Diphenylmethane Diisocyanate: Balancing Progress and Responsibility in Chemistry
Looking Back: How Diphenylmethane Diisocyanate Changed the Materials World
Diphenylmethane diisocyanate (MDI) grew out of a push to build better plastics, starting in the early 20th century. Early polyurethane pioneers in Germany first produced related molecules during the 1930s while searching for alternatives to natural rubber. After the war, companies ramped up commercial production, seeing the growing need for flexible foams, insulation, and durable coatings. MDI soon became a backbone for everything from building insulation panels to cushioning in car seats. Its rise mirrored a changing world—one hungry for comfort, safety, and efficiency. Watching how fast MDI-based materials spread through industries, it’s easy to see the deep connections between chemistry’s innovations and the massive changes that have shaped modern life. Each shift in how MDI is prepared or applied reflects evolving priorities—cheaper processes, more energy-efficient homes, or smarter uses of raw materials.
Product Details: What Sets MDI Apart
MDI stands out as an aromatic diisocyanate with two -NCO groups hanging off a central diphenylmethane core. It comes in several forms, from pure monomeric MDI to polymeric blends. At room temperature, it usually appears as a pale yellow liquid or a shiny solid, depending on how the molecules are chained together. This chemical’s unique reactivity with compounds containing active hydrogens, especially polyols and water, made it a workhorse for crafting tough yet flexible polyurethane networks. What strikes me is how its somewhat sharp, nutty odor becomes an instant reminder of the precision, and sometimes messiness, of this kind of industrial chemistry. MDI’s mix of high reactivity and relative stability gives engineers the leverage to fine-tune foam hardness, adhesive tack, or the toughness of sealants. This control over finished properties led to a revolution in product design, especially in the construction, automotive, and appliance sectors.
Physical and Chemical Traits: Strengths and Cautions
MDI sits at a crossroads between powerful chemistry and practical design. Its melting point usually falls near forty Celsius, but proprietary blends can nudge this slightly up or down. Unlike its cousin TDI, MDI stays less volatile and presents a lower risk of airborne exposure. That’s a big factor in why so many factories moved to MDI systems after stricter worker protection rules came in. In the lab or on a factory floor, one thing demands respect—the strong tendency of MDI to react with water and amines, giving off carbon dioxide and heat. If foam production goes wrong, that heat can spike and trigger violent expansion. Its compatibility with lots of chemical additives creates both opportunities and headaches: designers enjoy more choices, but small formulation tweaks can swing product performance in hard-to-predict ways. For anyone handling MDI, glove and goggle habits quickly become second nature—not just for safety, but because even trace splashes or fumes can leave a stubborn sting.
Technical Specifications and Product Labeling: Trust and Transparency
The need for clarity around what MDI contains has only grown as customers, regulators, and activists push for transparency. It’s hard to forget the shift, years ago, when labeling on drums changed from cryptic codes to full chemical names and clear hazard icons. Industry norms establish limits for NCO group content, permissible trace contaminants, and batch-to-batch consistency. For those producing or specifying MDI, every percentage point in the NCO index can make or break a formulation. Shipping and storing this material introduces challenges: containers must stay tightly closed to block moisture, and stock rotation keeps old, thickened MDI from wrecking downstream quality. Labs and plants have learned the hard way that losing track of a tank’s temperature or loading schedule can cost dearly in scrapped product or, worse, health problems for workers. Robust technical standards matter not only because of legal demands but because they anchor trust at every level.
Making MDI: From Benches to Factories
The backbone of industrial MDI manufacturing lies in the reaction of aniline with formaldehyde to make methylenedianiline, then converting this key intermediate with phosgene. This process—gritty, reliant on tight controls—packs real industrial risk. I’ve seen how rare slip-ups, like a vented phosgene line, can produce chaos. But with careful systems, it delivers a steady stream of MDI that feeds the downstream needs of foaming lines, adhesives, and coatings. Over the past decade, research has chipped away at the waste and hazards, swapping older, labor-intensive batch steps for continuous, closed-loop processes. Each improvement—less phosgene, better catalysts, tighter emissions capture—has reduced both environmental load and worker exposure. These successes didn’t erase the risk; instead, they forced a more careful matching of technology and practice. It's not hard to admire the engineers who kept dialing in better yields, all while keeping an eye on the chemical’s potent hazards.
