of ethyl chlorothionoformate shows how even behind-the-scenes tools shape both innovation and responsibility.
What is the chemical formula of Ethyl Chlorothionoformate?
Not Just a Bunch of Letters: C3H5ClO2S at Work
Ethyl chlorothionoformate, with the formula C3H5ClO2S, plays a bigger role in industrial chemistry than most folks realize. Out in the world, small molecules like this show up behind the scenes in specialty manufacturing. This compound, used as an intermediate, rarely makes the news, but it matters for plenty of skilled workers and researchers in laboratories.
Chemistry Runs on Details
I remember long hours in a campus lab, piecing together reaction mechanisms, always paying close attention to every atom. Overlooking a chlorine or switching up a functional group in a molecule like ethyl chlorothionoformate means the reaction train runs off the rails. Precision matters—one little change and the results could be toxic gasses or nothing useful at all. That formula—three carbons, five hydrogens, one chlorine, two oxygens, one sulfur—drives countless projects forward. It's not just about knowing the formula, but really seeing how every piece interacts with the others.
Not All Chemicals Are Created Equal
Industries use chemicals like ethyl chlorothionoformate to stitch together new substances, especially in agriculture and pharmaceuticals. For example, some pesticide makers rely on this compound to build active molecules targeting pests. Its reactive chlorine and sulfur parts help launch tricky chemical reactions that create more complex molecules. Pharmaceutical research labs sometimes turn to it while making building blocks. The chemistry textbooks might write a few lines about the risks and handling issues, but from experience, you learn real caution working with reactive chlorinated substances like this. One careless moment, and there’s trouble—from harsh vapors to dangerous byproducts.
Safety Comes from Respect and Good Practice
Many folks outside labs think of chemicals as mysterious threats. That’s not far from the truth with thionoformates. The secret is treating everything with respect. My old mentor insisted on double-checking every label, every time. Industry standards set by groups like OSHA serve the same purpose—never assume familiarity replaces careful habits. Proper training, trusted safety gear, and ventilation systems reduce risk, but culture counts too. Reminding coworkers to slow down, securing containers, and logging each transfer—those human moments make the real difference.
Better Alternatives and Research Drive Change
Chlorinated compounds, despite their usefulness, often raise eyebrows due to environmental and health effects. Chemical engineers have started exploring greener syntheses, trying to replace leaky reagents with safer options. After years working with both old and new chemicals, I see the need for substitution isn’t just a buzzword. Each new project brings a checklist—do we really need this compound, or is there a better way? More funding for research opens up choices. Universities and companies compete to find new routes, faster reactions, or cleaner breakdown pathways, and this push often starts from small, local solutions spread through mentorship and collaboration.
People Create the Safeguards and Innovation
The real story behind C3H5ClO2S goes beyond the formula itself. Expertise grows through hands-on experience and open sharing of practical safety and technique. Industry and research culture can nudge everyone toward safer processes, cutting risks while keeping innovation alive. Modern labs run best on more than just regulations—they thrive on a mix of care, curiosity, and constant learning, all sparked by paying attention to the details that matter.
What safety precautions should be taken when handling Ethyl Chlorothionoformate?
The Everyday Risks in Chemical Handling
Ethyl Chlorothionoformate sits among the chemicals that demand respect. Walk into any lab, and you quickly realize this isn’t a substance where shortcuts go unnoticed. Most people chat about best practices, but real safety begins before the bottle’s even open—thinking through what could go wrong and gearing up properly.
Why Personal Protection Really Matters
You’re not just suiting up for show. Direct exposure to Ethyl Chlorothionoformate causes skin burns and eye injury, plus harsh irritation in airways. If a friend walked in without goggles, gloves, and a proper lab coat, I’d stop them at the door. Nitrile gloves work much better than latex here, and chemical splash goggles shield against surprise droplets. Skip the sandals; stick with closed shoes—one splash to the foot can end a shift early.
Avoid Breathing in Trouble
This chemical doesn’t ask for much air time to become dangerous. Small spills put off vapors that can irritate lungs. Tossing open a window helps, but real control comes from working inside a chemical fume hood. I’ve seen people try to “hurry up” on the benchtop instead, which isn’t worth the risk. Even a short exposure leaves people coughing and uncomfortable.
Spill Response: Actions Over Excuses
Spills never wait for a good moment. If your hand slips, solid preparation changes everything. Keeping an absorbent spill kit nearby isn’t overkill—it’s smart. Most kits rely on activated carbon or inert material. If the spill reaches fabric or skin, rinsing with water right away helps limit damage. Don’t forget to report it. Every missed incident means a chance for others to learn and update their own practices goes wasted.
Storing with Sense, Not Hope
Where a chemical sleeps at night makes a difference. Storing Ethyl Chlorothionoformate away from heat, moisture and open flames keeps things predictable. I always use a tightly sealed glass container. Moisture means trouble here—hydrolysis ramps up the risk inside plastic or loosely capped bottles. Keeping it with separate from bases, acids, and strong oxidizers slashes the chance of unexpected reactions.
