Diphenyldiselenide: A Closer Look at a Compelling Compound
Historical Development
Back in the early days of organoselenium chemistry, researchers chasing new molecules for both curiosity and practical needs landed on diphenyldiselenide. It didn’t come out of nowhere. Organic chemists noticed that tweaking molecular frameworks with selenium held promise, dating as far back as the work of Paul Lebeau in the early 20th century. The evolution of synthetic techniques and the availability of elemental selenium fueled more accessible routes to organoselenium compounds. Diphenyldiselenide entered the picture due to both its unique properties and its relative stability compared with many other selenium-containing compounds. For decades, it has served as a go-to classic for chemists developing new reactions and investigating biological mechanisms that might hinge on selenium’s quirks.
Product Overview
In the lab, diphenyldiselenide features two phenyl groups straddling a diselenide linker. The molecule strikes a balance between stability and reactivity, which is probably why so many organic chemists reach for it in the pursuit of new reactions. Unlike some of the stinkier, more volatile selenium products, diphenyldiselenide can actually sit around on a shelf for reasonable stretches without breaking down. Its structure lends itself to activity as a source of electrophilic selenium and as a building block for more exotic molecules.
Physical & Chemical Properties
Diphenyldiselenide appears as a yellow to orange crystalline solid at room temperature. It melts near 61-62 °C, and fresh crystals display a gentle luster under light. Solubility leans toward organic solvents; good choices include chloroform, dichloromethane, and ether. Chemically, this compound stands out because the selenium-selenium bond is reactive but not too fragile for bench work. The weak Se–Se bond means that diphenyldiselenide can split into phenylselenyl radicals or cations—handy species for both synthesis and mechanistic exploration.
Technical Specifications & Labeling
Quality often comes down to purity and crystal form. Authentic samples should show clear melting behavior and a characteristic NMR pattern. Contaminants like elemental selenium or phenylselenol can mess with both reactivity and safety, so diligent warehouse practices and valid labeling have real meaning here. Responsibility for handling and storage falls squarely on both manufacturers and the hands of scientists, who need to watch for degradation or accidental exposure.
Preparation Method
One reliable route involves the oxidation of benzeneselenol under carefully controlled conditions. With air or mild oxidants, the selenol dimerizes, and crystals of diphenyldiselenide separate out of the reaction mixture. Some syntheses use elemental selenium and organometallic intermediates, though those can pull in different types of impurities depending on the method. The preparation demands good ventilation, gloves, and a real respect for the toxicity of selenium species, because things can get hazardous fast if exposure is not managed.
Chemical Reactions & Modifications
Diphenyldiselenide plays multiple roles in organic chemistry. It serves as a source of PhSe groups in substitution or radical addition reactions. Its ability to release phenylselenyl cations or radicals under the right conditions supports a slew of transformations, including allylic selenenylation and oxidative rearrangements. Once attached to a molecule, the PhSe group can often be converted into various useful functionalities, such as alcohols or aldehydes, through oxidation or elimination. This flexibility made the compound a star in the toolbox of organic synthesis for decades, and its consistent reactivity patterns help researchers sketch out and execute new synthetic plans.
Synonyms & Product Names
Diphenyldiselenide carries a few alternate names, including the shorthand PhSeSePh and 1,2-diphenyldiselenium. Some databases and supply catalogs list it as diselenobis(benzene) or bis(phenyl) diselenide. The different monikers reflect both formal systematic chemistry naming and the kind of language that circulates among working scientists.
Safety & Operational Standards
Selenium compounds can be harmful, and diphenyldiselenide deserves respect in the lab. Direct skin contact, inhalation, and accidental ingestion all rank as genuine risks, given the acute and chronic effects associated with selenium exposure. Adequate ventilation, the right gloves, and safety glasses aren’t just formalities; they help prevent problems that linger. Waste disposal also catches a lot of attention because organic selenium waste stands out as both persistent and toxic if released into the environment. Strong professional training and clear protocols make a difference, and these precautions also open doors to responsible research and safer industrial use.
