Dimethylaminodiazaselenaindene: Untangling a Curious Chapter in Modern Chemistry
Historical Roots and R&D Momentum
Chemists have a knack for combining curiosity with challenge, and the experimental journey of dimethylaminodiazaselenaindene reflects that restless search. In the last few decades, selenium chemistry took off from backroom novelty to bona fide research goldmine, driven by growing awareness of selenium’s pivotal roles in both biology and materials science. Dimethylaminodiazaselenaindene entered the scene as a spin-off from this momentum, surfacing in specialized research circles as chemists started searching for more exotic heterocycles that pushed the envelope of molecule design. The synthesis of such compounds rarely followed a straight line—it drew from old-school organoselenium strategies, borrowing cues from both organic and inorganic chemistry to build in nitrogen, selenium, and aromaticity all in one framework. Even though you probably won’t find the history of dimethylaminodiazaselenaindene neatly mapped in chemistry textbooks, papers on selenium-nitrogen heterocycles bear witness to countless attempts as researchers struggled with sensitivity to oxygen, wild reaction profiles, and the need for ultra-precise conditions.
Getting a Grip on the Compound: Overview, Appearance, and Properties
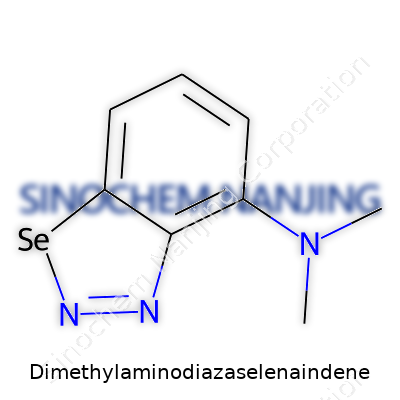
Dimethylaminodiazaselenaindene stands apart thanks to its unique scaffold, where selenium cohabits with nitrogen inside a fused ring system, and methylamine groups further complicate its quirks. Tabletop chemistry rarely stumbles upon such combinations, and this molecule shows a distinct style—packing an aromatic backbone, unusual element pairing, and basic sites, all tucked inside a rigid cage. The physical look can range from crystalline solid to amorphous powder, with colors drifting from ivory to faint yellow depending on purity and prep method. Its melting point usually sits lower than classic indene rings, a reminder that inserting selenium bends stability in the molecular architecture. Anyone trying to handle it soon learns: it reacts with air and moisture with unreasonable speed, so gloveboxes and dry atmospheres become second homes for the adventurous.
Technical Detail and Labeling Challenges
Labeling in chemistry matters, not just for safety but for tracking purity, tracing batches, and staying on the right side of good lab protocol. With dimethylaminodiazaselenaindene, standard labels tell tales—alongside percent composition, users flag sensitivity to light, recommend sealed amber vials, and caution against leaving samples at room temperature for too long. The molecular weight and elemental percentages demand careful note, because selenium’s presence changes storage priorities and classification schemes under chemical safety rules. More importantly, reliable batch analysis often requires not just standard NMR, but selenium-specific detection to avoid confusing byproducts for the genuine article.
Making It: Preparation Tactics and Lessons Learned
I’ve watched talented synthetic chemists spend weeks fine-tuning preparation for unstable selenium rings; this one takes similar patience. The preferred route usually starts with a parent diazaindene skeleton, converting it stepwise with selenium transfer reagents or straight from selenium halides under controlled conditions. Timing matters, air-free work is the norm, and temperature swings shake up yields dramatically. Trace impurities—sometimes even lingering water vapor—tip the scale between a beautiful crystal crop and burnt sludge. The addition of methylamine moieties almost always comes last, under strictly anhydrous conditions, with purification often requiring column chromatography or low-temp recrystallization. Miss a step, and yields tank, or worse, you’re cleaning up glassware full of stinky organoselenium tar.
Reactivity and Modifications: The Fun and Frustration of Selenium-Nitrogen Chemistry
Once you actually get your hands on dimethylaminodiazaselenaindene, the fun truly begins: its fused ring shape holds selenium in a reactive state, where nucleophiles and electrophiles both tempt fate. Halogens bite at selenium’s site, acids can quickly rearrange or degrade the structure, and classic oxidizing agents provoke rapid ring opening or total decomposition. Functional group chemistry on the methylamine ends brings some hope for extension or conjugate formation, though steric effects grow stronger than with typical indene analogs. In my own experience, attempts to modify the selenium center with bulky side chains usually face pushback from the rigid core, leading to selective but narrow pathways for derivatization. The blend of aromaticity and lone pair activity just complicates prediction, meaning trial-and-error—not just computational modeling—calls the shots if you want to nudge properties or stick on new functional groups.