Disposal: No Shortcuts Allowed
Chemical disposal can’t rely on wishful thinking. Most municipal drains or trash bins aren’t cut for the job. Neutralization procedures run best with expert guidance. Most labs keep a clear log for hazardous waste pickup, making sure nothing lingers where it shouldn’t. If you’re not sure about the right steps, a quick call to environmental health and safety clarifies things before mistakes happen.
Training Builds Long-Term Safety Habits
No one picks up perfect habits overnight. Working side by side with seasoned chemists taught me as much as any handbook. Refresher courses, emergency drills, and honest reviews after incidents set the standards. Everyone benefits from honest conversations—less finger-pointing, more focus on making sure we all head home safe.
Looking Ahead: Keep Learning, Stay Ready
Experience inside a busy lab demonstrates there’s never a finish line for safety. Handling Ethyl Chlorothionoformate brings real hazards, but sensible preparation reduces those risks. By working smart, sharing good habits, and updating protocols as we learn, safety becomes part of the lab’s daily rhythm—not just a line in a manual.
How should Ethyl Chlorothionoformate be stored?
Straight Talk on a Risky Chemical
Ethyl chlorothionoformate, a reagent with bite, finds frequent use in organic synthesis labs. My first experience with it sent an unforgettable whiff through the fume hood, poking at my nose, reminding me that this is not a chemical to handle like table salt. Its sharp, choking odor and corrosive fumes say enough—treat every bottle with caution.
Conditions That Matter
This liquid breaks down with moisture, kicking up toxic byproducts—hydrogen chloride and similar nasties. Keep water out at all costs. I always reach for containers made from glass or high-grade plastics, with tight, chemical-resistant seals. Polyethylene just doesn’t cut it here. For several months, our lab used borosilicate bottles stored beneath a layer of nitrogen gas. No sweating over leaks or unpredictable reactions—the liquid kept its bite but didn’t go wild in storage.
Heat spells trouble. Above typical room temperature, I’ve seen vapor pressure climb, leading to swollen seals and—once—cracking caps that nearly ruined a chemical cabinet. Stash ethyl chlorothionoformate between 2°C and 8°C. Dedicated refrigerators labeled for hazardous chemicals work best. Housekeepers won’t swipe your lunch, but don’t put your sandwich there, either.
Ventilation Isn’t Optional
Fume hoods aren’t just for show, especially after a bottle is opened. Any spill or even vapor escape fills the air with corrosive fumes. Researchers with experience know not to store bottles above head height or in shared spaces. Accidents always find their moment when someone rushes or cuts corners. Separate, ventilated cabinets, well away from acids and bases, ensure that incompatible neighbors don’t kick off an accidental reaction.
Labels, Logs, and Limits
Label every container with full chemical names, date received, and the hazard class. I’ve seen new students relabel bottles using abbreviated names, and confusion leads to mistakes—sometimes costly ones. Daily checks, at least for active bottles, help catch leaks or degradation before it gets out of hand. For labs or warehouses storing larger quantities, a simple logbook can prevent overstock and expired material from piling up behind newer stock.
Handling Spills—Experience Counts
A prep gone wrong once sent a spill racing down the bench, fizzing as it hit a damp rag. We cleared the lab, donned full-face respirators, and swept up dry—moisture can turn a tiny leak into a dangerous gas release. Keep spill kits close, stocked with compatible absorbents and neutralizers (not water!). If you’ve never walked through a safety drill for high-risk chemicals, ask for one. Training saves panic and injury.
Respect This Chemical—A Culture of Safety
Ethyl chlorothionoformate doesn’t forgive shortcuts. The goal—protect health, prevent accidents, and keep a clean track record—relies on habits, not fancy technology. Reliable refrigeration, attentive labeling, routine inspections, and prompt training all add up to safe storage. As someone who has cleaned up more than one avoidable mess, I can say: Store it right, and your future self will thank you.
Building Safer Labs, One Bottle at a Time
Safety isn’t a checklist. It’s a culture. Respecting chemicals like ethyl chlorothionoformate each day prevents disasters that never make headlines. The best labs I’ve worked in run on routine, clear communication, and unfussy discipline. Store it cold, dry, and separate. Let no one in your team forget that.
What are the potential health hazards of Ethyl Chlorothionoformate?
Understanding What’s at Stake
Walking through a chemical plant or lab, it’s easy to overlook some of the things people use every day. Ethyl chlorothionoformate probably doesn’t ring a bell for most folks, but for anyone working around chemicals, it pays to know the health hazards tied to it.
What Happens When You’re Around It
Ethyl chlorothionoformate gives off a sharp, irritating odor, which is often the first sign the chemical’s around. Eyes start to water and noses burn. Inhaling even a bit of those fumes can set off headaches, nausea, and dizziness, and in high doses, lead to coughing, chest pain, or shortness of breath. Many people who have handled industrial chemicals can recall that rush to leave the room after getting one whiff of something they shouldn’t have breathed.
