Dl-2-Amino-4-Pentenoic Acid: A Deep Dive into Its Role and Future
Historical Development
Long before synthetic chemistry made its mark on pharmaceuticals, amino acids played silent roles in the body’s complexity. Dl-2-amino-4-pentenoic acid, known in laboratories as a less-common analog of familiar amino acids, emerged from this intricate backdrop in the 20th century. By the late 1900s, researchers in organic synthesis explored this molecule as both a building block and a probe. Early experiments, especially in European labs focused on peptide synthesis, unveiled not just the structure of this compound but teased out its utility in modifying natural biochemical sequences. Through several decades, chemical suppliers and academic groups kept up the pace, expanding methods of production and application, often targeting disease models or protein research. Oil shortages and advances in catalysis influenced how scientists sought to optimize production, adapting lab methods for larger-scale use.
Product Overview
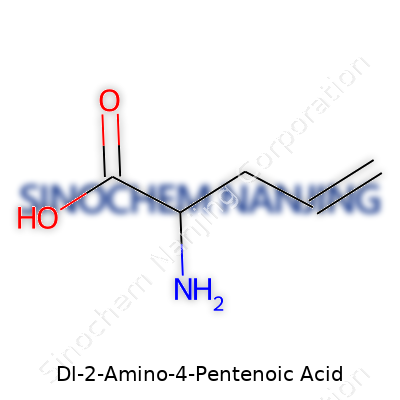
Dl-2-amino-4-pentenoic acid, also known as DL-allylglycine, stands out with a straightforward, five-carbon backbone and a vinylic group attached near the tail. Manufacturers and catalogues frequently provide both racemic and optically pure forms, delivering the raw material to peptide researchers, neuroscientists, and those designing enzyme inhibitors. The appeal here rests not just in its novel structure; this compound threads a careful needle between chemical curiosity and biological significance. Chemical catalogs list it for its unique ability to substitute in polypeptide chains, distorting or enhancing biological activity for investigative purposes. Plenty of research institutions turn to this amino acid’s analogs, searching for subtle changes in receptor function or enzyme kinetics.
Physical & Chemical Properties
This compound typically crystallizes as a white, free-flowing powder that dissolves readily in water. Melting points reported by various labs land between 179 and 185°C, giving it some stability for handling and storage in standard chemical facilities. The double bond in the side chain breaks away from most standard amino acids, granting it potential sites for further transformation—either through catalytic hydrogenation or radical chemistry. Its solubility in standard buffers matches glycine and alanine, making it easy to incorporate into biological assays or synthetic protocols. This structure welcomes nucleophilic attack at the carbon-carbon double bond, opening up a playground for organic modifications.
Technical Specifications & Labeling
Commercial suppliers provide DL-2-amino-4-pentenoic acid with purity values upward of 98 percent, usually determined by high-performance liquid chromatography. Typical vials carry hazard labels indicating low toxicity but recommend avoiding inhalation and contact with mucous membranes. Labels show batch numbers, structure, and storage conditions—dry and cool, away from light. Certificates of analysis detail specific rotation, melting range, and spectrum data, matching regulatory expectations for lab-based chemicals. Tracking lot-to-lot consistency poses occasional headaches, as different methods of production sometimes yield tiny shifts in crystal habit or trace impurities. Reliable documentation builds confidence for new users and recurring research programs.
Preparation Method
Synthesis begins with chloroacetic acid, which serves as a base for introducing amino and allyl groups. The Strecker synthesis anchors many preparative routes, running through a cyanohydrin intermediate. Reduction with hydrogen gas over palladium splits away unwanted functional groups, with followed up purification by recrystallization or ion-exchange chromatography. Some universities optimize yields with continuous-flow systems, while older batch protocols still see routine use in bench-scale operations. Handling the vinylic group without unwanted side reactions challenges even skilled chemists, as exposure to acids or high heat can polymerize or decompose the compound. The reproducibility of these reactions often depends on the skill of the bench chemist monitoring pH, temperature swings, and the timing of quenching steps.
Chemical Reactions & Modifications
Beyond its synthesis, this molecule thrives as a reagent and substrate. The allyl group entertains a wide range of addition, oxidation, and reduction reactions. In peptide chemistry, direct coupling via standard protection-deprotection cycles introduces dl-2-amino-4-pentenoic acid at defined points. Exposure to ozone cleaves the double bond, producing aldehyde functionalities for further manipulations. Radical-initiated transformations allow custom-tailored side chains, adjusting hydrophobicity or reactivity. Some medicinal chemists transform the parent structure into cyclic analogs, hunting for leads against resistant microbial strains or stubborn cancer targets. N-alkylation, carboxyl modifications, and ring-closing reactions all illustrate the handling versatility within both research and commercial labs.
