Dinitroresorcinol: Lessons from a Complicated Compound
Historical Development
Ever since scientists began tinkering with aromatic compounds in the late 1800s, dinitroresorcinol has sparked curiosity and respect in the lab. Chemistry students often run into dinitroresorcinol during advanced coursework—usually with a dose of caution from faculty, since pioneers like Hofmann, Hepp, and others queued up early syntheses and pointed to its stubborn yet fascinating chemistry. The nitro group, with its unpredictable punch and bright hues, drew explosives experts and dye makers alike over the years. Anyone digging through chemical archives will see how industrial revolutions made materials like dinitroresorcinol an attractive, if sometimes dangerous, proposition. The compound’s notoriety among classic nitroaromatics continues to color its reputation.
Product Overview, Physical & Chemical Properties
If you’ve held dinitroresorcinol in your hand, you may remember its yellow-to-orange appearance, but most scientists avoid open handling. At the molecular level, this compound packs a benzene ring dressed in two nitro groups and two hydroxy groups. Structurally, this pushes strong electron-withdrawing effects—which brings reactivity but also instability. Its melting point falls in a range showing moderate stability, but as heat increases, runaway decomposition may occur. Dinitroresorcinol dissolves poorly in cold water yet responds better to warm solvents, which influences how researchers approach extractions or reactions. Thanks to the nitro groups, its vibration spectra and color reactions prove useful for analytical chemists needing a marker for further studies.
Technical Specifications & Labeling
On a chemical bottle, dinitroresorcinol doesn’t need pageantry. The CAS number often sits front and center to avoid confusion, as synonyms pop up from different suppliers and national catalogs. Most professional labels warn about potential risks up front, from skin sensitivity to energetic decomposition. For storage, common sense and industry experience say keep it cool, dry, and far from reducing agents or sources of ignition. Each lot comes with data showing assay, moisture content, and relevant impurities—since one flawed batch could spell a ruined experiment or worse. Laboratories maintain careful records to comply with safety regimes, echoing lessons learned the hard way by past researchers.
Preparation Method
Remarkably, dinitroresorcinol owes its existence to the strong hands of nitration chemistry. Experienced hands combine resorcinol with concentrated nitric acid—sometimes with sulfuric acid lending a catalytic boost—under steady temperature control. Even so, literature shows that yields wander widely based on the nitro groups’ orientation or the temperature dialed in. Heated mixtures demand respect, not just for lab safety but for the fate of the product. Post-reaction, separation of the target compound from isomers and unwanted byproducts depends on patience, cooling, and a few rounds of crystallization. Looking at bench notes, achieving decent purity requires confidence in every wash, not just a recipe from an old textbook.
Chemical Reactions & Modifications
Dinitroresorcinol keeps surprising chemists willing to explore its reactivity. The molecule’s hydroxy groups open doors for etherification or esterification, while the nitro groups set the stage for reduction to amines. This allows tailored modifications for new functional materials, although caution remains constant—sometimes a bright idea in the lab turns hazardous if the nitro groups are stirred too briskly or heated too long. Those who favor organic synthesis know that subtle tweaks in conditions can flip reactivity, sometimes producing useful intermediates for pharmaceuticals, sometimes causing a mess best left to the fume hood. Attempts at metal complexation or oxidation produce enough variety to feed doctoral theses, each step tested one variable at a time for safety and yield.
Synonyms & Product Names
Anyone leafing through catalogs will notice dinitroresorcinol masquerades under several aliases, not unlike members of classic detective novels. Common names in the chemical literature include 2,4-dinitroresorcinol and DNRes. Cautious chemists always check CAS numbers to avoid ordering the wrong position isomer or comparable nitrophenol, since one vendor’s branding may conjure confusion for another. Products from different decades or languages often cross-reference older nomenclature like 2,4-dinitro-1,3-benzenediol, highlighting why product traceability matters in research and safety circles.
Safety & Operational Standards
Veterans of the chemical industry give dinitroresorcinol a berth measured in meters, not centimeters. With two nitro groups eager for reduction or decomposition, prudent lab operators keep quantities small and shields close. Typical rules restrict open heating, open flames, and mechanical shock near this compound for good reason—case reports reach back decades showing how minor mishandling led to major injuries. Gas masks and gloves aren’t window dressing, and ventilation systems get checked religiously. Even waste disposal runs on well-documented protocols, with local, national, and international regulations echoing stories from labs that ignored the dangers once. These operational standards do not change with fashion.
