Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate: A Closer Look
Historical Development
Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate has roots in mid-20th century organic chemistry, branching from early organochlorine research and the hunt for stable industrial intermediates. Chemistry textbooks used to highlight the compound's parent pathway, not just for its novelty, but for the attention that dichlorinated aromatic rings earned back in those days. Researchers meticulously forged this molecule in labs, building on the popularity of its analogs in agriculture and materials science. Students and chemists drew upon early breakthroughs to open doors for broader applications: whether through medicinal chemistry, environmental science, or polymer production. Years of hands-on bench experimentation and published academia shaped a clearer understanding of its nuances—and lessons learned about chlorinated aromatic substances shifted the focus toward responsible synthesis and handling.
Product Overview
Chemists recognize Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate as a white crystalline solid, sitting somewhere between intermediate and specialty product categories. Its defining feature comes from those two 4-chlorophenyl rings flanking a hydroxyacetate group, and this configuration bestows a distinct balance of reactivity and stability. You seldom find it sitting alone on a shelf; lab personnel pull it out for targeted derivatizations, not for broad-spectrum use. Its structure attracts attention in synthetic planning, and many protocols feature it as a stepping stone toward more elaborate molecules. Professionals handling it have always respected the meticulous documentation required for controlled chemicals of this nature, relying on batch records and supplier certifications before proceeding with use.
Physical & Chemical Properties
With a molecular weight in the neighborhood of 355 grams per mole and a melting point often reported just above room temperature, this compound offers a tactile sense of density when you handle a sample. The dual presence of chlorinated aromatic rings makes the molecule more robust under mild acidic or basic conditions. You can spot a faint aromatic odor in larger batches, not unlike other halogenated organics handled in a well-ventilated chemical space. Solubility swings depending on solvent: ethanol or methanol tend to work, water less so, which can drive decisions during process development. The high partition coefficient reflects its moderate hydrophobicity, signaling caution for those thinking about solvent selection in both analytical and synthetic workflows.
Technical Specifications & Labeling
Bottles carrying Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate feature meticulous labeling, as expected for organochlorine compounds. Labels list CAS numbers, purity percentages (often 98% or higher), storage requirements—typically dark glass containers, cool temperature, no exposure to light. Shipping manifests highlight UN classifications and precautionary statements required by regulatory authorities. Safety Data Sheets (SDS) explain everything from eye irritation risks to combustion products, laying out a roadmap for safe move from delivery to benchtop and—eventually—to disposal. Proper tracking through inventory systems prevents accidental double orders or unauthorized access, a lesson learned from years of regulatory tightening following high-profile incidents with similar compounds.
Preparation Method
The process starts with ethyl glyoxylate or its equivalents and introduces substituted chlorobenzene derivatives under controlled conditions, sometimes under reflux, sometimes with a catalyst. Every bench chemist who prepares it feels that moment when the crystallization occurs, marking a successful coupling. Purification follows—often by recrystallization out of an ethanol-water mixture or via column chromatography if purity thresholds dictate. In-process controls determine completion, tackling questions around by-product management and solvent recovery. Researchers strive to minimize chlorinated waste, thanks to environmental pushback against organohalogen pollution. Educational labs emphasize hazard reduction just as much as yield optimization, reflecting lessons learned from earlier decades when waste disposal got less scrutiny.
Chemical Reactions & Modifications
Reaction planning around this compound involves a sturdy aromatic core and a somewhat reactive hydroxy group. Electrophilic aromatic substitution, despite the electron-withdrawing chlorines, can tweak new ring functionalities. Chemists sometimes convert the ethyl ester moiety to carboxylic acids or amides, and these tweaks make the molecule suitable for downstream pharmaceutical or agrochemical building blocks. Researchers also introduce various alkyl or acyl groups to shift physical or biological activity, remembering to monitor for diastereomeric separation. Cross-coupling reactions with boronic acids or stannanes open more doors—classic Suzuki and Stille reactions—showing the breadth of modern methodology versus the limited toolkit of previous generations.
