Disodium Hydrogen Arsenate: A Commentary on Its Place in Science and Industry
Historical Development
Early chemists poked and prodded with dangerous substances, and Disodium Hydrogen Arsenate found its way onto laboratory benches through sheer curiosity about the elements. The groundwork for arsenic chemistry stretches back centuries, but systematic production picked up steam in the 1800s as metallurgists refined methods for separating and processing ores. As glassmaking and electronics developed, the compound’s unique traits earned it a spot in manufacturing manuals. Over time, regulations toughened, and handling protocols took shape. Looking back, society’s relationship with arsenic shifted from open handling in fields and factories, to carefully controlled applications shaped by lessons from accidental poisonings and mounting environmental concerns.
Product Overview
Disodium hydrogen arsenate crops up in discussions about water treatment, laboratory reagents, and chemical synthesis. It carries the formula Na2HAsO4 and appears in white crystalline form. Producers rely on its ability to act as a source of arsenate ions for a variety of reactions. Whether in powdered or granular form, this compound feeds supply chains for both scientific investigation and select manufacturing steps, bridging the gap between academic study and practical use.
Physical & Chemical Properties
Disodium hydrogen arsenate forms transparent, odorless crystals, dissolving easily in water and giving a clear, colorless solution. It melts at relatively low temperatures, and its solubility makes it suitable where even dispersal matters. Chemically, the compound consists of sodium, hydrogen, arsenic, and oxygen. The substance reacts as both a weak acid and a weak base, and it stands up to modest heat without breaking down quickly. Moisture doesn’t phase it much, though it needs airtight storage to avoid clumping or caking. These basic features suit it to roles five minutes into a lab experiment or weeks into industrial processing, provided care gets taken at every touchpoint.
Technical Specifications & Labeling
Manufacturers stamp packages with tight content ranges: sodium, arsenic, and water at traceable ratios, reflecting the demands of research, technical, and commercial buyers. Impurities get specified down to fractions of a percent. Labeling never spares the hazard warnings—large, clear, and unmistakable, anchored by international standards (such as the GHS). Arsenic content, solubility, pH range, and melting point all merit attention on technical data sheets. Lot numbers and expiration monitoring matter, especially when storing bulk reserves near sensitive production lines. In practice, experienced lab staff approach containers with thick gloves, goggles, and a mental checklist, confirming authenticity before anything gets weighed out.
Preparation Method
Trusted methods for preparing Disodium hydrogen arsenate typically build from controlled reactions between arsenic acid and sodium carbonate or sodium hydroxide, under carefully managed temperature and humidity conditions. The process involves gradual mixing, monitored heating, and scrupulous pH testing to coax sodium and hydrogen ions into balanced union with arsenate. After reaction, controlled cooling brings out the crystals, which can then be filtered, dried, and milled. Waste products require swift neutralization and separate containment, reflecting the compound’s unpleasant toxicity. Facility engineers plan air handling and recycling systems, not just for their own safety but because regulations demand airtight compliance at every stage.
Chemical Reactions & Modifications
Chemists use Disodium hydrogen arsenate as both a starting point and an intermediate. By reacting with acids, bases, or metals, it sheds or picks up ions, making it a stepping stone to materials from pigments to insecticides. Under controlled conditions, heating cycles drive dehydration to produce related arsenate compounds. Thanks to its ability to shuffle between hydrogen arsenate and meta-arsenate states, processes can fine-tune end products with reasonable predictability. With oxidizing agents, its arsenic content facilitates synthesis of organic arsenic compounds—tools for specialized research or, in earlier times, for semiconductors. The long list of modifications proves why seasoned chemists value precision in everything from weighing out stock solutions to adjusting reaction times.
Synonyms & Product Names
Industry jargon and catalogs list Disodium hydrogen arsenate under a handful of other names, including sodium arsenate dibasic, disodium acid arsenate, and sodium hydrogen arsenate. Product codes and abbreviations change by supplier, with CAS registry numbers tracking back to the original chemical definition. European and American chemical distributors tag this compound with hazard pictograms and multiple language translations, giving safety teams a fighting chance at catching mistakes before a scoop hits the mixing bowl. Researchers, meanwhile, stick to sodium arsenate in lab records, trusting that spelling out the CAS number avoids confusion when inventory crosses borders.