Synonyms & Product Names
Chemical repositories and vendor catalogs recognize several aliases: DL-allylglycine, 2-amino-4-pentenoic acid, and α-allylglycine. Some use historical names like 2-amino-4-vinylpentanoic acid. On custom-synthesized product lines or specialty research lots, you might see it stamped as AG5 or AGLY. These variants enable straightforward ordering but can confuse databases that try to cross-reference structures. Recognizing this, some researchers take care to include structural formulas alongside plain-language names in reports to keep communication clear.
Safety & Operational Standards
Lab users handle this amino acid under routines that protect against dust inhalation and skin contact. Safety data sheets list mild irritation risk and guide cleanup procedures for accidental spills. Goggles, nitrile gloves, and fume hoods factor into nearly every research protocol, not just as compliance but as practical habits. Waste management involves aqueous washes and collection for insoluble residues, feeding into streams compatible with existing lab disposal standards. Vendor-supplied documentation often includes additional warnings for working with its derivatives, especially those generated in situ during peptide synthesis. Institutional safety committees check that users remain up to date, and annual reviews keep protocols fresh and responsive to new information.
Application Area
Researchers seize on dl-2-amino-4-pentenoic acid’s unorthodox structure for probing neurotransmitter systems. Neuroscientists use it in animal models to mimic glutamatergic dysfunction, studying underlying mechanisms of epilepsy and neurodegenerative conditions. In the world of peptide engineering, this molecule opens doors to analogs with altered receptor affinity or stability. Agrochemical developers study its unusual chain as a model for nitrogen flow and metabolism in plants. A handful of teams investigate its derivatives as potential biosensors, linking changes in side-chain reactivity to sensor output. Scientific literature points to nearly fifty years of varied approaches, each one amplifying or tweaking the context in which this small compound reveals big effects.
Research & Development
Grants for synthetic biology and medicinal research keep the wheel turning. Labs in Europe, China, and North America chart new routes for incorporating this molecule into longer polypeptide chains or as a competitor in enzyme assays. Funding agencies look for promises of utility, pushing chemists to demonstrate practical use cases right alongside mechanistic insights. Besides biomedical directions, startups tinker with this molecule for novel material platforms, testing mechanical strength and chemical responsiveness in synthetic polymers. Conferences dedicated to amino acid innovation set roundtables for presenting new modifications, many times sparking collaborations that echo across continents. Young scientists cut their teeth optimizing purification protocols, building on decades-old methods while looking for green chemistry solutions that scale without unnecessary waste.
Toxicity Research
Groups studying dl-2-amino-4-pentenoic acid’s toxicity typically focus on animal models. Early mouse work flagged seizures as a repeatable consequence at moderate to high doses, owing to mimicry of neurotransmitter imbalances. Detailed pathways show elevated risks for neurological effects when administered directly to brain tissue, but oral and dermal exposures register much lower risk at small quantities handled in standard research settings. Modern studies pay close attention to metabolite profiles and organ specificity, measuring whether chronic, low-level exposure accumulates or clears. Regulatory frameworks now require thorough dossiers outlining both acute and chronic risk data, especially for labs using these materials beyond milligram scales. Responsible research hinges on paying attention to these details, not just to comply but to protect both workers and the environment.
Future Prospects
Over the next decade, demand for dl-2-amino-4-pentenoic acid in neuroscience and synthetic biology looks ready to expand. Advances in high-throughput screening platforms and peptide library synthesis amplify both consumption and inventive uses. Companies build out customized manufacturing based on trends pointing to novel inhibitors and biochemical tags. Some emerging fields, like soft robotics and smart materials, show interest for incorporating amino acid analogs into responsive frameworks. Environmental screening tools could pick up cues from modifications of this molecule, pushing its utility well past biochemical research alone. As knowledge deepens and industries broaden their reach, cross-sector standards will matter more than ever. Direct engagement between chemists, toxicologists, and manufacturers points to a future where regulatory clarity and technical creativity support promising new products while putting health and safety at the forefront. Tackling these practicalities with experience and a commitment to fact-based decision-making keeps the field both sustainable and innovative.