Synonyms & Product Names
Trade names and synonyms in supplier catalogs reflect regional naming practices. You’ll find it under common aliases like "Ethyl DCCA hydroxyacetate," or closer to its IUPAC roots for formal research paperwork. Chemists switching suppliers learn to match catalog numbers and molecular diagrams to prevent misordering, since structural isomers and analogs crowd the stockroom shelves. Legacy procedural notes often use older naming conventions, tying today’s work to a lineage that predates current regulatory formats. Documentation habits in the lab lean towards erring on the side of clarity, translating raw chemical names into concise inventory records for future reference.
Safety & Operational Standards
Lab veterans tell stories of close calls with organochlorines, and these stories drive home the need for double gloving, fume hood work, and spill containment. Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate demands care at every step. Eye and skin contact never get shrugged off; even trace amounts prompt cleanup with dedicated spill kits and detailed documentation. Waste containers sit nearby, labeled for halogenated organics. Regulatory training programs reinforce the rules behind PPE, storage, and documentation. Experience reminds everyone to check for ventilation and to keep emergency eyewash stations functioning—a lesson shared countless times in hands-on training and safety reviews.
Application Area
You see this compound surface in medicinal chemistry labs as a precursor for more elaborate pharmacophores, especially those exploring anti-inflammatory or antiparasitic potential. Industrial formulators may pull it into play when synthesizing specialty polymers or as a template for experimenting with chlorinated resin additives. Environmental researchers keep tabs on such compounds when studying degradation pathways, since their chlorine content flags persistence in ecosystem models. Certain agricultural research uses it as a platform for structural analogs, developing leads on pest management tools with more selective activity profiles. Across these fields, the need for precise sourcing and thorough record-keeping proves just as critical as breakthrough discoveries.
Research & Development
R&D teams continue pounding out proposals that leverage the aromatic backbone of Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate for custom synthesis. Pharmaceutical innovation teams discuss SAR (structure-activity relationship) clusters, tweaking positions around the aromatic rings and side chains. Polymer chemists tinker with its reactivity, searching for ways to create harder, more weather-resistant plastics. Research publications detail advances in greener synthetic methods, aiming to trim chlorinated waste at every turn. R&D units working on new analytical standards cite this compound as a spiking agent or calibration substance, thanks to its distinctive, reliable behavior under chromatographic analysis. Every year brings new modifications and reactions, adding to the compound’s versatile history.
Toxicity Research
I’ve seen first-hand how important it is not to take organochlorine toxicity lightly. Researchers have tested Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate in vitro and in animal models, carefully documenting acute and chronic exposure effects. Dose-dependent outcomes steer toxicology reviews, flagging attention to liver and kidney endpoints. Concerns about bioaccumulation and metabolite persistence surface in institutional risk assessments, leading to calls for regular monitoring in facilities using substantial quantities. Regulators continue to demand strong evidence on safe handling procedures, pushing for better fume extraction systems and stricter waste controls. Toxicological findings influence both local protocols and international agreements, limiting where and how this substance finds industrial or research use.
Future Prospects
The future of Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate ties deeply to changing regulatory landscapes and technological advances in green chemistry. Research collaborations now aim for alternative syntheses that sidestep harsh halogenation steps, with renewed interest in recyclable catalysts and closed-loop manufacturing. Interest in environmental impact drives new separation techniques and remediation strategies for organochlorine residues, especially as governmental agencies clamp down on emissions and waterway contamination. Startup ventures search for ways to harness these molecules for specialty applications in electronics and materials science, targeting properties that traditional monomers can’t easily match. Looking ahead, major R&D centers work to balance the benefits of advanced organochlorine chemistry with lessons learned from decades of health and safety oversight, keeping the pathways open for safer, smarter chemical development.
What is the primary use of Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate?
Unpacking an Often-Misunderstood Chemical
Mentioning Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate in a room full of scientists triggers a conversation that circles more around legacy than new discovery. This chemical—better known in many circles as an intermediate for DDT (dichlorodiphenyltrichloroethane)—belongs to an era when agriculture and public health needs pressed hard for pest control at any cost. Today, most people have never seen the name printed outside regulatory documents or chemical supply catalogs, but its shadow still lingers in the stories of banned pesticides, polluted soils, and evolving research into toxicity.
The Main Use: DDT Manufacturing
Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate has kept its main purpose over the decades: serving as a foundational building block for synthesizing DDT. During the heyday of chemical pest control, DDT transformed global agriculture and helped reduce malaria. Scientists and manufacturers considered intermediates like this one as routine train stops in enormous production processes.
