Dimethyl 4-(Methylthio)Phenyl Phosphate: More Than Just a Chemical Formula
Tracing the Path: Historical Development
Following World War II, the chemical industry exploded with new molecular discoveries, some arriving by accident, others through patient, careful design. In those decades, interest in organophosphates intensified, mostly because of their ability to disrupt the nervous system in insects. Scientists soon realized that tweaks to the molecular backbone could bring about big shifts in activity and selectivity. Amidst this surge, compounds like dimethyl 4-(methylthio)phenyl phosphate emerged. Not as iconic as malathion or parathion, yet solid in its utility, it rode those waves of agricultural and pest control research, riding alongside better-known cousins but offering new possibilities for chemical modification and fine-tuning.
Product Overview in Context
Holding a flask of dimethyl 4-(methylthio)phenyl phosphate, the faint aroma reminds me of early mornings in the university lab, the air fogged with solvents, scents clinging to coat sleeves. This compound appears as a pale liquid or crystalline powder, depending on storage temperature and purity. Researchers in crop science know it as a promising candidate when formulating new blends for targeted pest management, though its profile stretches further. It stands out for its tight balance: enough reactivity to matter, not so sensitive that it surprises anyone mixing or diluting it in standard lab conditions.
Physical and Chemical Properties in Use
Molecules like this tend to float below the radar until you see them in action. Dimethyl 4-(methylthio)phenyl phosphate’s defining feature sits in its functional groups: the phosphate handles reactivity, and the methylthio group fine-tunes its interactions. Its melting and boiling points rest in the moderate range, which means heating it doesn’t risk runaway reactions, but it still needs respect: vapor pressure and flammability aren’t trivial. The solubility profile makes it versatile, dissolving well in common organic solvents, letting chemists work up reactions without endless trial and error. This gives it just enough chemical backbone to join in a wide spectrum of transformations, without the messiness of unstable analogs that hydrolyze at the sight of water.
Technical Specifications and Labeling from a Practical Eye
Any shelf in a well-run lab tells a story, and clear labeling does more than check boxes for compliance. It keeps teams safe, prevents costly mix-ups, and helps track research outcomes. For a compound like this, detailed labeling includes its full IUPAC name, hazard statements tied to organophosphate exposure, and reminders to use gloves, eye protection, and fume hoods. It’s easy to gloss over the paperwork, but as I’ve seen in shared university spaces, one missing sticker has forced entire projects back to square one. The move toward QR codes and digital safety data connects the world of legacy chemistry with streamlined, future-ready lab practice, holding everyone accountable.
Preparation Method: Through the Lab Lens
Building dimethyl 4-(methylthio)phenyl phosphate starts with the right aromatic starting material – 4-(methylthio)phenol or related compounds. A simple esterification using dimethyl phosphorochloridate under controlled conditions draws out the phosphate esters and locks in the desired chemical structure. A wash step removes hydrochloric acid, and a round of purification with standard techniques (silica gel, rotary evaporation) gives a sample ready for analysis. Every prep run I’ve watched depended as much on patience as on reagents. Rushed reactions, or ignoring the pH tweaks during workup, led to poor yields and contaminated product. Mastering this prep separates careful chemists from careless mistakes.
Chemical Reactions and Modification Pathways
Once in hand, chemists rarely leave a molecule untouched. Dimethyl 4-(methylthio)phenyl phosphate carries reactive sites at the methylthio and phosphate positions, opening routes for oxidation, hydrolysis, or conjugation with other active molecules. Experts in synthetic modification use this as a gateway, introducing groups to boost bioactivity or reduce toxicity. Field researchers adjust these analogs in search of new pest deterrents or pharmaceutical intermediates. Each tweak spins out a new angle for inquiry, sometimes leading to commercial leads, other times to basic science insight about how sulfur and phosphorus atoms influence molecular interactions.
Names and Synonyms: Clearing Up Confusion
Lab jargon trips up the best of us. This compound has walked under many names: dimethyl p-(methylthio)phenyl phosphate, O,O-dimethyl O-[4-(methylthio)phenyl] phosphate, and a handful of trade labels that obscure more than clarify. For researchers and regulators trying to compare notes, the mess of nomenclature often frustrates more than it helps. Public datasets and published literature rarely agree on one title, lending weight to the push for uniform chemical identifiers like CAS numbers or digital tags. Until that catches on, careful checking remains a daily fact of life.