What is Dl-2-Amino-4-Pentenoic Acid used for?
The Core of a Unique Molecule
Mention Dl-2-Amino-4-pentenoic acid to anyone outside of a chemistry lab and you’ll probably get a blank look. In my days slogging through organic chemistry textbooks, names like this sparked groans. Most people wonder why scientists fuss over these compounds. That changes the moment one stumbles into the world of pharmaceuticals or specialized research.
Practical Importance In Research
Dl-2-Amino-4-pentenoic acid isn’t popular at drug stores or health food aisles. Its value lies in research. Scientists turn to this molecule when working on neurological studies, especially those exploring neurotransmitter regulation. As an analog of gamma-aminobutyric acid (GABA), this acid interferes with nerve activity in very specific ways, helping researchers figure out how synapses function and which routes brain signals use.
Back in the university lab, I remember the buzz around new GABA analogs. The mood changed any time researchers reported a finding involving nerve blockers or anything that let us map the nervous system a little better. Dl-2-Amino-4-pentenoic acid gave some students the tools they needed to trace how nerve signals slow down or speed up. Without these chemical "probes," progress would crawl.
Drug Development: More Than A Building Block
Drug makers often start with molecules like Dl-2-Amino-4-pentenoic acid before creating medicines that ease anxiety, treat seizures, or counteract nerve pain. Scientists mix and match these basic molecules, learning how structure changes effects. With epilepsy, for example, patients struggle with runaway nerve signals. Dl-2-Amino-4-pentenoic acid's structure lets scientists see which changes stop those misfires. It’s not a medicine you’ll take with your morning coffee, but it has helped steer a few new drugs toward clinical trials.
Making medicines isn’t just about combining whatever chemicals sound good together. It’s slow work, testing each variation. But stepping stones like this acid act as blueprints. Take Vigabatrin—a drug that manages seizures. The research that led to its discovery drew on GABA analogs, including compounds similar to Dl-2-Amino-4-pentenoic acid. Knowing how the acid fits with enzymes in the brain matters, since it points the way to more effective treatments with fewer side effects.
Synthetic Chemistry and Industrial Applications
Some chemists love synthetic challenges. Crafting molecules like Dl-2-Amino-4-pentenoic acid hones skills needed to build rarer, more complicated chemicals. This kind of practice builds experience for chemists looking to break into specialized pharmaceutical or biotech work. On the industrial side, only niche companies invest in production. The small scale means higher purity for lab experiments, not mass-market sales.
Challenges and Solutions
Outside academic walls, access to this molecule remains a hurdle. Regulatory rules, safety barriers, and price can slow work to a crawl. Smaller labs often can’t afford large quantities. Partnerships between universities and chemical suppliers can help bridge the gap. Pooled resources enable creative research that might otherwise get stuck due to cost. Grant programs and open-access initiatives provide opportunities for smaller projects to get their hands on specialty molecules without emptying their budgets.
Research on molecules like Dl-2-Amino-4-pentenoic acid reminds us that progress in medicine stands on less obvious building blocks. Every new discovery depends on the work done with these foundation compounds. The future of neurological medicine, and a few other fields, owes plenty to small molecules that rarely make headlines.
What is the molecular formula of Dl-2-Amino-4-Pentenoic Acid?
Making Sense of the Formula
Curiosity about molecules sparks bigger questions about how science helps us build medicines, treat disease, and understand life. Take Dl-2-Amino-4-Pentenoic Acid, for example. Its molecular formula is C5H9NO2. That means it’s got five carbon atoms, nine hydrogen atoms, a single nitrogen, and two oxygens all packed together. That formula is not just a string of letters and numbers—it’s a ticket to exploring a whole world of chemistry, biochemistry, and practical uses in labs.
Why Attention to the Exact Formula Pays Off
Every day, chemists rely on these formulas to design reactions, predict what molecules might do, or flag potential safety issues. Something as simple as swapping out an atom can change a molecule’s properties—sometimes with dramatic results. If you get the formula wrong in research, data interpretation goes off track. Waste of time, resources, sometimes even risk to health. In my experience, little chemistry errors snowball into headaches later, especially in pharmaceutical labs or academic settings.