Application Area
Researchers most often reach for diphenyldiselenide in organic synthesis. Its introduction into a molecule can set the stage for downstream transformations, and the PhSe unit supports selectivity in complex molecule construction. Beyond basic synthesis, the compound has cropped up in medicinal chemistry projects, with selenium-containing molecules under investigation for antitumor, antioxidant, and antimicrobial properties. In the field of materials science, organoselenium compounds—including diphenyldiselenide—sometimes find their way into precursors for thin films or nanostructured materials, usually where unique electronic or optical properties are sought.
Research & Development
The track record for diphenyldiselenide includes both established roles and steady innovation. Chemists continually revisit classic reactions using this compound to seek out milder, more selective, or greener protocols. Interest in selenium’s unique bioactivity has prompted a steady trickle of medicinal chemistry explorations. The embrace of catalytic strategies leveraging selenium-based intermediates adds nuance and value, as scientists look to replace rarer or more troublesome transition metals with these more earth-abundant alternatives.
Toxicity Research
Selenium’s double-edged nature—essential in trace amounts, harmful in excess—demands a closer look. Animal studies show that high doses of diphenyldiselenide bring about liver and kidney issues, with neurological symptoms kicking in at higher concentrations. Metabolic studies uncover a link between organoselenium compounds and altered antioxidant enzyme activity. Researchers keep an eye on possible DNA or cellular damage, and these signals prompt careful handling standards. Not only do such results guide workplace safety standards, they also influence the exploration of therapeutic dose ranges in biomedical investigations. Some studies probe structural modifications aiming to dial down toxicity while preserving—or enhancing—biological action.
Future Prospects
The path forward for diphenyldiselenide ties closely to a better understanding of selenium’s place in therapeutic mechanisms and catalysis. Chemists see gaps where selenium chemistry can outperform heavier, scarcer elements in reaction schemes. On the biomedical front, deeper insight into structure–activity relationships might unlock compounds with reduced side effects and broader clinical utility. Environmental questions about selenium waste won’t fade away soon, so scaled-up applications must grapple with greener prep methods and efficient waste recycling. Academic groups, industry, and regulatory agencies all face the challenge of translating the distinctive properties of organoselenium chemistry into safer processes and more beneficial applications, grounded not only in scientific promise but in real-world accountability.
What is Diphenyldiselenide used for?
The Science Behind Diphenyldiselenide
Diphenyldiselenide doesn’t roll off the tongue or pop up in dinner table conversations, but it grabs the attention of chemists who care about hitting targets with fine precision. In the lab, researchers use this compound for its ability to transfer selenium atoms. Selenium doesn’t get as much spotlight as iron or calcium, but it shapes reactions in subtle yet powerful ways. By swapping selenium into organic molecules, scientists create new drugs or test out promising chemical reactions that wouldn’t work the same way with sulfur or oxygen.
Chasing Better Medicines
The hunger for better medical treatments means researchers never stop hunting for tools to tweak molecules just enough to make them stronger, last longer in the body, or behave differently against disease. Diphenyldiselenide helps with that tuning. Work published in Bioorganic & Medicinal Chemistry shows that diphenyldiselenide-derived compounds sometimes fight inflammation with less toxicity compared to popular anti-inflammatories. Animal studies indicate it can block or dampen pain signals, and some teams have pushed it as a possible helper in the fight against neurodegenerative diseases like Alzheimer’s.
Fighting Oxidative Stress
Cells feel the pressure of oxidative stress every day, as molecules called free radicals chip away at DNA and tissues. Selenoproteins in the body act like first responders, and synthetic molecules such as diphenyldiselenide mimic those built-in protectors. In small doses, this compound sometimes acts like glutathione peroxidase, an enzyme that shields cells from damage. It's no magic bullet, but understanding how it works as an antioxidant opens up new ways to protect nerves and reduce cell death in animal models.