Safety & Operational Standards
Handling this compound puts safety at the forefront. Regulatory watchdogs—OSHA, REACH, GHS, and local agencies—dictate airtight ventilation, mandatory PPE, and strict training. Gloves, aprons, and splash goggles make up the lowest rung of protective equipment. Facilities commit to spill kits, emergency wash stations, and clear signage in multiple languages. Waste must travel through properly labeled containers, and transport falls under dangerous goods rules. Storage protocols favor low humidity and separate zones for arsenic compounds. Training drills reinforce what can go wrong, and regular audits keep lazy habits from creeping in. Working with arsenic-based materials remains a privilege earned by teams devoted to detail and mutual protection.
Application Area
Disodium hydrogen arsenate plays its role most often in laboratory settings, where reliable arsenate sources shape everything from agricultural tests to electronics research. Historians record use in early herbicides and wood preservatives—a legacy that lingers as older infrastructure gets replaced or demolished. Modern regulatory frameworks largely push it toward research, water treatment pilot studies, and tightly controlled industrial chemistry. Arsenate ions help establish standards for detection of heavy metals in soils and waters, where researchers need benchmarks to calibrate delicate instruments. While many former applications faded as health and environmental burdens became clear, its foundational chemical properties keep it on supply lists in pharmaceutical synthesis, rare organic reactions, and the teaching of advanced inorganic chemistry.
Research & Development
Ongoing research keeps Disodium hydrogen arsenate in the conversation around water purification and arsenic mitigation. Scientists investigate how its solubility, reactivity, and detectability can inform safer methods to trap or neutralize arsenic contamination in groundwater. Teams also explore new uses in analytical chemistry: the compound’s stable arsenate group provides a handy calibration point when comparing the effectiveness of new filtration membranes, sorbents, or bioremediation strains. Sophisticated labs experiment with encapsulating its arsenate ions, aiming to reduce mobility and toxicity for safe storage. These efforts reflect a broader push—to clean up environmental legacies and build technologies robust enough to stay ahead of shifting regulations.
Toxicity Research
Disodium hydrogen arsenate puts real pressure on toxicologists. Studies show arsenic compounds disrupt enzyme function and damage DNA over time, with links to cancer, organ damage, and developmental problems. Cell cultures, animal studies, and long-term epidemiological tracking all agree: repeated exposure can’t be brushed aside. Data collection tracks residues in workplace air, drinking water, and trace environmental samples to build a complete picture of risk. Regulatory limits for exposure run down to parts per billion, and researchers deploy atomic absorption spectroscopy and mass spectrometry to sniff out even the smallest amounts. Safety sections on technical sheets pull no punches, laying out step-by-step actions for exposure, ingestion, or accidental spills. Understanding the full weight of its potential harm shapes everything from hiring practices to waste disposal protocols.
Future Prospects
Today’s landscape sees Disodium hydrogen arsenate balanced on a knife edge between laboratory necessity and environmental burden. Demand for arsenic-free technology holds sway in electronics, agriculture, and construction, but research labs hold onto it because certain reactions just cannot proceed otherwise. Green chemistry initiatives target safer substitutes and tighter regulations aim to minimize community and ecosystem exposure. Yet, as new methods emerge for arsenic detection, recovery, and safe storage, some see possibilities for repurposing existing stocks and knowledge for remediation, testing, and teaching. The story of this compound reflects the thin line chemistry walks between risk and discovery, and its future rests on the ability of science to innovate responsibly without forgetting hard-earned lessons of the past.
What is Disodium Hydrogen Arsenate used for?
What Is Disodium Hydrogen Arsenate?
Disodium hydrogen arsenate belongs to a group of arsenic compounds used mainly in industrial and laboratory applications. Its chemical structure features arsenic bonded with oxygen, sodium, and hydrogen atoms. People don’t usually bump into it in everyday products. Instead, you’ll find it in specialized environments that deal with arsenic chemistry.
Why Would Anyone Use It?
This compound shows up most often in labs interested in chemical analysis or producing other arsenic-based materials. Scientists use it to prepare certain reagents and solutions needed for experiments, especially where understanding arsenic’s effects or tracking arsenate levels matters.