Application Area
Researchers still find niches for dinitroresorcinol in analytical chemistry, dye synthesis, and sometimes energetic materials research. Despite strict handling rules, the scientific appeal hasn’t faded—aromatic nitro compounds stand out for their color and reactivity. In my research group, a colleague tinkered with boron complex formation, hoping for new optical properties. Chemists also explore its transformations for tracing reaction mechanisms or as indicators for strong base solutions. Commercial uses shrink compared to the golden era of synthetic dyes, but universities and niche industries keep dinitroresorcinol on hand for targeted studies requiring distinct chromophores or reactive centers. Each project weighs the benefits against real risks.
Research & Development
University labs and industrial R&D clusters still turn to dinitroresorcinol for unlocking questions about aromatic substitution, nitro group migration, or new coupling reactions. Complexation studies, especially with transition metals, keep cropping up in journals, confirming that chemists crave both knowns and unknowns. Instrumental methods continue to refine how researchers detect minuscule trace levels in pharmaceutical intermediates. Modern R&D, though, puts more muscle into green chemistry and sustainability; I’ve seen younger researchers searching for ways to redesign the nitration process to cut down acid waste or minimize byproducts. These advances come from labs willing to take small, careful steps—acknowledging the weight of past mistakes.
Toxicity Research
To call dinitroresorcinol a mild irritant misses the point. Studies dating back decades warn of acute toxicity for skin and eyes, and ingestion sits off the table for obvious reasons. Animal studies and in-vitro research describe disruption in enzymatic pathways, with some reports noting methemoglobinemia after exposure. Plant studies show that even small environmental leaks impact growth, so environmental science tags dinitroresorcinol for strict controls. Having worked with similar nitroaromatics, I understand the draw of these compounds for advanced synthesis, but the mounting evidence around mutagenic potential keeps regulatory agencies interested. Modern toxicology now checks for chronic, subtle effects, not just acute thresholds.
Future Prospects
Opportunities for dinitroresorcinol in the years ahead look like a contest between caution and curiosity. On one hand, the growth in organic electronics and materials could carve out new uses for its color and reactive core structure. On the other, chemical safety regulators now scrutinize every handling and disposal step, driving demand for safer alternatives. Green chemistry holds promise for cleaner synthesis, though progress is slow due to inherent hazards. Universities continue to fold lessons from dinitroresorcinol into advanced curricula, highlighting both fascinating chemical behavior and the reality that every discovery carries risk. The next breakthroughs may come from teams committed to transparency, safe innovation, and learning from the old rules instead of rewriting them out of convenience.
What is Dinitroresorcinol used for?
From Factory Floors to Research Labs
Dinitroresorcinol doesn’t grab headlines like other industrial chemicals. It offers a story that stretches through medical labs, manufacturing sites, and even national defense. Most folks won’t see it on a store shelf, but it plays a role in projects that reach far beyond what meets the eye.
Behind the Scenes in Science
Walk into any university chemistry department and you'll find researchers looking for new ways to detect metal ions or track chemical reactions. Dinitroresorcinol often acts as an indicator in these labs. Drop it into a sample, watch the color shift, and suddenly trace amounts of metals like nickel or copper appear where detection once proved tricky. That tests lab skills and sharpens our understanding of what’s lurking in industrial waste or groundwater. Its reliability gives chemists a way to spot issues before they spiral into problems.
Serving Medicine and Diagnostics
Medical labs value compounds that help measure substances in blood, urine, or even pharmaceuticals. Dinitroresorcinol pops up in test kits built for this job. Years ago, when I worked under a hospital’s chemistry department, we leaned on color indicators to quickly estimate glucose and protein levels, guiding doctors who demanded answers fast. Manufacturers keep refining these tests, but the backbone stays the same—simple, effective color chemistry.
