Dimethyldioxane: Exploring Its Role in Chemistry and Industry
Historical Development
Dimethyldioxane sits among those chemicals that have quietly shaped modern industry without ever making headlines. Derived from dioxane, its origins go back to the post-war era, right as the world leaned into more complex organic synthesis. Back then, chemists began recognizing how slight modifications to the dioxane structure could offer new routes in everything from plastics to pharmaceuticals. The story of dimethyldioxane reads like a footnote in older chemical engineering textbooks, a reminder of the long tail of innovation that stretches from simple molecules to multi-step syntheses. Early research focused on maximizing yields and simplifying purification steps, and, for a time, it saw modest use in solvent systems and as an intermediate in other chemical processes. Progress in analytical techniques during the late twentieth century brought new scrutiny and raised fresh questions about the properties and safety profile of this lesser-known compound.
Product Overview
Most chemists working in labs have encountered compounds like dimethyldioxane as part of complex synthesis pipelines or as niche solvents. With two methyl groups connected to the dioxane ring, this molecule earned a reputation for offering selectivity and fine-tuned solvency in certain reactive environments. Applications might rank as specialized, but for chemists looking for that precise reaction condition, dimethyldioxane sometimes supplies the missing piece. In the commercial sector, it never saw the bulk demand of simpler solvents, yet retains value in carefully circumscribed technical segments—especially where ordinary dioxane or ethers do not cut it.
Physical and Chemical Properties
Dimethyldioxane stands as a clear, combustible liquid, not unlike many close relatives in the ether and dioxane families. Its boiling point sits above many basic solvents, providing a useful window for processes demanding elevated temperatures. The polarity and low viscosity make it a candidate for dissolving a broad array of chemicals—oils, resins, certain polymers. In real-life lab work, properties like vapor pressure or hygroscopic tendencies mean as much as any catalog listing. You see the difference when a reaction curve stays flat or when a separation goes smoother than expected. The chemical stability of dimethyldioxane under neutral conditions pairs with its willingness to lend oxygen during oxidative reactions, earning it fans among those who need controlled reactivity.
Technical Specifications and Labeling
What matters on a label often comes down to purity and contaminant profiling. Dimethyldioxane, supplied for chemical synthesis, often arrives with GC or NMR certificates outlining water content, peroxides, and residual solvents. Labs judge sources by their willingness to publish actual spectra and not just a purity percentage. Right-sized drums and bottles show up with color-coded caps, sometimes under plain chemical names and sometimes with trade names, but always with prominent flammability and health risk warnings. Shipping requires paperwork that respects international rules, and small changes in labeling over the years have come from stricter environmental and health regulations.
Preparation Method
The preparation of dimethyldioxane usually follows a path involving acid-catalyzed condensation or etherification reactions, starting from diols or glycols and working up with methylation reagents under controlled heat and pressure. Commercial processes tend to run in stainless reactors with in-line monitoring, both to catch unwanted side products and to safeguard workers against runaway reactions. Purification often employs fractional distillation, which relies on the fine temperature gap between dimethyldioxane and near-boiling impurities. Quality control insists on repeated tests for peroxide buildup—a risk with most ethers and dioxanes.
Chemical Reactions and Modifications
Dimethyldioxane fills its niche not by what it can tolerate, but by what it can become. Chemists frequently explore its use in ring-opening reactions, selective oxidations, and as a template for making more complex heterocycles. In some syntheses, it gives up methyl groups in the presence of strong acids, adding flexibility for those chasing novel intermediates. Many who work with such cyclic ethers spend time testing reactivity at the oxygen atoms, comparing ring strain and substitution outcomes to see if this compound offers a new shortcut or protection pattern. Its stability under basic conditions opens up alternative reaction planning when more traditional routes get tangled.
Synonyms and Product Names
Across catalogs and textbooks, dimethyldioxane goes by a mix of common and systematic names. Chemists reference 2,5-dimethyl-1,4-dioxane in formal settings, but shorthand and trade names still float around older stockrooms. Anyone searching chemical inventories soon learns to check CAS numbers to avoid confusion. This variety in labeling comes from decades of fragmented naming habits, reflective of how chemical supply chains have grown. Digital databases help now, but for years, the jumble of synonyms could frustrate even seasoned lab managers tracking down old reports or ordering new stock.