Safety and Operational Standards: Lessons Learned
Safety isn’t a formality here. The thioether and phosphate groups make organophosphates a class of concern in any protocol discussion. Long sleeves, double gloves, and sharp ventilation pull their weight in daily routines. Toxicity to non-target organisms, slow breakdown in aquatic systems, and potential risks for neurotoxicity mirror the hazards associated with cousins in this chemical family. Routine gets interrupted by rigorous training and periodic drills. Risk mitigation hinges on simple habits: labeling containers, segregating waste, cleaning up spills without shortcuts. For me, those repetitive signs in lab corridors underscore the personal stakes in industrial settings and core research labs alike.
Applications: From Field to Laboratory
Dimethyl 4-(methylthio)phenyl phosphate finds work in far more places than basic synthesis. Chemists look to it as a building block for new organophosphate pesticides, tweaking its structure for better pest targeting and slower environmental breakdown. Material scientists explore its additive effects in specialty polymers or coatings. Over time, niche uses have popped up – sometimes as intermediates for drug synthesis, sometimes in selective biological probes. Every year, industry and academia invest time hunting for safer, more effective compounds, but the landscape keeps shifting as new evidence comes in about long-term toxicity or unforeseen side effects.
Research, Development, and a Look at Toxicity
Digging through journals and reviews, it’s clear this molecule inspires equal parts curiosity and caution. Toxicology teams run bioassays to map out safe exposure limits and break down precisely how metabolic pathways process its functional groups. Some studies focus on breakdown products, which often pose more subtle risks than the parent compound. Regulatory boards scrutinize every new data point, balancing crop protection needs against the push to safeguard pollinators, farmworkers, and water supplies. I’ve watched peers work overtime screening for metabolites, realizing that a single methyl group can swing toxicity in unexpected directions. It’s this ever-moving boundary – between usefulness in the lab or field and risk in the broader environment – that shapes so much of the research and debate.
Future Prospects and Potential Solutions
Talk inside industry roundtables and research labs often lands on one challenge: balancing the demand for effective chemical tools with the moral and legal obligations to protect human health and natural systems. The next generation of organophosphates looks different from those of the past, shaped by new insights from computational modeling, green chemistry, and regulatory hurdles that demand cleaner degradation pathways. Dimethyl 4-(methylthio)phenyl phosphate may not make front-page news tomorrow, but the history of its use – and the push to modify and rethink its reactivity and persistence – keeps it at the center of chemical development efforts. Solutions grow from linking toxicology data, real-world field trials, and transparent communication among scientists, regulators, and end users. Following those threads, modest compounds can nudge whole industries in safer, more innovative directions.
What is Dimethyl 4-(Methylthio)Phenyl Phosphate used for?
Key Uses in Agriculture and Industry
Dimethyl 4-(methylthio)phenyl phosphate often turns up in discussions about crop protection. You see it in the chemical makeup of certain organophosphates, a class of compounds used by farmers and pest control professionals. They rely on these substances to fight pests that threaten yields. It’s not just about the science in a lab; on working farms, reliable crop harvests can mean the difference between profit and loss. This compound helps to form the backbone of insecticide formulas, mainly because of its ability to disrupt the nervous systems of harmful insects.
I remember walking through a greenhouse filled with tomato plants, watching workers carefully apply products to save their crops. Pests can wreak havoc overnight. Having a dependable tool to tackle infestations can protect the work of months. Still, it’s never only about wiping out bugs. People living near farms or eating produce expect safety, both for themselves and the environment. Regulations keep products containing such chemicals in check, but it takes constant vigilance from everyone involved.
Safety Concerns and Environmental Impact
Organophosphates, including derivatives like dimethyl 4-(methylthio)phenyl phosphate, have come under scrutiny for their toxicity. I’ve spent time reviewing reports from agencies like the World Health Organization and the EPA. There’s a reason these substances get re-evaluated every few years. Some problems from improper handling include water contamination, health risks for farm workers, and potential harm to non-target species such as bees. My own neighbors in rural areas have reported headaches when these chemicals drift over their property on the wind.