Dl-2-Amino-4-Pentenoic Acid, with that structure, falls into a family related to amino acids. Each element matters: carbon forms the backbone for most organic molecules, hydrogen keeps the structure stable, nitrogen introduces the amino group necessary for forming proteins, and oxygen atoms help create acid groups that play a crucial role in biological reactions. Missing just one atom could change everything—the way the body handles the substance, how a drug gets absorbed, or whether a new chemical is toxic or safe.
Real-World Relevance: Beyond the Classroom
Many textbooks gloss over the importance of precise molecular formulas, but in the real world, they shape the development of new materials, foods, and medicines. I remember helping a team test alternative amino acids for potential use in treating metabolic disorders. Getting the formula wrong delivered faulty results, cost us weeks, and required re-running experiments. Patience for detail and respect for molecular structure save resources and lives, no exaggeration.
With more biosynthetic techniques on the horizon and research pushing into new territories, people need to trust that the science on the label matches what’s in the beaker. Dl-2-Amino-4-Pentenoic Acid might not be a household name, but its backbone tells stories about enzyme reactions, pharmaceutical innovation, and ways to mimic or block activity in the human body. That’s why the formula C5H9NO2 gets top billing.
Building Good Habits in the Lab
Checking formulas avoids mix-ups and paves the way for deeper discovery. In professional settings, digging into the numbers, drawing the structures, and double-checking sources keep experiments on a steady path. I’ve caught mistakes in papers and patents just because the number of hydrogens didn’t match up—a simple cross-check prevented bigger errors down the road.
For students or junior researchers, learning to appreciate the small details—like getting the right formula—opens up opportunities to spot trends, understand molecular behavior, and predict outcomes. Effective teamwork in the lab depends on everyone being on the same page about what molecules are floating around in the test tubes.
Solutions for Future Research
More accessible resources for molecular formulas, better digital tools, and collaborative peer review in scientific publishing can reduce mistakes. Encouraging double-checking, fostering partnerships between chemists and computational scientists, and publishing corrections when formulas are mishandled all help raise the bar.
Knowing the correct molecular formula for chemicals like Dl-2-Amino-4-Pentenoic Acid isn’t just about passing an exam—it’s at the core of doing science that can be trusted, shared, and built upon.
Is Dl-2-Amino-4-Pentenoic Acid available in different purities?
The Range of Purity in Dl-2-Amino-4-Pentenoic Acid
Anyone who has handled specialty chemicals like Dl-2-Amino-4-Pentenoic Acid knows purity never stays the same across the board. Research-grade labs demand tight tolerances, sometimes chasing 99% or higher. Reagent suppliers stick to specifications set out in catalogs, but real people doing real science often find those advertised numbers only tell half the story. Actual purity relies on the entire production chain—raw materials, cleaning steps, storage—all of it affects quality in the bottle.
Quality Isn’t Just a Number
During my own years in the lab, I’ve witnessed how a few percentage points make or break an experiment. Once, a colleague picked up a bottle labeled “97% pure” for a routine synthesis. Everything looked good until his reaction stalled—trace impurities shut it down. We tracked the problem to a vendor swap that introduced a slightly dirtier batch. Three percent changes a lot: color, odor, crystal shape, and above all, performance.
This is where experience comes in. High-purity chemicals cost more, but the extra cost translates into less troubleshooting, fewer failed batches, and less time wasted. Pharmaceutical work, for example, almost never accepts compromises on purity. Analytical chemists juggling sensitive measurements feel the same pressure. So while catalogues might list technical, reagent, or analytical grades, buyers read between the lines, sometimes grilling vendors for certificates of analysis, batch histories, or additional third-party tests.
Why Lower Purity Appeals in Some Cases
It’s tempting to dismiss anything below 98% as subpar, but economics shapes plenty of decisions. Farmers dosing animals or engineers making simple resins sometimes pick lower-purity options to stretch their budgets. The trick is learning where contaminants start causing trouble. In industrial settings, so long as impurities don’t poison the process or foul equipment, less-than-perfect product gets the job done.
I’ve also watched startups balk at high costs, only to discover quality shortcuts raise risk. Propagation of tiny errors at the chemical stage snowballs later, causing entire projects to stall. Investing upfront in better grades—whether 99%, 98%, or the elusive “ultra-pure”—pays off in reliability and credibility.
How Buyers Can Sort Through Options
Transparency is the fulcrum of trust here. Trusted vendors put out clear documentation, not hand-waving claims. Lab teams can demand batch-specific data sheets, sometimes running their own spot-checks using NMR or HPLC to confirm the vendor’s claims. In my lab days, we routinely split purchases, holding some back for baseline runs just in case a new shipment veered off course.
