Dimethylglyoxal: More Than a Chemical Footnote
A Journey Through Its Roots
If you’ve poked around in a chemistry lab or read up on obscure organic compounds, dimethylglyoxal might have caught your eye. It’s not a headline-stealer, but its past and present quietly shape more corners of industrial chemistry than people give it credit for. Tracing the arrival of dimethylglyoxal to lab benches, you can find roots tangled in late nineteenth-century organic synthesis. Lab pioneers first stumbled on it during experimentation with glyoxal derivatives, realizing fairly quickly that changing just a couple of methyl groups could open doors to new reactivity, especially in the dye and pharmaceutical fields. Its structure—two methyl groups perched next to a double carbonyl—started serving as a springboard for bigger molecular builds. As synthesis got more precise, chemists found more ways to tweak reaction conditions, getting cleaner yields or novel side products along the way.
Getting to Know the Stuff: Properties That Matter
Anyone who’s ever handled dimethylglyoxal knows you can’t mistake it in the lab. At room temperature, it likes to linger as a yellowish liquid, carrying a sharp, pungent odor reminiscent of vinegar and metal polish—the kind that clings to your gloves. Its formula, C4H6O2, wraps up a taut little molecule that doesn’t waste much space. Dimethylglyoxal dissolves readily in water and many organic solvents, and has a knack for picking up hydrogen bonds wherever it lands. These traits make it handy as a reagent, yet also make containment important. The double ketone arrangement means it reacts on command with nucleophiles, which makes it popular in the hands of synthetic chemists but also encourages caution whenever open flames or bases are nearby. It’s volatile enough for safety-minded folks to want the fume hood humming whenever bottles come out. Its reactivity drives not just curiosity but also practical applications, especially where precise changes to carbon skeletons are needed in organic synthesis.
How Do We Label and Measure Up?
Dimethylglyoxal doesn’t hide its quirks, so technical specs and labeling take center stage in safe handling. On a production label, purity often gets top billing—usually above 98 percent for professional use. Specific gravity, refractive index, and boiling range offer clues to confirm what’s in the bottle. Safety symbols also matter, warning against inhalation and skin contact, pointing to the low flash point and moderate toxicity profile. These markings aren’t decoration—they back up every bottle on the shelf, ensuring anyone pulling dimethylglyoxal for a procedure knows it warrants respect. Batch testing, whether the solvent is used for dye precursors or cross-linking agents in new polymer blends, takes its cues from these numbers. Labs that don’t cut corners here suffer the consequences: contamination, yield loss, or worse, an avoidable accident.
From Raw Material to Useful Compound: Making Dimethylglyoxal
No lengthy production chain here—dimethylglyoxal usually springs forth from the oxidation of its cousin, pinacol, or directly via controlled oxidation of mesityl oxide. Both synthetic routes depend on a decent oxidant (potassium permanganate, for example) and fine-tuned temperature controls. Here’s where things can get fussy: too warm or too much oxidant, and byproducts start showing up or yields fall through the floor. Some methods draw on air oxidation, making for a cleaner, greener story, but this introduces slowdowns unless carefully managed. For peering deeper into structure and side reactions, researchers turn to derivatization—playing with amines or alcohols—to coax out crystalline forms or build bridges to even more exotic molecules for study. Each tweak in the recipe echoes in the bottle—one batch’s impurity haunts the next synthesis down the line.
Believe the Reactivity Hype: Chemical Modifications
Dimethylglyoxal isn’t shy about joining reactions. Expose it to amines and imine products pop up without much coaxing. These imines, sometimes called Schiff bases, underpin countless chemical investigations and drive pharmaceutical intermediate production. Under alkaline conditions, aldol reactions become possible, letting chemists play molecular Lego and build bigger frameworks for dyes, pigments, or drug candidates. Methylglyoxal’s tautomeric flexibility also fuels isomerization studies—scientists have spent years mapping how this small molecule hops between different forms, influencing reactivity, stability, and even how it gets detected in complex mixtures. For me, it’s almost like molecular origami—open to endless folding and refolding in the right chemical hands.