Chemical Reactions and Modifications: Unlocking Value
MDI’s real magic comes alive in its versatility under different chemical reactions. Make contact with polyether or polyester polyols, and the result is a solid, springy polyurethane foam fit for mattress makers or automotive seats. Pair it with chain extenders and crosslinkers, and coatings grow tough enough to stand up to rough factory floors. Researchers keep pushing the envelope, coaxing new reaction pathways to yield specialty elastomers or modified resins with added functionality. Each chemical tweak unlocks modified products—from temperature-stable insulation boards to noise-dampening panels in power plants. The isocyanate groups in MDI, so eager to react, demand not just precision in mixing but discipline in cleanup. Those lucky enough to watch a slab of foam rise out of a well-calibrated mold feel the thrill of chemistry as both craft and science. That sense of mastery keeps scientists and production workers pushing for each incremental improvement.
Names, Synonyms, and Trade Lingo
In the world of formulation and commerce, MDI can wear many hats. Someone looking for 4,4’-MDI, pure MDI, or polymeric MDI all lands in the same chemical family but sees real differences in performance. Some trade names flash across spec sheets in construction or footwear production circles, but under the brand surface beats the same core compound. This knot of trade vocabulary often keeps newcomers guessing, yet it underscores the sheer reach of MDI technology. Having spent time flipping between technical data from different countries, I recognize the confusion—and the sharp need for consistency to avoid costly mistakes in cross-continental supply chains.
Safety, Operations, and Responsibility
The story of MDI wouldn’t make sense without facing up to the hard realities of chemical safety. Isocyanates, by nature, hit hard on lungs and skin. Repeated, careless contact can trigger asthma or lingering skin problems. Regulations on workplace air, splash shields, and even medical monitoring stepped in due to real stories—those workers who lost their health through short-sighted shortcuts. There’s no getting around the need for PPE and good habits. Automated dispensing, closed transfer lines, and indoor air controls now dominate well-managed plants. Each extra safety step—right down to training newcomers on what spilled MDI smells like—represents lessons learned, often at real human cost. No amount of technical prowess excuses neglect on this front.
Applications That Touch Everyday Life
It’s tough to spend a day without brushing up against MDI’s legacy. The foam inside a running shoe, insulation behind a wall, steering wheel grip, even the tough finish on a school gym floor—all likely owe their properties to well-tuned MDI chemistry. The construction industry leans hard on MDI-based insulation to meet ever-tighter energy standards. Car makers build lighter, more comfortable interiors by integrating MDI in seats and dashboards. Appliance makers, seeking efficiency, rely on rigid polyurethane foam to cut energy loss. Medical device companies harness specialized grades to make splints and prosthetic liners. For many of these uses, success isn’t just about pure chemistry; it’s about how well teams mesh production, logistics, and design, delivering reliable performance in a demanding world. I’ve seen how the smallest shift, like a new curing agent or blowing agent, can ripple through costs, regulatory compliance, and product warranty claims. This push and pull between innovation and reliability defines MDI’s ongoing impact.
Research, Innovation, and Creative Problem-Solving
If you spend any time with chemists or engineers working on MDI, a shared attitude emerges: a mix of confidence and caution, always probing the boundaries of what this compound can do. Research labs keep targeting lower-odor grades, bio-based feedstocks, and faster curing times. Plenty of startup teams see a future where less phosgene feeds the process, or carbon-capturing technologies squeeze more value out of MDI plants. Everyone knows the risk of complacency: as regulations tighten and public awareness grows, there’s constant pressure to make MDI safer, less toxic, and greener. Past attempts to replace petro-based aniline with renewable alternatives have made uneven progress—more out of practical hurdles than lack of will. Still, each advance gives hope that industry’s environmental footprint can shrink without losing the performance that makes MDI indispensable. Most striking is how collaboration between industry, academia, and regulatory groups has gotten stronger, bringing new problem-solvers to the table.