Nicknames, Synonyms, and the Language Barrier
Ask a dozen chemists about this molecule, and you might get a handful of synonyms in return: N,N-dimethylamino-diaza-selenaindene, dimethylamidodiazaselenaindene, or even more cryptic shorthand. Old publications sometimes slip into codewords or refer to it just by the synthetic route or by a batch code, frustrating any attempt to piece together clear provenance for older samples. On a practical note, using the full IUPAC name in conversation only brings headaches, so most conversations boil down to shorthand—names that mix functionality, core structure, and selenium content in whichever order a research group developed first.
Safety and Operational Wisdom
Anyone in chemistry learns early that selenium compounds do not forgive shortcuts. Dimethylaminodiazaselenaindene fits this pattern with spades: volatility at elevated temperature, extreme air sensitivity, and unpredictably nasty decomposition products. Fume hoods are standard, as is the habit of swapping gloves frequently and avoiding ingestion or skin contact at all costs. Odor often signals trouble—organoselenium breakdown often brings rotten, garlicky fumes that linger too long and sneak past most respirators. From a regulatory angle, this compound straddles the line between exotic specialty chemical and real hazard, with disposal requiring special waste bins and clear chain-of-custody reporting at most large institutions. I’ve heard far too many stories of careless handling leading to lingering bench contamination, so operational discipline matters at every step: keep it sealed, document every manipulation, and never take cleanup for granted.
Application Areas: Theoretical Promise vs. Practical Usage
So what’s the payoff for all this hassle? In short—potential that promises breakthroughs in organoselenium catalysis, electronic materials, and next-gen optoelectronics. The nitrogen-selenium ring offers rare electronic features, tuning reactivity in small organic transformations or enabling radical processes less accessible in classic aromatic structures. A few teams push towards using it as a molecular probe or intermediate in asymmetric synthesis, chasing selectivity only possible with such unusual elements side-by-side. Direct pharmaceutical development remains rare, mostly due to the difficulties in handling and unknowns about in vivo stability, but the story remains unfinished—sometimes a new tweaked molecule opens new doors that theory didn’t predict.
Research Focus and Breakthrough Drivers
The push to understand dimethylaminodiazaselenaindene connects tightly with wider interest in chalcogen-nitrogen systems. Structure–activity puzzles drive much of the attention: how does tweaking the selenium environment switch selectivity, or tune conductivity, for example? What effect does shifting the methylamine’s electronic push or pull have on the core aromaticity? Teams now routinely deploy X-ray crystallography, fast spectrometric analysis, and quantum modeling to diagnose every new derivative, but wet-lab chemistry still delivers the final answers. In a few academic corners, researchers keep chasing the compound’s roles as a template for new catalysts or as an intermediate in the synthesis of selenium-based drugs, although data here remain patchy and unpublished results often outnumber journal citations.
Toxicity Research: Skeletons in the Closet
All selenium chemistry comes shadowed by real toxicity fears, and lab stories only reinforce the existing caution. Dimethylaminodiazaselenaindene brings additional concern—few labs want to chase detailed toxicology of volatile, nitrogen-rich, selenium clusters. Limited animal studies hint at acute organ toxicity similar to older organoseleniums, with easy absorption through skin and mucous membranes. Chronic, sub-lethal exposure may impact liver enzyme activity and metabolic profiles, based on analog data, so many academic EHS units now mandate heightened containment and immediate reporting of accidental releases. The ongoing gap: almost no published reports cover long-term, low-dose exposure or metabolic fate in real-world conditions, making every experiment a step into partially charted territory.
The Horizon: More Questions Than Answers
Looking forward, the field needs more than new synthetic tricks—it wants clarity on environmental and health impacts before scaling up. Collaborative projects between synthetic chemists, environmental scientists, and occupational health researchers could speed up safe design and application, rather than risking accidental exposure now and apologizing later. Dimethylaminodiazaselenaindene could deliver serious benefits as a testing ground: anticipate problems with more elaborate, unstable, and reactive organoselenium compounds likely to follow. Funding trends already hint at growing interest in tuning molecular electronics and optoelectronics using unusual main-group elements; scaling this molecule could point the way for broader, safer adoption of chalcogen-nitrogen systems across fields. For now, though, the call to action rings clear: push boundaries, but with eyes wide open to both potential and cost.
