The Story of Dipentyl Phosphate: More Than Another Chemical
The Turns in Historical Development
Chemistry always surprises those who look close. Dipentyl phosphate, once a silent lab product, now nudges its way into a range of industries. Years ago, most talk around phosphate esters focused on greases and detergents, but over time, researchers realized that swapping out short alkyl chains for bulkier ones brought fresh results. In the 1960s, laboratories chasing better plasticizers and safer flame retardants began prodding at molecules like dipentyl phosphate. While some people shrugged at its modest origins, the attention from polymer researchers and process engineers grew, thanks to the compound’s stable behavior and satisfying performance during demanding applications.
Getting a Handle on the Product
Anyone who’s dealt with esters knows they seldom keep still; they find new uses each year. Dipentyl phosphate, built on a backbone of five-carbon chains connected to a phosphate group, falls into this evolving group. The chemical wins favor in places where stability under heat and exposure to strong acids or bases becomes essential. Compared to smaller esters, its pentyl arms deliver a mix of moderate solubility and respectable resistance to hydrolysis, letting it blend into tougher conditions without falling apart. No giant headline announces its entry, but makers of specialty lubricants, plastic additives, and even antistatic agents keep a keen eye on consistency, and that’s where dipentyl phosphate usually holds its ground.
Physical and Chemical Properties Unpacked
Look at a sample of dipentyl phosphate. The liquid pours out a clear but slightly viscous stream, usually colorless or with a faint yellow tint. One sniff catches a sharp, reminiscent tang found in other phosphate esters. Water doesn’t tempt it much — the longer alkyl chains mean low water solubility. The boiling point heads up past 300°C, and the flash point lands beyond what base solvents ever reach, which grants users some breathing room when handling or blending. Chemists appreciate its lower volatility in comparison to lighter analogs, cutting down unnecessary losses during processing. This same resistance to evaporation lets it stick around longer in high-temperature formulations. Still, as with most organophosphates, dipentyl phosphate does not shrug off strong oxidizing agents or sustained alkali treatment without breaking down.
Technical Specifications and Labeling in Practice
Anyone working with dipentyl phosphate has probably sifted through technical sheets looking for purity, acid value, and residual alcohol content. Customers want to see assays above 95 percent. Color may sound cosmetic, but in specialized films or fluids, even slight tinting signals batch variation. Safety labels focus on irritancy and potential skin hazards, never a surprise where phosphates turn up. Tracking batch numbers and dating each container offers insurance if any recalls hover on the horizon. Strong labeling becomes the last defense against confusion — even for skilled operators who cut corners on routine checks in tight production schedules.
From Bench to Bulk: The Preparation Method
Those years tinkering at the bench hammer this point home: synthesis matters just as much as sales claims. Standard lab routes for dipentyl phosphate start with phosphorus oxychloride, chased with pentanol under basic conditions, and finished with hydrolysis. Each step has its signature quirks. Getting the alcohol-to-phosphorus ratio right prevents leftover reactants, which can spoil finished batches and spark waste issues. Reaction temperatures stay moderate — too high, byproducts bloom, too low, yields drop. Continuous processes now edge into scale-up, as steady demand for electrostatic control agents and biodegradable additives rises. Waste handling trails every preparation step, because downstream customers increasingly raise an eyebrow at byproduct footprints.
Chemical Reactions and Simple Modifications
Anyone chasing modified profiles experiments on basic phosphate esters. In the right hands, dipentyl phosphate tacks on to alkyl or aryl groups for tailored solubility or new flame retardant properties. Heating it with longer alcohols nudges it into a hybrid, while acid chlorides turn it toward other phosphoric esters. Under certain catalysts, transesterification swaps alkyl side chains with surprising ease. Each twist brings both promise and headaches — mixed esters don’t always play nice in standardized testing. But for patent hunters and materials scientists, these variations unlock fresh business.
Synonyms and Commercial Product Names
Every industry seems to have its private dictionary. Dipentyl phosphate often turns up as pentyl phosphate, dipentoxy phosphate, or in more formal literature as phosphoric acid, dipentyl ester. Trade names rarely stick since the substance often disappears into blends or proprietary mixes. End users sometimes call it by catalog number or in shorthand, keeping the precise name only for regulatory or shipping purposes.