Safety and Operational Standards
Handling dimethyldioxane takes more than goggles and gloves—it teaches respect for vented hoods, dry containers, and regular peroxide tests. Safety protocols owe much to lessons learned from fires and health scares involving similar ethers. Strict storage means clear dates on bottles and regular checks for signs of polymerization or peroxide formation. Modern standards recommend spill kits that neutralize rather than disperse, as well as continuous education about the hidden dangers of chronic exposure. In labs, periodic drills check not only equipment, but also staff response in case of fire or leak. Regulatory frameworks from OSHA and REACH shaped much of the current practice, but experienced hands add a layer of caution grounded in experience—like never storing open bottles over the weekend or combining even trace amounts with strong oxidizers.
Application Area
Dimethyldioxane never became a household word in major manufacturing, but in specialty synthesis, it still earns its spot on the shelf. Paint chemists sometimes reach for it to solve formulation headaches where other ethers fall short. Pharmaceutical research turns to molecules like this as reaction media for tricky, water-sensitive pathways. Some companies test its compatibility with certain pesticides or processing aids, looking for small improvements in yield. Specialty polymer research sometimes finds a use during chain propagation or termination steps, but these are not mass-market applications. Often the biggest value comes from very specific transformations, or in analytic laboratories where baseline contamination must stay as low as possible.
Research and Development
Research momentum shifts over decades, but cyclical interest in alternative ether solvents has kept dimethyldioxane in play among chemical engineers and academic groups. Efforts to curb environmental impact have led researchers to profile its breakdown products, bioaccumulation potential, and remediation possibilities. Analytical chemists explore detection thresholds in air and water, developing techniques for faster, more precise monitoring. Biotechnology sometimes explores derivatives or modified dimethyldioxane groups in enzyme studies, looking for better mimicry or control over complex biological reactions. Newer environment-sensitive processes have spurred attempts to develop less toxic analogs or safer derivatives, though few have made it to mainstream adoption.
Toxicity Research
Decades ago, safety reviews for dimethyldioxane ran on little more than acute toxicity tables. Studies gradually built up a clearer—and less comfortable—picture. Animal studies revealed dose-dependent liver and kidney effects, raising red flags for occupational exposure. Metabolic breakdown pointed to formation of simpler, sometimes mutagenic compounds, leading to tighter restrictions and better air handling in labs. Long-term studies on chronic inhalation and dermal exposure are less complete than desired, so risk management leans on analogy with close relatives like dioxane and THF, which share metabolic pathways. Environmental monitoring, especially around chemical plants, measures groundwater and air for trace levels, acknowledging both the persistence and movement of volatile ethers. These findings led to stronger recommendations on air filtration, personal monitoring, and employee health checks.
Future Prospects
Looking ahead, demand for dimethyldioxane hinges on what researchers decide in the push for greener chemistry. As regulators clamp down on hazardous solvents and target persistent chemicals, only those compounds with strong performance and manageable risk profiles will outlast the shift. Some research groups believe modified dioxanes, with built-in routes for easier breakdown or safer handling, may one day swap in for the traditional molecules. Industry investment in biodegradability and closed-loop recycling could chart a path for dimethyldioxane’s continued presence, but that depends on credible safety and environmental data. For now, chemists who know the quirks of this compound will continue leveraging its strengths in narrow, highly controlled contexts, and perhaps guide the next generation in safer, smarter ways to use molecules like it.
What is Dimethyldioxane used for?
Looking Inside the Chemistry
Dimethyldioxane, usually cropping up in discussions about ingredient safety, pops up in all sorts of places. This chemical gets talked about most as a byproduct from manufacturing processes, especially where ethoxylation is involved. Ethoxylation turns up in making everything from household cleaners to shampoos. The big deal comes in because if you ever look over the back of your bottle of dish soap, you might see ingredients like sodium laureth sulfate. That stuff gives us foam and bubbles, makes soap feel nice, and gives laundry detergent its power. As companies make this ingredient, dimethyldioxane sometimes appears as an unintended hitchhiker.
Household Cleaners and Cosmetics: How Did We Get Here?