Toxicity and Public Health: Facing Tough Facts
For all its utility, MDI comes with risks that no responsible business can ignore. Inhalation and skin exposure, sometimes experienced by untrained workers, have led to long-term health issues—especially in settings where safety protocols get ignored or underfunded. Studies link chronic exposure to increased risks of occupational asthma, lung inflammation, and sensitization, making strict industrial hygiene crucial. Companies responding to these realities deploy air monitoring, personal protective gear, and routine medical checks. Industry watchdogs and public health experts keep a close eye on claims about MDI’s environmental fate—tracking potential soil and water contamination, along with how efficiently waste products get treated. These public debates highlight the wider implications of chemical innovation. Worker training, community outreach, and transparent reporting of exposure incidents all move the needle toward better stewardship.
What Comes Next for MDI: Sustainability and Beyond
The next chapter in MDI’s story will likely be defined by the push for sustainable chemistry. Consumer pressures drive manufacturers to search for bio-based routes and tighter recycling loops. More regulations will land, nudging--or forcing--factories to cut emissions, improve waste management, and invest in better ventilation and containment technologies. The role of advanced analytics and digital controls promises more precise tracking, possibly leading to lower worker exposure and better batch quality. Some startups target circular-economy breakthroughs, aiming to reclaim polyurethane waste streams back into MDI feedstocks, or substitute greener ingredients without losing performance. As climate change and resource shortages weigh more heavily, companies face both an obligation and opportunity to rethink their relationship with foundational chemicals like MDI. This story—and our shared future—depends on matching industry’s pace with a new sense of responsibility.
What is Diphenylmethane Diisocyanate (MDI) used for?
A Key Ingredient in Comfort and Construction
MDI changes daily life in ways most people never think about. I remember pulling up old carpet in my first apartment and seeing an ugly yellow foam pad underneath. That strange-smelling pad didn’t seem impressive, but it quietly kept my place warm in winter and made walking barefoot a little softer. That’s polyurethane foam, and MDI helps make it.
Foam for Sofas, Beds, and Cars
MDI plays a big part in making soft, flexible products like couch cushions, mattresses, and car seats. Ever noticed how springy a fresh mattress feels or how a new armchair bounces back when you stand up? That’s the result of MDI’s role in shaping the structure of flexible polyurethane foam. The foam adjusts to different weights and holds up for years. A seat cushion in a budget car and a plush theater chair both owe their comfort to chemistry that starts with MDI.
Improving Energy Efficiency
Walk into any newly built home. The walls usually hide a thick layer of rigid foam insulation inside, which relies on MDI. This closed-cell foam keeps heating and cooling costs down, blocks moisture, and can even slow down a fire. Builders trust it because it works well and speeds up the job. The Environmental Protection Agency reports that buildings swallow about 40% of total energy use in the United States. Small pieces like these foam boards, made possible with MDI, help chip away at those bills, making homes and businesses cheaper to run.
Making Stronger Everyday Products
Polyurethane adhesives—glues for wood floors or construction—stick so well because of MDI. The same goes for coatings and sealants that last through rain and heat, thanks to the tough bonds this chemical produces. Factories turn to MDI-based adhesives over older glue types because they grip tightly but stay flexible through years of use.
Workplace Risks and How to Manage Them
Contact with MDI can bother workers’ skin, eyes, and lungs. Some jobs expose people to airborne dust and vapor, which can trigger asthma or even severe allergic reactions. I once toured a furniture plant and saw that every worker wore gloves and filtered masks. Management installed full-room air ventilation to grab stray fumes. Safety data backs this up—regular air checks and personal protective equipment limit harm, so experienced sites run routine training and upgrades.
Seeking Safer and Greener Paths
Communities now care more about health and pollution than ever. Companies experiment with safer forms of MDI, recycling old foam, or mixing in bio-based alternatives from vegetable oils. These solutions matter since polyurethane wastes pile up fast, and harsh chemicals can move from factories into water or air. California lists MDI as a chemical to handle with special care, and many global groups set strict workplace exposure limits.
What Really Matters
MDI-built products last longer and work better, from comfy furniture to weather-tight buildings. That said, factories can’t ignore the dangers that come with handling and disposing of this chemical. Solutions like safer equipment, recycling, and investing in new materials count just as much as scientific progress. People want comfort, savings, and safety all at once, and the real progress with MDI shows up in how well those hopes balance over time.
What are the health and safety risks of handling MDI?
MDI Isn’t Just Another Everyday Chemical
Living with news about harmful chemicals used in industry doesn’t always hit home until you stand beside a drum of MDI—methylene diphenyl diisocyanate. I’ve heard from friends who work in foam factories and construction. Most know MDI helps build insulation, paint, and adhesives. Fewer recognize what can go wrong if people treat it lightly.