A solid grounding in organic chemistry reveals that changing a molecule’s small part can mean life or death for entire crops, or health for whole communities. In the lab, chemists combine this compound under controlled conditions with other reagents, ending up with a batch of DDT. That final product packs a punch against mosquitos, lice, and pests spanning a wide range. For many years, factory workers and farmers didn’t stop to ask how each ingredient in the mix might behave later in the food chain or water supply.
Lingering Issues and the Call for Solutions
Although DDT got credit for saving millions of lives, especially in the 1940s and 1950s, ongoing use led to a legacy of pollution. Environmental groups started making noise when eggshells weakened in birds of prey and when traces of DDT began appearing nearly everywhere—from arctic ice to breast milk. Scientists worked their way up the chain and realized that even starting materials, including Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate, showed up in soil and water in some regions.
Tighter regulations on both end products and intermediates soon followed. The Stockholm Convention in 2001 brought a wave of bans and phasedowns around the globe. On paper, that meant less demand, but the reality remains more complicated. Small-scale production and illicit trade thrive in pockets where malaria still claims lives or crop loss wrecks economies. Old factories sometimes sit abandoned or leak chemicals, affecting drinking water and local agriculture.
Building a Better Path Forward
The chemical’s journey tells a bigger story about the choices involved in fighting disease and feeding the world. Full-scale bans without real alternatives can put already struggling communities in a tougher position. Research focuses on safer pest control, targeted mosquito traps, and biopesticides. Fact remains, any new approach must pass not just scientific muster, but also win trust in villages and on farms.
People living near legacy contamination sites need governments and companies to take cleanup seriously. Remediation isn’t glamorous, but it matters. Experience shows me that trust comes slow when past decisions have poisoned wells or fields. Open data on soil and water testing, honest public meetings, and steady investment in community health programs start to patch the wounds. Most who live near these sites want the mess cleaned up, not arguments about why it happened.
As the world looks for workable solutions, scientists need to keep the past in view but steer work toward better, safer answers. Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate sits etched in a complicated chapter, not yet finished. Facing that story with honesty gives everyone a firmer footing for smarter action down the road.
Is Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate safe to handle?
Getting Familiar With the Compound
Ethyl 2,2-bis(4-chlorophenyl)-2-hydroxyacetate can trip up anyone who doesn’t work in a chemistry lab. Most people see a mouthful of a name like this and tune out. Dig a little deeper and the story gets interesting. This chemical stands out not just because of its structure, but because it's related to DDT, a pesticide banned around the world for its toxic legacy. And here’s where experience kicks in—the closer a molecule sits to something infamous, the more tightly we should grip our gloves around it.
Toxicity Concerns and Handling Risks
Long chemical names can sometimes mean very little; in this case, the fine print matters. Research on the compound itself turns up less than you might expect, but its family ties raise red flags. Chlorinated aromatic compounds, especially those linked to DDT, have earned a rough reputation over decades of use and study. Their persistence in the environment and in living tissue isn’t an old wives’ tale. At Rutgers, we ran soil tests showing how tiny traces of substances like these turn up in places untouched by direct spraying for decades.
What does that mean for a person weighing if it’s “safe to handle”? Gloves and goggles aren’t extra credit—they’re table stakes. Ethyl 2,2-bis(4-chlorophenyl)-2-hydroxyacetate doesn’t have a glowing record for safety. It can break down into chemicals that don’t leave the body for years. Lax safety habits can get you a dose that lingers. Even outside the lab, older agricultural sites fling these molecules back up from the dirt season after season. It doesn’t take a lot for them to build up in your system; slow poison is still poison.
Scientific Consensus and Facts
Google Scholar data shows most peer-reviewed research on compounds like this focuses on environmental persistence, effect on wildlife, and toxicology projections. Workers in chemical plants know how poor ventilation and carelessness led to illnesses and lawsuits in the past. Research on similar esters projects they can act as endocrine disruptors. Animal models have shown birth defects, cancer risks, or neurological symptoms, although results vary based on exposure and dose. It's not just rodents; studies tracking pesticide workers link long-term exposure and increased rates of metabolic and neurodegenerative diseases.