Dimethyldioxane caught my attention after reading a few studies and safety reports while shopping for baby soap. It isn’t added on purpose. Manufacturers end up with traces because of how certain ingredients are made. The catch is, even small amounts of this chemical, which counts as a probable human carcinogen according to agencies like the EPA, can show up in things people use every day. I've noticed a growing number of brands put "1,4-dioxane free" on their products to give shoppers confidence, which says a lot about changing priorities.
Manufacturers have relied on its presence for years, not because they want it, but because older chemical processes weren’t built with consumer safety in mind. Legislation and consumer demand have started to push things in a better direction, but it takes big industry changes and some public awareness to see real improvement.
Water Supplies and Environmental Concerns
Beyond household cleaners and soaps, talk about dimethyldioxane usually leads straight to drinking water. Several states, including New York, have tested water supplies and found detectable amounts that came from industrial runoff. Drinking water shouldn't come with a side of chemical byproducts. Once this stuff gets into groundwater, towns might face expensive cleanup projects. Our environment—and public health—both pay for shortcuts in manufacturing years ago. One solution that’s picking up steam comes through advanced water filtration, but that doesn’t fix the source. Cleaner manufacturing needs to become the rule, not the exception.
What Companies and People Can Do
Reading labels more closely shifted the way I pick bathroom products, and I encourage others to do the same. Brands that opt for safer alternatives typically use different manufacturing methods, or invest in extra purification steps to filter out dimethyldioxane. Customers have a louder voice than ever. By choosing products that are transparent about their ingredients and sourcing, shoppers push companies to invest in cleaner processes. Plenty of advocacy groups keep up pressure with petitions and education, making life a little safer one step at a time.
Calling for Smarter Choices
Dimethyldioxane doesn’t belong in soaps, your water, or anywhere near your skin. I’ve seen how regulation and a shift in buying habits move the industry, even if slowly. There’s momentum around outlawing or strictly limiting this chemical in consumer products, and it feels overdue. Science keeps finding links between trace chemicals and long-term health concerns, so cutting them out before they start seems wise. As someone who wants a safer home, I watch this space and vote with my wallet. If more of us do, perhaps companies will leave dimethyldioxane behind for good.
What are the safety precautions when handling Dimethyldioxane?
Respecting the Risks
Dimethyldioxane isn’t some harmless lab curiosity. I learned pretty quickly in my career that certain chemicals demand extra care, and this one tops the list. Back in my graduate days, one whiff of its sickly-sweet odor told me there’s danger lurking. Breathing it in, spilling it on skin, or even having it in a poorly ventilated workspace pushes luck the wrong way. Cancer warnings connected to dimethyldioxane aren’t theoretical—they’re written on the label for a reason. The EPA and IARC both flag it as a probable carcinogen. Over time, exposure can mess with the liver and kidneys. Short-term, even a small splash burns and irritates eyes and lungs.
Safe Habits in the Lab and Workplace
I’ve always believed in over-preparing, so I never handle dimethyldioxane without reliable ventilation. Fume hoods pull vapors away from my breathing space—a basic rule for every chemist with their career in mind. Not every lab skips this step, which puzzles me. The chemical loves leaking into the air, so I treat open bottles as emergencies unless they’re capped right away.
Gloves, goggles, and a solid lab coat aren’t suggestions—they’re my uniform. Latex gloves don’t hold up well against dimethyldioxane, so I use nitrile or heavier-duty barriers. Any contact with eyes needs a trip to the eyewash station, no stalling. Long sleeves matter, too. In the industry, people sometimes forget splash risk, and it’s usually hands and arms that pay the price.
Storage and Spill Control
Locking bottles in a cool, dry, fireproof cabinet stays crucial. This chemical isn’t just flammable—it’s volatile enough to worry about sparks or static near storage. Every container gets labeled, even the tiny samples. Once, after a rushed shift, I stumbled across an unlabeled vial; panic set in until I tracked down the logbook. No one likes guessing games with toxic substances.
If a spill hits the floor, I tell coworkers and clear the area. Absorbent pads—never sawdust or paper—make cleanups safer, since this stuff can start a fire before most realize it. Using water alone can spread the mess. Properly disposing of soaked materials keeps trash cans from becoming fire hazards.
Personal Responsibility and Worker Training
It surprises me how often people skip safety briefings or ignore posted warnings until after an accident. Training sessions on chemicals like dimethyldioxane aren’t busywork; they prevent long-term health emergencies. New hires can’t just shadow seasoned techs—they need to see every emergency shower and eyewash station, learn evacuation routes, and run through scenario drills.
