Disodium 2-Oxopentanedioate: Unpacking the History, Science, and Future Potential
Historical Development
Disodium 2-oxopentanedioate, long a staple in laboratory and industrial chemistry, owes its relevance to both academic curiosity and real-world necessity. Chemists in the early 20th century first isolated this compound while mapping metabolic intermediates in the citric acid cycle. Back in those days, any compound showing potential as a chelating agent or metabolic regulator got immediate attention. Researchers tested it, tried a variety of reactions, and soon cataloged it as a reliable standard for work in organic synthesis and biochemistry. As culture shifted and technology stepped up, factories began to produce the compound in bulk, not only for research but for industrial processing, preservation, and analytical work. Reliability and predictability quickly pushed it past the bench into the mainstream.
Product Overview
Today’s market lists disodium 2-oxopentanedioate in high-purity crystalline form, sometimes as a buffered solution. Manufacturers stress stability and shelf life, often sealing product in air-tight, opaque packaging to stop moisture and light from triggering unwanted breakdown. Price and accessibility now mirror mature supply lines and heavy demand, especially from chemical manufacturers, labs, and pharmaceutical developers. My own time in the lab brought me face to face with this compound more often than I could count—sometimes as a reactant, other times as a stability agent—never once feeling out of place in research kits or standardized test sets.
Physical & Chemical Properties
Most lots of disodium 2-oxopentanedioate appear as white granules or crystalline powder with a faintly tart taste. The compound dissolves clear in water, displaying good solubility—kids in science fairs can vouch for its use in simple solubility demos. Melting point readings usually fall around 270°C (decomposition). Chemically, this salt registers a neutral to slightly basic pH in solution, rarely giving off gas or reacting violently outside extreme conditions. Chemists count on the presence of two sodium cations for interactions with other metal ions and organic species, an asset for any researcher working at the interface of organic and inorganic processes.
Technical Specifications & Labeling
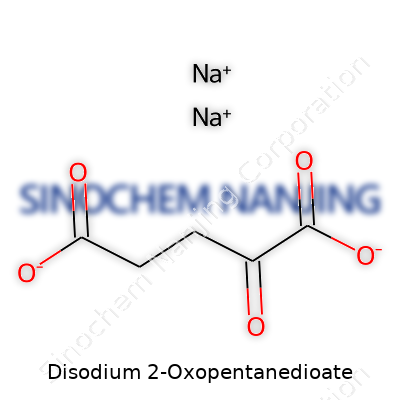
Bottles and drums of disodium 2-oxopentanedioate arrive with lot numbers and certificates of analysis, detailing purity (often greater than 99%), loss on drying, water content, and microbial levels. Safety data sheets spotlight the handling precautions, which echo most other sodium salts: avoid inhalation, wear gloves, and keep dry. Reliable vendors label packages with chemical formula (C5H4Na2O5), structural diagrams, and proper hazard symbols. These practices help buyers trace every product batch back to the source, building confidence in research, production, and regulatory compliance.
Preparation Method
Production frequently starts with glutaric acid, treated with sodium carbonate or sodium hydroxide under controlled acidity and temperature to create the disodium salt. The process leans on water as solvent, and agitation helps speed things along. After filtering out impurities, producers evaporate the water, then recrystallize from alcohol or another low-boiling solvent to tighten up purity. Plants running at scale can churn out hundreds of kilos per week, largely by optimizing filtration systems and recycling unused sodium reagents. I remember hearing seasoned plant operators say, “Keep it simple, keep it clean,” and their process has not changed much in decades.
Chemical Reactions & Modifications
Put disodium 2-oxopentanedioate in the middle of a synthetic pathway and it acts with modesty, rarely dominating a reaction scheme unless paired with an active ester or acid anhydride. Common reactions include reduction to related hydroxy acids, or oxidation with strong agents to generate more reactive intermediates. It plays well in buffer systems, and you can substitute both sodium ions with potassium or lithium if your process demands it. People blend it with amino acid derivatives during peptide synthesis because the carbonyl function integrates smoothly under mild or basic conditions. Years ago, colleagues tested alternative counterions, only to circle back to sodium for its balance of reactivity and price.