What Safe Actually Looks Like
Personal experience walking chemical plants says labeling everything “dangerous” helps nobody. Knowledge and respect for complex molecules does. Turning a blind eye because gloves “get in the way” or a mask “seems like overkill” opens the door to accidents. Many injuries never made the papers—the quick trip to rinse a splash in the eye, the unexplained rash a few weeks later. These moments teach real lessons. Every Material Safety Data Sheet (MSDS) pushes standard best practices: splash goggles, nitrile gloves, fume hoods. Lab training always drove home how a shortcut can cut a career short.
What Industry Can Do Better
Employers shouldn’t treat MSDS compliance as the endgame—safe work culture means talking about risk, not just posting warning signs. Real training, not just paperwork, saves hands and lungs. Companies using or disposing of chemicals like this owe it to workers and neighborhoods to maintain airtight containment, top-notch ventilation, and responsible waste management. It takes investment, but fines and lawsuits cost more. Everyone from seasoned chemists to recent hires deserves to know the truth about the risks, the right equipment for the job, and the stories where shortcuts led to disaster.
What are the storage requirements for this compound?
Checking Storage Needs: Safety Matters
Safe storage of any compound never comes down to guesswork. I’ve seen otherwise careful labs slack off on simple details: a mislabeled container, a shelf too close to a heat source, a faulty lock. Mistakes like these open the door to risk. Many chemical incidents start not with explosions or dramatic events, but with a little moisture leaking in where it shouldn’t, or a solvent vaporizing about fifteen degrees too soon.
Temperature and Humidity Are Top Priorities
In practice, most compounds behave differently outside their preferred comfort zones. A chemical that clumps or cakes from ambient humidity loses accuracy for dosing or reactivity, and this can send an otherwise reliable process off the rails. If the material wants refrigeration, a tight-sealing fridge that won’t dip below freezing does the trick. Freezer-burn damages some heat-sensitive samples just as much as a warm bench. Dry, dark, and cool—those three standards keep more trouble at bay than any high-tech system ever could, and yet many people cut corners on the basics.
Why Labeling and Separation Make a Difference
Professional labs rely on crisp, clear labels with hazard symbols that shout instead of whisper. “What’s in this bottle?” never belongs in a storage room. Chemicals of wildly different reactive potential have no business sitting together on the same shelf. Acids and bases, oxidizers and reducers—combine the wrong pair and you’re looking at a mess or, worse, an emergency. Smart practice puts incompatible neighbors far apart, preferably with separate secondary containment. Regular audits make sure nothing sneaks into the wrong spot over time.
Containers and Security: Not Just for Show
Flimsy caps or old, deteriorating seals let air and contaminants spoil sensitive stock. Glass stays neutral longer than plastics, but some aggressive solvents chew through even specialized polymers. Metal drums rust and leach, so proper lining always gets my vote for bulk storage. Access matters as much as what’s inside: limited entry protects both the stock and the people using it, especially in shared facilities. A secure storage cabinet with well-maintained locks reminds everyone to stay detailed—no shortcuts, no surprises.
Regular Checks and Emergency Plans
Every couple of weeks, I do a walk-through and check thermometer readings, shelf arrangements, and expiration dates. Cleaning up an expired spill or tossing a bloated canister beats explaining a full-blown accident to the fire chief or the local news. A solid inventory helps catch slow leaks or forgotten bottles before issues snowball. For every container, I keep spill kits and fire extinguishers nearby. Training goes beyond boxes on a form; people should know how to use the tools and when to get help. It’s one thing to write out evacuation routes and quite another to practice them so anyone can move fast if alarms sound.
Small Steps, Big Impact
I’ve learned that attention to storage requirements pays off long before problems happen. It saves money, keeps systems running smoothly, and protects everyone nearby. No fancy gadget replaces staying organized, keeping meticulous logs, and actually putting tools back where they belong. Most incidents start with small lapses; discipline with storage stops risks right where they start. For anyone handling potentially dangerous materials, setting up good storage habits from the start means you’re less likely to need the safety equipment you keep close by.
How should Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate be disposed of safely?
Understanding the Risks Behind the Name
Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate doesn’t show up in the daily vocabulary of most folks, but it matters in science and industry. We’re talking about a compound related to persistent organic pollutants, and if you’ve spent any time around labs or chemical plants, you’ve seen the fallout from chemicals like this getting out into the world. Exposure can spell trouble for both people and the environment, since breakdown often leads to even nastier byproducts. So, tossing this chemical in the trash or pouring it down the drain doesn’t cut it.