Good management culture steps in where personal responsibility leaves off. I’ve seen places where leadership talks safety on paper but not in the halls. Supplies run low, or inspections get skipped to save time, until someone ends up hurt. Companies actually lower costs in the long run by treating chemical safety as a core value, keeping injuries and lawsuits out of the mix.
Developing a Safety Mindset Beyond the Rules
Dimethyldioxane won’t forgive carelessness, so I treat procedures as living habits. Beyond following guidelines, I watch out for those around me, knowing everyone benefits from shared vigilance. If a process looks unsafe or the equipment doesn’t work, I speak up. Protecting health in the lab and shop starts with respecting every risk and holding the line on proven precautions. That mindset has paid dividends in every project and every team I’ve worked with.
Is Dimethyldioxane hazardous to health or the environment?
What Is Dimethyldioxane?
Dimethyldioxane pops up now and then in chemical industry conversations, especially around solvents and manufacturing. The full chemical name—1,4-dioxane, 2,3-dimethyl—doesn’t trip off the tongue, but the substance plays a role in industrial cleaning agents, plastics, and sometimes even cosmetic formulas. The stuff rarely makes headlines, but folks who work with chemicals might recognize the risk hiding behind such a quiet name.
Health Risks: What Science Tells Us
Researchers who spend their days in labs have linked many dioxane variants, including dimethyldioxane, to significant health problems. Just breathing in the vapor in high enough concentrations can lead to dizziness, drowsiness, and irritation of the eyes and throat. At higher levels or after longer exposure, the substance can start to affect your liver and kidneys. Animal testing—still the backbone of many toxicity studies—often highlights tumors and organ damage when exposure drags on.
Regulators in the United States and Europe keep 1,4-dioxane on their list of “probable human carcinogens.” No direct link in humans, but there's enough evidence from mice and rats to warrant caution. I’ve talked with people who have worked in manufacturing sites where solvents like this drifted through the air. They still worry, even years later, as the long-term risks linger.
Nobody wants their drinking water tainted by chemicals. The U.S. Environmental Protection Agency reports that dioxane contamination sometimes creeps into groundwater near industrial sites and waste disposal areas. Traditional water filters at home won’t touch this stuff. Residents depend on proper cleanup and strict monitoring from authorities.
Environmental Consequences
If the only issue related to dimethyldioxane involved workers in a lab or factory, the story would be easier to contain, but this chemical refuses to stay put. Dioxane in general dissolves easily in water and travels quickly through soil. Once it gets into a river or an aquifer, it spreads, making cleanup expensive and technically challenging.
Most bacteria in natural environments can’t break the substance down fast enough. So, dimethyldioxane lingers, sticking around long enough for fish and wildlife to pick it up. I’ve seen researchers standing knee-deep in creeks downstream from factories, sampling for stubborn traces years after a spill. Most studies show these compounds resist light and natural processes, sticking around much longer than anyone would like.
Possible Paths Forward
Industry doesn’t have to rely on risky solvents—not completely. Many places transition to safer substitutes or closed-loop manufacturing to keep evaporation and waste down. Governments have set strict workplace exposure limits and call for regular monitoring in water sources near manufacturing.
For cleanup, a growing number of engineers back advanced methods like pump-and-treat systems, or using special bacteria to break the molecule apart—a process called bioremediation. These aren't perfect fixes. They cost money, and the public must keep pressure on both companies and regulators to keep sites clean.
Dimethyldioxane isn’t a household word, but its risks ripple out far beyond the lab. Addressing the problem means keeping up with the research, demanding accountability, and making choices that protect both people and ecosystems. I've watched small towns band together to demand better testing after local contamination. Without public watchdogs and firm rules, the chemical’s harms outlast any single generation.
How should Dimethyldioxane be stored?
Everyday Safety in the Lab and Warehouse
People in chemistry, whether in research, manufacturing, or storage, always wind up with a few substances that keep them on high alert. Dimethyldioxane absolutely belongs to that group. This isn’t just a matter of OSHA compliance or ticking off a safety audit. Storing it right protects health and avoids fire hazards—real, day-to-day concerns for anyone sharing space with volatile chemicals.
