Dimethyl Oxalate: Much More Than Just a Chemical Compound
Stories Written in the Lab: A Brief Historical Walk
Chemistry has always fascinated me for its patience. Take Dimethyl Oxalate — scientists first isolated it over a century ago through basic esterification, using methanol and oxalic acid. Over time, the process shifted from small glassware to industrial reactors. People recognized its value pretty early, realizing it did a lot more than simply fill another bottle on a shelf. The drive to find low-cost and efficient production routes started just as the chemical industry ballooned in the twentieth century. China, in particular, has invested in large-scale production, using it as a critical step on the path to polyester manufacture. Over the years, the global trade of Dimethyl Oxalate has tracked the spread of synthetic fibers, solvents, and even new forms of energy conversion. Its journey reflects the tug-of-war between innovation, demand, and regulation in chemical manufacturing.
Down to Details: What Dimethyl Oxalate Really Is
Dimethyl Oxalate, known to chemists as methyl oxalate or just DMOX, shows up as a colorless liquid or solid, depending on temperature. Open the bottle and a faint, pleasant smell escapes, not unlike the solvents stocked in college labs. Each bottle should carry a clear label: C4H6O4, molecular weight 118.09, melting point between 54-56°C, and boiling at 162°C. DMOX dissolves well in alcohols and many organic solvents, but stays away from water. It’s those two methyl groups on the oxalate backbone that give it its unique edge in synthesis work. Keeping this compound pure takes real attention to detail — trace impurities radically change its reactivity and even risk safety.
Listing the Technical and Safety Ground Rules
Every lab technician learns early on to check purity, density, refractive index, and moisture content. Reputable suppliers provide test sheets and safety data, but real trust comes from experience and repeated analysis. The chemical, by its very nature, isn’t just flammable — it will release toxic fumes if handled carelessly or heated above its boiling point. Wear gloves, work in a fume hood, and never underestimate the risk of skin or eye contact. In the workplace, storage standards call for cool, dry, well-ventilated rooms. Proper labeling isn’t just a regulatory hoop — it protects anyone who stumbles on an unlabeled bottle after hours. The difference between a clean lab record and a workplace accident comes down to respecting these straightforward rules.
On the Bench: Making Dimethyl Oxalate
Industry uses two routes most of the time: direct esterification of oxalic acid with methanol, catalyzed by strong acids; or oxidative carbonylation of methanol, which skips over oxalic acid and goes straight for the product. The first method, a classic staple of undergraduate organic chemistry, generates water as a by-product. Industrial plants recover and recycle methanol streams, always moving toward lower waste and energy use. The oxidative carbonylation process — using carbon monoxide, oxygen, and metal catalysts — has excited a lot of interest in China. It fits in with large-scale coal-to-chemicals infrastructure, making DMOX from coal-derived methanol cheaply at vast scale. Each route has fans, but the allure of new technology, cost savings, and climate policy keeps research on production methods lively.
Reactivity and Modification: Turning Potential into Products
Look past its basic structure and Dimethyl Oxalate becomes a quiet workhorse for organic transformations. Its two ester groups react with nucleophiles to make diesters, polyesters, and custom-designed building blocks for medicines. In the plastics industry, it acts as an intermediate for ethylene glycol by hydrogenation, which then flows into everything from PET bottles to antifreeze. The solvent properties matter, too: DMOX finds a niche in formulations where traditional solvents are too toxic or don’t evaporate at the right rate. With strong bases or organometallics, the compound serves as a source of the oxalate group, which orchestrates coupling reactions and material modifications. As a synthetic chemist, I appreciate how its precise reactivity cuts down on by-products.
Sifting Through Names and Synonyms
Like many semi-obscure chemicals, Dimethyl Oxalate answers to several names. Methyl oxalate and oxalic acid dimethyl ester pop up in technical papers. In regulatory filings you might find “dimethyl ethanedioate” or its Chemical Abstracts number, 553-90-2. This web of aliases can trip up even seasoned researchers if they jump between suppliers or regulatory environments. For safe transport, containers also carry hazard symbols for toxicity and flammability. The careful tagging of names and identifiers protects both scientific progress and basic human safety.