Synonyms & Product Names
On a packaging label or certificate of analysis, don’t be surprised to spot alternative handles such as disodium glutarate, pentanedioic acid disodium salt, or glutaric acid, disodium salt. International chemical catalogs list CAS number 2836-32-0 in parallel, but the real-world buyer knows to crosscheck supplier brochures for all known synonyms. If you’ve worked in regulatory affairs, as I have, you learn to scan not just for one name but a string of synonyms that reflect local preferences and translation quirks.
Safety & Operational Standards
Typical safety training covers exposure through skin or inhalation. Workers insist on gloves, lab coats, and splash-proof goggles. The substance doesn’t pose the same high risk as strong acids or alkalis, but dust clouds can trigger respiratory irritation. Storage calls for cool, dry, and well-ventilated shelves, away from acids or strong oxidizers. Disposal partners with local regulations—neutralize any spills with water and sweep up dry powder carefully. My own training once drilled home the importance of clear spill protocols, and every well-tuned lab adopts checklists for peace of mind.
Application Area
The range of uses for disodium 2-oxopentanedioate grew by leaps and bounds over time. Specialty chemical processing uses the compound as a buffer and intermediate for higher-value materials. Food scientists experiment with it as a sequestrant for minerals. Cosmetics manufacturers test it for pigment stabilization, relying on its mild compatibility with organic solvents and skin-safe pH. Pharmaceutical research taps into it for controlled-release formulations and as a chelating agent for precise metal regulation during drug synthesis. Even wastewater treatment plants take note—engineers lean on its reliability, using small doses to bind up metals in effluent streams, a trick I picked up from a veteran environmental chemist years ago.
Research & Development
Ongoing research keeps uncovering nuances in this compound’s reactivity and stability. Teams working on drug delivery hunt for better excipients and matrix agents, and so they push chemical modifications on the basic structure. Analytical chemists look for tighter detection limits and faster assays by using the compound as a model analyte. University groups in Europe and North America test substitution reactions and new salt forms to meet niche applications. Many projects quietly target medical or environmental health, hoping to find a link between trace exposure, metabolic compatibility, and long-term impacts—driven by curiosity, but also regulatory tightening.
Toxicity Research
Low acute toxicity helps explain why this compound earns broad acceptance. Animal studies typically show mild effects at high doses, mostly limited to gastrointestinal irritation. Genotoxicity screens remain negative at reasonable exposures, though nobody discounts the need for wider surveillance as long-term and chronic exposure data gather. Regulatory agencies in the US and EU hold back classification, preferring ongoing monitoring over outright bans. Food and beverage applications undergo extra scrutiny, especially as analytical methods become more sensitive. Every seasoned analyst keeps an eye on cumulative exposure, especially in closed, high-use environments.
Future Prospects
Looking ahead, there’s every sign the role of disodium 2-oxopentanedioate will grow along with material science and pharmaceutical innovation. New salt forms, smarter packaging, and custom functionalizations wait on the horizon. As digital process control rolls out in industry, tighter control over reagent quality promises even more reliable results for downstream users. Efforts in green chemistry target minimal waste, greener reagents, and closed-loop production cycles. Meanwhile, regulatory landscapes tighten, making product provenance and analytical traceability more crucial than ever. From my own work in pharmaceutical development and regulatory affairs, tighter integration between supplier, manufacturer, and lab stands out as the best path forward for quality and safety.
What is Disodium 2-Oxopentanedioate used for?
Understanding Its Role in Food
Walk down any grocery aisle. Flip over a package of instant soup or a bag of chips. Chances are, you’ll spot tongue-twisting names in the ingredients, one of those being disodium 2-oxopentanedioate. Food companies look for ingredients that enhance taste and help products keep a consistent texture. This chemical, sometimes called disodium alpha-ketoglutarate, helps with that. It isn’t there to deliver nutrition, but it supports flavor and product stability, two things shoppers notice even if they don’t talk about them.
