Dysprosium Nitrate: From Rare Earth Curiosity to Industrial Commodity
Tracing the Growth of Dysprosium Nitrate
Dysprosium nitrate carries an interesting story that stretches across more than a century. Long before it showed up in technical catalogs or lab shelves, dysprosium itself took decades to isolate from the mineral mixtures of the late 1800s. The industrial rise of magnets and nuclear power changed things. By the 20th century, researchers started synthesizing dysprosium compounds, including the nitrate, in response to growing demand in electronics and energy applications. Dysprosium nitrate moved from obscure chemistry sets into processes tied to lasers, lighting, and new materials. Its story follows society's shifting focus—starting as scientific curiosity and winding up as a targeted ingredient in very modern technologies.
What Sets Dysprosium Nitrate Apart
You don’t stumble across dysprosium nitrate among kitchen supplies or hardware store shelves. What stands out is its crystalline form: it lands as off-white to pale yellow crystals, typically produced as the pentahydrate. This stuff dissolves well in water but keeps clear from most organic solvents. You can smell faint acid when the bottle is opened—thanks to the nitric acid component clinging to the rare earth metal. Unlike its plain oxide sibling, dysprosium nitrate slips into liquid processes and reacts easily with other chemicals during material synthesis or etching routines. The formula Dy(NO3)3 matches its chemical form, holding three nitrate units welded to a dysprosium atom.
Specs, Labels, and What’s in the Bottle
Buyers care about more than the name. Good suppliers give technical specs covering purity (usually 99.9% or higher), moisture content, absence of hazardous metal impurities like lead or iron, and crystal size range. The labeling includes batch number, date of manufacture, CAS number (10031-49-9), plus emergency contact stuff. If you check the actual lot, you’ll find granules or powder sealed against air and moisture, often double-bagged. Some researchers prefer their samples ultrapure for laser crystals or magnetic studies, while others settle for standard-grade batches in ceramics or glass fiber work.
Making Dysprosium Nitrate: From Ore to Lab Bench
Dysprosium rarely shows up pure in nature. Extracting it involves processing bastnäsite or monazite minerals, first busting the ore with acids, then laboriously separating dysprosium from other lanthanides through multi-stage solvent extraction or ion exchange. To make the nitrate, the metal oxide (Dy2O3) reacts with nitric acid, usually under gentle heat, until the solution goes from dull to clear. Drying the solution delivers the hexahydrate or pentahydrate crystal. Each cycle demands careful handling—nitric acid has no mercy for careless skin or lungs, and rare earth oxides can be pricey. In practice, large producers fine-tune their process for scale, tweaking temperature, acid ratios, and wash techniques to lift yield and cut down on contaminants.
What Dysprosium Nitrate Does in the Lab
Chemists appreciate dysprosium nitrate for its versatility. Add strong heat or a reducing agent, and the compound drops the nitrate group, forming dysprosium oxide or other specialty chemicals. Reactions with organic ligands let researchers build custom complexes for catalysis or luminescent applications. It holds value in preparing advanced ceramics, magnetic powders, or as a dopant in glass and fiber lasers. Some academic labs focus on modifying its structure or bonding new organic moieties to unlock fresh optical or catalytic functions. Its nitrate part offers both reactivity and a gentle handle to carry dysprosium into solution, critical for uniform mixing and targeted reactions.
Other Names, Same Compound
This compound pops up under a basket of synonyms. If you spot terms like "dysprosium(III) nitrate," "nitrate de dysprosium," "dysprosium trinitrate," or "nitrato de disprosio," you’re staring at the same stuff. Lab catalog listings and import/export paperwork sometimes use the CAS, EINECS (233-225-4), or even customs codes, but the core material remains unchanged. These alternate names mostly help cross-check sources or locate the product across languages and regulatory systems, especially when sourcing for international research or manufacturing projects.