Propellants and Explosives: Risks and Realities
The defense industry gets complicated. Dinitroresorcinol can serve as a component for explosives and propellants. Safety teams tread carefully because its energy content means small mistakes have big consequences. If left unsecured, it threatens more than just property. Regulators keep close watch, and every ounce needs storing under strict rules. These controls reflect bitter lessons from the past, when oversight slipped and injuries followed. It reminds us that chemical innovation never travels far without its share of risk.
Challenges That Demand Respect
Handling dinitroresorcinol isn’t for the unprepared. I remember one project where we faced skin irritation and inhalation hazards. Just a splash left a bright stain and an itchy arm. Respirators and gloves went from annoying to mandatory that day. Accidents like those teach safety more clearly than any manual. Factories storing it in bulk constantly review emergency plans. Nothing ruins a productive shift faster than complacency around chemicals like this.
Looking Ahead: Balancing Use and Safety
People keep asking for safer, greener compounds that do the same jobs. Chemists answer by testing alternatives with fewer health risks or less environmental impact. Some universities have started sharing these advances to help small labs catch up and cut down on incidents. It won’t happen overnight, but teamwork between industry, academia, and regulators moves everyone closer to safer work.
Dinitroresorcinol probably won’t become a household name. Even so, its roles in science, health, and industry mean most of us have benefitted from its presence—usually without knowing. An informed approach to handling and replacing chemicals like this not only protects workers but also helps us steer toward a future where innovation and safety can live side by side.
What are the safety precautions when handling Dinitroresorcinol?
The Everyday Risks Sitting in the Lab
Dinitroresorcinol might look like just another chemical in a bottle, but there’s some real risk packed in its yellow crystalline powder. I remember the tang in the air the first time I walked by a bench after someone spilled a small amount. No mask, no gloves—he just froze, realizing what happened. No one feels invincible around this stuff for long.
Let’s talk about why folks give it respect: Dinitroresorcinol carries both toxic and explosive hazards. Even small exposures leave a mark. Swallowing or breathing its dust sends toxic compounds straight into the blood. It doesn’t take a lot for your body to face tremors, headaches, or more serious trouble. Fumes irritate lungs, while any skin that’s not protected turns red or starts to itch. This isn’t an academic warning—every year, people land in the emergency room because basic gear sat unused next to the chemical cabinet.
The Basics: Gear and Setup
A good pair of nitrile gloves, wrapped wrists, and an old lab coat—these keep drama low. I’ll never forget the sting from a single drop on my skin before I switched to goggles and a face shield. Big, clear lenses protect better than designer glasses. Even if you think you’re only opening a bottle, a lab coat and decent shoes beat shorts and sandals every single time.
Ventilation stands out as the unsung hero. Fume hoods suck up dangerous vapors and micro-particles before you breathe them in. The difference feels obvious on a busy bench: no headache, no tickle in the nose, just clean air. Labs that settle for windows or regular fans miss the point—tiny particles linger, and masks alone won’t filter them out.
Storage Isn’t Optional
The thing about Dinitroresorcinol—heat, sunlight, or a spark pushes it from calm to chaos. One careless toss into a regular trash can sparks more than just a small fire risk. Storage rules aren’t just legal red tape. Use containers with tight-fitting, non-reactive lids. Fireproof cabinets—set far away from acids, bases, or anything with potential for static electricity—matter a lot more than you think. I’ve seen ruined benches and firefighting foam sprayed across the floor by folks who took shortcuts.
Training Saves More Than Just Lab Equipment
Hands-on training beats online slideshows every time. Watching a demonstration, handling spill kits, and running mock evacuations—this sticks in memory much longer than reading instructions on the wall. Real drills shake off nerves and sharpen reactions. Inexperienced hands often grab water for a chemical fire, spreading the mess. Team practice builds the muscle memory to grab the right extinguisher or reach the safety shower before things get worse.
Disposal and Cleanup: No Shortcuts Here
Any leftover Dinitroresorcinol sticks around as a hazard. Pouring down the drain, even in tiny amounts, contaminates water and creates dangers for sewage workers. Professional chemical waste contractors might charge, but the peace of mind and community safety pay back every cent. Spill kits built for chemicals, not all-purpose ones, absorb and neutralize accidents fast. Bleach or detergents mix poorly with Dinitroresorcinol—stick to the recommended cleanup agents.