Tools for Research, Not for Daily Life
Every time I’ve spoken with chemists or spent time reading lab protocols, diphenyldiselenide pops up as a favorite for specific organic syntheses. It doesn’t have a role in consumer products, and I haven’t found it used directly in food, cosmetics, or any day-to-day setting outside the lab. Handling it means careful storage, fume hoods, and gloves, since high doses can prove toxic. The limited availability and safety profile block its use in mainstream products, and that’s for the best.
Where Safety and Promise Collide
Stories of chemical breakthroughs always bump up against tough regulations and real risks. Diphenyldiselenide offers a way forward for certain kinds of drug research, but using it on a bigger scale means tackling tough safety questions. Exposure risks encourage proper disposal and extra protective equipment in labs, not something to shrug off or ignore.
Improving Outcomes Through Smarter Chemistry
Scientists don’t work with diphenyldiselenide for novelty—they turn to it because it fills specific gaps that other chemicals simply don’t. To cut risk, labs run tight quality controls and keep a close watch on exposure. For society to benefit, research teams keep pushing for less toxic, more targeted compounds that keep selenium’s helpful traits without carrying as much baggage. Looking ahead, the goal stands to design even better molecules and learn more from selenium’s quirks, building stronger, safer medicines for people who need them most.
What is the chemical formula of Diphenyldiselenide?
Breaking Down the Structure
Diphenyldiselenide shows up as a bright yellow powder, and you’d spot its chemical formula written as C12H10Se2. It comes from two phenyl rings bridged by a pair of selenium atoms, which you can see if you write it out as (C6H5Se)2. So much in chemistry boils down to structure, and here, knowing there are twelve carbons, ten hydrogens, and two selenium atoms shapes everything from reactivity to safety.
Why Selenium Matters
Selenium can play both sides: vital nutrient on one hand, risky with too much exposure on the other. With diphenyldiselenide, you’re not looking at something that belongs in a multivitamin, but instead a compound that researchers have eyed for its antioxidant abilities and its use in organic synthesis. In many laboratories, chemists choose diphenyldiselenide because it transfers selenium atoms smoothly during reactions. That predictability links directly to its chemical makeup.
Experience in the Lab
I’ve worked with selenium compounds a few times, and you can’t ignore their smell—sharp and telltale, even in tiny quantities. The clear structure of diphenyldiselenide helps with planning safe handling because you know exactly what’s involved. In research, I’ve always counted on the stability of those phenyl rings and the distinct “Se-Se” bond for reliable results in synthesis. This is not just theory but real world laboratory work, where mistakes can mean trouble.
Health and Environmental Considerations
People sometimes overlook the risks with organoselenium compounds—just because they’re useful doesn’t mean they’re harmless. Diphenyldiselenide does not escape that truth. It can irritate, and too much exposure leads to problems for both workers and the environment. According to the U.S. National Library of Medicine, selenium toxicity can affect major organs when mishandled. The formula’s transparency allows chemists and safety officers to plan proper ventilation, use of gloves, and strict waste disposal. That’s a big step forward from the days when experience counted as the only shield.
Safer Laboratory Culture
Regulatory bodies such as OSHA and EPA keep setting clearer boundaries for how labs handle selenium compounds. In practice, that means eyewash stations nearby, well-marked containers, and comprehensive training for everyone. The detailed nature of the chemical formula helps teams assess risks quickly, train students thoroughly, and keep accidents at bay.
Pushing Toward Solutions
Safer alternatives to selenium-based reagents keep emerging as green chemistry goals come into focus. Still, for some critical reactions, diphenyldiselenide remains indispensable. One step for improvement might involve more research into reusable or recyclable selenium sources, lessening dependence on fresh, raw chemicals. Educational outreach can boost understanding well before a bottle ever gets opened for the first time.
Bottom Line
For chemists, the chemical formula C12H10Se2 doesn’t just represent another compound. It brings clarity, directs safe practice, and reminds us that every bottle holds both promise and responsibility. With good training and strict attention to detail, labs can keep making discoveries without sacrificing safety or stewardship.
How should Diphenyldiselenide be stored?