Safety Rules from the Real World
Dysprosium nitrate never gets shipped without its safety data sheet. It’s not radioactive, but it deserves respect. Don’t breathe the dust—rare earths as a class can irritate lungs and mucous membranes. Since it comes laced with nitric acid residues, the powder stings cuts or burns eyes on contact. Gloves, goggles, and ventilation turn into daily routine for anyone dispensing it in a lab or industrial setting. Storage should be air-tight and dry, far away from organic combustibles. Shipping classifies the compound as hazardous, requiring careful packaging, labeling, and sometimes even special permits. Mishaps lead to corrosive spills and potential skin or respiratory injury, so first-aid plans belong front and center where the chemical is used.
Where Dysprosium Nitrate Ends Up
On the application side, dysprosium nitrate anchors itself in fields where regular reagents fall short. Scientists reach for it in crafting phosphors for lighting, doping fiber optic cables, boosting magnet strength in wind turbines, or etching laser elements. In the electronics industry, makers use it in growing rare earth-doped crystals for solid-state lasers and medical imaging. Its role in magnetic cooling technology stands out for energy-efficient refrigeration—fields eyeing new ways to cut carbon impact. Defense and aerospace sectors use it in specialized ceramics or magnets that work under crushing pressure or extreme heat. Every end-user has their preferred grade and format, reflecting the tough demands of lasers, magnets, or high-grade optical components.
R&D and the Push for New Properties
Academic and industrial researchers keep stretching dysprosium nitrate’s value. Labs design new synthetic routes to help it blend with nanoparticles, liquid crystals, or organometallic agents. Others probe ways to recycle rare earth streams, cutting back on environmental mining damage tied to primary extraction. Some groups run quantum calculations, hunting for tweaks that might make dysprosium complexes more effective as MRI contrast agents or quantum dots for displays. The field never holds still for long—a small change in the reaction formula opens whole new families of materials, sometimes with dramatically better performance in optics, magnetism, or energy.
Digging Into Its Toxicity
People handling dysprosium nitrate want straight answers on risks. Acute toxicity sits in the low-to-moderate range, with rare earths causing skin, eye, or lung irritation but rarely threatening fatal poisoning at research-scale exposure. Chronic risks emerge if inhaled dusts linger over months—animal studies suggest issues like lung or liver disruption, though most observed at higher doses than lab or factory workers would normally see. The nitrate part brings typical acid hazards; accidental ingestion hits stomach lining and may cause vomiting or pain. Smart users store it safely, train regularly, and install spill response plans before accidents happen because rare earth compounds deserve serious precaution due to their persistent, cumulative nature in environmental settings.
Looking Ahead: Where Dysprosium Nitrate Might Go
The outlook for dysprosium nitrate links right back to the rise of green technology, advanced manufacturing, and quantum electronics. Researchers see it as a bridge to better rare earth magnets, which affect everything from electric vehicles to national power grids. Cleaner, smarter chemistry emerges as more labs adopt closed-loop recycling methods, slashing the carbon footprint and pollution from rare earths. Meanwhile, regulatory changes and supply bottlenecks push some teams to invent new synthetic routes, sometimes blending dysprosium nitrate with bio-based ligands or using renewable solvent systems. As big companies step up demand for robust magnets and miniaturized sensing materials, dysprosium nitrate sits in a growth lane. Its future depends on how well society manages resource security, environmental impact, and the next wave of technology that will need this oddball but critical ingredient.
What is Dysprosium Nitrate used for?
What Dysprosium Nitrate Means for Modern Tech
Dysprosium nitrate, with its pale yellow crystals, doesn’t catch much attention outside labs. Out in the real world, though, this chemical plays a bigger role than most folks realize. Every time somebody uses a smartphone or drives a hybrid car, there’s a good chance a piece of that tech runs thanks to materials like dysprosium. This compound doesn’t show up in the end product, but it helps shape the world around us.
Crucial Ingredient for High-Performance Magnets
In my years following advances in clean energy, I found dysprosium at the center of lots of cutting-edge projects. It’s used to make high-strength magnetic alloys, which show up in electric motors, wind turbines, and advanced vehicles. Magnets made with dysprosium keep their strength even at high temperatures—something standard magnets struggle to do. If you ever noticed how smoothly an electric car accelerates or how a wind turbine spins out by the highway, that performance often traces back to rare earth elements like this one.