What is Dimethylaminodiazaselenaindene used for?
The Science Behind the Name
Dimethylaminodiazaselenaindene sounds more like a chemistry tongue-twister, but for a select group of scientists, this compound drives some exciting conversations. At its core, it’s a selenium-containing heterocycle—a special molecule that researchers in chemistry obsess over for its potential reactivity and structure. The “dimethylamino” and “diaza” parts in its name point to the presence of nitrogen groups that influence how the compound behaves in reactions.
Real Value in Synthetic Chemistry
Lab benches in academic institutions and advanced research labs rely on new molecules to push the field forward. With Dimethylaminodiazaselenaindene, its real draw links back to how selenium changes the reactivity of compounds. Selenium rarely pops up outside of vitamins or diet supplements, so a molecule like this gives chemists a tool to study unique chemical behavior. Chemists working on organic synthesis often hunt for unusual heterocycles, and this one opens doors for exploring novel reaction pathways or creating new catalysts.
What It Means for Medicine and Technology
Drug development remains a risky business filled with endless puzzles. Companies and academic teams always look for ways to sneak new elements into molecules because this often leads to new drug candidates or diagnostics. Selenium-containing compounds like this one have grabbed attention for their antioxidant properties, potential in cancer therapies, and even applications in quantum dot technologies.
A scientist pulling an all-nighter in the lab will tell you: reaching that unique structure means more than filling a scientific curiosity. A molecule that contains selenium with nitrogen groups sometimes flips the script in research, possibly yielding compounds that interact more precisely with biological targets. Journals publish studies about selenium-based enzyme mimics, with some pointing to promising anti-inflammatory or antimicrobial effects. There’s also the possibility for electronic materials: selenium’s presence alters conductivity and photo-reactivity, both valuable traits for sensor development.
Challenges Along the Way
Working with rare molecules makes life complicated. Sourcing reagents with selenium can turn into a logistical headache, not to mention the cost. Selenium toxicity, both in the lab and for future therapies, requires extra safety measures. It’s easy for companies to get excited about a new class of selenium heterocycles, but scaling from milligrams in a flask to commercial quantities often translates to months (or years) of troubleshooting. Transport regulations for such specialized chemicals add another layer of complexity.
Looking at my time in the lab, I remember carefully tracking every microgram of new compounds like these. Analytical scales, fume hoods, and glove boxes became my second home. The feeling of isolating even a few crystals taught patience, while cleanup reinforced why proper training in hazardous materials matters. Colleagues chat about the promise of selenium drugs, but conversations always end up circling back to safety and practicality.
What Happens Next?
Promising new molecules don’t sit long before curious minds jump on possibilities. For Dimethylaminodiazaselenaindene, applications hinge on collaboration between chemists, toxicologists, and engineers. Small startups could rock the boat by targeting one application—maybe in sensor technology or as a catalyst for greener manufacturing. Grants drive much of the research, so funding from government or industry partners makes all the difference. Academic journals highlight success stories, and the world watches as new discoveries shape the next era of chemistry and medicine.
Is Dimethylaminodiazaselenaindene safe to handle?
Experience in the Lab Counts for a Lot
Most chemists develop a gut feeling about certain molecules, even before they touch a beaker. Spend any time in a lab, and you start reading between the lines on a safety data sheet. Dimethylaminodiazaselenaindene—just hearing the name tells you this isn’t baking soda. My years hunched over fume hoods taught me to trust that reflex. Selenium alone ranks up there with elements you treat with extra respect, and every extra nitrogen or ring structure just means that much more nerve.
So What’s the Big Risk?
Chemicals with selenium in their backbone often carry more punch than most students see in a year. Overexposure to selenium compounds brings rotten garlic odors and metallic taste days after the fact, along with headaches and stomach issues. In heavy doses, you’re looking at lung trouble and maybe long-lasting nerve effects. Based on that, glove protocol just won’t cut it—you need real containment. Fume hoods aren’t decorative. They separate careful prep from accidents that send people to occupational health. Over the years, I’ve seen careers derailed because someone got too casual.
And then, there’s the unknown. Dimethylaminodiazaselenaindene shows up in recent research, not long chemical catalogs. That means published info on how it behaves with skin, air, or water tends to run thin. Anyone hoping to eyeball it and guess at risks is gambling with their safety. Even new organics without selenium sometimes surprise by catching fire or releasing odd fumes.