Demand in the Real World
Over the years, the range of uses for Dimethyl Oxalate has shown just how flexible this compound is. Factories feed hydrogen over it to make ethylene glycol, which lands in bottles, fibers, and films. Chemists rely on it for pharmaceuticals and agrochemicals, where precision and purity affect human health outcomes. In high-tech industries, Dimethyl Oxalate appears in custom solvents and specialty polymers. Clean-up and recovery operations sometimes make use of its solvent power. Markets swing with oil prices and green chemistry trends. The growth of coal-to-chemicals sectors in Asia has thrown new light on its potential as an energy-bridge molecule. The sometimes-overlooked importance of this molecule in everyday materials — from sportswear to electrical insulation — demonstrates just how central reliable chemical supply chains are to modern life.
Pushing Further: Research and Development
In research, Dimethyl Oxalate acts almost like a test case for new reaction pathways. Investigators use it to study hydrogenation catalysts, aiming to squeeze more glycol out of less raw material. Startups now look at bio-based or CO2-to-chemicals processes that rewrite the rules on feedstock and waste. Real advances often hinge on catalyst lifespan, tolerance to impurities, and energy consumption. Teams working in green chemistry use Dimethyl Oxalate to probe safer or cleaner reaction conditions. At conferences I’ve seen exciting work on using dimethyl oxalate as an intermediate in biodegradable plastics synthesis, linking sustainability and industrial demand in noticeable ways.
Learning from Toxicity Studies
Every chemical carries risks. Toxicity research for Dimethyl Oxalate shows limited acute poisoning in animal tests but flags skin and eye irritancy, potential for organ damage at high doses, and dangers from combustion by-products. Chronic exposure data sits thinner than some would prefer, especially for workers in factories running at several tons per day. The compound’s toxicity profile demands personal protection and tight engineering controls, from gloves and goggles to air extraction. Community awareness near production sites remains patchy — a problem for policymakers aiming to balance economic growth with public health. Researchers call for detailed monitoring of air and effluent streams around production sites to ensure everyone from plant operators to local residents avoids unnecessary exposure. Many older plants lacked detailed safety protocols, but modern facilities face stricter audits and real-time monitoring.
Peering into the Future
Trends in chemical manufacturing now circle around circular economy principles, and Dimethyl Oxalate’s story reflects these pressures. New methods promise less CO2, less energy use, and more feedstock flexibility — even using captured carbon from other industrial processes. As regions shift away from oil, DMOX could play an even larger role bridging traditional and emerging value chains, especially with investments in alternative energy and chemical recycling. Future opportunities include making fully bio-based polyesters that reduce environmental toll, supported by policy, consumer demand, and technical innovation. On the regulatory side, stiffer rules around workplace exposure and environmental releases will only increase pressure to manage every production step with care. I see real potential in open scientific exchange: the team that cracks high-yield, zero-waste DMOX synthesis could set new standards for sustainable chemical practice.
What is dimethyl oxalate used for?
A Chemical Workhorse Few People Notice
Spend some time around a chemical plant or even just pay attention to the labels on industrial products, and dimethyl oxalate might pop up more than expected. It gets overlooked compared to splashier materials, but its impact carries real weight—especially in how modern polymers and fuels come to life.
Drilling Down: Where You’ll Find Dimethyl Oxalate
For the average consumer, dimethyl oxalate stays behind the curtain. Once inside a science lab, or a factory focused on plastics, the picture changes. Producers value this compound as a source of methyl glycolate and methyl oxalate, both steps along the pathway to making ethylene glycol.
Ethylene glycol isn’t just some obscure lab product; it ends up in antifreeze, polyester fibers, and resins. Chances are, anyone wearing athletic gear or driving a car with modern coolant owes something to chemistry that began with dimethyl oxalate. China, in particular, uses a large-scale process where this molecule steps in for traditional petrochemical routes, tapping into coal-based feedstocks rather than crude oil.
Lowering Oil Dependence
Countries searching for alternatives to petroleum have turned to dimethyl oxalate as an answer. This route, usually called the coal-to-ethylene glycol (CtEG) process, grew fast in regions with abundant coal reserves. Plants can take synthesis gas from coal or natural gas and convert it into dimethyl oxalate, then push on to ethylene glycol.