Nuclear Reactors and Safety
Dysprosium nitrate lands on the shopping list for nuclear reactor operators. Most folks never think about what keeps a reactor safe. In this field, dysprosium helps control the reactions inside, since it absorbs neutrons better than many other materials. Reactors need this property to avoid overheating or going out of balance. Keeping nuclear plants both efficient and secure takes solid science backing every step, and dysprosium nitrate supports that aim.
Research and Laser Technology
I spent some time in university research labs, where new ideas become real-world products. Scientists often use dysprosium nitrate when working with advanced optics and laser technology. The element’s unique properties help tune lasers for surgery, communications, and precision measurement. People might not see it, but work in these labs produces tools that end up in hospitals, research centers, and even satellites. Many of these breakthroughs start with compounds just like this one.
Environmental Impact and Supply Chain Issues
Every material with value runs into supply challenges. Dysprosium comes from a handful of countries, China leading the pack. That creates risk—not only for manufacturers but also for the countries trying to build up their clean energy sectors. There’s been international debate about how to secure sources, recycle used materials, and invest in alternative chemistries. I’ve watched companies dig deep to find substitutes or push for more recycling of rare earth elements. Progress doesn’t come easy, yet the push is there because so much relies on these hidden ingredients.
Moving Forward: Sustainable Use and Innovation
We’ll keep hearing more about rare materials like dysprosium nitrate as the world leans into renewable energy and electric vehicles. Scientists focus on ways to reuse old magnets, and some engineers look for alternative elements when possible. Industry leaders now emphasize transparent sourcing and recycling projects. More investment in mining, refining, and innovation helps limit bottlenecks and reduce tensions over trade. For folks outside high-tech labs, dysprosium nitrate still might sound distant, but its story connects to cleaner energy, safer power, and smarter products right in our pockets.
What is the chemical formula of Dysprosium Nitrate?
Understanding Dysprosium Nitrate
Most folks don’t wake up thinking about rare earth elements, but dysprosium nitrate quietly powers plenty of modern tech. The formula, Dy(NO3)3, looks simple, but each part packs a punch. That “Dy” stands for dysprosium, an element often pulled out of the ground in far-off places, mostly in China and a handful of other countries. The nitrate part, three times over, gives the compound properties that scientists and engineers chase for their projects.
Why Chemical Formulas Actually Matter
Chemical formulas keep things honest. Getting a single number or letter wrong can turn a chemical from something useful into something dangerous. Dy(NO3)3 contains a dysprosium atom paired with three nitrate groups, not two or four. That means different chemical behavior, from how it dissolves in water to how it reacts when heated. Mistakes here in a lab setting could lead to failed experiments, wasted money, or even safety risks.
Dysprosium’s Real-World Presence
Growing up near an industrial city, I learned quick that complex-sounding chemicals shape everyday products. Dysprosium nitrate doesn’t land on grocery store shelves, but it winds up in high-performance magnets, sometimes even in electric cars. The nitrate compound serves as a bridge, helping manufacturers get dysprosium into the right form for making permanent magnets strong enough to drive motors in everything from hybrid vehicles to wind turbines. These devices don’t work without materials refined to the right purity and composition, which always circles back to having the right chemical formula.
Issues Surrounding Production and Supply
Manufacturing parts with dysprosium nitrate can dig up questions about sustainability and fair trade. Rare earth mining takes a toll on natural landscapes and puts pressure on local communities near mining sites. Health risks rise when workers don’t have proper protection. As with many chemical compounds, improper storage or disposal of dysprosium nitrate can harm water supplies. The mining countries sometimes tighten the tap, controlling the world’s access in a way that leaves some industries scrambling.
Ideas For Safer, Smarter Use
It doesn’t pay to turn a blind eye to sourcing and handling. Companies should keep a closer eye on how they get dysprosium, choosing suppliers who invest in safer mining and cleaner processing. Regulations for shipping and storing chemicals like dysprosium nitrate need teeth, not loopholes. Recycling tech magnets and batteries could help reduce reliance on new mining, cutting down environmental harm as demand for electric cars and green energy grows. Consumers can ask more questions about where the materials in their tech come from, nudging the market in a cleaner direction.