Reliable suppliers don’t hide test results or duck questions. Companies with a track record usually have processes in place to catch and flag outliers, and their support staff fields technical calls willingly. Sourcing from “gray market” or unvetted overseas outlets comes with obvious hazards—sometimes a discount means nothing but unrecoverable sunk costs when batches fail downstream.
Improving the Landscape
Merely selling purer forms isn’t enough. Industry can do better by making third-party testing routine, sharing data openly, and answering technical questions promptly. Scientists and buyers can advocate for clearer standards, not just “technical” or “analytical” jargon, but hard evidence about what’s inside the bottle.
In the end, high purity isn’t just about numbers—it’s about people protecting their work, their reputations, and the safety of anyone using the result. Making the extra effort here never goes to waste.
What are the storage conditions for Dl-2-Amino-4-Pentenoic Acid?
Understanding the Significance
Dl-2-Amino-4-Pentenoic Acid isn’t just another white powder in a glass flask. It plays a role in chemical synthesis and research. With any amino acid, but particularly one with a double bond like this, light, air, and moisture can change its structure and ruin experiments. Over the years in labs, I’ve seen careful planning go to waste simply because a compound sat out on the bench for a day longer than it should have.
The Essential Storage Conditions
Storage starts with temperature. Most suppliers, and my own experience, point to storing this compound in a refrigerator, typically around 2-8°C. Keeping it in the fridge slows down reactions with air or water vapor, which can break double bonds or cause side reactions. Some researchers choose the freezer, especially for long-term storage. This makes sense if you’re ordering in bulk or expect to work with the material over months or even years.
A dry atmosphere makes a difference. Moisture is a real risk, and nobody’s immune to humidity—particularly in the summer or in basements. To help, keep the product tightly sealed in its original bottle. If that’s not available, turn to amber glass bottles with tight caps, and always toss in a packet of desiccant. Silica gel is cheap and does the job. Avoid plastic bags; they often leak air and water vapor over time.
Light can break the amino acid’s double bond, easily missed if you use clear glass or leave materials out under bright lab lighting. Whenever possible, store it in dark or opaque containers. Sometimes, the manufacturer ships it in amber vials—don’t transfer to clear glass if you can avoid it. This simple step has saved samples more than once in academic and industry labs.
Risks of Poor Storage
Neglecting these precautions brings trouble. Exposure to air leads to oxidation, especially in compounds that don’t have strong stabilizing groups. If left open or on a bench, the powder can also absorb water and clump. I’ve pulled samples from the shelf before that turned sticky because someone left the cap loose. Not only is the compound likely to degrade, but analytical data becomes unreliable. That wastes both money and valuable lab time.
Research from reputable chemical suppliers backs this up. Sigma-Aldrich and TCI list “store at 2-8°C, protect from light and moisture” right on their labels. Publications describing syntheses of related amino acids have come to similar conclusions through trial and error. Strong evidence supports these routines—not just fussy techniques, but steps grounded in basic chemistry.
Best Practices for Everyday Handling
Keep an inventory log to prevent forgotten bottles from sitting unused for years. I set reminders to check the dryness of desiccant and the tightness of bottle caps every month. Always return the compound to the fridge promptly, whether after weighing or sampling. Label bottles with opening dates, as some degradation happens even under ideal conditions.
If you split material among colleagues, agree on rules: label everything, record each removal, and never return spilled or exposed powder to the storage container. Sharing responsibility ensures safety and protects the integrity of everyone’s work.
By being diligent about light, temperature, and moisture, Dl-2-Amino-4-Pentenoic Acid keeps its quality for the next important step in research.
Is Dl-2-Amino-4-Pentenoic Acid toxic or hazardous to handle?
The Basics of Dl-2-Amino-4-Pentenoic Acid
Dl-2-Amino-4-pentenoic acid has a lot of attention from labs and industry. Some folks call it an analog of amino acids, a fact that stirs curiosity among chemists. Using chemicals like this in daily research means questions about safety pop up for anyone in a lab coat.
Understanding Toxicity
This compound shares features with known neurotoxins. Structurally, it looks a lot like DL-allylglycine, which can mess with gamma-aminobutyric acid (GABA) production in nerve cells. GABA serves as a natural brake pedal in our brains—disrupt it, and seizures can follow. Rats exposed to high doses under controlled conditions end up with seizures. Eager chemistry students quickly learn that structure and effect often run together.