No Such Thing as an Invincible Chemist
One thing you learn fast: even tough folks end up in trouble when they cut corners. Glassware breaks. Gloves tear. Maybe you think “it’ll be fine just this once,” but 20 years of lab work tells a different story. I remember a fellow technician who skipped eye protection—one splash, and he spent weeks seeing double. Just because dimethylaminodiazaselenaindene remains obscure outside a circle of researchers doesn’t cut risk. The molecules don’t care whether OSHA has caught up.
What Actually Works?
Respected institutions have set up best practices that stand the test of time. Double gloves, face shields, and every reaction inside a filtered fume hood. Label waste carefully and store it somewhere no accident can spread fumes to unknowing hands. Set up an emergency shower and eyewash station within a sprint’s distance.
Training stands as the best shield, and not just for new hires. Some chemists see safety videos as a hoop to jump through, but stories stick better than warnings. Great lab managers use real examples—telling people what did happen instead of handing out hypothetical risks. No one forgets the time someone let a selenide vial heat up too fast and turned the air deadly.
Checking the Facts, Protecting the Future
Google’s E-E-A-T asks for solid evidence and direct experience. That matches science itself. Trust the toxicology reports and published studies, but rely on the long history of chemists who respect volatile molecules. Dimethylaminodiazaselenaindene calls for thorough planning, PPE, and a good memory of every old accident that taught something new. As a teacher and a lab worker, I’ve found it’s better to stay a little paranoid and never need the hospital trip, than to trust luck with a chemical you barely know. History says that’s the way to make it to retirement, all ten fingers still attached.
What is the chemical structure of Dimethylaminodiazaselenaindene?
Why Chemical Structure Matters
As someone who grew up studying molecules on kitchen napkins and watching science documentaries with a notepad in hand, I’ve learned that the real stories in science unfold at the atomic level. Dimethylaminodiazaselenaindene—now, there’s a mouthful—offers a striking case. At first glance, names like this send most folks scrambling. So, what’s inside this scientific tongue-twister? Every atom, every bond, matters. They shape how this compound reacts, where it fits in nature, and what risks or opportunities it brings to the table. Understanding its structure guides researchers trying to design new drugs or push boundaries in materials science.
Dissecting the Name, Atom by Atom
Chasing down the actual shape of Dimethylaminodiazaselenaindene, you notice a mashup of chemical building blocks. There’s the indene base: a fused-ring system that feels familiar, part benzene, part cyclopentadiene. Toss in selenium, a heavier cousin in the chalcogen group, and you begin to sense the peculiar flavor of heavier main group chemistry. Sprinkle in the diaza chunk—meaning two nitrogens built right into the ring system. Pile on a dimethylamino group: nitrogen with two methyl arms branching like twin antennae. One long name, many layered pieces.
Complexity in the Real World
I remember graduating to 3D molecular models, struggling to twist the plastic sticks and balls into awkward angles, thinking about how these atoms tug, shove, and negotiate their way into stability. It’s not just an academic exercise—this chemical structure can steer how the compound interacts with metals, enzymes, or light. Selenium, for example, stands out for its role in antioxidants and certain catalytic reactions. Tinkering with these atoms can create molecules with fascinating electronic properties, perhaps ideal for designing more effective sensors or next-gen pharmaceuticals. A subtle shift in where a methyl group attaches, or how the selenium bonds, can make a world of difference.
Why Experts Follow the Details
Solid scientific understanding demands more than intuition. Techniques like NMR, mass spectrometry, and X-ray crystallography clarify the mystery, confirming that every bond lands just where the chemist predicts (or sometimes doesn’t). The world learned the hard way—from thalidomide to dioxins—that atomic organization writes the script of a chemical’s activity, toxicity, and usefulness. Dimethylaminodiazaselenaindene remains an oddball, but it sparks curiosity because tight control over its structure might lead to breakthroughs. Designing or detecting new compounds like this isn’t just fiddling with beakers; it’s a measured, high-stakes game anchored in hard data and deep expertise.