Many processed foods aim for a consistent taste year-round—even when main ingredients change between batches. Disodium 2-oxopentanedioate steps up as a flavor enhancer and stabilizer in packaged foods, keeping the finished product tasting familiar every time you open the box. I used to work in a deli, and watching foods come in from various suppliers, I noticed small brands struggled to keep broths or sauces tasting the same. Large food makers could deliver that uniform taste consistently, often thanks to these specialized additives.
Applications Beyond the Kitchen
This compound finds a home in dietary supplements. It relates to metabolism and cellular energy, linking to the Krebs cycle, which every high-school biology student remembers as a key process for energy production. By supporting amino acid synthesis in the body, disodium 2-oxopentanedioate appears in some supplements aimed at athletes or people recovering from surgery or illness. Some research from nutrition journals suggests it might help support muscle maintenance and recovery, especially in the elderly.
In hospitals, medical foods often reach for compounds like this to meet the nutritional challenges of certain patients. Individuals with kidney or liver concerns sometimes rely on specialized powders and drinks containing this ingredient to supplement their intake, especially when everyday eating isn’t possible. Registered dietitians know these ingredients can make a difference during recovery or for people with chronic conditions who struggle to keep weight on.
Is It Safe to Consume?
Whenever an unfamiliar chemical pops up in food, people want to know—is it safe? Food safety agencies, including the FDA and EFSA, give guidance here. Disodium 2-oxopentanedioate has a track record in food and supplements without major red flags emerging from studies to date. Some people feel wary of additives, and I share that caution. A smart shopper reads labels, sticks with whole foods when possible, and keeps an honest eye on ingredient lists—but shouldn’t panic just from spotting a long name.
In my own household, we favor scratch cooking, but busy days call for shortcuts sometimes. I recognize that regulated additives have backed safety reviews behind them. Still, moderation stays key—processed foods help in a pinch but shouldn’t crowd out fruits, vegetables, beans, and other basics.
Finding Better Balance in Modern Food
The challenge of balancing convenience and nutrition stays front-and-center in most homes. Ingredients like disodium 2-oxopentanedioate land in packaged goods for a reason: without them, products spoil faster or lose their appeal. Food makers could experiment more with natural alternatives, such as yeast extracts or plant-based flavor boosters, though these come with tradeoffs around shelf life and cost.
I’ve found that the most practical solutions come from a place of balance. Anyone concerned about unfamiliar additives can cook more often from scratch, rely on fresh produce, and buy fewer ultra-processed snacks. Food will always include some chemistry, but being informed and choosing purposefully gives us the most control over what feeds our families.
Is Disodium 2-Oxopentanedioate safe for consumption?
What is Disodium 2-Oxopentanedioate?
This rather long name refers to a food additive more commonly called disodium alpha-ketoglutarate. You’ll spot it hiding in ingredient lists on processed foods, supplements, and sometimes even in sports nutrition products. It’s easy for the average shopper to overlook an unremarkable chemical name, but scrutiny is growing as more people want cleaner food and fewer artificial extras in their diets.
Food Additive Scrutiny
People want to trust what they eat, and suspicion often grows around synthetic-sounding substances. I remember standing in my local store grocery aisle, peering at snack labels with a skeptical eye, and finding myself stuck on unfamiliar words. If it’s in the food, it’s fair to ask if it’s safe.
What Do Scientists Say?
Research matters. Disodium 2-oxopentanedioate isn’t just made up in a lab without oversight. It’s related to alpha-ketoglutaric acid, a compound that plays a role in the body’s metabolism. Food scientists and toxicologists look for problems, running studies on rats or cell cultures long before an ingredient winds up in your crackers or capsules.
European Food Safety Authority (EFSA) and the U.S. Food and Drug Administration have not flagged disodium 2-oxopentanedioate as a hazard for consumption in regulated amounts. Published toxicological reviews, including those in journals like Food and Chemical Toxicology, haven’t pointed toward glaring red flags under current usage. There’s also no record of serious allergic reactions or widespread toxicity cases in the public database.