For many years, compounds like disodium hydrogen arsenate played a role in agriculture. Insecticides and herbicides contained arsenate salts because they could kill pests to protect crops. Over time, health and environmental risks pushed people away from this route. Research linked arsenic exposure to serious health problems, even cancers. Governments set tight limits on arsenic in pesticides and many countries banned their use in farming.
The chemical still serves a purpose where researchers need a reliable source of arsenate ions. In water treatment studies and materials science experiments, knowing how arsenates react and behave helps scientists track arsenic contamination or invent safer ways to remove this toxic element from soil and drinking water. Testing equipment and protocols call for certain reference materials, and disodium hydrogen arsenate gives consistent results.
Risks That Come With Handling Arsenate
No one treats this compound lightly. Breathing or eating even small amounts can lead to poisoning. People handling it wear protective gear, follow strict protocols, and limit access to only essential staff. Most workplaces using arsenic compounds have special ventilation, emergency showers, and spill kits ready to handle accidents.
Old uses—particularly in agriculture—left a legacy of contaminated soil. Communities still deal with these risks today. Long-term exposure from polluted water or crops will damage health over time. The Environmental Protection Agency places arsenic among its top priorities for cleanup and monitoring.
Challenges in Reducing Risks
Completely removing arsenic from the lab and industry is tough because some processes depend on its unique properties. Replacing it means finding substances that match its behavior, which isn’t always possible or practical. Safer protocols, locked storage, tighter regulation, and ongoing worker education all help cut down misuse or accidental exposure.
One approach involves developing better detection tools. Quicker, more accurate tests for arsenate contamination let people react before the risk gets out of control. Reducing the need for new disodium hydrogen arsenate in industry also means reclaiming and recycling arsenic from old stocks or waste, lowering demand for fresh production.
What People Can Do Next
Communities living near old industrial or agricultural sites benefit from regular soil and water checks. Where contamination shows, cleanup teams dig out polluted earth, treat water, and teach people how to avoid risk. Ensuring the right disposal channels for all chemical waste—especially arsenic compounds—protects the wider environment.
Educational outreach matters, too. Being open about the risks, current rules, and safe handling tips helps everyone involved, from workers to neighbors. Proper transparency supports trust and quick action if something goes wrong.
Final Thoughts
Disodium hydrogen arsenate reflects a bigger story about industrial chemistry, public health, and environmental responsibility. Keeping its use safe takes science, strict rules, and a clear understanding of lessons from the past.
Is Disodium Hydrogen Arsenate toxic or hazardous?
Recognizing Toxicity in Everyday Chemicals
Most people never hear about disodium hydrogen arsenate unless they're working in a lab or digging through old textbooks. This chemical, which includes the notorious element arsenic, deserves attention. Why? The simple answer is toxicity. Arsenic outranks many other elements for harm to living things, and its compounds—disodium hydrogen arsenate included—carry real risks.
What Makes Disodium Hydrogen Arsenate Risky?
Disodium hydrogen arsenate contains arsenic in a form that the body can absorb. Arsenic exposure, even at fairly low levels, links to problems like skin lesions, digestive issues, cancer, and problems with nerves. The World Health Organization lists arsenic among the top ten chemicals that threaten public health. Anyone handling arsenic compounds in agriculture, mining, or scientific research faces strict rules to keep exposure low. For instance, the U.S. Environmental Protection Agency enforces tough limits on arsenic in drinking water and in soil used for growing food.
The dangers of disodium hydrogen arsenate go deeper than its chemical makeup. The substance dissolves in water, making it easier for it to spread through the environment. Spills or improper storage can send arsenic compounds into rivers and groundwater. People who touch contaminated water or breathe in dust risk arsenic poisoning. Chronic exposure can lead to numbness, skin changes, and a much higher chance of cancer.
Personal Safety and Community Health
Working with hazardous chemicals has shaped my own views on the topic. Protective gear, real-time air monitors, and strict handling procedures created a culture of safety in every lab I’ve been part of. The risks aren’t just theoretical. One spill or a careless moment in disposal can have long-term effects not only for workers but for their families and neighbors.
Arsenic poisoning events from industrial accidents and contaminated water in places like Bangladesh, West Bengal, and parts of the western United States show what’s at stake. Farmers who used arsenic-based pesticides saw contamination persist in soils for years, haunting future generations. Even small mistakes with storage or disposal of chemicals like disodium hydrogen arsenate cascade into bigger problems.