Laws Shape Every Step
As someone who’s stepped through more than a few lab safety briefings, one point never changes: local and national regulations decide what you can and can’t do. The U.S. Environmental Protection Agency (EPA) and European Union’s REACH laws both list derivatives linked to DDT, which tells you straight off this compound is no joke. That means ordinary waste services won’t take it. Take it from someone who’s been grilled on proper labeling—hazardous chemicals like this belong in trusted hands.
On the Front Lines: Real-World Handling
Lab and industrial workers rely on trained hazardous waste professionals. These folks suit up, gather the chemical in tightly sealed containers—think materials that stand up to leaks, corrosion, and tumbling during transit. They tag it with clear hazard symbols. Their risk assessments decide on chemical compatibility and storage. In smaller labs, every milliliter of solvent and byproduct might go into a safety can before a scheduled pick-up.
Open air burning or flushing poses real threats. Animal studies have linked chlorinated organics with cancer, reproductive issues, and genetic damage. Waterways near improperly managed dump sites cycle those problems back into the food chain. Fish, birds, and eventually, humans, pay the price.
Destroy, Don’t Dump
Destruction through high-temperature incineration ranks as the method that actually eliminates the hazard. Good incinerators break these molecules down—temperatures over 1,100°C, scrubbers in place to trap volatile byproducts. Some licensed facilities pyrolyze waste to break it into safer fragments. No backyard fire or steel barrel can manage this. From personal experience, waiting for the quarterly pickup from a certified processing center always beats facing an OSHA fine or a ruined ecosystem.
Reducing the Pile in the First Place
Upstream, researchers and companies can swap in greener alternatives for synthesis or opt for smaller production runs. For years, green chemistry has chipped away at the industrial reliance on persistent pollutants. My time in academic labs showed that waste audits, limited stockpiling, and better purchasing practices shrink disposal headaches. The less of this chemical produced, the less risk for accidental exposure in labs, storerooms, and the spaces where people live.
Community Accountability
Neighbors want transparency, and so should anyone working with chemicals at scale. Regular environmental monitoring, open reporting, and honest risk communication build public trust. When accidents do happen, clear emergency plans protect workers, clean-up crews, and anyone downstream from spill sites. It’s not about fear, but about recognizing responsibility. Safety around chemicals starts long before the chemical needs disposal.
What is the chemical structure or formula of Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate?
Unpacking a Structure with Weight
Ethyl 2,2-Bis(4-chlorophenyl)-2-hydroxyacetate might sound like a term best left to the depths of a chemistry lab, but the structure packs meaning far outside textbooks. This molecule stands as a near relative of DDT, one of the most controversial pesticides of the 20th century. It’s easy to overlook why structure matters, yet the atoms and their arrangement make all the difference—affecting not just what the compound does, but how it persists in the world.
Decoding the Formula
The formula for Ethyl 2,2-Bis(4-chlorophenyl)-2-hydroxyacetate is usually written as C16H14Cl2O3. Picture it: two benzene rings, each with a chlorine hanging at the “para” position (directly across from each other), tethered to a central carbon. Add a hydroxy (-OH) group and an ethyl acetate stringing off that same carbon. That central carbon becomes a hub, tying together pieces that define the molecule’s behavior in the environment and in chemical synthesis.
Why the Details Make a Difference
This isn’t just chemistry trivia. Chemical structure guides everything—resistance to breakdown, movement through soil and water, potential effects on living tissues. History handed us a tough lesson through DDT: Small changes in a molecule’s arrangement can spill into big ripples for public health, wildlife, and ecosystems. The presence of those chlorinated rings means this compound carries not only strength as a chemical tool, but also stubbornness. Chlorine on a benzene ring shields it from bacteria and sunlight, so it tends to linger wherever it goes.
Experience from the Field
I remember seeing older textbooks putting diagrams side by side—DDT’s core and this derivative—reminding students there’s a thin line between what sits locked in a shelf and what gets unleashed in a field. Farmers, scientists, doctors: all became linked by the results of how these molecules behave. Even casual gardeners started wondering what was drifting into their soil. That's why the way atoms connect rings so loudly beyond labs.