Why People Still Worry
Fears aren’t always rational, but they’re worth addressing. Many consumers draw a sharp line between natural and artificial, worrying that long-term effects aren’t fully explored. Historical cases, like the change in public attitude toward aspartame and artificial colors, bring out caution. Even though regulatory authorities set limits after rigorous review, skepticism lingers, especially because new data can change guidance down the line.
Personal experience reminds me — people don’t just read studies, they watch what happens after eating certain foods. Reports of upset stomach or headaches often circulate anecdotally online, yet these have never been directly traced to disodium 2-oxopentanedioate in controlled studies. Still, some prefer to stay clear just to avoid any perceived risk.
Supporting Claims With Facts
Trust builds when science and transparency walk hand in hand. Studies published over the last two decades have examined breakdown products of disodium 2-oxopentanedioate; these prove to be regular components of the body’s energy pathways. In therapeutic areas, its close cousin alpha-ketoglutarate has been tested as a supplement for kidney health and sport performance, with no consistent evidence of harm at recommended doses.
Finding a Balance
The safest approach seems to involve moderation and a diverse diet. Heavy reliance on processed foods, regardless of one additive, carries bigger health risks than exposure to any single synthetic compound. Industry watchdogs, scientists, and public health officials must keep updating the public, making it easy for consumers to track any updates about safety.
For now, sticking with regulatory guidelines and focusing on whole foods will keep risks low. Food labels shine a light on what’s inside, and checking those lists is something every consumer deserves to do confidently.
What is the chemical formula of Disodium 2-Oxopentanedioate?
The Simple Formula with Complex Uses
Disodium 2-oxopentanedioate goes by a straightforward formula: C5H4Na2O5. The name gives a hint about its structure—two sodium ions paired with the 2-oxopentanedioate ion, which is better known to chemists as the salt form of alpha-ketoglutaric acid. It’s easy to gloss over chemical formulas and miss their impact, but this compound finds its way into more places than most people expect.
Understanding the Structure
Alpha-ketoglutaric acid, the backbone behind this sodium salt, plays a vital role inside the human body. Disodium 2-oxopentanedioate’s formula (C5H4Na2O5) shows just how sodium replaces protons that would normally attach to the acid’s carboxyl groups. This swap makes the compound far more stable, especially when solutions require less acidity. Industries benefit too, especially when purity matters and pH must be carefully controlled.
Importance to Metabolism and Science
Through school and work, I learned that alpha-ketoglutarate isn’t just any molecule—it’s a key player in the citric acid cycle, also called the Krebs cycle. Every cell with a mitochondrion uses this chemistry to turn food into usable energy. Sports supplements sometimes include alpha-ketoglutarate salts for this reason. That’s where the disodium variant steps in, because the body can handle sodium much more easily than pure, acidic alpha-ketoglutaric acid.
Laboratories rely on disodium 2-oxopentanedioate for buffer solutions that need a predictable pH range. The sodium ions prevent rapid shifts, keeping everything steady for accurate experiments. I’ve mixed this very compound while running enzymatic assays, trusting that its consistent dissolution helps keep results trustworthy.
Safe Handling and Transparency
Chemical formulas like C5H4Na2O5 hold more than letters—they carry safety concerns, too. Every time I open a jar labeled with such a formula, I keep in mind the right precautions. Disodium 2-oxopentanedioate usually appears as a white powder with low toxicity compared to stronger acids or many organic solvents, but inhaling dust or getting it on bare skin is never a good idea. Reading the safety data sheets, wearing gloves, and using good ventilation cuts down on risks. Industries that use this chemical in food or supplements must follow strict regulations and provide transparent labeling so users understand what’s included in any product.
Looking Toward Practical Solutions
Unlabeled ingredients frustrate consumers with allergies or special dietary needs. When every ingredient follows scientific naming with clear chemical formulas, anyone can know exactly what lands on their plate or in their supplements. Making these details easily available, not just buried in technical sheets, builds trust. Providing education around common compounds like disodium 2-oxopentanedioate helps lift some of the confusion many face about what goes into scientific work, processed food, or nutrition products.
I’ve seen mistakes happen just from overlooked or misunderstood formulas. Keeping chemical information visible and easy to check stops small errors from turning into big issues, in the lab or out in the world. This simple sodium salt teaches that a little knowledge really can make a major difference.