The safety data sheets on this compound mark it as hazardous. Touching or inhaling powder risks irritation. Swallowing it or leaving it on your skin too long means exposure to something the nervous system doesn’t like. There are no claims it causes cancer, but animal tests show neurological risks. That’s enough for anyone serious about lab safety to wake up.
Protecting Yourself in the Lab
Safety procedures matter. Real lab veterans know gloves and lab coats let them work without constant fear, but that goes double for a compound with ties to neurotoxicity. Goggles, well-ventilated fume hoods, proper labeling—those steps are non-negotiable. It’s not dramatic to say that stories exist of even seasoned researchers absent-mindedly skipping goggles and later regretting it.
Disposal also demands respect. Drains and regular garbage cans have no place in this process. Chemical waste should always land in labeled, secure containers picked up by professionals who specialize in hazardous material. Students and new lab techs sometimes think rinsing off a bench with water fixes everything—but neuroactive substances belong nowhere near common sewer systems.
Lessons from the Literature
Case studies don’t lie. Reports in the Journal of Neurochemistry and Toxicology tell about animal nervous system effects and irritation in the lungs and eyes. Mistakes happen, but recognizing risk means fewer hospital trips. Even if the regulatory agencies haven’t rolled out strict Standard Operating Protocols for Dl-2-amino-4-pentenoic acid like they have for cyanide or mercury, the building blocks are the same: treat unknowns with care, treat knowns with more.
A Better Way Forward
Anyone working with neuroactive or possible irritant amino acid derivatives needs proper training, not just handouts repeating generic warnings. Hands-on workshops get the message across far more than stacks of printed safety sheets. Regular safety drills remind teams that shortcuts carry consequences. Knowledge and vigilance beat overconfidence every time.
Moving forward, labs should keep pushing for clearer research on long-term health effects, not just acute toxicity. If documentation lags behind discovery, risk increases. Being open about what we know and what we don’t protects everyone in the chain, from students to experienced researchers. Using caution and building a culture of curiosity—rather than carelessness—lets innovation and safe practice go hand in hand.
| Names | |
| Preferred IUPAC name | 2-Aminopent-4-enoic acid |
| Other names |
DL-Homoallylglycine 2-Amino-4-pentenoic acid Dl-2-Amino-4-pentenoic acid |
| Pronunciation | /diːˈɛl tuː əˈmiːnoʊ fɔːr pɛnˈtiːnoʊ ɑːsɪd/ |
| Identifiers | |
| CAS Number | 19764-30-8 |
| 3D model (JSmol) | `CC\C=C(C(=O)O)N` |
| Beilstein Reference | 1209261 |
| ChEBI | CHEBI:85161 |
| ChEMBL | CHEMBL142873 |
| ChemSpider | 21318 |
| DrugBank | DB08338 |
| ECHA InfoCard | InfoCard: 100.136.255 |
| EC Number | EC 205-532-6 |
| Gmelin Reference | 74114 |
| KEGG | C01752 |
| MeSH | D000687 |
| PubChem CID | 10447 |
| RTECS number | UY1050000 |
| UNII | 7E25H96W3Y |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C5H9NO2 |
| Molar mass | 115.15 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 1.160 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -2.1 |
| Vapor pressure | 0.0 mmHg (25°C) |
| Acidity (pKa) | 2.39 |
| Basicity (pKb) | pKb 3.39 |
| Magnetic susceptibility (χ) | -5.53 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.495 |
| Dipole moment | 5.91 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 133.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | Not found |
| Pharmacology | |
| ATC code | N03AG01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS07, Warning, H302, H315, H319, P264, P270, P280, P301+P312, P305+P351+P338, P337+P313 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P363, P501 |
| NFPA 704 (fire diamond) | 1-2-0-健康 |
| Flash point | 102.4 °C |
| Lethal dose or concentration | LD50 oral rat 1310 mg/kg |
| LD50 (median dose) | LD50 (median dose): Mouse oral LD50 > 681 mg/kg |
| NIOSH | AW7525000 |
| PEL (Permissible) | Not Established |
| REL (Recommended) | 0.1 mg/m3 |
| Related compounds | |
| Related compounds |
L-2-Aminobutyric acid L-Homoserine L-Norvaline L-Norleucine |