Breathing It In Isn’t a Small Matter
MDI vapor or dust can irritate the nose, throat, and lungs. Shortness of breath, coughing, or wheezing can pop up after just a single shift in a poorly ventilated room. People can develop asthma after repeated exposure, even at levels below the legal workplace limits. Research from OSHA and NIOSH confirms that isocyanates like MDI rank among the top causes of occupational asthma. That’s not a risk any employer wants to see on the shop floor.
Skin and Eyes Aren’t Safe, Either
A splash on the skin seems harmless—until redness or a rash shows up hours later. With repeated exposures, that rash can turn into chronic eczema. Touching MDI might cause permanent skin allergies after enough contact. Eyes face real trouble, too. A mist or a bit of dust can cause pain and damage that’s hard to heal.
Long-Term Consequences Get Overlooked
I’ve heard complaints from workers who ignored a sore throat or patch of irritated skin, only to watch the problem get worse month after month. Unlike a simple cold or cut, the symptoms fade slowly, if at all. At its worst, MDI-induced asthma doesn’t disappear with just a job change. Everyday life becomes a challenge.
Simple Steps Can Prevent Harm
No one claims that removing MDI from production floors is easy. Since bans aren’t around the corner, the responsibility falls on everyone in the supply chain. Engineering controls, such as fume hoods and proper ventilation, do make a difference. I’ve visited sites where open windows and good air movement dramatically cut down on worker complaints.
Gloves, goggles, and long sleeves feel awkward in summer. Most workers would rather skip them. Still, the science is clear—protective gear cuts the risk by a huge margin. Regular training helps: people forget, and new faces need to learn from the start.
Everyone Gains from Clear Communication
Misunderstandings come quickly when a chemical looks just like everything else on the job. Clear warning signs in plain language—not just codes or numbers—help people react fast if something spills. Easy access to safety showers and washing stations can limit damage from accidents. Companies that build a culture around speaking up about risks see fewer long-term illnesses.
Looking to the Future
Alternatives to MDI might eventually take its place in the industries that depend on it. Until that day, every step forward rests on shared responsibility—between companies, supervisors, and everyone working with these chemicals daily. Real change means listening, staying alert, and caring about coworkers’ health more than any daily deadline. When people band together to enforce safety, the risks shrink—and so do the accidents and illnesses nobody wants.
How should Diphenylmethane Diisocyanate be stored and transported?
The Reality of Working With Chemicals Like MDI
Anyone who has worked in manufacturing or construction has probably been around diphenylmethane diisocyanate, often called MDI. This chemical makes up the backbone of many polyurethane foams, insulation boards, and sealants. Looking at it in a drum or tanker truck, you wouldn’t guess it can affect health or property so much. But ask anyone who has had a spill or a leaky drum—things get serious in a hurry.
MDI and Health: What’s at Stake
MDI doesn’t mess around. Inhaling the vapors can trigger asthma or allergic reactions that sometimes never go away. Skin exposure may lead to irritation and, over time, even permanent sensitivity. Every person I’ve talked to who’s suffered a reaction remembers it for life. Facts back up those stories—the US National Institutes of Health and OSHA both list MDI as hazardous, and say to keep it away from air and skin contact as much as possible.
Taking Storage Seriously
Temperature swings create big problems. MDI reacts with moisture. Water contamination causes the chemical to cure inside the drum, forming solids that block valves and ruin batches. All it takes is a humid warehouse or a leaking roof for a bad day. I’ve walked past storage sites in summer, and it’s not hard to guess which ones are dry, shaded, and controlled. Temperature-controlled, well-ventilated warehouses extend the shelf life dramatically. Preventing contact with water often requires dehumidifiers and sealed containers. It sounds simple until storage gets crowded, pipes sweat, or people rush and forget to double-check lids.
Transport Goes Beyond Loading a Truck
Transporting MDI presents another set of problems. During transit, containers should always stay upright, properly strapped, and away from sources of moisture or heat. Federal regulations call for labels, placards, and manifests, not just to keep up with paperwork but to help first responders if an accident happens. My neighbor hauls freight for a living, and he always double-checks the placards—one wrong label can invite fines or worse, endanger others.