This shortcut bypasses naphtha cracking—long the old standard for making ethylene glycol. The shift opens up independence from the oil market’s roller-coaster prices, but it doesn’t come without tradeoffs. Supporters argue it extends the life of coal assets and gives a lifeline to local economies tied to mining. The flip side points to greenhouse gas emissions tied up in coal-to-chemical plants.
Making Plastics Stronger and Cheaper
Plastic manufacturers lean on dimethyl oxalate to tweak properties in certain resins. Polyesters and polycarbonates get used for everything from soda bottles to smartphone cases. Starting with high-purity intermediates, makers have tighter control over the resin’s final strength and flexibility. Having a stable, predictable input like dimethyl oxalate means the difference between brittle, throwaway goods and materials that outlast tough use.
Stepping Stones to Valuable Chemicals
Dimethyl oxalate doesn’t just vanish after it’s done its job in glycol production. It can act as a base for producing other chemicals, such as methylamine and even some pharmaceuticals. In agriculture, it provides the building blocks for certain pesticides, though this sector sees closer regulatory scrutiny to protect health and the environment.
Over years working in labs that handle industrial organics, I’ve seen strict tracking and safety guidelines kick in the moment dimethyl oxalate enters the picture. Toxic if inhaled or ingested, chemists treat its handling as seriously as that of better-known hazards.
Room for Improvement
Cleaner technologies stand as a real need for every part of the chemical supply chain. Dimethyl oxalate’s role in coal-based production begs big questions about long-term climate goals. Research groups in Asia and Europe race to find catalysts and processes that lower emissions, cut waste, and use greener starting materials. Consumers rarely see this effort, but shifts here shape the cost and eco-footprint of everything from coolant in their car to the fibers in their clothes.
Living alongside new standards for carbon emissions and tougher global rules, producers experiment with renewable feedstocks and closed-loop recycling. Regulations push industries to limit releases, while customer demand for “greener” products pushes corporate labs to squeeze every efficiency from trusted old reactions, including those started with dimethyl oxalate.
Is dimethyl oxalate hazardous to health?
Understanding Dimethyl Oxalate
Dimethyl oxalate comes up in conversations about chemical manufacturing, plastics, and solvents. It's a clear liquid, sometimes with a fruity smell, and it appears in more places than most people realize. Used as an intermediate in polyester production and as a solvent or additive, its presence often sits quietly behind the products on supermarket shelves.
What Science Says About Safety
Safety around chemicals always brings up tough questions. The short answer for dimethyl oxalate: treat it with respect. This compound irritates the eyes, skin, and respiratory system. Industrial users have long relied on gloves, masks, and tight work practices to limit direct contact. Even brief exposure can lead to watery eyes, coughing, and skin redness. In my time touring chemical plants, I noticed that well-trained workers never cut corners with personal protective equipment around this stuff, even for routine tasks.
Swallowing it or breathing in concentrated fumes can be much more serious. Larger doses can trigger nausea, vomiting, even kidney damage in extreme examples. Studies show that dimethyl oxalate converts to oxalic acid in the body, which can disrupt calcium levels and harm organs. Safety datasheets—required at every plant—carry clear emergency instructions in case of splashes or spills precisely because the risks are real.
Environmental and Community Concerns
Accidental release causes more than a minor headache. Water systems cannot easily break down this chemical. Accumulated spills add to local pollution burdens. I’ve seen communities near chemical hubs become wary of unexplained runoff and fumes—not just from personal discomfort, but out of concern for long-term well-being. Chemical storage and haulage always spark heated meetings in those towns, and dimethyl oxalate is part of those discussions. People want better monitoring, emergency planning, and transparency from industry leaders.
Balancing Industry and Public Health
Factories can’t run on fear, but ignoring legitimate dangers only leads to mistrust. Occupational limits put out by governments and international bodies come from stacks of research. The American Conference of Governmental Industrial Hygienists and Europe’s ECHA have detailed recommendations for exposure—numbers that workplaces can measure with real instruments, not guesses. Tracking air quality and spill records must move from paperwork to open, digital platforms. Community right-to-know laws proved their worth decades ago, and companies that keep neighbors in the dark only stoke suspicion.
What Can We Do Better?