Avoiding harm takes clear rules and careful monitoring — not only checking residue on produce, but teaching workers about safe application and personal protective equipment. Modern tracking has improved, with everything from digital apps to government spot-checks, but challenges remain, especially where training or enforcement falls short.
Balancing Yield and Responsibility
Most growers can’t risk unchecked infestations, but pressure also comes from consumers who want less chemical residue on their food. Retail chains set stricter limits, and more buyers prefer eco-labels or organic options. That shift puts a spotlight on how essential chemicals like dimethyl 4-(methylthio)phenyl phosphate should be used — if at all — and sparks fresh investment in biological alternatives.
Newer approaches, such as integrated pest management, combine targeted use of synthetic chemicals with natural pest predators and scout-based monitoring. I’ve spoken with growers experimenting with these blended strategies. Yields stay consistent, and less product winds up in groundwater or on work boots. That gives rural families a bit more peace of mind as their kids play in the fields.
Technological and Policy Solutions
Regulators tend to respond fastest when science uncovers risks, or when citizens demand higher standards. In several countries, tighter rules have pushed chemical companies to develop less toxic alternatives. Drones and remote sensors help pinpoint areas that need attention, so only a fraction of the landscape gets sprayed. Some farm cooperatives test water downstream of their operations and share data to build trust.
All tools should be held to the same standard: improve productivity without threatening health. The conversation around compounds like dimethyl 4-(methylthio)phenyl phosphate will always involve trade-offs, but open eyes, clear data, and more investment in alternatives guide the industry in a smarter direction.
What are the safety precautions when handling this chemical?
Respect for Chemicals Starts on Day One
Walking into a lab or industrial site, someone new might notice the weird gloves, goggles, and bright signs before anything else. Those aren’t just for show. I learned the hard way during my first month on a job in manufacturing. A drop of solvent landed on my sleeve. By lunchtime, the sleeve was stiff and my skin was itching. It’s easy to brush off small spills, but small mistakes can leave a lasting impact.
Protecting Skin, Eyes, and Lungs
Every chemical isn’t created equal, so personal protective equipment changes, too. When I handled hydrochloric acid, nitrile gloves and a splash guard stopped burns. My co-worker tried to shortcut with thin gloves, figuring he’d “be quick.” He felt the sting two minutes later. Basic gear isn’t up for every job. Reading the label and grabbing the right pair of gloves or safety glasses turned out to be a simple way to avoid a trip to the nurse’s office. The Centers for Disease Control reports thousands of chemical exposure injuries every year; nearly half could have been stopped by using the right protection.
Ventilation Isn’t Just a Box to Tick
I spent a few months working with epoxy resins. Even though the air looked clear, I could still smell fumes. Cracking a window did nothing. The supervisor explained that fumes sink and gather in low spots. He showed me proper exhaust hoods and fans. I started noticing how my head felt after a few hours. I once skipped proper ventilation just to meet a deadline—it cost me the whole afternoon and a pounding headache. Acute exposure sometimes lands workers in emergency rooms, so keeping air moving with exhaust fans or local hoods keeps people out of danger without anyone realizing.
The Importance of Labels and MSDS Sheets
Some containers look clean and safe but carry surprise risks. Mislabeled bottles get people into trouble. The Material Safety Data Sheet (now called Safety Data Sheet) tells not just the obvious risks, but small details like how to handle spills, what to do if you get some in your eyes, and storage temperatures that prevent explosions. I once found toluene stored in a fridge—the vapors sparked a sensor that set off the fire alarm. Reading and following those sheets felt tedious at first, but those facts come from real-world accidents. OSHA sets out strict rules because real harm follows small oversights.
Housekeeping and Emergency Readiness
I’ve learned the value of cleaning up every small spill and resetting the workspace. A coworker slipped on a patch of oil and broke his wrist. Later, I kept cat litter and neutralizer handy for acid spills. Some folks want to ignore emergency showers or eyewash stations. Cuts and splashes really happen, and fumbling with stuck caps wastes time no one has in a crisis. Training on drills and knowing the escape route kept me calm once when a barrel leaked in storage. Fumbling through a fog of panic in that moment can be deadly.