Dealing With Spills and Leaks
I’ve seen what happens when a drum leaks—sticky, pungent fumes fill the air, and hazmat suits show up quickly. Emergency gear, from absorbent pads to breathing protection, should stay within reach wherever MDI sits or moves. Regular training may feel like overkill until someone forgets the steps during a spill and panics. Most workplaces now do annual drills for chemicals like this, and walkthroughs show where problems crop up before something goes wrong.
What Works and What Could Improve
It still surprises me how many facilities improve simply by fixing doors, adding climate control, or writing up checklists for drivers. Clear procedures save time and keep people from guessing. Automated climate controls—while expensive up front—usually make up for the cost by reducing waste and medical incidents. Technology helps here, but the biggest impact still comes from a crew who understands why these steps matter. If managers reinforce the message, folks tend to pay attention.
Every step, from choosing the storage spot to loading a trailer, shapes whether MDI serves its purpose or shows its dangerous side. I’ve watched companies step up, accept help from chemical suppliers, and build systems that keep accidents rare. It’s the kind of behind-the-scenes work that never shows up in headlines but keeps people and products safe every day.
What is the chemical structure and formula of MDI?
What Is MDI, Really?
MDI stands for methylene diphenyl diisocyanate, a substance that pops up in more places than most folks might realize. At its core, MDI brings together chemistry and practicality. It forms the backbone for a wide range of polyurethane products, from insulation panels to the soft cushions in car seats.
The Nuts and Bolts: Breaking Down the Structure
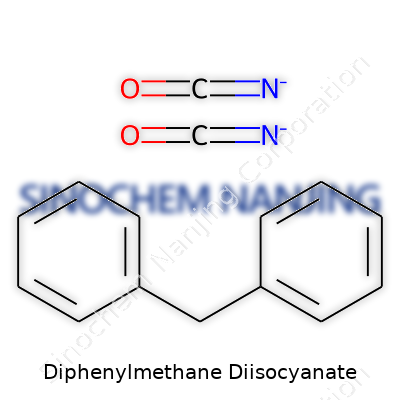
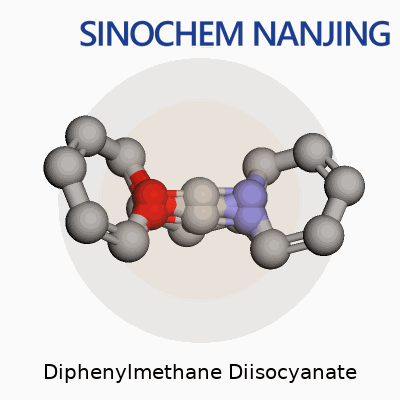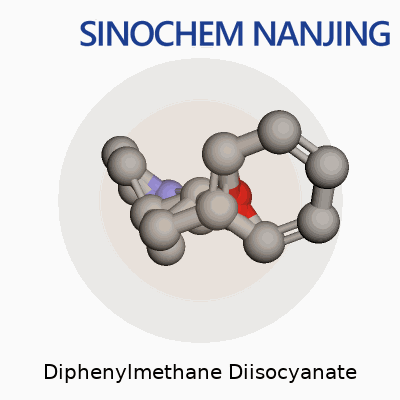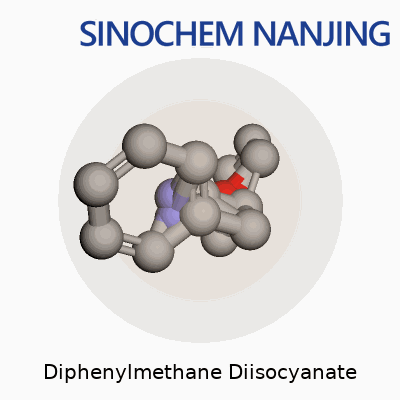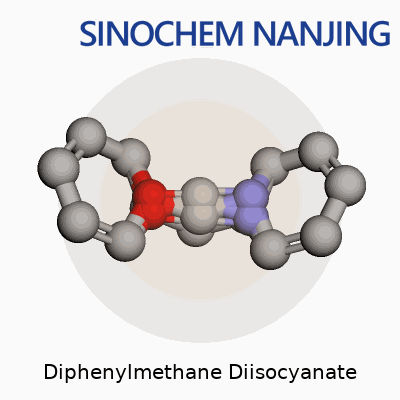
The structure of MDI isn’t just academic. It's tangible. You can feel its effect in the durability of shoe soles or the resilience of construction materials. Chemically, MDI features two isocyanate groups (-N=C=O) bonded to a benzene ring through a bridging methylene group (–CH2–). The formula for the standard or “pure” version is C15H10N2O2.