Substitution stands out as a real fix. Shifting to safer solvents and additives, or designing new materials that skip hazardous intermediates, brings lasting results. Some manufacturers already follow this path—using alternative chemistries when possible, at least for non-critical applications. Waste handling and recycling processes also need constant updates, since old infrastructure can crack or leak. It’s worth pushing regulators for routine simple checks, not just fancy risk assessments every few years.
Open conversations change attitudes, both on the factory floor and in city council meetings. Demands for stricter labeling, better emergency drills, and real-time air sensors come not from panic, but from hard-earned experience. That’s the best lesson from years spent in both industry and worried communities—knowledge keeps people safe.
What is the chemical formula of dimethyl oxalate?
Chemical Formula and Structure
Dimethyl oxalate stands out in the world of industrial chemistry. Its formula, C4H6O4, describes a molecule formed by swapping both hydrogens on oxalic acid with methyl groups. It looks simple as a string of carbon, hydrogen, and oxygen atoms—four carbons, six hydrogens, and four oxygens. The structure follows as (COOCH3)2, showing off those ester linkages that chemists and engineers come to recognize right away.
Importance in Industry and Synthesis
In the classroom years ago, I first came across dimethyl oxalate during an organic lab. The instructor explained that it’s more than an academic curiosity. Real-world uses stretch from the production of plastics, pharmaceuticals, and sometimes even as a step in turning coal or natural gas into everyday products like antifreeze or polymers. This reaction, often called oxidative carbonylation, avoids some of the nastier byproducts found in older manufacturing routes and uses less energy, reducing the environmental footprint.
Why the Formula Matters
Knowing the precise formula C4H6O4 goes beyond memorization. Chemical industries rely on tight process controls. Factories calculate quantities of starting materials and expected yields using exact formulas. If the numbers are off—even by one atom—reactions take detours, products lose their purity, and waste piles up. Each oxygen, hydrogen, and carbon has a job. Dimethyl oxalate’s formula ensures chemists get the reaction they want, not some hazardous or useless byproduct.
Safety and Environmental Concerns
Every chemical brings risk. Dimethyl oxalate falls in a middle ground. It isn’t the worst actor in the lab, yet, as with all esters, skin contact or inhalation can irritate. Factories carry strict procedures and ventilation systems. Spills on the shop floor mean careful cleanup, not just a towel and a shrug. For disposal, hydrolysis and controlled incineration have become common, sidestepping old-fashioned options that poisoned water and soil.
Innovation and Future Directions
For designers of greener processes, dimethyl oxalate represents a step away from more polluting chemistry. Its role as an intermediate lets industries avoid harsh chlorination or dangerous heavy metals seen in past decades. Some companies now experiment with plant-based feedstocks, cutting ties with fossil fuels. It’s not common yet, but pilot plants have shown C4H6O4 can be made from waste biomass, opening up a circular approach to manufacturing instead of the dig-and-burn mentality of the last century.
Finding Smarter Solutions
Better safety training, robust equipment maintenance, and continuous research into alternative feedstocks point to a world where industrial dimethyl oxalate isn’t just another commodity. It’s a linchpin for safer, more sustainable production. Universities and private laboratories continue to improve yields, trim waste, and squeeze more value out of each molecule. It takes people willing to adapt, from those on the factory floor to research chemists and regulators.
Conclusion
Dimethyl oxalate—a string of C4H6O4—offers more than a textbook example. It showcases the power of precise chemical understanding and a path toward safer, smarter, and cleaner industry. Paying attention to the molecular level rolls out real-world benefits far beyond the lab.
How is dimethyl oxalate stored and handled?
What Dimethyl Oxalate Brings to the Table
Dimethyl oxalate serves a purpose in industrial chemistry, mostly in making pharmaceuticals, plasticizers, and sometimes in fuel additive production. This clear, colorless liquid seems pretty harmless at first, but experience shows it demands caution and respect in a work setting. Its sweetish odor doesn’t help warn anyone about real risks that go with poor storage and sloppy handling.
Potential Hazards Lurking in the Drum
People working with dimethyl oxalate have learned never to underestimate its dangers. Vapors irritate eyes, skin, and airways. Longer exposure leaves a person feeling unwell, and spills on skin bring burns. A direct hit to the eyes pushes straight into emergency territory. Most chemicals in this family pack a punch, and this one doesn’t break the pattern.