Why Awareness Matters
A lack of awareness and training remains a dangerous blind spot. In many labs and on job sites, new workers often hear more about how to fill out forms than how to handle real risks. A focus on hands-on demonstrations, using real-life stories, helps people understand that regulations exist for good reasons. Posting basic reminders in high-risk areas, and making it normal to speak up if something doesn’t seem right, stops accidents.
Where Do We Go From Here?
Tighter controls on how disodium hydrogen arsenate gets used and stored would go a long way toward easing risks. Updates in labeling, better worker education, and new technologies for real-time monitoring help catch problems early. Communities benefit when companies and regulators share information about hazardous substances. People who understand what chemicals they’re using, and why the rules matter, get the power to protect themselves.
Disodium hydrogen arsenate might not make headlines, yet ignoring its risks has serious consequences. With knowledge and respect for the hazards, accidents decline and health improves—one careful step at a time.
What are the storage requirements for Disodium Hydrogen Arsenate?
Understanding Disodium Hydrogen Arsenate
Disodium hydrogen arsenate stands out as a white, crystalline solid with toxic and potentially carcinogenic effects. Found on shelves in labs or tucked away in chemical supply rooms, its risks reach beyond the label. This compound gets used in some research, industrial, and agricultural settings. Since arsenic compounds are not forgiving—one mistake can cause serious health problems or environmental contamination.
Personal Experience Shapes Perspective
Anyone who has walked through a lab, seeing containers with poison signs, knows the uneasy feeling that comes with handling dangerous chemicals. Years ago, working with legacy reagents, I saw firsthand what neglect does. Spilled white powders, faded hazard stickers, bins stacked with cracked lids—it only takes one poorly stored bottle to turn a workplace into an emergency site. Disodium hydrogen arsenate deserves respect.
Safe Storage Conditions Are Not Optional
A room with good ventilation always gives peace of mind. This chemical should sit only in areas with controlled temperatures, away from sunlight and heat sources. Dry conditions help, since moisture can make it clump and raise the risk of accidental release. Labs that follow these practices see fewer accidents and less loss, and staff avoid unnecessary risks to their lungs and skin.
Glass, plastic, or polyethylene containers with tight seals work best. I always check chemical inventory stored in original packaging or sturdy chemical-resistant bottles. No dents. No corrosion. Clear and durable labeling protects everyone, even visitors who don’t work with these toxins every day. Every year, I run into horror stories where staff, thinking they grabbed table salt or another non-toxic powder, spilled toxic compounds. Old, fading labels caused it more than once.
Why Segregation from Other Chemicals Matters
Disodium hydrogen arsenate never goes near acids, food, or organic material. Cross-contamination opens doors to dangerous reactions—arsine gas, for example, forms with acids, threatening anyone nearby. Segregating arsenic compounds reduces the chance of these accidents. Separate shelves in locked chemical storage cabinets or designated poison rooms help, not just in big companies but in small research outfits as well.
PPE and Handling Procedures Back Up Safe Storage
Even the most organized storage setup needs solid habits. Proper gloves, lab coats, eye protection—these cut risk when handling the compound. Any spills or dust demand an immediate response. Knowledge carries weight in these environments: the best teams practice drills and refresh training. Waste management does not get overlooked; this chemical never goes into regular trash or down the drain. Labs coordinate with licensed hazardous waste handlers. The paper trail, the logs, the signatures: they seem tedious, but they mean fewer headaches down the road.
Building a Culture of Safety
Every time a lab takes an audit seriously, reviews its emergency procedures, and updates chemical inventory lists, the risk drops. Keeping disodium hydrogen arsenate secure means safer workers, cleaner spaces, and less chance for legal or health troubles. Storage rules make sense not because of bureaucracy, but because mistakes with arsenic compounds last a long time. Sharing these lessons with newcomers and holding each other accountable keeps everyone safer day after day.
How should Disodium Hydrogen Arsenate be handled safely?
Understanding Where Hazards Come From
Disodium hydrogen arsenate finds its way into various laboratories and some industrial settings. The big deal here isn’t just about having it on the shelf—it’s about how toxic arsenic compounds affect people and the planet. Toxicology reports and decades of chemical accident data point to its risk of long-term health problems, including cancer, if the compound gets inhaled, swallowed, or even absorbed through skin contact.