Why Storage Gets Overlooked
Chemical storage sounds like a background task. No one brags about their careful shelving or labeling habits. Still, for compounds like diphenyldiselenide, smart storage saves labs money, headaches, and sometimes, safety hazards. My own slip-ups with moisture-sensitive chemicals have ruined expensive bottles more than once.
Understanding Diphenyldiselenide’s Temperament
Not every compound sits quietly on a shelf. Diphenyldiselenide reacts to air, light, and improper temperatures in ways that can disrupt experiments or put people at risk. Its pungent smell doesn’t just indicate presence—it points to volatility. Once, I left a sample on the bench overnight. Next day, the lab reeked, and my intended synthesis was a flop.
Reliable Storage Methods
Air-tight containers earn their keep. Glass bottles with PTFE-lined caps lock out air and moisture. Ignore cheap containers—any leak lets this compound degrade. Polypropylene falcon tubes often get used for convenience, but glass resists reaction best.
Keep light out of the equation. Amber or opaque bottles block light and help prolong shelf life. On open shelving or where light sneaks in, wrap bottles in aluminum foil. Even indirect sunlight speeds up decomposition. Years ago, I stored a bottle close to a window, and three months later, it was useless.
Cooler is better but avoid freezing solid. Standard lab refrigerators usually hold 2–8°C. Storing diphenyldiselenide there slows decomposition. Never freeze unless absolutely necessary, since thawing can introduce moisture even inside a capped bottle. Fluctuating cold and warm cycles wreak havoc over time. My colleagues once tried deep-freezing, thinking it stopped every reaction, but handling frost-damaged samples just led to sticky messes.
Label everything clearly. Whatever gets opened—big supplier bottle or a transferred aliquot—needs a date-down label. Use permanent markers, not stickers that peel in the cold.
Don’t store chemicals above head height. Spill accidents happen, and reaching for a leaky bottle never feels good. One spill in a fume hood lingers for days, and you don’t want that smell following you home.
Cutting Down on Waste
Loose storage practices often lead to wasted chemicals and strained budgets. Buying large amounts to “save money” only works if samples last. Order only what you’ll reasonably use. I’ve seen unopened bottles thrown out after a year, all because the chemical inside turned brown or clumpy.
Staying Safe: The Bigger Picture
Diphenyldiselenide carries hazards beyond property loss. Small exposures can build up over time. Fumes—if allowed to escape—pose health risks. Store only in fume hoods or designated flammable cabinets, not on bench tops. Keep updated safety data sheets nearby, not buried in a binder no one checks.
Colleagues new to selenium compounds sometimes forget: gloves and goggles matter every time. Even if you’re just rummaging for a bottle, splash risk remains. A near-miss from a leaky cap taught me this lesson fast.
Maintaining Progress in the Lab
Proper storage rarely grabs attention, but it shapes every experiment’s outcome. Those small daily habits—checking seals, rotating stock, labeling with dates—turn complicated chemistry from unpredictable to reliable. Respect for each compound’s temperament, especially diphenyldiselenide, protects people and keeps projects from derailing. Most good chemistry comes down to simple routines executed well.
Is Diphenyldiselenide hazardous to health?
Understanding the Basics
Diphenyldiselenide usually shows up in chemistry labs, not in kitchen cupboards. Scientists lean on it as a reagent for organic synthesis, thanks to its selenium content. Folks might stumble across the compound in research settings, especially in pharmaceutical or chemical manufacturing environments. Its color, yellow-orange, seems pretty harmless to the untrained eye, but that doesn’t give a pass on safety.
What Science Says About Exposure
Researchers have spent years uncovering the effects of organoselenium compounds. Animal studies suggest diphenyldiselenide can enter the body through skin, inhalation, or ingestion. Selenium itself is an essential trace element, playing a role in defending cells from oxidative stress. Too much, though, puts a person at risk for toxicity. The concern jumps when dealing with concentrated or synthetic forms like diphenyldiselenide rather than dietary selenium.
Exposure in the lab or during manufacturing can affect the nervous system, liver, and lungs. Signs of overexposure might include symptoms like dizziness, fatigue, or even problems with coordination. Chronic high exposure to selenium compounds sometimes leads to hair loss and fingernail changes, not to mention gastrointestinal distress.