MDI’s full chemical name, 4,4'-methylenediphenyl diisocyanate, points right to its backbone: two phenyl rings connected by a methylene bridge, both holding an isocyanate group. This symmetry and rigid structure lock in physical properties that resist heat and chemicals.
MDI isn’t a one-trick pony. Manufacturers create several related molecules under the MDI umbrella, each tweaking the number or position of isocyanate groups or benzene rings. But most applications rely on the classic 4,4'-MDI for its predictable, tough performance.
Why Structure Matters to Everyday Life
The magic of MDI’s structure really hits home once you look at its impact on comfort, safety, and energy efficiency. Polyurethane foam relies on those double isocyanate arms to cross-link with compounds containing active hydrogen—often water or polyols. This cross-linking turns liquid ingredients into foams, adhesives, or coatings in minutes.
Energy bills shrink for people living in homes built with MDI-insulated walls. Cars last longer on the road thanks to robust polyurethane bumpers and seats. Wearing protective gear, I’ve seen MDI-curing foam stop water leaks in minutes during repairs, saving tons of hassle.
There’s no way around the health side of the equation, though. Breathing MDI in raw form can create real health risks. Anyone working with it needs proper protection, because once MDI cures, it stabilizes, becoming non-volatile. This history of caution comes from both personal experience and a mountain of regulatory guidance—OSHA sets exposure limits, and industries invest in strong ventilation systems for good reason.
Responsible Use and Alternatives
MDI itself isn’t going away anytime soon. The world depends on it for everything from sneaker soles to fridge insulation. Still, safer handling protocols keep people out of the ER, and the push for greener chemistries shows promise. Universities and companies chase bio-based polyurethanes and less toxic isocyanate reactions. One emerging route involves developing hybrids that keep performance up while dialing back potential health risks.
As demand for sustainable choices grows, expect shifts in how MDI products get made and handled. Education, clear labeling, and enforcement of safety standards support both factory workers and end consumers. In my own work, providing up-to-date training and easy access to protective gear made a real difference in both morale and safety records.
The specifics of MDI’s chemical structure aren’t just ivory tower facts—they shape comfort, efficiency, and responsibility every day in the field, workshop, and home.
What personal protective equipment (PPE) is recommended when working with MDI?
Understanding MDI Risks
Methylene diphenyl diisocyanate (MDI) turns up in a lot of workplaces—especially where insulation, adhesives, or polyurethane are around. It doesn’t take long to hear stories about coughing fits, itchy skin, or even more serious breathing problems after handling this chemical. MDI gets into air easily, especially if temperatures rise or there’s poor ventilation. The reality is, just a little contact can bring big health headaches. Decades of research show regular MDI exposure can cause asthma or even permanent lung damage. Not enough people know how sneaky this stuff is: it can soak in through skin as well as the air. PPE builds a last line of defense, but the story doesn’t end there.
Where Safety Matters: Hands, Eyes, Lungs, Skin
You can spot a seasoned worker by checking their gear at the start of a shift. Gloves come first. Not every glove blocks MDI; thin nitrile or latex gloves break down after just a few minutes of contact. Research from the CDC points towards thicker nitrile or butyl rubber gloves for proper blocking power—they keep skin from itching or burning later in the day. Hands tend to swipe across faces or rub eyes without a second thought. Any trace of MDI left behind spells trouble.
Eye protection can seem excessive, but anyone who has felt that sudden sting when a drop lands near the eye knows the value of a snug set of chemical splash goggles. In one busy facility, a quick spill ended with a coworker racing to an eyewash station—goggles make the difference between an annoying interruption and permanent damage.