Why Protective Gear Is No Luxury
Direct experience in chemical labs shows that gloves, goggles, and lab coats make a mammoth difference. I still remember my first faculty mentor saying, “If you wouldn’t pour it in your morning coffee, don’t let it touch your skin.” Vinyl or nitrile gloves, wraparound eye protection, and closed-toe shoes cut down dangerous exposure. A fitted respirator matters in situations where dust could go airborne. Not every lab’s air quality hits the same mark. Dry powders, no matter how safely stored, end up in the air if mishandled—even with what someone might think is a gentle pour.
Smart Storage Isn’t Optional
Chemical storage mishaps have real costs, and not just for the person making the mistake. A spill can affect entire buildings or water systems. Keeping disodium hydrogen arsenate inside a labeled, tightly-sealed container in a well-ventilated, locked chemical cabinet keeps hands, lungs, and the environment safer. I’ve seen emergency cleanups where a wrongly stored container leaked into a classroom. The aftermath meant whole teams spent hours in hazmat suits—and that’s with the alarms working and training in place.
Dealing With Accidents and Spills
Anybody who’s been in a lab more than a few months knows mistakes find a way to happen. Quick access to a spill kit with absorbents, heavy gloves, and a dust mask matters. In my experience, moving slowly and not overreacting keeps the dust down while collecting the material. Double-bagging contaminated waste and labeling it as hazardous lowers the odds of someone untrained getting exposed. The local hazardous waste disposal office should always get a call. Pouring anything with arsenates down the drain has made its way into far too many “Don’t Ever Do This” safety slides, all because rivers and municipal water have suffered the consequences.
Training and Personal Accountability
Reading safety data sheets isn’t just bureaucratic hoop-jumping. These documents spell out what science and the law already know about arsenic toxicity. Regular training makes more of a difference than new equipment. I once watched a well-funded group hire new people without proper orientation, and it ended with preventable skin rashes. The most careful teams talk through their workflow before opening any bottle involving substances with an arsenic warning.
Improving Safety Culture
Promoting a culture where folks feel comfortable stopping work to ask about safety does more for real-world health outcomes than posters on the wall. Even small labs with limited budgets can take safer steps by reducing how much disodium hydrogen arsenate gets kept on hand and by swapping out for less hazardous materials when the science allows. Transparency about errors and learning from close calls builds a safer workplace—no matter how many years of experience someone racks up.
What is the chemical formula and molecular weight of Disodium Hydrogen Arsenate?
Getting Clear: The Formula
Disodium hydrogen arsenate sounds like something only a chemist finds interesting, but understanding simple chemical information can offer fresh insight, even outside the science classroom. This compound has the chemical formula Na2HAsO4. The way the formula comes together tells us a lot about the elements inside. We see two sodium atoms, a single hydrogen, arsenic anchoring the group, and four oxygen atoms. Sodium brings its reactivity, arsenic catches attention for its toxicity, and the oxygen and hydrogen turn it into a salt.
Molecular Weight: Not Just a Number
Molecular weight plays a role in everything from transportation to disposal. For Disodium hydrogen arsenate, the molecular weight comes out to roughly 185.91 g/mol. Here’s how it adds up: sodium (Na) brings 22.99 g/mol each, hydrogen offers 1.01 g/mol, arsenic weighs in at 74.92 g/mol, and oxygen counts for 16.00 g/mol times four. Adding these gives us the total: 2 x 22.99 + 1.01 + 74.92 + (4 x 16.00) = 185.91 g/mol.
Back in college labs, every student watched scales carefully, not only for correct measurements but also because proper weight matters for safety. If you weigh out the wrong amount, you risk exposure that healthy bodies can’t handle. With a compound carrying arsenic, mistakes quickly become health hazards. The weight isn’t abstract: it shapes how labs or industries package, store, ship, or neutralize the chemical.
Why People Care About This Compound
Arsenic doesn’t have the best public reputation. Think groundwater warnings, historic poisons, and agricultural debates. Disodium hydrogen arsenate gets used as a reagent in the lab, but its presence in any workplace means tight protocols and clear rules. It highlights the need for regulatory bodies like OSHA to keep workplaces safe, and demands discipline from practitioners.