Are there any known side effects of Disodium 2-Oxopentanedioate?
What Is Disodium 2-Oxopentanedioate?
Disodium 2-Oxopentanedioate, better known as disodium alpha-ketoglutarate, ranks among those food additives that seldom make headlines despite being found in nutrition supplements and sometimes in specialized hospital formulas. Most folks bump into it because it pops up on an ingredient list, and they wonder if their daily scoop of fortified powder might bring trouble.
What Science Says So Far
I’ve spent time digging into ingredient labels. School cafeteria food, late-night vitamins, and even fitness supplements have taught me to double check what’s inside. As a naturally occurring compound in the body, alpha-ketoglutarate plays a role in amino acid metabolism. The U.S. Food and Drug Administration gives disodium 2-oxopentanedioate the thumbs up as “generally recognized as safe” when it’s used the way it’s approved. So far, scientific studies haven’t turned up a pattern of serious side effects when it’s used at appropriate doses.
That said, years living with a sensitive stomach trained me to respect even so-called “safe” additives. Although overwhelming evidence points to disodium 2-oxopentanedioate as safe, a handful of reports mention mild stomach upset, loose stool, or bloating in some people taking high doses of alpha-ketoglutarate as a supplement. The chances of running into problems climb as soon as anyone exceeds the recommended amount. Anyone with kidney issues, or on a sodium-restricted diet, could need to be especially mindful—That extra dose of sodium adds up fast and could set off blood pressure swings.
Paying Attention to Labels and Reporting Issues
Reading every label matters more than most of us want to admit. Over the years, health habits come and go, but keeping an eye on what goes into your body never goes out of style. Transparency from supplement makers and food producers makes this easier. Clear labeling about all excipients and additives, including disodium 2-oxopentanedioate, lets people living with medical conditions—such as hypertension or chronic kidney disease—make safer decisions. Full disclosure builds trust, plain and simple.
People notice reactions that the scientists haven’t caught yet. That’s why reporting even minor symptoms—nausea, gut ache, swelling, weird rashes—not only helps your own doctor track down what’s going on, but it also adds to the global record. The National Institutes of Health, FDA’s MedWatch, and consumer safety networks all rely on ordinary people flagging their experiences with additives.
Moving Toward Fewer Surprises
Curiosity about what we eat, drink, and swallow helps prevent health surprises. Some additives drift through unnoticed, but the few studies that follow people using disodium 2-oxopentanedioate long-term still leave open questions. A call to the family doctor never hurts when new symptoms pop up after trying a novel supplement.
Balancing the straight facts—minimal side effect reports in healthy adults at reasonable doses—with the reality of individual differences underlines a timeless message: Health isn’t a guessing game. If manufacturers, regulators, and consumers all keep the conversation honest and evidence-driven, the chances of a negative surprise from something like disodium 2-oxopentanedioate start dropping. As research continues, we’ll see if the list of known effects stays short—but informed choices always outlast fads.
Where can I buy Disodium 2-Oxopentanedioate?
My first encounter with chemical names like Disodium 2-Oxopentanedioate ended in confusion. Speaking to researchers and lab techs, some call it disodium glutarate. This compound turns up mostly in specialized labs, not your average store shelf. If you’re hunting for it, there’s a good reason for some caution—regulations aren’t just paperwork. Trusted supply matters a lot, both for research quality and safety.
Finding a Trustworthy Seller
Plenty of online marketplaces let you browse chemicals, but not all sellers track how they source or store compounds. Easy shopping sometimes leads to unreliable or dangerous products, which isn’t a risk worth taking, especially with chemicals. For those who care about lab safety and experiment reliability, I’d focus on verified chemical suppliers. Strong vendors include Sigma-Aldrich, Thermo Fisher Scientific, and Alfa Aesar. They specialize in lab-grade materials and maintain strict quality checks.
Buying from these companies isn’t just about brand names. They offer Certificates of Analysis, give clear batch information, and explain purity levels. For many labs, these details spell the difference between clean data and hours of troubleshooting. You’ll almost always find the most recent safety information as well, which matters if compliance or liability comes up.