Fires and explosions may not occur every day, but dimethyl oxalate catches flames fast. Vapors form flammable mixtures with air, especially when storage spaces heat up. It becomes even more concerning once you know that, in busy facilities, a bit of carelessness sets the stage for big mishaps.
Storage: No Corners Cut
Over years spent on busy plant floors, it becomes clear: storing chemicals safely requires a plan and follow-through. Drums and containers must keep a tight seal. Any sign of corrosion, bulging, or leaking means those containers have to go. Storing this liquid away from sparks, heat sources, and direct sunlight reduces risk. Warehouse crews who skip these basics end up with spills, fires, or health scares.
Stashing dimethyl oxalate alongside acids, strong oxidizers, or reactive metals spells trouble. Workers checking the chemical register prevent these mix-ups. Lessons learned from near-misses get passed down in safety briefings for a reason.
No chemical vault works without good ventilation. Fume hoods and exhaust fans keep vapor levels low. Regular checks and working alarms solve half the problems before they grow. Fire extinguishers always stand by—never gathering dust.
Safe Handling: Trained Hands Only
Walking into a plant where people wear goggles, gloves, and sturdy coats, it’s easy to spot the teams who know what’s at stake. Training sessions aren’t treated as paperwork—they’re a lifeline that keeps people out of harm’s way. Those who get complacent put others in danger, too.
Spill kits sit in plain view. Any splash gets cleaned up fast, since delays create breathing risks and slippery hazards. Used gloves and rags head straight into marked bins, because old stories of fires from oily waste stick with every safety officer.
Emergency showers and eyewash stations stand close to storage and work areas. Years of experience show that not a second can be wasted in those first few moments after an accident. Workers need to know escape routes and emergency contacts by heart.
Supporting Safety with Policy and Oversight
Reliable safety comes from managers caring about their people—not just checking boxes for compliance. Clear labeling and up-to-date safety sheets let everyone know exactly what they’re dealing with. Regular audits and surprise inspections provide accountability. Skipping these tough conversations opens the door to preventable disasters.
Regulators weigh in here, setting hard rules to keep standards high. Following government guidance and learning from industry best practices raises the bar for everyone. By making a habit out of vigilance, companies save lives and steer clear of fines that pile up when corners get cut.
Getting storage and handling right goes beyond any single job—it honors the lesson that health and safety always matter, every single day.
What are the physical properties of dimethyl oxalate?
A Clear Picture of What Dimethyl Oxalate Looks and Feels Like
Dimethyl oxalate pulls its own weight in everyday chemical settings, even if most people outside a lab have little reason to think about it. This stuff shows up as a colorless, crystalline solid, and if you’ve ever spent time around organic solvents, you’ll recognize the sweet, slightly fruity smell drifting from an open container. Crystals like these don’t weigh much; in fact, dimethyl oxalate has a density around 1.1 grams per cubic centimeter. That means it floats just below the surface in water but still packs more heft than many lighter organic solvents you might find sitting nearby.
Melting and Boiling: Key Temperatures in Use
If you left dimethyl oxalate in an open dish at room temperature, it’d stay solid, not running off like some volatile compounds. Warm it up a bit, and at about 54 degrees Celsius, it starts to melt into a clear liquid. Let the temperature rise to just under 190 degrees Celsius, and you’ll see it boil off, sending its vapors swirling up and away. In a manufacturing plant, workers have a good idea how careful they need to be during distillation because of these numbers. No need to worry about it catching fire easily; the flash point sits at about 75 degrees Celsius, so it doesn't go up in flames as fast as, say, ether or acetone.
Solubility and Everyday Handling
Anyone who’s tried dissolving dimethyl oxalate in water quickly learns it breaks down a bit but doesn’t disappear. The solubility lands around 5 grams per 100 milliliters at room temperature. Mix it with alcohol or ether, and the story changes: it dissolves right away. This trait lets it get along fine in organic reactions, especially in processes where water just gets in the way. In a lab, you’ll find flasks with solutions of this chemical in ethanol, which makes it easy to manage during transfers and reactions.