Understand What You’re Dealing With
Dimethyldioxane’s chemical structure loads it with flammable vapors, and its tendency to form peroxides makes it more than a predictable fire risk. That’s what pushes safe storage front and center. A few years ago, I worked in a university lab where everyone took flammable liquids for granted, but a single spill during a hot summer afternoon made us rethink our habits. Experience makes one remember how this compound can turn a casual mistake into a visit from the fire department.
Temperature and Ventilation Matter
You never want to leave dimethyldioxane near a heat source. Half the fires in chemical storage rooms start with someone leaving a bottle near sunlight, steam pipes, or an overworked fridge motor. The goal is always the same: keep it cool, ideally below 25°C (77°F), in a spot that stays the same temperature throughout the day. Good ventilation becomes non-negotiable too. Fumes shouldn’t linger, and airflow helps whisk away any vapors that slip from seals or during bottle transfers.
Choose the Right Containers and Cabinets
Glass is a solid bet for short-term storage, but tight-sealing metal cans with chemical-resistant linings work best when you plan on holding it longer. Dimethyldioxane eats through old or incompatible plastics in no time, so it pays to read up on what the supplier recommends. Flammable liquids belong away from oxidizers—never let curiosity lead you to stack everything by alphabetical order instead of chemical compatibility. Fire-rated cabinets aren’t just for show; I’ve watched one contain a small explosion once, saving everyone serious grief.
Daily Practices that Make a Difference
In the rush to get experiments done or to clean up after a busy shift, small steps like labeling, regular checks for leaks, and keeping just enough on hand save hassle later. Steam from a broken HVAC system can change a storage area overnight. A quick walk-through each morning, with an eye out for swelling bottles or crust around the caps, picks up trouble before it gets serious. Containment trays under shelves catch drips, and safety data sheets should sit within arm’s reach.
People Are the Biggest Safety Investment
A locked cabinet doesn’t replace training. Every new person in a lab or warehouse should see, up close, what mishandled dimethyldioxane can do. Over the years, I’ve learned that people who understand the risks stop bad habits before they become routine. Face-to-face training, realistic drill scenarios, and open talk about close calls create a shared sense of responsibility. Everybody wins when staff show up to work confident, not complacent.
Laws and Insurance Line Up with Smart Storage
Local fire marshals and federal guidelines don’t just write rules for their own sake. Insurance adjusters follow the same logic—if you keep flammable chemicals stored properly, you avoid headaches after an accident. Regular self-inspection, paired with the right paperwork, cuts down on unexpected costs and regulatory trouble.
What are the chemical properties of Dimethyldioxane?
Understanding the Basics of Dimethyldioxane
Dimethyldioxane turns up in labs and factories with a very specific set of quirks. This compound falls under the category of cyclic ethers, made up of a six-membered ring with two oxygen atoms. Picture it as a distant cousin of more famous ethers, but with a couple of methyl branches that change the game. Those seemingly simple tweaks to its structure end up mattering a lot, especially in day-to-day work with organic solvents or in discussions about industrial safety.
Reactivity Under Different Conditions
Dimethyldioxane shows off its ether nature by being relatively stable under moderate conditions. Ethers, in my experience, don’t usually catch fire at the drop of a hat, but if you heat or expose them to a strong acid, trouble follows. Dimethyldioxane carries a potential to form peroxides on standing in air, something every chemist learns to keep an eye on—peroxides can build up over time, becoming dangerously explosive. Its methyl groups help it shrug off attacks from most bases, but strong acids can break open the ring, sometimes leading to pretty messy mixtures of byproducts.
Solubility and Compatibility
This molecule dissolves well in most organic solvents, including alcohols and common hydrocarbons. Water doesn’t blend with dimethyldioxane, not unlike other ethers. Working in the lab, I noticed that it floats as a separate layer, making for simple separations if you know what you’re doing. Mixing isn’t always the goal—sometimes you want your solvent to stay apart during extractions or purifications, and dimethyldioxane reliably respects that boundary.
Volatility and Flammability Risks
Dimethyldioxane comes with a low boiling point and high vapor pressure, so you get intense fumes at room temperature. Anyone who has opened a bottle will recognize the strong smell. In a poorly ventilated space, it doesn’t take much for fumes to become overwhelming, or worse, flammable. Industrial accidents often connect back to ignoring these properties—sparks, open flames, or hot surfaces can all set off a flash fire with dimethyldioxane. From what I’ve seen, keeping a tight lid and working in a fume hood can head off most problems.