Regulations and Responsible Sourcing
Buying chemicals brings some paperwork. Professional chemical suppliers will ask for business details, lab affiliation, or proof you’re qualified to order. These steps can frustrate buyers, but they help keep dangerous materials out of the wrong hands and ensure safe shipping and handling. I’ve witnessed people hit walls where suppliers refuse to ship to home addresses or non-institutional buyers. If you’re affiliated with a university or licensed company, getting paperwork together ahead of time makes a difference.
Some who search for niche chemicals run into sites that skirt rules—think drop-shippers and shady middlemen. Plenty of people get burned by expired, impure, or entirely incorrect products. If the seller can’t provide lot numbers or earlier customer reviews, that’s a serious red flag. One of my old colleagues purchased a solvent from a barely-legible site and spent days cleaning up the mess. He later stuck only with vetted distributors.
Safe Handling and Storage
Chemical safety can’t be an afterthought. Disodium 2-Oxopentanedioate usually arrives in tightly-sealed bottles, often with warnings. Proper lab storage guidelines should be followed, including labeling and separation from incompatible reagents. Disposal adds another step—a quick call to a waste management company or university safety office usually helps line up safe, legal disposal options.
What If You’re Not a Researcher?
People sometimes look up Disodium 2-Oxopentanedioate for do-it-yourself chemistry or alternative uses. Without a lab setup and training, this is not safe. Many chemical suppliers refuse orders from private individuals for good reason. For anyone who only needs the chemical for theoretical knowledge or curiosity, public science libraries or reputable online sources offer plenty of information without any risks attached. Real-world experience shows that patience and solid sourcing beat shortcuts every time—especially when health and safety are on the line.
| Names | |
| Preferred IUPAC name | Disodium 3-oxopentanedioate |
| Other names |
Disodium alpha-ketoglutarate Disodium 2-oxoglutarate Disodium glutarate-2-one Disodium 2-oxopentanedioate Disodium 2-oxopentanedionate |
| Pronunciation | /daɪˈsoʊdiəm tuː ˌɒksoʊˌpɛntəˈdaɪəʊeɪt/ |
| Identifiers | |
| CAS Number | 20813-60-7 |
| 3D model (JSmol) | `9JY6GNH5JJJYBO-UHFFFAOYSA-L` |
| Beilstein Reference | 1740246 |
| ChEBI | CHEBI:132965 |
| ChEMBL | CHEMBL1201655 |
| ChemSpider | 16218695 |
| DrugBank | DB03744 |
| ECHA InfoCard | 19d6cca2-f597-4695-992d-d0bfa6a5a4ef |
| EC Number | 222-538-8 |
| Gmelin Reference | 10434 |
| KEGG | C01853 |
| MeSH | D020123 |
| PubChem CID | 86610173 |
| RTECS number | PY9335000 |
| UNII | V1Q0O96L5X |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID9034095 |
| Properties | |
| Chemical formula | Na2C5H4O5 |
| Molar mass | 188.05 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 1.73 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -2.1 |
| Vapor pressure | 0.01 hPa (20 °C) |
| Acidity (pKa) | 7.59 |
| Basicity (pKb) | pKb: 6.69 |
| Magnetic susceptibility (χ) | -43.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.478 |
| Dipole moment | 7.7 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 222.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1179.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1468.5 kJ/mol |
| Pharmacology | |
| ATC code | A12CC06 |
| Hazards | |
| Main hazards | H319: Causes serious eye irritation. |
| GHS labelling | GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | Precautionary statements: P261, P264, P280, P301+P312, P305+P351+P338, P337+P313, P501 |
| NFPA 704 (fire diamond) | NFPA 704: 1-0-0 |
| Lethal dose or concentration | LD50 Oral Rat 4,417 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 12,000 mg/kg |
| NIOSH | UE8575000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| Related compounds | |
| Related compounds |
Monosodium 2-oxopentanedioate 2-oxopentanedioic acid Diethyl 2-oxopentanedioate 2-oxoglutaric acid Glutamic acid |