The Many Names and Faces of Dimethylglyoxal
Chemists rarely stick to one nickname. Dimethylglyoxal may show up as biacetyl, 2,3-butanedione, or butanedione in the literature. Chefs unwittingly recognize it as the compound lending buttery smoothness to some foods—though the levels there remain far less concentrated than anything pulled from a chemical drum. This shape-shifting nature can confuse those new to handling “flavors and fragrances” chemicals, underscoring the importance of translating between IUPAC nomenclature and trade jargon. If you’re searching chemical catalogues, these synonyms help catch all relevant data, but it pays not to be caught off guard by an unfamiliar alias before cracking open a bottle.
Rules of the Game: Safety and Operations
Dimethylglyoxal doesn’t play nice if overlooked. Anyone handling it suits up with gloves, goggles, and lab coats, because direct skin contact stings or worse. Inhalation risks remain real—symptoms like sore throat, cough, and headaches can develop if ventilation slacks. Spills demand swift attention: absorbent pads plus proper disposal pipelines keep contaminants away from the environment and ensure compliance with local waste handling laws. Not every company enforces the same protocols, but in regulated settings, Safety Data Sheets (SDS) function as gospel, outlining required storage (cool, dry, away from acids and bases) and spill cleanup. As a volatile organic compound, it also draws scrutiny under atmospheric release regulations—too much vapor or careless drain disposal can prompt fines or audits.
Where the Chemistry Hits Home: Who Uses Dimethylglyoxal?
It shows up in more places than most people suspect. Beyond its famed food flavoring credentials—imparting that fake-butter zing to popcorn, margarine, or dairy-free spreads—dimethylglyoxal finds work in dye manufacturing, photosensitive film production, and as a cross-linking agent for tougher plastics and polymers. The pharmaceutical sector values its carbonyl duo in retrosynthetic planning, where introducing methylglyoxal derivatives can streamline assembly of target molecules. Analytical labs hold dimethylglyoxal up as a calibration standard, or use it to derivatize broader classes of reactive carbonyls for measurement and detection. My experience tagging along on facility tours showed me its reach: from paint factories in the Midwest to flavor houses in Northern Europe, this chemical connects dots you can’t always see from the outside.
Digging Deeper: Research, Development, and Toxicity
Academic curiosity about dimethylglyoxal hasn’t waned in decades. Early studies mapped out its spectral signatures—UV, IR, NMR—and these details now feed AI-driven chemical analysis tools. Toxicologists flag it for moderate risks; ingestion or contact with high levels damages tissue or disrupts cellular metabolism. Research in the ‘80s pushed for stricter safety levels in consumer products. Modern studies touch on its role as a biomarker in certain diseases (including diabetic complications), opening a window for clinical diagnostic tools. Green chemistry efforts target alternative synthesis that swap noxious solvents for safer, renewable feeds. Despite its long laboratory history, each year still delivers a new twist or discovery—from making more stable imine complexes to exploring antioxidant activity.
Gazing Down the Road: Possible Futures
Dimethylglyoxal’s future holds both promise and challenge. Expect tighter regulatory standards, especially in industries moving toward lower-emission or clean-label products. Analytical demands will climb, too, requiring purer product and more detailed traceability. With biotechnology advancing, fermentation-derived dimethylglyoxal could supplement or replace traditional petrochemical synthesis. If current research on antioxidant and diagnostic uses pans out, doctors and biochemists might soon rely on the molecule in ways that were unthinkable just a decade ago. For anyone interested in organic synthesis or industrial safety, keeping up with these shifts promises both headaches and breakthroughs. Dimethylglyoxal may not be the most famous molecule, but its legacy keeps evolving—sometimes quietly, sometimes at the center of tomorrow’s next chemical innovation.
What is Dimethylglyoxal used for?
What Is Dimethylglyoxal?
Dimethylglyoxal, a chemical with the formula C4H6O2, turns up in chemistry labs and industrial settings more than most folks realize. I remember bumping into the stuff during a college organic chemistry course, where its pungent, sharp smell stuck with me long after class ended. Ever since, its versatility has caught my attention, and it's worth understanding how it ends up touching more lives than you might think.