Breath Matters Most
Lungs respond fast to MDI. Mask use makes a solid line of defense, especially when jobs stir up dust or mist—think of spraying, mixing, or cleaning up spills. Air-purifying respirators with cartridges rated for organic vapors filter out much of the harm, giving workers a way to breathe safely. PAPR (powered air-purifying respirators) show up in places where ventilation stinks or jobs run long. If you’ve ever breathed through a respirator on a hot, sticky day, you know wearing it isn’t fun. Still, the alternative means wheezing or hospital visits down the line. A survey from NIOSH (National Institute for Occupational Safety and Health) showed major drops in asthma cases just by adding recommended masks and training on their use.
Clothing: The Overlooked Shield
Exposure is sneaky—sometimes it sneaks through seams or cuffs. Coveralls or chemical-resistant aprons solve most of these problems. Washable work clothes, kept away from home laundry, stop chemical traces from reaching family members or car seats. Boots need closed tops, so spills run off and feet stay safe. Simple habits—changing out of gear before leaving—score big points for health. A bit of discomfort now beats months of skin rashes or breathing treatments later.
Fixing the Gaps: What Else Helps
PPE isn’t magic. The most careful setup only works when people learn how to wear gear properly and recognize when it wears out. Employers—big shops or small teams—can build a culture where speaking up feels normal and everyone checks one another’s masks and gloves before the job starts. Stocking proper sizes and styles—a range of glove sizes, goggles that fit faces with glasses, or respirators suited for long beards—helps real workers keep protection in place, not hanging on a hook in the shed. Regular checks make sure no one cuts corners when they get busy or tired. Those small, steady actions do more for health than any form on a clipboard ever could.
| Names | |
| Preferred IUPAC name | Benzene-1,1'-methylenediisocyanate |
| Other names |
MDI Methylenediphenyl diisocyanate Methane diphenyl diisocyanate |
| Pronunciation | /daɪˌfiːnɪlˈmɛθeɪn daɪˌaɪsəˈsaɪəneɪt/ |
| Identifiers | |
| CAS Number | 101-68-8 |
| Beilstein Reference | 1276050 |
| ChEBI | CHEBI:83108 |
| ChEMBL | CHEMBL1432287 |
| ChemSpider | 15749 |
| DrugBank | DB14516 |
| ECHA InfoCard | 13f4683c-b684-4641-a208-5b39d3efb8d8 |
| EC Number | 202-966-0 |
| Gmelin Reference | 82255 |
| KEGG | C06434 |
| MeSH | D003972 |
| PubChem CID | 6616 |
| RTECS number | PA8050000 |
| UNII | 9JCM0L2A90 |
| UN number | UN2078 |
| CompTox Dashboard (EPA) | DTXSID2020286 |
| Properties | |
| Chemical formula | C15H10N2O2 |
| Molar mass | 250.25 g/mol |
| Appearance | White to pale yellow solid or liquid |
| Odor | Musty; Earthy |
| Density | 1.23 g/cm³ |
| Solubility in water | Insoluble |
| log P | 3.9 |
| Vapor pressure | 0.0002 mmHg (25 °C) |
| Acidity (pKa) | 22.1 |
| Basicity (pKb) | 11.26 |
| Magnetic susceptibility (χ) | -49.0e-6 cm³/mol |
| Refractive index (nD) | 1.574 |
| Viscosity | 150 - 250 mPa·s (25 °C) |
| Dipole moment | 2.78 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 355.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -94 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6304 kJ/mol |
| Pharmacology | |
| ATC code | D08AX |
| Hazards | |
| Main hazards | Harmful by inhalation, causes skin and eye irritation, may cause sensitization by inhalation and skin contact. |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS02,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H315, H317, H319, H332, H334, H335, H351, H373 |
| Precautionary statements | P201, P202, P260, P264, P271, P272, P280, P284, P302+P352, P304+P340, P305+P351+P338, P308+P313, P333+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-1-1-W |
| Flash point | 199.4 °C |
| Autoignition temperature | 600°C |
| Explosive limits | Explosive limits: 0.4–11.0% |
| Lethal dose or concentration | LD50 (Oral, Rat): > 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral > 2,000 mg/kg |
| NIOSH | NIOSH: PMDI0000 |
| PEL (Permissible) | 0.02 ppm |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | 75 mg/m3 |
| Related compounds | |
| Related compounds |
Toluene diisocyanate Hexamethylene diisocyanate Naphthalene diisocyanate Polymethylene polyphenyl isocyanate Isophorone diisocyanate |