People sometimes overthink chemical safety or treat it as an afterthought. Gloves, goggles, ventilation, mindful storage, and practiced cleanup—these keep both newcomers and veterans safe. Each caution comes from a story, a mishap, or someone else’s close call. I’ve seen enough to know that a little preparation outclasses any rush to save time. Safety isn’t just a policy—it’s a habit that keeps the science going strong.
What is the chemical structure of Dinitroresorcinol?
Unpacking a Complex Phenol Compound
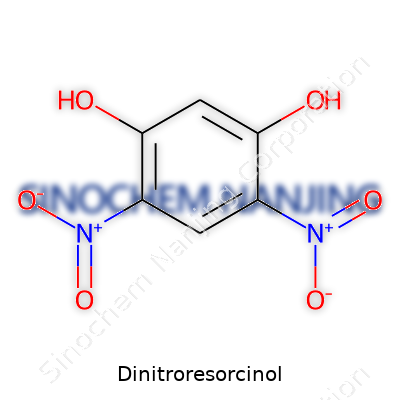
Dinitroresorcinol catches the attention in chemical discussions because of its tidy structure—and the fact that it comes with more than a whiff of risk thanks to those nitro groups. Draw it out, and you find a benzene ring dressed up with two nitro groups (–NO2) and two hydroxyl groups (–OH), typically found at positions 2 and 4 for the nitro groups and at 1 and 3 for the hydroxyls. You’ll see this structure called 2,4-dinitroresorcinol, a name that calls back to its roots as a derivative of resorcinol, which itself is simply 1,3-dihydroxybenzene.
Why Structure Matters in Real Life
In labs, chemists don’t look at these arrangements just for art. The tight placement of those nitro and hydroxy groups raises its reactivity. Nitro groups pull electrons away from the benzene ring, while hydroxyl groups push some back. That tug-of-war shapes the compound’s behavior, showing up in properties like solubility, acidity, and the way it responds when mixed with other chemicals. Handle dinitroresorcinol right, or watch it go from benign to hazardous; that's a lesson anyone in a synthesis lab learns fast.
Lessons from the Lab Bench
Every chemist I’ve spoken with keeps a wary eye on nitro-aromatics. Dinitroresorcinol underscores that caution. There’s a direct link between chemical structure and safety: nitro groups heighten both explosive risk and toxic potential. The molecule doesn’t just pose risks for fun, either. Industrial stories about nitro compounds—like TNT—show the sharp edge of this chemistry. Lab accidents have driven home that one stray spark or bit of friction around dry nitro compounds can bring real danger. That risk makes proper labeling, ventilation, PPE, and handling routines more than just red tape; testing, storage, and disposal all scale up in care when nitroresorcinol comes through the door.
Talking Facts, Not Fears
A look at published hazard data turns claims about toxicity and safety from stories to numbers. Dinitroresorcinol can cause skin and eye irritation, and inhalation or ingestion brings on symptoms ranging from headaches to more severe responses like methemoglobinemia, where oxygen delivery in blood gets impaired. Studies list LD50 values in ranges that spark respect for even small quantities. Notorious for environmental persistence, dinitro-compounds tend to stick around—something that keeps regulatory agencies monitoring their use and disposal closely.
Solutions That Count
While there’s never a magic fix for chemical risk, prevention pays. Substitution becomes the smartest starting point. Using alternatives with fewer nitro groups, or with lower environmental impact, keeps everyone safer. Training stands as another pillar; nothing replaces hands-on time learning the quirks of nitro compounds. Up-to-date safety data sheets and emergency protocols lower the odds of a mishap turning into a disaster. Waste reduction, like neutralizing residues and using closed systems, steers this story toward greener ends. Keeping labs and factories open by respecting the structure and potential of compounds like dinitroresorcinol is all about facing facts and choosing care over shortcuts.
Looking Forward in Chemistry
The structure of dinitroresorcinol offers a lesson: small tweaks in a molecule echo in both the lab and the world outside. Modern chemistry carries a responsibility to understand those ripples, blending curiosity with care. Lessons learned from handling this compound push the field forward, one well-studied molecule at a time.
How should Dinitroresorcinol be stored?