Recent headlines linked to arsenic contamination in rice or baby food have sparked community frustration. Disodium hydrogen arsenate, while not exactly the form found in grains, shares the arsenic root. Consumer protection groups rely on fundamentals, like knowing the substance’s molecular weight, to draft laws and guide enforcement. Nobody wants to see health crises come from overlooked details.
Solutions and Smarter Choices
One smart step: training people who handle chemicals to think like chemists, whether they went to grad school or not. Reading a label like “Na2HAsO4” shouldn’t be mysterious. Clear communication, color-coded storage, and ready access to safety data sheets help everyone. Stronger policies in industrial sites, and community investment in water testing, lead to fewer accidents or long-term health impacts from arsenic compounds.
On the industry side, staying current means tracking chemical shipments, regular environmental monitoring, and supporting research that moves away from hazardous substances. Plenty of technology companies work on better ways to neutralize arsenic waste, or on greener alternatives. Regulatory frameworks rooted in deep, straightforward chemical knowledge protect more than just the people in the lab. They also shield surrounding neighborhoods from slow, unnoticed exposure.
Why Details Matter
Simple facts like a compound’s formula and weight aren’t small talk—they shape how we approach problems in public health, workplace safety, and science. If people know what’s inside everyday chemicals, they take smarter steps, ask better questions, and trust systems that keep their water, food, and air safer. Disodium hydrogen arsenate reminds us that attention to detail saves lives, not just test tubes.
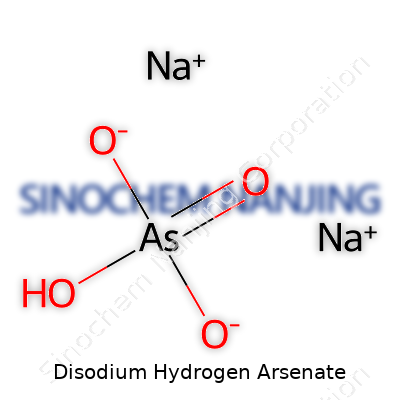
| Names | |
| Preferred IUPAC name | disodium hydrogenarsenate |
| Other names |
Disodium arsenate Sodium hydrogen arsenate Disodium acid arsenate |
| Pronunciation | /daɪˌsoʊdiəm ˌhaɪdrəˈdʒɛn ɑrˈsiːneɪt/ |
| Identifiers | |
| CAS Number | ''10048-95-0'' |
| Beilstein Reference | '385296' |
| ChEBI | CHEBI:48601 |
| ChEMBL | CHEMBL1201647 |
| ChemSpider | 16219150 |
| DrugBank | DB13704 |
| ECHA InfoCard | ECHA InfoCard: 03-211-9692262-37-0000 |
| EC Number | 231-993-9 |
| Gmelin Reference | 110144 |
| KEGG | C02344 |
| MeSH | D000077197 |
| PubChem CID | 25136 |
| RTECS number | CG3325000 |
| UNII | DTA7B4196M |
| UN number | UN1605 |
| Properties | |
| Chemical formula | Na2HAsO4 |
| Molar mass | 222.00 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.87 g/cm³ |
| Solubility in water | Highly soluble |
| log P | -2.05 |
| Vapor pressure | Negligible |
| Acidity (pKa) | pKa ≈ 2.29 |
| Basicity (pKb) | 11.5 |
| Magnetic susceptibility (χ) | -55.0e-6 cm³/mol |
| Refractive index (nD) | 1.341 |
| Dipole moment | 2.54 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 184.6 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1567.2 kJ/mol |
| Pharmacology | |
| ATC code | A01AB10 |
| Hazards | |
| Main hazards | Toxic if swallowed. May cause cancer. Causes damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS02,GHS06,GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P260, P262, P264, P270, P271, P273, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P314, P320, P330, P391, P501 |
| NFPA 704 (fire diamond) | 3-0-2-ox |
| Lethal dose or concentration | LD₅₀ Oral - Rat - 40 mg/kg |
| LD50 (median dose) | LD50 (median dose): 6 mg/kg (oral, rat) |
| NIOSH | CG2450000 |
| PEL (Permissible) | PEL: TWA 0.01 mg(As)/m³ |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | 5 mg As/m3 |
| Related compounds | |
| Related compounds |
Sodium dihydrogen arsenate Arsenic acid Sodium arsenite |