Stability: What Happens Over Time
Dimethyl oxalate shows decent stability if kept out of strong acid or base environments. Given enough time or the wrong kind of conditions, it can break down to release dangerous gases, including carbon monoxide, so you can’t just stash it anywhere. I’ve found that sealed containers, kept in a cool, well-ventilated spot, give peace of mind. Poor storage can mean slow decomposition, sometimes unnoticed until that tell-tale odor isn’t so sweet anymore.
Safety Concerns Tied to Physical Properties
In industrial settings, the solid form of dimethyl oxalate can float as dust if spilled or handled roughly. That’s a problem because breathing in dust doesn’t do anyone any good. Workers pull on N95 masks, and storage requires closed bins, since the powder flows easily and likes to cling to surfaces. Because of its melting point, it’s easy to handle as a solid at room temperature, but once processes heat up, any leaks or spills can mean quick vapor release, so good ventilation makes a difference.
Finding Better Solutions and Practices
Experience has taught me to treat dimethyl oxalate’s physical properties as more than numbers on a datasheet. Each one hints at either a safe, simple lab day or a troubleshooting session no one wants to face. By training staff, setting out clear rules for storage, and using proper personal protective equipment, facilities avoid most of the risks. Engineers who design reactors and piping that stay far below that boiling point end up saving headaches. Even though the molecule might look simple, a little respect for these fundamental properties keeps work safe and processes steady.
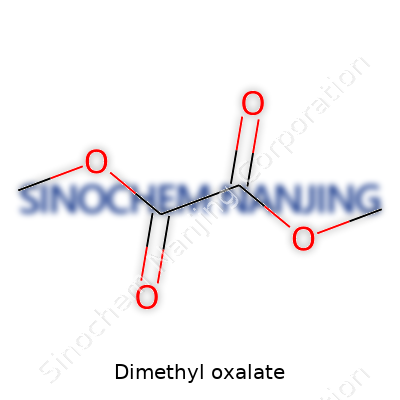
| Names | |
| Preferred IUPAC name | Dimethyl ethanedioate |
| Other names |
Dimethyl ethanedioate Oxalic acid dimethyl ester Dimethyl oxalate (DMO) DMO |
| Pronunciation | /daɪˈmɛθ.ɪl ɒkˈsæ.leɪt/ |
| Identifiers | |
| CAS Number | Dimethyl Oxalate CAS Number is '553-90-2' |
| 3D model (JSmol) | `Dimethyl Oxalate JSmol string: "dimethyl oxalate"` |
| Beilstein Reference | 878125 |
| ChEBI | CHEBI:85144 |
| ChEMBL | CHEMBL16360 |
| ChemSpider | 16313 |
| DrugBank | DB14049 |
| ECHA InfoCard | 08d1a981-94e2-48b5-b75f-b52c3f2ead13 |
| EC Number | 203-743-0 |
| Gmelin Reference | 3974 |
| KEGG | C01575 |
| MeSH | D008976 |
| PubChem CID | 7924 |
| RTECS number | MO1925000 |
| UNII | GQ463U8T1K |
| UN number | UN1162 |
| Properties | |
| Chemical formula | C4H6O4 |
| Molar mass | 118.09 g/mol |
| Appearance | Colorless transparent liquid or white crystalline solid |
| Odor | fruity |
| Density | 1.145 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.4 |
| Vapor pressure | 0.24 kPa (at 25 °C) |
| Acidity (pKa) | pKa = 13.1 |
| Basicity (pKb) | 12.52 |
| Magnetic susceptibility (χ) | -51.0e-6 cm³/mol |
| Refractive index (nD) | 1.381 |
| Viscosity | 1.19 mPa·s (25 °C) |
| Dipole moment | 1.07 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 233.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -587.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1244.4 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H319 |
| Precautionary statements | P210, P261, P280, P302+P352, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 54 °C |
| Autoignition temperature | 397 °C |
| Explosive limits | 4.1–22.0% |
| Lethal dose or concentration | LD50 oral rat 3300 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 3300 mg/kg |
| NIOSH | WX8575000 |
| PEL (Permissible) | PEL: 5 mg/m3 |
| REL (Recommended) | 50 mg/m³ |
| IDLH (Immediate danger) | IDLH: 500 mg/m³ |
| Related compounds | |
| Related compounds |
Oxalate Dimethyl carbonate Dimethyl malonate Methyl oxalate Ethyl oxalate |