Safety Habits and Operational Standards
It doesn’t take long handling organophosphates to figure out safety means more than thick gloves. Few accidents chime in with flashing alarms — most creep up. Dipentyl phosphate calls for goggles, ventilation, and sensible chemical resistant clothing. Spills leave a slick stain unless wiped quickly. If skin makes contact, prompt rinsing knocks down irritation. The compound doesn’t drive acute systemic toxicity in healthy adults at manageable concentrations, but one shouldn’t let routine dull judgment. Stockpiling safety data sheets, running periodic air quality checks, and scheduling refresher training help everyone stay alert. Conversations around new safety guidelines take time and patience, but these discussions become the backbone of zero-incident workplaces, especially when so many custom blends rely on common raw chemicals behaving as expected.
Areas Where Dipentyl Phosphate Gets Work Done
Break open product catalogs in manufacturing, electronics, or plastics: dipentyl phosphate keeps popping up. In plasticizers, it softens PVC without weakening heat resistance. Power cable insulation specialists use it to manage static and stretching issues during long service life. In industrial lubricants, the compound dials up flame resistance and improves long-term stability at the cost of a bit more effort during blending. Some researchers nudge it into the world of niche detergents and cleaning fluids, chasing lower foam and milder residue. Many of these uses travel under the radar, never headlining glossy trade fairs, but this chemical finds steady work supporting other substances. What often gets overlooked is its quiet success in research formulations: its resistance to hydrolysis makes it a candidate for outdoor use, where rain, moisture, and UV light break down weaker esters.
Push for Research and Continuous Development
Labs everywhere trade information on the next tweak or improvement, and dipentyl phosphate remains a lively subject. Researchers now look beyond traditional uses, eyeing performance under stress, biodegradability of byproducts, or compatibility with bio-derived plastics. Regulators sharpen their focus on downstream effects, kicking off deeper toxicity and environmental fate studies. Meanwhile, the push to minimize hazardous side products sends production teams back to the drawing board, refining batch sizes and exploring greener alternatives. Patent records fill with ideas for blending it into fire-resistant coatings or eco-friendly plasticizers. These cycles of invention and revision guarantee that knowledge around this molecule stays fresh and adaptable.
What Toxicity Studies Teach
Skepticism leads my approach here because claims outpace data in countless chemical catalogs. Toxicity studies on dipentyl phosphate remain thin compared to more notorious organophosphates, but available data points to mild irritancy without high systemic harm at standard exposures. Researchers track biodegradation in water and soil, noting that longer-chain esters like this one resist fast breakdown but don't persist as menacingly as some heavier industrial contaminants. No one expects this chemical to replace ultra-safe lubricants any time soon, but its toxicity profile matters more as it enters contact with everyday products. Until deeper, more transparent studies stack up, prudence still guides most lab and plant operations.
Thinking Forward: Prospects and Pitfalls
Market needs rarely stay static. The ongoing campaign for safer, greener, and more flexible chemicals pushes dipentyl phosphate into the spotlight. Material scientists see space for improvement — better synthesis routes, safer blending, smarter lifecycle tracking. As hybrid plastics and electronics evolve, the demand for high-performing, low-toxicity additives grows. Still, calls for more research should outpace shortcuts; no one benefits from rushing untested blends into mass circulation. Through collaboration among industry, academia, and regulators, future applications may stretch far beyond what the inventors of phosphate esters ever pictured. Keeping a level head, sharing new data, and listening to small but real user feedback will shape the next chapter for dipentyl phosphate, just as it has for many behind-the-scenes specialty chemicals.
What is Dipentyl Phosphate used for?
Where You’ll Find It
Walk through a chemistry lab or look into the world of industrial processing, and you might spot dipentyl phosphate on a list of ingredients. This compound, rooted in the world of organophosphates, shows up in places that don’t usually make headlines. Its primary use revolves around extraction and separation, especially for metals. Refining rare earth metals or separating uranium from phosphates — that’s where this compound comes into play.
Why Industries Care About Dipentyl Phosphate
Experience shows that real life problems rarely have glamorous solutions. Mining and metallurgy might lack the polish of tech, but they build the gadgets we love. Separating tiny amounts of useful metals from mountains of waste becomes ever more important as the demand for energy storage, computers, and green tech soars. Dipentyl phosphate steps in here. People use it as an extraction agent, helping sort out the material people actually want from all that mess.