Responsible Chemistry and Next Steps
Chemists and regulatory bodies now look more closely at molecular makeup before signing off on widespread use. The lessons written into C16H14Cl2O3’s blueprint call for better screening—predicting persistence, testing bioaccumulation, and mapping out unintended effects up front. The data backs it up: Chlorinated aromatics tend to resist natural breakdown, so designing molecules for purpose and safety isn’t optional. The EPA and similar agencies recommend stricter evaluation based on both structure and behavior, not just effectiveness.
Design for the Future
Tools exist, but they need regular updates: molecular modeling, computer-aided screening, real-world deployment, and environmental monitoring. Teaching younger chemists and product designers to look not just at what a molecule does, but what it could become, flips the script. Pushing for alternatives with less environmental hangover keeps the industry moving forward without repeating the past. Watching science evolve, from DDT’s dawn to new generations of safer compounds, underlines why structural details deserve everyone’s attention—not only the person mixing at the bench.
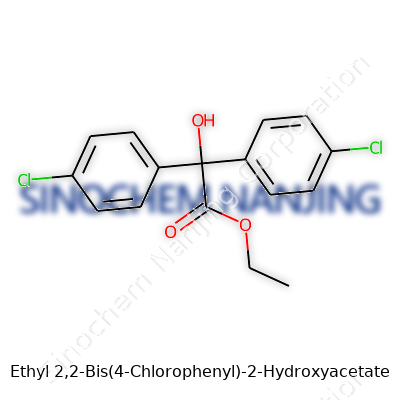
| Names | |
| Preferred IUPAC name | Ethyl 2,2-bis(4-chlorophenyl)-2-hydroxyacetate |
| Other names |
Ethyl bis(4-chlorophenyl)hydroxyacetate Ethyl 2-hydroxy-2,2-bis(4-chlorophenyl)acetate ESTONIC ACID Ethyl ester of DDA (Dichlorodiphenylacetic acid) Ethyl bis(p-chlorophenyl)hydroxyacetate |
| Pronunciation | /ˈiːθɪl ˈtuː tuː ˈbɪs fɔːr ˈklɔːrəˌfɛnɪl tuː ˈhaɪdrɒksi əˈsiːteɪt/ |
| Identifiers | |
| CAS Number | 24453-76-3 |
| 3D model (JSmol) | `JSmol` string for Ethyl 2,2-Bis(4-Chlorophenyl)-2-Hydroxyacetate: ``` CCOC(=O)C(O)(C1=CC=C(C=C1)Cl)C2=CC=C(C=C2)Cl ``` |
| Beilstein Reference | 3737039 |
| ChEBI | CHEBI:8087 |
| ChEMBL | CHEMBL3611255 |
| ChemSpider | 21853417 |
| DrugBank | DB07522 |
| ECHA InfoCard | 03b42a63-efde-4bb2-a11a-8915b9924981 |
| EC Number | 205-535-0 |
| Gmelin Reference | Gmellin 83387 |
| KEGG | C06698 |
| MeSH | DDT |
| PubChem CID | 11404 |
| RTECS number | GV5950000 |
| UNII | 63F918552G |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C16H14Cl2O3 |
| Molar mass | 367.18 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.43 g/cm3 |
| Solubility in water | Insoluble in water |
| log P | 2.85 |
| Vapor pressure | 1.4E-6 mmHg at 25°C |
| Acidity (pKa) | 11.58 |
| Magnetic susceptibility (χ) | -0.00012 |
| Refractive index (nD) | 1.600 |
| Viscosity | 1.0127 g/mL at 25 °C |
| Dipole moment | 3.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 489.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -471.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1518.9 kJ/mol |
| Pharmacology | |
| ATC code | N06AA02 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P305+P351+P338, P333+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | Flash point: 196°C |
| Lethal dose or concentration | > LD50 oral rat 2500 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3500 mg/kg (rat, oral) |
| NIOSH | PC3325000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | Not established |
| Related compounds | |
| Related compounds |
Dichlorodiphenyldichloroethane (DDD) Dichlorodiphenyldichloroethylene (DDE) Dichlorodiphenyltrichloroethane (DDT) Ethyl 2-hydroxy-2-phenylacetate Bis(4-chlorophenyl)methanol |