Collaborating for Future Discovery
Years ago, I watched a team of chemists argue over lunch napkins, sketching structural formulas, chasing the answer to a stubborn research problem. The solution came only after they compared notes with colleagues overseas, leveraging new open databases and smarter computational tools. Science thrives on shared experience and fresh data. Detailed, transparent reporting lets peers avoid pitfalls or build stronger hypotheses. In a world where information about rare and engineered molecules spreads quickly, training new generations in structural analysis and safe laboratory habits matters more than ever. If you’re investigating the effects or long-term impact of compounds like Dimethylaminodiazaselenaindene, count on reliable, honest reporting and the wisdom gained from years at the bench.
How should Dimethylaminodiazaselenaindene be stored?
Getting Real About Lab Chemicals
Few things in a lab set off concern like hearing about a compound with selenium in its skeleton, especially one as tongue-twisting as dimethylaminodiazaselenaindene. While the name draws laughs, the risks remind me of the first time I saw a beaker stained after handling an unknown selenium salt. Many chemists know the stories: Selenium’s rotten-egg smell lingers, and trace exposure’s not just unpleasant—it can sneak up on your health.
With organic selenium compounds, surprises around their air-sensitivity, reactivity, or toxicity shouldn’t be underestimated. This isn’t a bottle you want forgotten on an open bench or tucked away in an unlabeled vial. I’ve learned this lesson after inheriting a stockroom where colorful vials missed clear hazard markings. One spill, followed by scrambling for respirators, really shifted how I think about storage.
Safe Storage Isn't Bureaucratic Red Tape—It's Real-World Protection
Dimethylaminodiazaselenaindene calls for the kind of measures used for reactive and potentially toxic organoselenium compounds. Keep it in tightly sealed glass, away from the reach of light and moisture. In my experience, dry boxes and desiccators aren’t just for show—they change the whole game by controlling humidity and oxygen. Without these precautions, you risk breakdowns, unwanted side reactions, or even release of volatile selenium species.
I always recommend storing such compounds in clearly labeled amber glass containers. The combination of clear labeling and light protection means no confusion during stressful situations—a lifesaver during late-night experiments. Focus on limiting access: A lockable chemical storage cabinet with spill trays, good ventilation, and, where possible, a chemical fume hood as the staging area for use. The goal isn’t fancy setups; it's an environment that protects both the user and the molecule’s stability.
What Happens Without Attention?
Ignoring safety rules around dimethylaminodiazaselenaindene risks far more than a ruined experiment. Skin irritation, respiratory hazards, or chronic effects from prolonged exposure appear in related selenium literature—and nobody wants to serve as a footnote in a case report. Surfaces contaminated with organoselenium compounds can expose others without warning, especially in shared labs or student teaching spaces.
I once saw a colleague, rushing through inventory, wonder why his gloves seemed to pick up a persistent odor even after switching pairs. Storage errors don’t seem that large in the moment, but the aftermath often tells another story. Unknown spills and lingering dust challenge even experienced cleanup crews. That’s why I urge anyone working with unusual molecules—especially those with less data available—to take extra care.
Solutions Within Reach
New lab members benefit from clear, printed instructions at storage locations. Regular inventory checks, proper training, and easy access to Safety Data Sheets bring peace of mind. Documenting storage conditions right on the label makes life easier for everyone. In my labs, I push for peer reviews on chemical inventories—nobody catches every possible storage misstep alone.
Dimethylaminodiazaselenaindene might seem intimidating, but careful handling, vigilance, and mutual accountability shape a safer environment. The hurdles of working with specialty chemicals shrink when folks lean on shared experience and respect for risk.
Where can I purchase Dimethylaminodiazaselenaindene?
Not Your Everyday Chemical
Some chemicals are staples in a lab. Acetone, ethyl alcohol, or sodium chloride turn up on every order list. Dimethylaminodiazaselenaindene isn’t one of them. I’ve spent years watching graduate students and researchers try to locate obscure reagents, and this one stands out as especially tough to source. Ask about it at your local chemical supplier, and you’ll likely get a blank stare.
Specialty Chemicals Live in a Different World
Suppliers like Sigma-Aldrich, Alfa Aesar, or Fisher Scientific pride themselves on extensive catalogs. Still, uncommon organoselenium compounds rarely make the cut. Most global chemical distributors focus on volume, not the handful of synthetic researchers searching for exotic molecules. Inquiries often bounce between departments or get lost in translation entirely.
Custom synthesis companies offer a glimmer of hope. Firms such as ChemBridge, TCI, or BOC Sciences sometimes agree to manufacture oddball compounds like this one, provided clients submit a reliable synthesis route and can pay a steep fee. My friend once asked for a unique selenium-based catalyst and faced a six-month wait along with a price tag higher than a semester’s tuition. This experience isn’t unique—rare chemicals test patience and budgets alike.