Why This Matters
Safety gaps lead to real accidents. I remember my early days of lab training where our group handled unfamiliar chemicals without respecting these deeper risks. Nobody showed us how vapors can sneak past cheap gloves or how spills hang around on surfaces just waiting to be picked up by unsuspecting hands. Diphenyldiselenide, with its stability and oily consistency, lingers longer than people expect.
Even more concerning, information can be scarce or filled with dense jargon. People outside of research rarely see hazard data. Arguments downplay risks since few human exposure cases appear in public records. That doesn’t mean the threat isn’t there—it means mishaps haven’t made headlines.
Industry Practices and the Current System
Labs consider chemical safety training part of the onboarding process, but I've seen how easy it is for shortcuts to become regular habits. Fume hoods left off, gloves reused, spill kits gathering dust—maybe because the urgency fades without a front-page incident.
Looking at guidelines, OSHA and NIOSH list selenium compounds as hazardous. The risk level depends on handling practices and exposure duration. European agencies echo this, setting workplace exposure limits that show how seriously authorities view these chemicals. These guidelines exist because stories of chemical accidents, mild and severe, still happen every year in settings with experienced professionals.
Solving the Problem
Prevention starts with access to information that’s easy to digest. Clear, honest risk disclosures work better than buried PDF safety sheets. Proper ventilation and protective equipment keep risk manageable, but only if workers trust their gear and use it consistently. Supervisor buy-in matters just as much as line worker caution. Routine air monitoring and personal exposure tracking hold companies accountable.
Small changes make the biggest difference. I've seen teams that shared firsthand stories about close calls change their attitudes overnight. Simple things like pre-shift safety briefings or quick “what went wrong” reviews after incidents remind people that safety isn’t a chore, it’s a necessary deal for everyone in the building.
Final Thoughts
Diphenyldiselenide deserves respect. Underestimating it—just because it isn’t a household name—misses the point. Health hazards aren’t always about acute symptoms; long-term exposure carries real weight. Staying curious and demanding strong safety culture will keep workplaces much safer, one shift at a time.
What are the handling precautions for Diphenyldiselenide?
Understanding What Diphenyldiselenide Can Do
Diphenyldiselenide isn’t something most people will find in their homes, but it lands on the benches in plenty of chemistry labs. This organoselenium compound brings some unique reactions to the table. Still, these same properties can turn risky fast. Just smelling its sharp odor hints that something potent is at play. Most folks who’ve worked around these chemicals know even the tiniest mistake can turn routine experiments into something much worse.
What Makes Diphenyldiselenide Risky
Se compounds don’t pull punches. Spend enough time in a lab, you’ll see people talking about their headaches, stomach trouble, and sometimes breathing issues, especially after poor handling. Selenium’s toxicology is well-documented; it disrupts enzymes and causes inflammation when absorbed. Skin contact or inhaling fine particles raises the stakes. Labs have issued more than their share of incident reports after minor spills, especially when proper protection gets neglected.
PPE is the First Line of Defense
Gloves, goggles, and lab coats aren’t just for show. Nitrile gloves handle the job better than typical latex, and goggles prevent accidental splashes. After a spill years ago, I’ve taken to wearing thicker gloves when working with Diphenyldiselenide. Sometimes it feels over-cautious until you remember that facial splashes or seeped gloves can mean days of complications, not to mention the paperwork.
Ventilation and Handling
Proper airflow limits how much vapor lingers. Fume hoods prove their worth every time Diphenyldiselenide gets measured out. From personal experience, carrying out transfers outside the fume hood—even “just for a moment”—leads to headaches that last until well after you leave the lab. If a work space smells like garlic or has that chemical tang, something went wrong with the setup.
Storage Practices That Matter
Diphenyldiselenide asks for a tight cap and a dark place. Exposure to air and light helps this substance break down, releasing more volatile compounds. One time, a bottle left under direct light developed crystals that clogged the cap, increasing the odds for messy accidents. Now, storing it in amber jars and dedicated cabinets keeps things safer and easier to track. Labeling bottles with the date and full chemical name goes a long way, especially during safety audits.