Real Solutions: Culture, Not Just Rules
Building a safety culture where everyone looks out for each other stops trouble before it starts. Safety isn’t a one-and-done routine. Refresher courses and safety meetings aren’t red tape—they’re reminders that lives depend on good habits. The right gear, proper training, and strong habits always matter more than luck or shortcuts. In my experience, even one moment of carelessness can change a career or a life. Respecting proven safety steps every single day keeps chemical hazards from turning into real-world disasters.
What is the chemical formula and molecular weight of this product?
Why Details Make All the Difference
A chemical formula tells a precise story. It doesn't leave room for wild guesses or ambiguity. Folks who work in labs, factories, kitchens, or gardens all count on knowing exactly what’s inside the bottle or bag. A molecular formula, say C6H12O6, isn't just a bunch of characters. For a chemist, that translates directly to glucose—a foundation of energy in biology. The numbers matter because the tiniest difference can change everything, from how a substance behaves to whether it's safe to use.
From the Classroom to the Supply Chain
Lessons from chemistry class stick with those who work in manufacturing or science. Mixing up NaCl for NaBr doesn't sound like a big deal unless you’re the one consuming it. Sodium chloride, the salt everyone adds to food, looks a lot like sodium bromide on paper, but swap them and you’ve got a recipe for disaster. Even formulas that appear close, like H2O2 and H2O, create radically different substances—one cleans wounds, the other keeps us alive. This is where trust and accuracy replace shortcuts.
The Hard Numbers: Molecular Weight Tells Its Own Story
Every atom brings its own weight to the table. Hydrogen, oxygen, carbon—all these add up to a molecular weight, usually measured in grams per mole. Nobody wants to pour twice the amount needed just because the label didn’t mention the right number. Errors on this level aren’t just embarrassing; they drive up costs or worse, create safety hazards. Real-life stories fill trade journals from pharmaceutical missteps and industrial accidents all linked to wrong figures or lazy oversight. The stakes climb if you’re shipping across borders where customs and regulations demand ironclad documentation.
Why Real Experience Still Counts
A digital catalog with formulas and weights helps nobody if the numbers aren’t checked or understood. I've watched new staff on the factory floor pick up a drum with the wrong label—once, it stopped a process dead for hours. It isn’t just about lost time; everyone in line learns that chemistry doesn’t forgive mistakes. Folks in the field know you don’t trust vague paperwork. They reference the MSDS, check for the CAS number, and verify the formula against a second source. If the name doesn’t match the code stamped on the paperwork, nothing moves.
Demanding More from the System
Fixing the problem means training staff so they read, not skim, key facts. Company leaders can bring in regular audits, invest in smarter labeling, and direct teams to double-check every step of the way. Regulatory bodies step up by punishing repeat offenders and publishing public lists—no business wants to land there. Transparency comes from open data; everyone down the line, from scientist to end user, deserves the real numbers. Reputable suppliers already know their reputation depends on this.
Consumers and Professionals Alike Rely on Accuracy
In a world where the wrong chemical might endanger health or halt production, nobody should shrug off the fine print. People shop smarter, industries run leaner, and communities stay safer when chemical formulas and molecular weights come straight from trusted, transparent sources. It may seem like fine detail, but the difference is life-changing—sometimes literally.
How should Dimethyl 4-(Methylthio)Phenyl Phosphate be stored?
Understanding the Risk
Over the years in chemical labs, certain substances always demanded extra attention. Dimethyl 4-(methylthio)phenyl phosphate sits in that group. The reason goes beyond simple caution—this chemical tends to show sensitivity to light, air, and temperature swings. Relying on makeshift storage or guesswork often turns a manageable substance into something dangerous. My lessons from lab safety incidents say clear protocol beats clever shortcuts.
Ideal Storage Conditions
Anyone working with organophosphate compounds learns one truth quickly: don’t leave them out in the open. Dimethyl 4-(methylthio)phenyl phosphate reacts poorly to moisture, so dry conditions win every time. High humidity or leaks in a storeroom encourage slow breakdown. In my facility, dry cabinets with silica gel packs kept things stable. We checked the gel color often because even a slight pink tinge warned us adsorption dipped.