Facts For Reference
A single hybrid car motor can use several grams of dysprosium in its magnets. Worldwide, factories use thousands of tons of rare earth compounds each year, and dysprosium nitrate is just one form that keeps the supply chain moving. Handling and labeling with the correct chemical formula keeps everyone safe and the production lines running.
How should Dysprosium Nitrate be stored?
The Real-World Challenge of Handling Rare Earth Compounds
Walk through any lab that deals with rare earth materials and you’ll quickly realize these compounds ask for more than casual oversight. Dysprosium nitrate sits high on that list. It delivers valuable magnetic and luminescent properties but demands disciplined storage. In my years involved in technical environments, a misstep with nitrate salts rarely ends well. Too often, ignoring the basics leads to ruined samples—sometimes much worse.
Respecting Its Nature—Why Storage Details Matter
For dysprosium nitrate, water is both a friend during synthesis and an enemy in storage. This compound pulls moisture from the air, clumping up or turning into a sticky mess if someone leaves the cap off a bottle. Laboratories in humid regions fight this battle all year. Even if you avoid an outright spill, these salts manage to find their way onto hands or bench tops, and their oxidizing nature can start slow, unwanted chemical reactions.
Fire hazards aren’t a scare tactic. Nitrate ions promote combustion and can make an otherwise small lab accident much worse. Tossing this material into a cabinet with everyday flammables or organics borders on reckless.
Best Practices—Lessons from the Field
Practical experience counts for more than a shelf of safety manuals. Here’s what ongoing practice shows. Glass containers with tight screw-caps—sometimes with PTFE liners—stand up to nitrate salts far better than plastic, which can get brittle over time. Storing the substance in a cool, dry room keeps it from degrading or absorbing too much water from the local atmosphere. A dedicated, labeled desiccator removes most of the risk from the start. Silica gel or anhydrous calcium chloride inside the desiccator create a dry microclimate, giving the material a better shot at a long shelf life.
Staff turnover brings new risks. Chemical inventory procedures work best when they’re more than a box-ticking exercise. Regular checks on how many people actually understand the risks and participate in refresher training have a bigger impact than a dozen warning signs on a cabinet.
Supporting Safe Work—Not Just for the Big Players
Even small startups handling dysprosium nitrate find value in treating storage protocols as non-negotiable. Having a chemical hygiene plan that outlines exactly where and how this salt will be stored saves a lot of guesswork. Keeping up-to-date safety data sheets within easy reach isn’t just bureaucracy—it’s about making sure everyone knows the hazards and emergency procedures.
Laboratory fire departments and regulatory agencies share plenty of horror stories about what happens when chemicals get mixed up, mislabeled, or forgotten in the wrong spot. Relying on clear labeling, regular audits, and shared responsibility makes more sense than trusting chance.
Solutions for Today’s Laboratories
As supply chains bring more rare earths to labs worldwide, solid storage habits matter as much as technical skill. Investing in humidity monitors, double-checking how well desiccators are maintained, or simply double-bagging containers for transport can save huge amounts of time and money later. Cross-training teams and spreading knowledge help prevent missteps, especially as labs scale up.
Dysprosium nitrate isn’t a household chemical. It doesn’t forgive mistakes, and taking shortcuts does not work out. Respecting its chemical temperament and enforcing good storage discipline proves far easier than cleaning up after a mistake that should have been avoided.
Is Dysprosium Nitrate hazardous or toxic?
Understanding the Risks
Chemical names often sound intimidating, but safety always comes down to how a substance affects real people in real settings. Dysprosium nitrate is a compound that crops up in labs and some industries using rare-earth elements. The name alone doesn’t make it dangerous, but the facts about it deserve a close look.
On paper, dysprosium nitrate is labeled as hazardous. The main risks come from its corrosive qualities and its strong oxidizing nature. My own stint working in a research lab taught me that even chemicals used in small amounts can go from benign to problematic, depending on how they're handled or stored. Skin contact leads to burns, and inhaling its dust or mist may cause irritation or even respiratory trouble. The Material Safety Data Sheets don’t sugarcoat it—protective gloves and eyewear are not just helpful, they are essential.