Uses in Industry
The first thing most chemists mention about dimethylglyoxal is its role as an intermediate. In manufacturing, it acts as a key building block when making certain dyes and resins. Through simple chemical reactions, it helps produce colors for everything from textiles to plastics. These products wind up in everything from t-shirts to electronics cases, giving color and structure where it’s needed. Dimethylglyoxal's ability to step into a reaction and deliver reliable results makes it appealing in industrial settings where consistency holds major value.
Research and Laboratory Work
Researchers appreciate dimethylglyoxal for its reactivity. It reacts readily with metals, so chemists use it in analytical chemistry to isolate or identify nickel, copper, and a handful of other metals in samples. During my internship in the university research lab, we put it to work detecting trace metals in groundwater. Its telling reaction often meant the difference between a safe water report and a red flag. Getting this data matters deeply in places where industry meets community, like towns near old mining sites or factories.
Pharmaceutical Potential
Interest around dimethylglyoxal doesn't stop with dyes and tests. Pharmaceutical researchers keep an eye on it as a building block for active ingredients. Chemical groups from dimethylglyoxal can be assembled into more complex molecules. The hope is to craft new antibiotics or drugs that target hard-to-reach pathogens. Drug development always takes patience, but every useful starting block helps.
One study found that some derivatives show antimicrobial properties, though more testing is needed before anything will hit pharmacy shelves. Still, exploration in this space could give a leg up against infections that no longer respond to standard treatments.
Safety Considerations
Handling dimethylglyoxal requires respect. Its sharp smell hints at its irritating effects on the skin, eyes, and lungs. The lab where I worked enforced strict safety rules, gloves and fume hoods included. The key is minimizing exposure and keeping clear labeling in busy environments. Some countries regulate its handling under chemical safety laws, so manufacturers and labs must track inventories and waste.
How to Push Forward
Responsible use and disposal should sit at the core of any operation using dimethylglyoxal. Simple steps like routine safety training and updating safety data sheets can avoid accidents. Investment in research focused on greener synthesis or safer substitutes could offer future benefits. Clear labeling and community outreach, particularly in areas with heavy industry, help address any local concerns.
The story of dimethylglyoxal underscores how even obscure chemicals shape modern life. Each application demands a careful balance of utility and responsibility. By staying informed and cautious, industries and researchers can use its strengths while keeping people and the environment protected.
Is Dimethylglyoxal safe to handle?
What Dimethylglyoxal Does in the Lab and Industry
Dimethylglyoxal shows up in organic chemistry labs, crop science, and sometimes production settings. I spent months studying reactions that used this tricky compound. Its sharp smell hits you as soon as you open a bottle, which always reminded me to approach with care. In my own experience, gloves and goggles aren’t optional – they’re the rule, and for good reason.
Health Risks with Direct Exposure
Direct contact with dimethylglyoxal brings problems. The chemical acts as a skin and respiratory irritant. Even vapors make the eyes water, and prolonged inhalation can inflame the airways. I saw a roommate handle it without a mask once; he coughed for hours and had to leave the room. Immediate skin contact often leads to redness, pain, or even blistering. If it splashes, it burns. All this trouble grows in poorly ventilated spaces.
Let’s not forget: the compound’s ability to form toxic by-products. Run-off or spills catch attention from environmental officers for a reason. Dimethylglyoxal’s reactions create more aggressive toxins and sometimes unstable intermediates, so a fume hood isn’t just fancy equipment — it keeps everybody out of the ER.
Regulations and Best Practices
Regulatory groups like OSHA and the EPA keep a cautious eye on chemicals similar to dimethylglyoxal. Official safety sheets spell out the protective equipment and protocols. I never worked anywhere that let people skip training, no matter how much chemistry they knew. Minimum rules: chemical-resistant gloves, lab coats, protective eyewear, and always some method to vent vapors outside. For clean-up, spill kits beat paper towels every time.
Labeling is not just for the sake of order. When you see a container marked with hazards, you think twice before moving it, and you won’t mistake it for something benign. I remember instructors repeating this lesson. One missed label landed a coworker with chemical burns.
Long-Term Hazards and the Bigger Picture
Long-term research into the effects of low-level dimethylglyoxal exposure suggests risks aren’t limited to acute burns or coughs. Repeated inhalation or contact probably raises cancer risk. Some animal studies point toward DNA damage with plenty of exposure. Now, few people off the street need to worry about this, but for lab techs, plant workers, or even students – this matters.