A chemical like this doesn’t like the sunlight—UV light can nudge certain bonds to break faster than expected. Direct light exposure almost always led to discolored samples or broken glass vials after a couple of months. Standard practice shifted to brown glass containers in a closed cabinet. Not all see-through plastics block UV, so glass brings more certainty. Mixing up solvents and containers just to save a few dollars invites real headaches down the road.
Temperature Control
Many chemicals handle room temperature, but experience shows swings up or down stress dimethyl 4-(methylthio)phenyl phosphate. We settled on refrigeration as the sweet spot—between 2°C and 8°C. Winters in northern states convinced us less than 2°C thickened the liquid and risked crystal formation. Freezing sometimes forced the cork loose or built pressure behind a cap. That taught us to skip regular freezers and go for a lab fridge, away from food and drink. Home fridge freezers just don’t offer precise environment control.
Separate and Label Clearly
Mixing this with oxidizers or acids multiplies risk. Storing each chemical family apart cuts down on unintended reactions. A separate shelf, backed up with lipped trays, stops spills from moving beyond a one-shelf accident. After seeing an old bottle with a faded label, I started double-marking each bottle with big, legible writing. Wearing out a marker beats squinting at a half-peeled-off strip of tape when seconds count.
Old habits stick—before leaving for the day, I’d swing by the cabinet and run a fingertip over the labels, checking for fading and checking seals for stickiness or flakes. Leaks catch up with you only if you let routine slide.
Staying Ahead of Trouble
Relying on MSDS sheets (Material Safety Data Sheets) pays off. Clear guidance on fire hazards, reactivity, and cleanup shortcuts the learning curve. I used to print out updated safety sheets and pin them near the storage area. Not everyone bothers, but on long shifts or in shared spaces, that habit handed out peace of mind.
Spill kits waited near the cabinet. Nobody wants to grab a mop or kitchen paper for a toxic chemical, so registered kits—including absorbent mats and proper gloves—always stood ready. Emergency wash stations nearby offered another layer of security. Investing time upfront in storage and emergency planning limited ugly surprises later.
Practical Approaches
Long weeks spent managing chemical stores convince anyone that safe storage takes steady effort. I kept a shelf for routine checks on every bottle. Change out faded silica packs. Inspect seals and double-check the log. Teach everyone in the lab, new and old, that patience with storage wins more than speed ever could. A tidy, cool, dry, and dark place—dedicated and well-marked—outperforms any high-tech solution that gets ignored or forgotten.
Is this product available in different purity grades?
Behind the Label: Purity Grades Explained
Sorting through scientific supplies or pharmaceuticals, the question about purity grades pops up often. Whether talking table salt, laboratory chemicals, or active ingredients in medicine, the purity of a product decides where and how it works safely. Companies offer products in different grades—sometimes called technical, food, or pharmaceutical grade—to help people match the right option to the job.
Why Purity Isn’t Just a Buzzword
No one opens a bottle of aspirin or grabs sugar for coffee thinking about impurities, but those details shape outcomes in the real world. A chemist in a pharmaceutical lab chases the highest purity—since tiny contaminants might trigger dangerous side effects. As someone who’s worked in a quality control lab, I saw that even common compounds, like sodium chloride, show up in several grades. Technical grade works for cleaning or basic industrial work. Food grade meets regulations on what’s safe to eat. Pharmaceutical grade—well, that’s where the strictest rules kick in. That difference in purity separates a batch that’s safe for medicine from one that’s not.
The Price of Precision
Sometimes people roll their eyes at paying more just for “the same thing” in a cleaner form. They imagine it’s a marketing trick. If you’ve scrubbed glassware with technical grade chemicals, you know it gets the job done without breaking the bank. Food-grade options crop up in kitchens, bakeries, and food factories, where regulators focus on what might go into our bodies. But labs working on sensitive or lifesaving treatments use the kind of ultra-pure compounds that cost more but leave less to chance. That’s more than a label—it means layers of filtering, extra testing, and tracking every batch back to its source.