Skin contact is another worry. This stuff isn’t gentle. Splashing even a drop can leave red, painful skin or even blisters. A buddy of mine once brushed up against a leaky valve and walked away with a nasty welt that stuck around for days. If it hits the eyes, trouble gets bigger: burning, redness, and even potential long-term injury. This is why goggles and gloves are part of any real chemical worker’s toolkit.
Chemical’s Toxic Nature
Chronic exposure or breathing in the vapors over time can do more than just make you sick for a day. The respiratory system takes a beating. Coughs turn chronic, sometimes leading to asthma-like issues. Studies have found signs of damage in liver and kidney tissues among workers exposed for long periods. The material safety data sheets paint a picture most would want to avoid, and for good reason.
People using ethyl chlorothionoformate in production or research don’t always have a choice, but the need for respect is real. The International Agency for Research on Cancer (IARC) suggests treating chemicals like this with extra caution. There isn’t an endless supply of lungs and livers to trade in after mistakes.
Reducing Harm and Protecting Workers
Simple habits make the difference. Ventilation works wonders—exhaust hoods, air purifiers, and regular maintenance help stop vapors from floating into noses and lungs. Personal protective equipment, especially gloves, goggles, and long-sleeved gear, is worth every penny. Most labs and plants enforce emergency showers and eyewash stations for good reason.
Clear labeling and regular safety training shouldn’t be an afterthought. New hires might not know their risks, but stories from old hands often stick: “I saw Bob lose a week from a chemical splash, and I’m not skipping my gloves.” Workers lean on each other, swapping tips for safe storage and cleanup. Having a plan for spills—spill kits, absorbent neutralizers, instructions posted nearby—keeps everyone on their toes and accidents under control.
Prevention means fewer hospital visits. Good ventilation, gear, and training aren’t just company costs—they’re investments in healthy, capable people. No one wants to swap a day’s pay for a stack of medical bills or years of chronic breathing trouble. With a bit of respect, and the right tools, most of these hazards stay manageable, and people get to go home safe.
| Names | |
| Preferred IUPAC name | Ethyl chlorosulfanylformate |
| Other names |
Chloroformic acid, ethyl thioester Ethyl chlorothioformate Ethyl chlorothionoformate Ethyl chlorothiocarbonate Ethoxythiocarbonyl chloride |
| Pronunciation | /ˌɪθ.ɪl klɔːˌroʊ.θaɪˈɒn.oʊ.fɔːr.meɪt/ |
| Identifiers | |
| CAS Number | 541-41-3 |
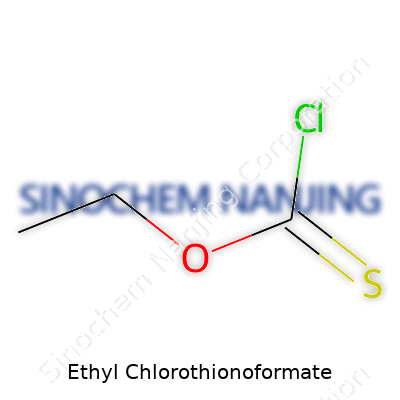
| 3D model (JSmol) | `JSmol('C(=S)(OCC)Cl')` |
| Beilstein Reference | 1718736 |
| ChEBI | CHEBI:38960 |
| ChEMBL | CHEMBL468443 |
| ChemSpider | 24736 |
| DrugBank | DB14274 |
| ECHA InfoCard | 03f166f4-3866-43ad-9c09-048e6abc7aaa |
| EC Number | 209-768-0 |
| Gmelin Reference | Gmelin Reference: 7775 |
| KEGG | C19235 |
| MeSH | D002490 |
| PubChem CID | 13940 |
| RTECS number | FG9625000 |
| UNII | LY943N0L0T |
| UN number | UN2672 |
| CompTox Dashboard (EPA) | DTXSID4039152 |
| Properties | |
| Chemical formula | C3H5ClO2S |
| Molar mass | 141.57 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | penetrating |
| Density | 1.41 g/cm³ |
| Solubility in water | Decomposes |
| log P | 1.99 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | pKa = -3.41 |
| Basicity (pKb) | Ethyl Chlorothionoformate has a pKb of 7.45 |
| Magnetic susceptibility (χ) | -58.41·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.508 |
| Viscosity | 1.217 mPa·s (20 °C) |
| Dipole moment | 2.01 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 327.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -97.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -751 kJ/mol |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06,GHS05 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H314: Causes severe skin burns and eye damage. H317: May cause an allergic skin reaction. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P210, P261, P271, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P311, P330, P337+P313, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | Flash point: 58 °F (14 °C) |
| Autoignition temperature | 518°C |
| Lethal dose or concentration | LD50 oral rat 55 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 60 mg/kg |
| NIOSH | WM6125000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Ethyl Chlorothionoformate: "0.05 ppm (ceiling) |
| REL (Recommended) | 1 ppm |
| IDLH (Immediate danger) | IDLH: 3 ppm |
| Related compounds | |
| Related compounds |
Methyl chlorothionoformate Isopropyl chlorothionoformate Phenyl chlorothionoformate Ethyl chloroformate Thioesters |