Health Concerns and Environmental Impact
Short-term exposure, mostly through inhalation, causes dizziness, nausea, and headaches. Prolonged contact will dry out and irritate the skin, which matches up with other solvents in this family. Long-term health effects of repeated exposure haven’t been as well studied, but the cautious approach—keep contact to a minimum and always wear gloves—makes a difference. Safe disposal becomes equally important, since this compound doesn’t break down easily and can linger in water or soil, polluting the environment. Treatment in chemical waste streams, incineration with high-temperature scrubbing, and tight containment all help reduce its footprint.
Finding Responsible Solutions
To work responsibly with dimethyldioxane, labs and factories must track storage conditions and monitor for peroxide formation. Regular testing and routine disposal of old stocks curb risks; one professor insisted we label each container with the date opened, making it easy to keep up. Substituting with greener solvents, where possible, keeps both the team and the planet safer. Still, for some reactions, dimethyldioxane can’t be swapped out so easily. That’s where tight regulatory controls and strong training play a major role. People need to know the risks, handle the material with care, and always choose methods that prioritize both health and safety above speed or convenience.
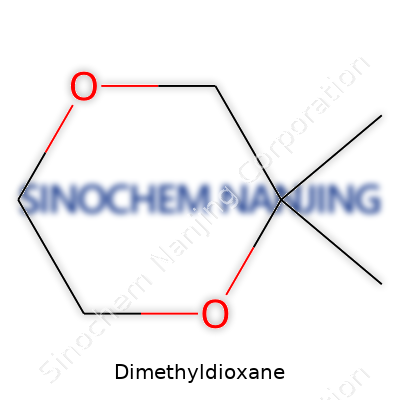
| Names | |
| Preferred IUPAC name | 2,2-Dimethyl-1,3-dioxane |
| Other names |
1,3-Dimethoxypropane 1,3-Dioxane, 2,2-dimethyl- 2,2-Dimethyl-1,3-dioxane Dimethyl-1,3-dioxane |
| Pronunciation | /daɪˌmɛθɪlˈdaɪ.ək.seɪn/ |
| Identifiers | |
| CAS Number | 5704-30-9 |
| Beilstein Reference | 1731166 |
| ChEBI | CHEBI:34718 |
| ChEMBL | CHEMBL52972 |
| ChemSpider | 21520 |
| DrugBank | DB13262 |
| ECHA InfoCard | 12f3a1c6-da3e-4932-a60a-493fdcddfd6a |
| EC Number | 205-575-3 |
| Gmelin Reference | 4399 |
| KEGG | C06508 |
| MeSH | D004102 |
| PubChem CID | 10845 |
| RTECS number | JG0875000 |
| UNII | 1F2M8P1A3V |
| UN number | UN1162 |
| Properties | |
| Chemical formula | C6H14O2 |
| Molar mass | 118.17 g/mol |
| Appearance | Colorless liquid |
| Odor | Faint ethereal odor |
| Density | 0.987 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 0.14 |
| Vapor pressure | 1.66 kPa (at 20 °C) |
| Acidity (pKa) | 14.55 |
| Basicity (pKb) | 4.15 |
| Magnetic susceptibility (χ) | -62.7·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.413 |
| Viscosity | 2.38 mPa·s (20 °C) |
| Dipole moment | 0.09 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 257.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -413.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −3345.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H226, H302, H319, H335 |
| Precautionary statements | P210, P280, P305+P351+P338, P309+P311 |
| NFPA 704 (fire diamond) | 2-3-2-₩ |
| Flash point | 41 °C |
| Autoignition temperature | 215 °C |
| Explosive limits | Explosive limits: 1.1–9.5% |
| Lethal dose or concentration | LD50 oral rat 3080 mg/kg |
| LD50 (median dose) | LD50 (median dose): 308 mg/kg (oral, rat) |
| NIOSH | JN8450000 |
| PEL (Permissible) | PEL: 100 ppm (380 mg/m³) |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
1,3-Dioxane 1,4-Dioxane Dimethoxyethane |