Energy generation, for example, owes a debt to chemicals like dipentyl phosphate. Uranium extraction for nuclear power plants depends on complex methods where organophosphates act as the middleman, latching onto uranium and leaving impurities behind. The whole industry relies on these processes to keep fuel clean and costs low.
Everyday Impact: Why It Matters
Folks rarely think about what goes into their lithium-ion batteries, their electric vehicles, or even their computers. But every piece of hardware rests on a complicated supply chain, stretching from ore deep underground to sleek consumer goods. Critical metals like rare earth elements, cobalt, nickel — processing these sustainably continues to challenge everyone from miners to governments.
Chemists and engineers reach for dipentyl phosphate not just for extraction power. They turn to it for selectivity. Materials recovery depends on picking out exactly what you want and leaving the rest. That’s no easy feat at scale, and mistakes lead to lost resources or waste that harms both pocketbooks and the environment.
Safety, Environment, and Transparency
No industrial chemical escapes scrutiny these days. Handling and disposal of organophosphates draw attention for good reason. Long-term exposure can impact workers’ health and pose a risk to waterways if not controlled. Governments and watchdog groups keep a close eye on chemical processing facilities for signs of sloppy practices.
The World Health Organization and EPA publish clear guidelines for allowable limits and workplace exposure. Responsible firms treat those standards not as an obstacle, but as a baseline for doing the right thing. Companies adopting new separation methods, waste recovery, and closed-loop cycles shrink the risk of pollution.
From my own discussions with engineers, the best results come when the people on the ground stay engaged, question old habits, and push toward safer substitutes or better containment systems. Engineers remember how improper disposal in the 20th century led to lengthy cleanups and lost public trust. That memory keeps many in the industry honest.
Looking Ahead: Better Recovery, Less Waste
Clean tech rests on reliable access to rare materials. Dipentyl phosphate and similar agents make recovery possible, but demand for even greener, safer alternatives keeps growing. Research teams around the world have begun looking for bio-based or less hazardous chemicals to do the same job with less risk.
People care not just about efficient extraction — they want to see companies taking real steps to minimize harm. Responsible sourcing, transparency, and genuine stewardship turn what used to look like a shadowy corner of industry into something people can support.
Is Dipentyl Phosphate hazardous to health?
Understanding Dipentyl Phosphate
The chemical world is full of obscure names, and dipentyl phosphate sounds like one of those under-the-radar substances. It often finds its place in labs, industry, and sometimes in products many people never notice. Digging into whether this stuff poses a threat to health doesn’t require scouring complicated scientific papers—sometimes, common sense backed by solid research goes a long way.
How People Come Into Contact
Workers in facilities using dipentyl phosphate for making specialty chemicals might run into it. Most folks outside a lab won’t deal with it directly. Still, workers mixing, transferring, or cleaning up after production get the closest to this chemical. That’s where the biggest risk sits. Breathing in fumes, getting dust on the skin, or an accident with a concentrated liquid brings questions about safety to the front row.
What the Research Tells Us
Scientific research gives clues about risk. Researchers say that dipentyl phosphate can irritate skin and eyes. Short-term exposure might mean redness or a burning feeling. Breathing in vapors in high concentration can irritate the airways. Some animal studies brought up concerns about effects on organs when the dose is high enough—usually, that's much more than a typical worker would face with proper controls.
One wider concern with phosphate compounds, including dipentyl phosphate, is their impact when released into water or soil. They can, in some cases, stress aquatic life. Regulations push companies to control emissions and waste to avoid environmental damage, because pollution can cycle back to affect people in food and water.
Workplace Safety and Exposure Limits
Big institutions like the European Chemicals Agency and the U.S. Environmental Protection Agency keep tabs on chemicals like dipentyl phosphate. So far, there’s not a flood of official risk data. That doesn’t mean there's no risk; it means most use cases have good controls in place already, and confirmed health crises haven’t been widely reported. In my experience visiting a chemical plant, glove use and proper ventilation left little room for real exposure. Training made sure no one skipped those steps because even a rare incident has big consequences.
What Can Be Done to Reduce Risk
Companies using dipentyl phosphate have the most power to keep people safe. Engineering controls—like using closed systems and local exhaust—make a big difference. Personal protective equipment (PPE) such as gloves, goggles, and masks add another layer. Good safety data sheets spell out exact steps for cleaning up spills and responding to exposures.