Why This Matters
It’s tempting to think that rare-earth chemicals only affect folks in white coats or hazmat suits, but that’s not the case. I’ve watched as old containers got knocked off a storage shelf, and everyone within arm’s reach needed to know how to respond, fast. Dysprosium nitrate releases nitrogen oxides and other toxic fumes when heated. These fumes don’t just vanish in air—they irritate the lungs and nose, and high doses have been linked to lasting respiratory issues.
Handling this stuff outside a fume hood puts everyone at risk, not just the person doing the mixing. I remember one safety drill that went sideways because someone didn’t realize how quickly fumes can drift. In less controlled settings, those lapses stack up and the risk multiplies.
Toxicity and Environmental Impact
Many chemicals labeled as hazardous also carry a big environmental footprint. Dysprosium nitrate ticks that box too. Spills seep into groundwater or soil and disrupt local ecosystems. Plants and aquatic life don’t have much tolerance for nitrates or rare-earth metals, and build-up over time leads to real-world harm. Some studies show that even low concentrations can change microbial communities or put stress on fish.
Workers and nearby communities end up shouldering the burden of improper disposal. I spent time near an old chemical plant, and I saw firsthand how a lax approach to waste management can poison drinking water and ruin farmland. The pain and expense linger for years.
Working Toward Safer Handling
Risks become manageable when information flows freely and training stays current. Any college chemistry course opens with safety lessons, but outside the classroom shortcuts sneak in. I’ve met experienced techs who skipped gloves or left open bottles on benches because the bottle “looked clean.” Posting reminders about proper procedures isn’t enough. Safety culture demands practice, accountability, and supervision that doesn’t cut corners.
Modern labs invest in training, better containment, and rapid response kits. Disposal gets handled by specialists who know local regulations for hazardous waste. There’s no substitute for real expertise or high standards. Those lessons spill over to other industries, helping to prevent small mistakes from growing into big disasters.
Looking Ahead
Dysprosium nitrate has real value in electronics and scientific research, but nobody gains from treating the hazards as an afterthought. Continuous training, strict protocols, and regular health monitoring create a buffer between useful work and lasting harm. My own missteps taught me to never roll the dice with hazardous materials—respect for the risks keeps everyone safer.
What are the physical properties of Dysprosium Nitrate?
An Element Hidden in Plain Sight
Most people can’t point to dysprosium’s place on the periodic table, but this grayish metal actually plays a critical role in some of our toughest tech. In the real world, few get to handle this element pure. The most common contact happens through salts, like dysprosium nitrate.
Looks and Texture
Crystals of dysprosium nitrate often shine with a pale, almost colorless appearance. Touch them and they feel gritty, sort of like table salt spilled dry on a countertop. Water brings out a different side; the crystals dissolve easily, making a clear, acidic solution. Most batches you find hold onto molecules of water—these hydrate forms are the bulk of what ends up in bottles worldwide.
Odor and Taste
Pure dysprosium nitrate doesn’t carry any scent you’d notice, and tasting it is strictly for the reckless or the foolish. Like many rare-earth compounds, even tiny slivers present real health risks—avoid the temptation to test it with your tongue.
Melting, Boiling, and Beyond
This chemical doesn’t melt like chocolate or sugar. Add heat and the solid hydrates decompose, not into a puddle, but into gases and other compounds long before seeing a proper liquid state. Heating leads to fumes of nitrogen oxides—think dark red or brown smoke—which you do not want to inhale for any reason. Good airflow and protection matter here.
It doesn’t stand up to fire the way some metals do. Instead, it’s the first to break down, giving off smoke and losing its crystalline form well below the temperature most metals will melt.
Solubility: The Water Test
Dysprosium nitrate passes the classic chemistry class test: drop it in water and watch it disappear, leaving no cloud or residue. This property—thanks to those hydrating water molecules—lets it move easily through solutions in labs and factories. Ethanol will take it up too, a useful trait for separation and purification work.
Density and Weight
Dysprosium itself is pretty dense, but the nitrate version, with its water of crystallization, holds up lighter than you might think. Pour these crystals off a scoop, and you’ll see how they flow without feeling overly heavy in your grip. It makes them manageable for researchers, not handling a lump of lead.