Understanding Dinitroresorcinol’s Nature
Dinitroresorcinol isn’t an everyday chemical you’ll find at the hardware store. Its bright yellow appearance can look almost harmless, but it comes with serious baggage. This compound poses real dangers—liable to explode if mistreated, potentially harmful if inhaled or absorbed, and even hazardous to the local environment if a leak gets out of control. I recall walking through a university chemical storeroom as a student, noticing unique labels and color codes for materials like this, a reminder that some things can't just sit on any old shelf.
Getting the Basics Right
Dinitroresorcinol always deserves respect, starting with where it gets stashed. Heat, friction, and even stray sparks create headaches. I learned from safety courses that chemicals of this type demand a cool, dry place. Direct sunlight ramps up temperature and increases risk. Rooms with steady, low humidity help fend off decomposition and prevent pressure from building up inside containers.
Secure, clearly marked cabinets made for explosives or reactive substances are the trusted locations. Locks keep curious hands away. These cabinets often line labs and storerooms where anything hazardous is present. People handling these supplies should double-check seals and packaging every time. If a cap feels loose or a seal looks worn, it’s time for a replacement—not a shrug.
Protecting People and Preventing Problems
There’s another rule that often gets hammered into new lab workers: separation. Dinitroresorcinol shouldn’t hang out with strong acids, alkalis, or anything easily oxidized. Placement away from flammable materials means if things go wrong, the fallout doesn’t multiply. Fire-resistant barriers help, offering extra minutes to respond during an emergency. Learning from industrial accidents in old case studies, chemical incompatibility often sets off the chain reactions that make headlines.
Inventory logs play a huge role, too. Trained staff keep accurate notes on arrival times, amounts, and package conditions. Outdated or unknown chemicals scare every safety officer. It pays to practice “first-in, first-out,” so what sits longest gets checked and removed before trouble starts. At my last lab job, quarterly safety audits caught several forgotten tins before they became a hazard.
Long-Term Safety Practices
Preparation goes beyond the storage room. Emergency equipment—fire extinguishers, spill kits, and personal protective gear—remains close by where Dinitroresorcinol lives. Workers stay ready for accidents by running drills, not just reading instructions. Response plans lie taped inside cabinet doors, so nobody scrambles for answers during a crisis.
Labels deserve a quick mention. Fact-based hazard notices, not generic warnings, let everyone know what’s in the box. I've seen good teams make checklists part of their routine, swinging by cabinets each week for fast inspections. Written guidance matters less if it doesn’t get reviewed, so hands-on training counts far more.
Solutions and Smarter Handling
Safe storage ties directly to good habits and honest culture. Shortcuts have no place in this business. Manufacturers can help by shipping Dinitroresorcinol in sturdy, leakproof, color-coded bottles with unmatched caps. Labs might consider installing ventilation systems in rooms housing high-risk chemicals, kicking out dangerous fumes before they create problems. Digital inventory controls can flag old stock and remind teams it’s time for a review.
Protecting health demands that everyone involved treats Dinitroresorcinol seriously, following clear rules forged by hard-won lessons, and refusing the temptation to make things easier at the cost of safety.
Is Dinitroresorcinol hazardous to health or the environment?
What Dinitroresorcinol Means for People
Most folks never handle dinitroresorcinol in daily life. Even so, this chemical finds its way into certain industrial labs. What’s the big concern? Dinitroresorcinol packs two nitro groups into a tight molecular ring, used as an intermediate for dyes or explosives. Anyone working near dinitroresorcinol faces a mix of real risks: skin burns, eye pain, and trouble breathing. Dust or residue drifting in the air doesn’t just irritate; it can harm. Lab accidents stick in my memory—friends who forgot gloves, or wiped sweat from their brow near the lab bench, sometimes paid the price with raw skin or itching eyes.
Science offers some data points. Inhalation or skin contact stirs up issues like dermatitis, headaches or dizziness. A 2022 occupational safety report from Germany flagged dinitroresorcinol among aromatic nitro compounds similar to dinitrophenol, raising red flags due to its toxic action in the body. If it gets past the skin or into the lungs, it can mess with blood oxygen levels, all without a strong smell to foreshadow trouble.