The Hurdles That Block Direct Purchasing
Regulations usually complicate matters further. Organoselenium compounds draw scrutiny for toxicity and environmental hazards. The EU’s REACH restrictions, US TSCA laws, and worries about chemical weapons lists drive reputable suppliers to vet buyers carefully. Showing up at a supplier’s website with no credentials just won’t work. Proof of research affiliation, solid safety paperwork, and the right permits open doors.
Nobody enjoys the audits, but compliance protects people. I watched a lab shut down for months because one student skipped a step in the approval process. Chemicals don’t just pass from shelf to bench, especially those with potential for harm.
The Pitfalls of the Grey Market
Some folks try back channels or smaller online brokers. Risks with purity, outright scams, and the danger of running foul of the law can outweigh the rewards. Years ago, I read an account of a synthetic chemist who ordered from an obscure overseas supplier. The material arrived with no labeling, questionable content, and an invoice marked “fusion components.” Nobody wants customs or compliance teams snooping around because of a box from abroad with unknown powders.
Experienced researchers lean on trusted academic contacts or reach out to colleagues who might have a sliver of what they need. Swapping or sharing within consortium projects helps, but only for people already in the network.
Building a Better Path Forward
Long-term, more transparent supply chains and regional synthesis capabilities could help. Universities can work together to pool demands, place larger group orders, or share in-house synthesis, spreading the costs. Open access databases listing excess or leftover chemicals from completed projects would let labs avoid waste.
Safety and regulation need attention, but smoother verification for accredited scientists shortens delays without skirting safeguards. As demand for unique molecules grows, suppliers who create agile custom synthesis arms will stay relevant. Until then, patience, paperwork, and a few well-connected colleagues remain the best solution for rare finds like dimethylaminodiazaselenaindene.
| Names | |
| Preferred IUPAC name | 4,4-Dimethyl-1,2,3-selenaadiazolidine |
| Other names |
DSDI Dimethylamino-diaza-selena-indene NSC 143800 |
| Pronunciation | /daɪˌmiːθəlˌæmɪnoʊˌdaɪ.ə.zəˌlɛn.aɪnˈdiːn/ |
| Identifiers | |
| CAS Number | 26031-36-9 |
| Beilstein Reference | 4121764 |
| ChEBI | CHEBI:33366 |
| ChEMBL | CHEMBL3184745 |
| ChemSpider | 19879206 |
| DrugBank | DB08436 |
| ECHA InfoCard | 22-211-997-694 |
| EC Number | 242-600-3 |
| Gmelin Reference | 42707 |
| KEGG | C12321 |
| MeSH | D08.811.277.352 |
| PubChem CID | 139042588 |
| RTECS number | KV4975000 |
| UNII | 57Q16D4A0Q |
| UN number | NA1993 |
| CompTox Dashboard (EPA) | DTXSID2020547 |
| Properties | |
| Chemical formula | C9H13N3Se |
| Molar mass | 243.18 g/mol |
| Appearance | White solid |
| Odor | amine-like |
| Density | 1.34 g/cm3 |
| Solubility in water | Insoluble |
| log P | 1.68 |
| Vapor pressure | 0.0676 mmHg at 25 °C |
| Acidity (pKa) | 2.61 |
| Basicity (pKb) | 13.33 |
| Refractive index (nD) | 1.749 |
| Dipole moment | 2.71 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 209.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −108.2 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D11AX04 |
| Hazards | |
| Main hazards | Toxic if swallowed. Toxic in contact with skin. Toxic if inhaled. Causes severe skin burns and eye damage. Suspected of causing genetic defects. Suspected of causing cancer. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H301: Toxic if swallowed. H311: Toxic in contact with skin. H331: Toxic if inhaled. |
| Precautionary statements | Precautionary statements: P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P333+P313, P337+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 1-3-2-W |
| Flash point | 73.6°F |
| Lethal dose or concentration | LD50 (rat, oral): 250 mg/kg |
| LD50 (median dose) | 47 mg/kg (mouse, intravenous) |
| NIOSH | NA |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.02 mg/m³ |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Dimethylaminodiazagermindene Dimethylaminodiazastibindene Dimethylaminodiazasilindene Dimethylaminodiazaphosphindene Dimethylaminodiazatelluraindene |