Transparency is just as important as engineering. On my last project near an industrial lab, open communication between safety officers, management, and workers meant faster fixes and fewer accidents. Workers who know what chemicals do—supported by regular, real-world training—spot problems early. Companies that own up to the chemicals they use build trust with nearby communities, not just regulators.
Families living beside plants have a right to know what comes out of the facility. Public reporting and environmental monitoring hold companies accountable. Communities that push for environmental protection and regular health checks for workers set a higher bar for safety and help catch problems before they spiral.
Putting It All Together
Dipentyl phosphate doesn’t act like a household chemical, and it shouldn't end up in situations where untrained people handle it. At high exposure, like many industrial chemicals, it can cause harm. Strong safety culture, up-to-date training, and transparent reporting turn those risks into manageable, well-controlled elements of modern manufacturing. The goal can't be to ban every chemical with a warning label, but to keep people out of harm’s way with practical, proven steps.
What is the chemical formula of Dipentyl Phosphate?
Why the Formula Matters
Chemistry isn’t just a field for those in lab coats hunched over beakers. The formulas found in everyday products carry real meaning, and dipentyl phosphate stands as a good example. Its formula, C10H23O4P, pops up in research, industry documents, and regulatory filings for good reason. Every element and bond in this formula comes with history, know-how, and quite a bit of scrutiny, especially as safety and environmental regulations tighten across industries.
The Building Blocks: C10H23O4P
Think about what makes up this molecule. With ten carbons, twenty-three hydrogens, four oxygens, and one phosphorus atom, dipentyl phosphate leaves a distinct signature. Two pentyl (C5H11) groups attach to a phosphate backbone. This structure isn’t just a curiosity—it gives the molecule its function and hazard profile.
These molecular features lead to dipentyl phosphate showing up in specific niche settings. For instance, it finds a place as a plasticizer and in specialized extraction processes, particularly as a reagent for separating metals in hydrometallurgy. Some folks in chemical engineering know this formula from hands-on troubleshooting, figuring out how this compound interacts with other chemicals, metals, and process equipment.
Importance in Chemical Manufacturing and Safety
Manufacturers and safety professionals watch this formula because of what it brings to the production floor. Dipentyl phosphate, like other organophosphate esters, requires careful handling. Organophosphates get flagged for their toxicity in some contexts—think pesticides or nerve agents—though dipentyl phosphate is milder in its effects. Knowing precisely what makes up the formula allows professionals to evaluate risks, set safe exposure limits, and design proper containment systems.
Experience in plant operation teaches that missing a detail in a chemical formula can lead to costly accidents or failed compliance audits. For example, the four oxygen atoms don’t simply stick around for show; they contribute to the molecule’s polarity, which affects everything from solubility to how a spill behaves on the shop floor. Phosphorus linking both pentyl groups means a spill can smell, and it attracts close regulatory attention due to environmental persistence. Not only does the formula guide responses in emergencies, but it also shapes day-to-day operational protocols.
Environmental Considerations and Practical Solutions
Attention keeps turning to organophosphates in waste streams, and dipentyl phosphate is not immune from that scrutiny. Its persistence in aquatic environments raises flags for toxicologists and wastewater treatment engineers. Having an exact chemical formula lets scientists build accurate models for predicting breakdown, movement, and long-term effects. I’ve seen firsthand how regulatory inspectors comb through process records, double-checking formulations to ensure that every molecule gets properly accounted for before discharge.
There’s no quick fix for the challenges these compounds bring. Yet, practical solutions often start with precise formulas. Closed-loop handling, better wastewater treatment using advanced oxidation, or even switching feedstocks where possible all start from knowing what's in the drum. The push for greener chemistry means chemists now build new molecules, aiming for similar results but with safer or more biodegradable structures. Lessons from compounds like dipentyl phosphate show the demand for innovation, but without glossing over legacy chemicals that still move through our supply chains.
The Role of Transparency and Accountability
Trust in chemical processes—whether in a small lab or a massive production line—rests on open, accurate sharing of information. Dipentyl phosphate’s formula serves as a foundation for building that trust. This detail is not just for regulatory forms. It helps guide safer alternatives, informs emergency response, and ensures that employees, neighbors, and consumers all know what’s in the products around them.