Environmental Red Flags
Industrial chemists know dinitro compounds feel stubborn in the environment. Bacteria don’t easily chew up these molecules. If dumped into rivers or soil, dinitroresorcinol lingers. Runoff after a leak can find its way into groundwater.
Few official studies directly measure dinitroresorcinol’s influence on wildlife, though the evidence from cousins like dinitrophenol makes it clear: aquatic life takes a hit. Fish and small critters show stunted growth or odd swimming if exposed even to low doses. In my hometown, local rivers bounced back eventually after a 1970s dye factory leak, but fishing never quite returned to old levels. Hard lessons stick; one careless chemical spill leaves scars on an ecosystem for decades.
Looking Toward Solutions
Safety gear always makes a difference. Gloves, splash goggles and fume hoods keep workers out of harm’s way. Clear training helps stop mistakes before they start. In 2019, I toured a university lab where students needed to scan a QR code before handling dinitro compounds. It forced a safety check. These little steps matter.
Beyond personal safety, companies need honest disposal routes. Incinerators designed for nitro residues do the job right. Old habits—dumping leftovers in open land—don’t cut it. Community watchdogs and environmental officers must keep tabs on factories and labs that buy dinitroresorcinol, and they do make a difference. In the Netherlands, routine inspections caught one plant skimping on waste treatment, stopping damage early.
Cleaner chemistry deserves more funding. Eco-friendly dyes or adhesives without persistent toxins sound like science fiction, but recent advances in green chemistry offer hope. Swapping hazardous intermediates for plant-based alternatives calls for investment, not talk.
Why Pay Attention?
Dinitroresorcinol does its job well in tightly controlled industrial settings, but health and environmental dangers travel quietly with it. I’ve seen what a single grain of a chemical can do to a finger, or a river, and it sticks with me. Respect for a substance like this only grows with experience. Safe habits, careful oversight, and investment in alternatives all stack up to keep trouble at bay.
| Names | |
| Preferred IUPAC name | 2,4-Dinitrobenzene-1,3-diol |
| Other names |
3,5-Dinitro-1,3-benzenediol 3,5-Dinitroresorcinol Etilon Dinitroresorcin |
| Pronunciation | /daɪˌnaɪtroʊrɪˈsɔːrsɪnɒl/ |
| Identifiers | |
| CAS Number | 603-39-0 |
| Beilstein Reference | 136507 |
| ChEBI | CHEBI:9267 |
| ChEMBL | CHEMBL55147 |
| ChemSpider | 13418 |
| DrugBank | DB02587 |
| ECHA InfoCard | ECHA InfoCard: 100.007.664 |
| EC Number | 208-096-2 |
| Gmelin Reference | 60795 |
| KEGG | C11177 |
| MeSH | Dinitroresorcinol MeSH: D017972 |
| PubChem CID | 9890 |
| RTECS number | DW3675000 |
| UNII | W7XZI6P76A |
| UN number | UN1320 |
| CompTox Dashboard (EPA) | DTXSID10123495 |
| Properties | |
| Chemical formula | C6H4N2O6 |
| Molar mass | 184.11 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.806 g/cm³ |
| Solubility in water | soluble |
| log P | 1.72 |
| Vapor pressure | 3.9 x 10^-7 mm Hg (25°C) |
| Acidity (pKa) | 3.0 |
| Basicity (pKb) | 7.48 |
| Magnetic susceptibility (χ) | -82.0·10⁻⁶ |
| Refractive index (nD) | 1.693 |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 216.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -92.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1141 kJ/mol |
| Pharmacology | |
| ATC code | D03AX03 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06 |
| Signal word | Danger |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | Precautionary statements: "P261, P280, P305+P351+P338, P309+P311 |
| NFPA 704 (fire diamond) | 1-3-2-Explosive |
| Flash point | 180°C |
| Autoignition temperature | 250°C |
| Lethal dose or concentration | LD50 (oral, rat): 160 mg/kg |
| LD50 (median dose) | LD50 (median dose): 240 mg/kg (rat, oral) |
| NIOSH | TT2975000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Dinitroresorcinol: Not established |
| REL (Recommended) | 10 mg |
| IDLH (Immediate danger) | IDLH: 20 mg/m3 |
| Related compounds | |
| Related compounds |
Resorcinol Styphnic acid Picric acid |