Waste Management Isn’t Optional
Disposal rules can feel strict, but for good reason. Diphenyldiselenide counts as hazardous waste. Double-bagging all contaminated materials and using properly labeled waste containers stops accidental exposure down the line. I’ve watched more than one rookie try to rinse equipment in a regular sink, only to get a firm correction from a supervisor. Labs process all waste for offsite handling rather than letting it mix with everyday garbage.
Preparing for Spills and Exposure
Fast action beats panic. Every lab should keep spill kits—neutralizers, absorbents, disposal bags—close at hand. Practice drills might feel unnecessary, but that muscle memory helps when someone knocks over a bottle. Immediate washing and notification, quick cleanup, and filling out exposures help avoid bigger problems. Emergency eye wash stations matter, and people remember to check them more often after near-misses.
The Bottom Line on Diphenyldiselenide
Safety isn’t just about boxes checked on paperwork; it comes from seeing what happens when rules get ignored. Protective equipment, good habits, and clear storage all build trust in a lab and help send everyone home healthy at the end of the day. No single step does the job alone—each adds up to real protection for people and the environment.
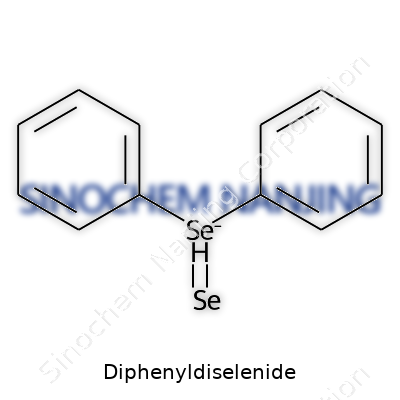

| Names | |
| Preferred IUPAC name | bis(phenyl) diselenide |
| Other names |
Diselenobis(benzene) Benzeneselenenic anhydride PhSeSePh Dibenzene diselenide Diphenyl diselenide |
| Pronunciation | /daɪˌfɛ.nɪl.daɪˈsiː.ləˌnaɪd/ |
| Identifiers | |
| CAS Number | 1666-13-3 |
| Beilstein Reference | 1363681 |
| ChEBI | CHEBI:2757 |
| ChEMBL | CHEMBL1233635 |
| ChemSpider | 20568379 |
| DrugBank | DB07636 |
| ECHA InfoCard | 03b186a4-6db2-4531-b439-580d5ba4d7c1 |
| EC Number | 220-129-8 |
| Gmelin Reference | 82138 |
| KEGG | C12431 |
| MeSH | D008934 |
| PubChem CID | 69048 |
| RTECS number | KV8400000 |
| UNII | QX4X86IWI7 |
| UN number | UN3077 |
| Properties | |
| Chemical formula | (C₆H₅)₂Se₂ |
| Molar mass | 315.98 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.55 g/cm3 |
| Solubility in water | insoluble |
| log P | 3.68 |
| Vapor pressure | 0.0000225 mmHg at 25°C |
| Acidity (pKa) | 12.5 |
| Basicity (pKb) | 1.47 |
| Magnetic susceptibility (χ) | -104.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.778 |
| Viscosity | 0.0086 Pa·s (25 °C) |
| Dipole moment | 1.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 393.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 42.3 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1608.4 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed or inhaled. Causes skin and eye irritation. May cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | `GHS05, GHS06` |
| Signal word | Danger |
| Hazard statements | H301 + H312 + H332: Toxic if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261, P264, P270, P271, P272, P273, P280, P301+P312, P302+P352, P304+P340, P308+P311, P312, P321, P330, P362+P364, P403+P233, P405, P501 |
| Flash point | 125°C |
| Autoignition temperature | 250 °C |
| Lethal dose or concentration | LD50 (rat, oral): 68 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 68.1 mg/kg |
| NIOSH | NT1225000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 300 mg/kg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Phenylselenol Phenylseleninic acid Selenium dioxide |