As industries strive to balance productivity with responsibility, formulas such as C10H23O4P act as reminders of the need for transparency. Decisions about chemicals, especially those with complex legacies, demand thorough understanding and straightforward communication at every step.
How should Dipentyl Phosphate be stored?
Understanding the Material
Dipentyl phosphate’s name might not pop up outside lab circles, but anyone who handles chemicals knows that each substance brings its own set of hazards and quirks. Many phosphates can leach or react, and people in the chemical field have seen what happens when storage gets sloppy—corroded shelves, warped labels, and the sickly tang in the air reminding everyone that not all control comes down to a locked cabinet. It pays to treat even the “routine” chemicals with some respect.
Keeping It Cool (and Dry)
Taking cues from material data sheets and industry best practices helps protect people and property. Dipentyl phosphate wants a cool, dry spot. Heat and moisture start trouble. A heated app room means pressure from flashes of vapor, or spilled liquid turning volatile faster than expected. Damp storage areas might seem harmless at first. Give moisture a way in, corrosion and container breakdown start quickly. Some labs run cheap hygrometers just to signal early signs of rising humidity.
A dry shelf with decent airflow handles most liquid chemicals well. Don’t get tempted to store it above eye level; accidents happen whenever a jug tips and you’re standing right where gravity wants it. Chemicals often get parked high up to save bench space or keep things “out of the way,” but most injuries start with one misstep or rushed afternoon. Old-timers put dangerous bottles closer to waist height, right where spills are easiest to contain.
Container Choice and Labeling
Plastic or glass usually gets the job done. Pick containers rated for acids or corrosives. Some older labs stick with thick-walled glass jars, but plastics, like high-density polyethylene, block leaks and won't shatter as easily during a drop. People sometimes forget, or even ignore faded labels, but there’s no faster way to invite confusion or mistakes. Mark the container clearly with the full name and date received or opened. Resisting the urge to abbreviate or skip labeling keeps everyone safer, including folks who sort stock months later.
Separation and Safety
Every safe storage setup separates acids from bases, oxidizers from reducers, and flammables away from sparks and heat. Dipentyl phosphate shares traits with other organophosphates; mixing by accident leads to reactions that nobody wants inside a crowded room. Acids stored near incompatible substances reach across shelves, and vapors combine in the air even if bottles never touch. Storing it away from strong bases, oxidizers, or sources of ignition takes little effort and pays off every day.
Bigger storerooms sit behind fire-rated doors with spill trays underneath, but even in small labs a simple polypropylene tray under the bottle catches everything—small leaks, vapor, or a sudden break during a rush.
Regular Checks and Community Knowledge
Forgetfulness creeps in after years of routine. Weekly walk-throughs catch frosting, cracking, swelling, or odd smells—subtle signs of trouble most folks miss on busy days. If the container looks off or the liquid seems cloudier than last week, it’s time to replace or neutralize it. Share what works and what goes wrong. Fellow lab users offer solutions not found in manuals—a trick for checking seals, a suggestion for shifting inventory when space gets tight.
Knowledge and good habits steer clear of the classic slip-ups. Established storage rules, combined with common sense and steady routines, keep both veteran chemists and beginners safe and productive.
What are the safety precautions when handling Dipentyl Phosphate?
Recognizing Real Risks
Working in a lab brings back memories of seeing bottles labeled “Dipentyl Phosphate” on the shelf. Friends who deal with chemicals daily often talk about hazards that get ignored because they don't seem "dramatic." Dipentyl Phosphate slides right into that category. It looks safe enough—clear, not especially smelly, not fuming—but ignoring safety routines invites trouble. Skin irritation, serious eye injuries, and unexpected inhalation can happen with one careless move.
Personal Protection Isn’t Optional
Splash goggles and gloves always hang at my station, and with a chemical like this, that habit pays off. The liquid can sneak right through thin latex gloves if you aren’t careful, so those thick nitrile ones make a real difference. Forgetting lab coats or rolling up sleeves feels tempting on hot days, but then a spill turns a minor task into a bigger deal. Safety eyewear becomes non-negotiable; a tiny splash in the wrong spot can mean an emergency eye-wash sprint.