Not Just Paperwork—Real Risks and Real Solutions
Mismatching a product’s grade with its use can jump from annoying to dangerous fast. I’ve seen confusion lead to expensive failures—like using a lower grade in a drug lab, then discovering contamination that wasted weeks of research. The 2008 melamine scandal in powdered milk showed how impurities or adulterants could spark a health crisis. Facts like these stress the need for honesty and traceability in supply chains.
Clear labeling helps everyone make informed decisions, but it’s only step one. Regular audits, third-party testing, and transparent records help protect both companies and the people counting on their work. Investing in staff training pays off, too—everyone from warehouse crew to scientists benefits from knowing why purity rules exist. It’s not just about checking boxes; it’s about safety, trust, and getting the job done right the first time.
Looking Ahead
As industries push for sustainability, faster testing, and tighter regulations, the push for clearer, more reliable purity information grows. Advances in digital tracking and rapid analysis hold promise, but so does a culture where no one treats purity grades as a footnote. Solid science, clear records, and honest conversation will shape the next chapter for purity in products that touch our lives every day.
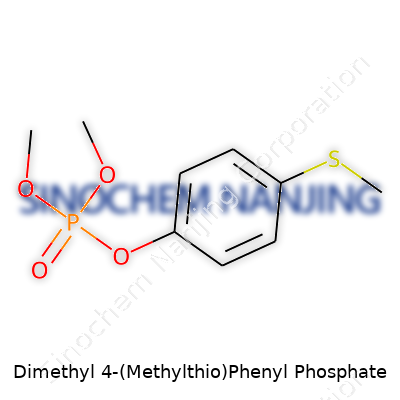
| Names | |
| Preferred IUPAC name | Dimethyl [4-(methylsulfanyl)phenyl] phosphate |
| Other names |
Phosphoric acid, dimethyl 4-(methylthio)phenyl ester Dimethyl p-(methylthio)phenyl phosphate O,O-Dimethyl O-(4-(methylthio)phenyl)phosphate p-(Methylthio)phenyl dimethyl phosphate Phosdrin-S |
| Pronunciation | /daɪˈmɛθɪl fɔːr ˈmɛθɪlˌθaɪ.oʊ ˈfiːnəl ˈfɑs.feɪt/ |
| Identifiers | |
| CAS Number | 2525-73-1 |
| Beilstein Reference | 1638734 |
| ChEBI | CHEBI:87135 |
| ChEMBL | CHEMBL2022177 |
| ChemSpider | 20370312 |
| DrugBank | DB08633 |
| ECHA InfoCard | ECHA InfoCard: 100.041.804 |
| EC Number | 26544-38-7 |
| Gmelin Reference | Gmelin Reference: 238972 |
| KEGG | C18602 |
| MeSH | D017967 |
| PubChem CID | 151118 |
| RTECS number | TG3150000 |
| UNII | X3B371KG3Z |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C10H15O4PS |
| Molar mass | 306.32 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.27 g/cm3 |
| Solubility in water | Insoluble |
| log P | 1.98 |
| Vapor pressure | 0.0003 mmHg at 25°C |
| Acidity (pKa) | 7.32 |
| Basicity (pKb) | 4.20 |
| Magnetic susceptibility (χ) | -62.97 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.562 |
| Viscosity | Viscosity: 2.1 mPa·s (25 °C) |
| Dipole moment | 3.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 395.6 J·mol⁻¹·K⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin and serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P270, P271, P273, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P337+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1-1-0-Health |
| Flash point | 180°C |
| Autoignition temperature | 250 °C |
| Lethal dose or concentration | LD50 oral rat 370 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1070 mg/kg (rat, oral) |
| NIOSH | TB6675000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Dimethyl 4-(Methylthio)Phenyl Phosphate is not established. |
| REL (Recommended) | REL (Recommended): NIOSH considers dimethyl 4-(methylthio)phenyl phosphate concentration in air to be 0.2 mg/m³ (10-hour TWA) |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Dimethyl phenyl phosphate Dimethyl 4-nitrophenyl phosphate O,O-Dimethyl O-(4-methylthio)phenyl phosphorothioate Diethyl 4-(methylthio)phenyl phosphate Dimethyl 4-methylphenyl phosphate |