Managing Dimethylglyoxal Responsibly
There’s no need to panic, just a clear requirement for respect and care. Training and repetition do more to prevent accidents than fancy gear. I still remember my first day learning chemical safety, and how simple reminders kept us out of trouble: store away from heat, never smell directly, and clean up thoroughly after every session.
Waste disposal gets careful attention too. You don’t pour leftovers down the drain. Licensed hazardous waste handlers collect purpose-labeled containers for neutralization or incineration.
For anyone working with dimethylglyoxal, it boils down to education, vigilance, and respect for the substance’s power. That approach has protected me and my colleagues through countless projects, and hasn’t failed yet.
What is the chemical formula of Dimethylglyoxal?
Why Dimethylglyoxal Draws Interest
Dimethylglyoxal isn’t the sort of compound you hear about every day, but for chemical researchers and folks curious about organic molecules, it carries importance. The formula for dimethylglyoxal reads as C4H6O2. With two methyl groups attached to a glyoxal backbone, this compound is part of the vast world of diketones. At first glance, this formula looks straightforward, but a closer look reveals how small changes in structure can influence how a chemical behaves in different environments.
Looking at the Structure
With C4H6O2, dimethylglyoxal contains four carbon atoms, six hydrogens, and two oxygens. Its skeleton features two carbonyl (C=O) groups sitting next to each other, giving it the tag of a vicinal diketone. Each carbonyl has a methyl group hanging off it. It’s these attachments that make dimethylglyoxal distinct from its close cousin, methylglyoxal, which holds just one methyl group.
Why Structure Matters
The arrangement of atoms shapes how a molecule interacts with other substances. That matters in research—whether in organic synthesis or atmospheric chemistry. Diketones, for example, can act as intermediates in several chemical reactions. Chemists often rely on structural details to predict reactivity, toxicity, and solubility. The double carbonyl system in dimethylglyoxal influences how it binds with metals, breaks down in sunlight, or reacts with other small molecules.
Dimethylglyoxal in Practice
Delving into its uses, dimethylglyoxal doesn’t turn up in consumer products on a regular basis. Instead, it draws attention in laboratories, where researchers examine its reactivity and role in atmospheric samples. Diketones like dimethylglyoxal have shown up in studies focused on the breakdown of larger molecules and the formation of complex organic aerosols in the atmosphere. In these settings, knowing the exact chemical makeup helps scientists trace air quality issues, giving clues about pollution or climate impacts.
Challenges and Safety Notes
Working with dimethylglyoxal calls for careful handling. Like many small aldehydes and ketones, it carries risks. Inhaling vapors or coming into contact with skin may lead to irritation. It’s smart to use gloves, goggles, and work in well-ventilated spaces. These aren’t just busywork protocols—long-term exposure to reactive chemicals has produced health problems in lab workers before. Information about toxicity often comes from animal studies or case reports, so many scientists watch for updates on safety standards.
Looking Ahead
Making sense of why a formula matters extends beyond knowing the numbers. Dimethylglyoxal, with its C4H6O2 structure, opens the door to bigger conversations about pollution tracking, chemical manufacturing, and safety in the lab. Advances in analytical chemistry, like gas chromatography and mass spectrometry, have made it easier to identify compounds in air samples and industrial waste. Tools like these, combined with a solid understanding of molecular structure, help chemists track problems and evaluate cleaner alternatives.
Making Better Choices in Chemistry
Focusing on the building blocks, like dimethylglyoxal, reminds us that safety and environmental impact can’t be afterthoughts. Safer alternatives, improved ventilation, and updated guidelines play a role in responsible chemical use. Awareness of the formula is just one step; keeping people and communities safe builds a foundation for better science and a cleaner environment.
How should Dimethylglyoxal be stored?
Why Safe Storage of Dimethylglyoxal Deserves Attention
Dimethylglyoxal often pops up in labs and industrial settings. This chemical brings real risks. It’s flammable, breaks down into stuff that can harm you, and even in small amounts, it irritates the skin and eyes. Letting storage slide doesn’t only threaten health, but also dents the budget if accidents, leaks, or spoilage happen. In my time around chemical storerooms, I’ve seen how a little carelessness multiplies trouble. Nothing sharpens focus on storage standards like a near miss with a leaky bottle or a surprise vapor whiff.