Ventilation Matters a Lot
An old professor used to crack a window instead of relying on the fume hood, but the smell sometimes stuck around. Dipentyl Phosphate doesn’t always announce itself with an obvious odor, so the fume hood has become my go-to. Proper ventilation gets overlooked as a small detail, yet a good airflow turns what could be a risky job into something routine. It also helps with long-term safety; exposure over time often creeps up, and proper air movement keeps concentrations lower.
No Shortcuts in Cleanup
Spills don’t wait for a good time to happen. There’s a temptation to wipe and move on, but that quick fix spreads residue to unexpected places. Absorbent pads, followed by neutralizing agents, save time in the long run. I saw someone mop up a spill with paper towels, forget to wash up, and then rub an eye—minutes later, it took the nurse and an eyewash station to set things straight. Rinsing affected skin with plenty of water, then washing with soap, goes a long way.
Labeling and Storage
Mislabeled bottles cause chaos. In one case, a mix-up delayed an experiment and left everyone wondering if invisible contamination happened. Permanent labels and strong seals help. Locking corrosive chemicals in a ventilated cabinet, far from heat or sunlight, keeps both people and materials safer. Having the Safety Data Sheet (SDS) right nearby isn’t just a formality—digging through computer files in an emergency takes longer than flipping through a binder.
Training Turns Caution into Habit
Hearing stories about accidents changes nothing if people shrug them off. Regular training, with real stories and hands-on demos, sticks with most folks. It all comes down to making safety a routine. Taking the time to review procedures and actually practicing how to use an eyewash or respond to a splash sharpens awareness, and keeps incidents from becoming disasters. No one plans for mistakes, but planning for a response always makes a difference.
Stronger Safety Pays Off
Chemicals like Dipentyl Phosphate don’t get headlines for spectacular accidents, but the little steps—protective gear, clean storage, clear labels—stack up. Solid habits make the difference between a close call and a trip to the ER.
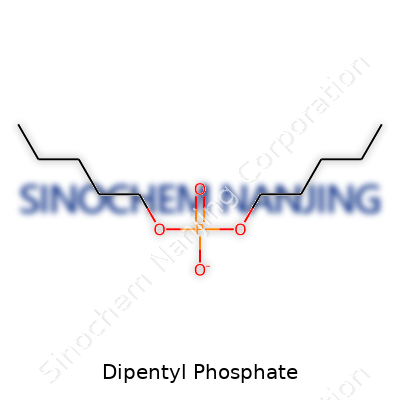
| Names | |
| Preferred IUPAC name | Bis(pentyl) hydrogen phosphate |
| Other names |
Phosphoric acid, dipentyl ester Dipentyl hydrogen phosphate Dipentylphosphate Dipentylphosphorsäure Pentyl phosphate |
| Pronunciation | /daɪˈpɛn.tɪl fəˈsfeɪt/ |
| Identifiers | |
| CAS Number | 2528-36-1 |
| Beilstein Reference | 1912438 |
| ChEBI | CHEBI:37958 |
| ChEMBL | CHEMBL490538 |
| ChemSpider | 21711 |
| DrugBank | DB08307 |
| ECHA InfoCard | 05b37ea7-635f-4d01-b1c5-7cf7177c8aa8 |
| EC Number | 213-997-4 |
| Gmelin Reference | 74363 |
| KEGG | C18254 |
| MeSH | D006701 |
| PubChem CID | 12421 |
| RTECS number | TH1050000 |
| UNII | E1U2L7K91I |
| UN number | UN2920 |
| Properties | |
| Chemical formula | C10H23O4P |
| Molar mass | 266.33 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Odorless |
| Density | 1.015 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 0.89 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.37 |
| Basicity (pKb) | 1.77 |
| Magnetic susceptibility (χ) | -78.8·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.438 |
| Viscosity | Viscosity: 15 mPa·s (20 °C) |
| Dipole moment | 3.59 D |
| Thermochemistry | |
| Std enthalpy of formation (ΔfH⦵298) | -1086.8 kJ/mol |
| Pharmacology | |
| ATC code | D08AX06 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | P280, P305+P351+P338, P310 |
| Flash point | Flash point: 113 °C |
| Autoignition temperature | Autoignition temperature: 400 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 3,600 mg/kg |
| NIOSH | Not established |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 500 mg/L |
| Related compounds | |
| Related compounds |
Methyl phosphate Dimethyl phosphate Diethyl phosphate Dibutyl phosphate Dioctyl phosphate |