Stability and Handling
Set a jar of this chemical out in a humid room, and it’ll start to clump, pulling water straight out of the air. This pulls the substance into bigger crystals or sometimes leaves it mushy—hardly ideal for storage. Sealed containers and dry rooms cut down on losses and keep the nitrate in ready-to-use form.
Safety and the Real World
Most chemical labs have strict rules for rare earths, and dysprosium nitrate is no exception. It’s labeled hazardous, capable of irritating eyes and skin badly if you get careless. Gloves, sealed bottles, and proper ventilation aren’t extras—they’re essentials. A few splashes into your local water source could disrupt aquatic life, so disposal means following every local guideline to the letter.
In my own work with rare earths, a colleague once underestimated how quickly these compounds grab moisture. In a few hours, an open jar turned from dry powder into a sticky lump, contaminating other samples and wasting valuable product. That memory sticks as a reminder that with powerful materials—the everyday stuff like air and water can cause as much trouble as the chemicals themselves.
Looking Forward
As demand for rare-earth materials grows, those physical quirks—deliquescence, solubility, sensitivity—require smarter storage and handling tools. Dry boxes, well-labeled sealed jars, and patience in weighing samples save headaches and money. Safety awareness must run deep, from scientist to technician, keeping both people and environment out of harm’s way.
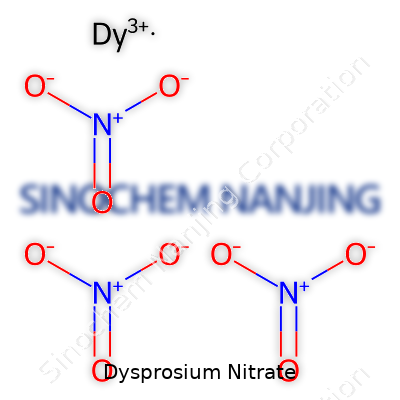
| Names | |
| Preferred IUPAC name | dysprosium(3+) trinitrate |
| Other names |
Dysprosium(III) nitrate Dysprosium trinitrate Nitric acid, dysprosium(3+) salt |
| Pronunciation | /daɪsˈprəʊziəm ˈnaɪtreɪt/ |
| Identifiers | |
| CAS Number | 10031-49-9 |
| Beilstein Reference | 3287662 |
| ChEBI | CHEBI:52070 |
| ChEMBL | CHEMBL4296618 |
| ChemSpider | 20396577 |
| DrugBank | DB14792 |
| ECHA InfoCard | echa.infocard.100.039.980 |
| EC Number | 236-978-9 |
| Gmelin Reference | 16316 |
| KEGG | C18674 |
| MeSH | Dysprosium Compounds |
| PubChem CID | 159423 |
| RTECS number | OP9625000 |
| UNII | SZR8XH6I1C |
| UN number | UN1466 |
| CompTox Dashboard (EPA) | 6ff6e3f2-74f9-4b01-8a5c-2ec6de15be45 |
| Properties | |
| Chemical formula | Dy(NO3)3 |
| Molar mass | 373.51 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.56 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.64 |
| Vapor pressure | Negligible |
| Basicity (pKb) | pKb ≈ 10.1 |
| Magnetic susceptibility (χ) | +11500e-6 cm³/mol |
| Refractive index (nD) | 1.515 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 213.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1646.0 kJ/mol |
| Pharmacology | |
| ATC code | V09XX04 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes serious eye irritation. |
| GHS labelling | GHS05, GHS07, GHS08 |
| Pictograms | GHS03,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H272: May intensify fire; oxidizer. H314: Causes severe skin burns and eye damage. H319: Causes serious eye irritation. |
| Precautionary statements | Precautionary statements of Dysprosium Nitrate: "P210, P220, P221, P264, P273, P280, P305+P351+P338, P370+P378, P501 |
| NFPA 704 (fire diamond) | 1-0-0-OX |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 788 mg/kg |
| NIOSH | WN3655000 |
| PEL (Permissible) | 10 mg/m3 |
| REL (Recommended) | 5 mg/m3 |
| IDLH (Immediate danger) | IDLH not established |
| Related compounds | |
| Related compounds |
Lanthanum nitrate Gadolinium nitrate Holmium nitrate Erbium nitrate Lutetium nitrate Yttrium nitrate |