Simple Steps, Serious Results
Storing dimethylglyoxal calls for more than tossing it on any old shelf. The label often reads “store in a cool, dry place, away from sources of ignition.” That’s not just red tape. Dimethylglyoxal catches fire under the wrong conditions. I once watched someone unthinkingly put it beside a window on a hot day—bad move. Direct sunlight pushes the temperature, and warmth plus vapors means danger. Better to pick a spot with steady cool temps. If you set up climate controls, you don’t just keep it safe, you cut down on accidents across the board.
Protect Your People and Property
The right container makes a world of difference. Glass bottles with secure, chemical-resistant tops keep vapors and liquids where they belong. Metal can react, especially with something as touchy as dimethylglyoxal—rust or chemical changes become a real headache. In shops where turnover is high, secondary containers lined with absorbent material trap leaks before they go from manageable to hazardous.
Personal experience tells me that clear labeling does more than tick a box for compliance. I’ve watched new staff reach for the wrong bottle more than once. Bright colors, simple warnings, and expiration dates foster smarter handling and storage. It’s easy to gloss over labeling as a boring detail—until someone’s hunting for an antidote instead of an explanation.
Plan for the Bumps in the Road
Even if everything else lines up, emergencies still happen. In the shops I’ve worked, investing in spill kits and fire extinguishers isn’t a nuisance — it keeps everyone working with peace of mind. A spill response plan saves panic or finger-pointing. Running drills helps everyone react fast. There’s no substitute for training—it stops injuries before they start and builds trust throughout a team.
Room for Improvement
One spot that gets overlooked is the paperwork. A chemical inventory, updated and double-checked, means you know what you have, what you need to get rid of, and where the risks hide. Connecting with local fire departments pays off, too. Sharing what’s on-site helps emergency crews come prepared if alarms sound.
Shelf life runs out fast with sensitive chemicals. Regular audits keep outdated material out of circulation. Safe disposal costs less than a full-blown incident. In my own work, these habits turn chemical storage from a guessing game to a solid foundation for safety.
What are the potential health hazards of Dimethylglyoxal?
What is Dimethylglyoxal?
Dimethylglyoxal isn’t a chemical you run into over the kitchen counter. This compound, used in labs and industrial settings, has properties that let it react with other substances to create products like pharmaceuticals or specialty chemicals. Folks working with Dimethylglyoxal usually know it’s a strong-smelling liquid, but the health hazards might not be obvious if you don’t spend much time with chemical safety data sheets.
The Direct Effects on Health
Whenever someone handles Dimethylglyoxal without gloves or eye protection, the risk starts before the first drop spills. Skin contact leads to irritation, redness, or a burning feeling after enough exposure. Breathing in vapors, even for a short period, results in a sore throat, coughing, or headaches. Longer exposure or a higher concentration could do even more harm to the nose, lungs, and possibly the central nervous system.
Spilled on the skin or splashed in the eyes, this stuff doesn’t just sting; it can cause damage. Eyes need immediate rinsing, sometimes even medical care if the pain or redness doesn’t settle. That’s something I learned firsthand years ago, working in a university research lab where one careless moment landed a colleague in the emergency room.
Looking at the Science
The scientific research over the past decade makes it clear that repeated or prolonged contact with Dimethylglyoxal could possibly sensitize skin, making later exposures even riskier. Animal studies show negative effects on organs when high doses are inhaled or ingested, with the liver and kidneys working overtime to clear it from the system. No one wants those organs burdened any more than necessary.
Clinical evidence from workplace monitoring shows that people exposed chronically to low levels often complain of headaches, nausea or fatigue. I’ve heard similar accounts from chemists and process technicians, who say poor ventilation turns a regular day’s work into a health hassle.
Why it Matters for Everyone
It’s not just people working directly with Dimethylglyoxal who need to understand the risks. Improper disposal or accidental leaks can send vapors into shared spaces, even outside lab environments. That’s brought concern among workplace safety experts, since safety culture often slips over time until a real emergency wakes everyone up. I’ve been around enough chemical plants to know that experience tends to lag behind good intentions.
Dimethylglyoxal reacts strongly with some acids and oxidizers, so accidental mixing isn’t just a paperwork issue. A small mistake could put whole shifts at risk. In situations where workplaces store dozens of reactive agents, even a well-organized lab can misplace a drum, especially during busy periods or staff turnover.
Reducing the Hazards
Solving these risks takes more than a page of safety instructions taped to the wall. Automated sensors and real-time air monitoring now play a bigger role than ever, catching accidents before workers develop symptoms. Training makes a difference, particularly for new staff who might never have seen a chemical like Dimethylglyoxal. Regular drills and honest reporting of close calls teach teams to recognize symptoms early.
Personal protective equipment—gloves, goggles, and good ventilation—still forms the backbone of daily defense. Health administrators now recommend medical surveillance for long-term workers, so issues get caught before they turn serious. Disposal rules have tightened, making sure even residue gets a full audit. From what I’ve seen, strong leadership and regular walk-throughs by experienced staff create a workplace where vigilance feels like the routine, not a nuisance.
Most folks outside the industry will never run into Dimethylglyoxal. Those who do should know it demands respect, not fear, and a steady hand when handling or cleaning up spills. A safe environment builds itself on more than policies; it grows from lived experience and mutual responsibility.
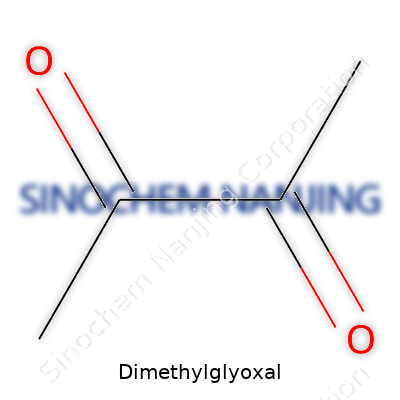
| Names | |
| Preferred IUPAC name | 2,3-dimethylpropanedial |
| Other names |
Biacetyl monoxime Dimethylglyoxime DMG |
| Pronunciation | /daɪˌmiːθəlɡlaɪˈɒksəl/ |
| Identifiers | |
| CAS Number | 600-14-6 |
| Beilstein Reference | 0112257 |
| ChEBI | CHEBI:17447 |
| ChEMBL | CHEMBL205486 |
| ChemSpider | 202789 |
| DrugBank | DB14073 |
| ECHA InfoCard | 03b8ce1d-1fef-4ce7-83ad-760c3b2266ab |
| EC Number | 205-049-5 |
| Gmelin Reference | 65734 |
| KEGG | C02802 |
| MeSH | D007944 |
| PubChem CID | 12541 |
| RTECS number | RR1925000 |
| UNII | GH90236BUX |
| UN number | UN2265 |
| Properties | |
| Chemical formula | C4H8O2 |
| Molar mass | 102.12 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Pungent |
| Density | 0.956 g/mL at 25 °C (lit.) |
| Solubility in water | Soluble |
| log P | -1.0 |
| Vapor pressure | 0.44 mmHg (at 25 °C) |
| Acidity (pKa) | 7.58 |
| Basicity (pKb) | 8.86 |
| Magnetic susceptibility (χ) | -33.3×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.394 |
| Viscosity | 1.77 mPa·s (25 °C) |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 296.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -285.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1045.3 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes severe skin burns and eye damage. May cause respiratory irritation. |
| Precautionary statements | P261, P264, P270, P271, P273, P301+P312, P304+P340, P305+P351+P338, P312, P330, P337+P313, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | 11 °C |
| Autoignition temperature | 287 °C |
| Explosive limits | Explosive limits: 2.4–13% |
| Lethal dose or concentration | LD50 (oral, rat): 3300 mg/kg |
| LD50 (median dose) | LD50 (median dose): 620 mg/kg (oral, rat) |
| PEL (Permissible) | PEL for Dimethylglyoxal: Not established |
| REL (Recommended) | Not established |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Methylglyoxal Glyoxal Dimethylglyoxime |

