Diisoamyl Ether: More Than Just Another Organic Compound
Historical Glimpses and Product Journey
Diisoamyl ether stands as a testament to the way chemistry persists in carving out useful niches in an ever-evolving industrial world. Its history runs back to an age when chemists favored practical hands-on methods over computational wizardry, searching for better solvents in both simplicity and performance. Diisoamyl ether—ether being a common class discovered in the haze of the nineteenth century—only appeared formally in the literature a little later, as workers explored new ways to build or separate molecules. Back in university days, talk of ethers always conjured memories of old labs reeking of solvents, and of lecture slides showing how such compounds took chemical separations to another level. Diisoamyl ether isn’t as famous as diethyl ether; it never had the glamour or the historical drama. Yet in workbenches that chase efficient extractions and neat separations, it keeps showing up, steady as ever.
What Sets Diisoamyl Ether Apart
You only need to pour out a sample and catch a whiff to know you’re dealing with something distinct. Diisoamyl ether offers up a faint, sweet aroma—reminds some old hands of pears—something common with alkyl ethers but sharper, owing to its branched structure. The chemical recipe is simple enough: it's an ether with two isoamyl groups stuck to oxygen. In its pure form, it's a colorless liquid, free-flowing and fast to evaporate, which makes handling it both a blessing and a hazard. It’s lighter than water, so if you ever mix the two in a separatory funnel, you get clear delineation—a good property for extraction work. The physical facts sit out there: boiling point hits just past 160°C, flash point below 50°C, and density floats lower than water’s. Unlike heavier ethers, diisoamyl ether doesn’t bring stubborn, resinous residues to glassware; it usually rinses clean, assuming you respect what you’re working with.
Technical Aspects: What Labels Can’t Tell You
Across different labs, you might catch the label "Diisopentyl ether" or sometimes "3,3-Dimethylbutyl ethyl ether," but they all point to this same molecule. Chemical codes and purity claims don’t tell the whole story. You get the essential identifiers: C10H22O and various regulatory numbers, sure, but as someone who’s watched more than a few researchers handle it, safety matters as much as a paper guarantee. Always, storage means tight sealing—oxygen creep and sunlight play havoc with ethers, forming peroxides over months. That’s a risk easy to ignore until an old bottle sparks trouble. Suppliers like to signal purity down to deciles of a percent, but for real work, purity matters most for synthesis and chromatography steps. In practice, minor impurities morph from trivia to trouble depending on what you plan to do—getting clean separations or prepping for NMR runs means different expectations.
How It’s Made: Behind the Reaction Glass
Diisoamyl ether doesn’t tumble off trees or emerge from the ground; chemists have to coax it into existence. Most commercial routes use dehydration—taking isoamyl alcohol (derived from fermentation side streams or refinery runs) and treating it with an acid catalyst. Sulfuric acid gets the call in most labs. It’s a straightforward reaction: mix, heat gently, and let the ether distill. Some folks detest the sulfur dioxide stink, but nothing else works as well at scale for this specific compound. Practicing hands know to mind temperature and keep oxygen at bay, because runaway side-reactions can turn product into tar. For those after even better yields, tweaks like continuous extraction or using fancier catalysts have seen the light, but for most labs, the old acid-catalyzed method remains the go-to.
Chemical Reactions and Molecular Life
In a world brimming with things that react at the drop of a hat, diisoamyl ether sits in a middle ground. Its main virtue, chemical inertness, gives it value in fields where people need stable media or gentle solvents. It doesn’t hand over protons or chase after nucleophiles like alcohols do. Still, given enough persuasion—say, strong acids or certain radicals—it can crack or split into smaller things, forming isoamyl alcohol, pentenes, or even heavier tars with heat and light. This resistance to reactivity means folks pick diisoamyl ether for processes where they’d rather not trigger side-winding reactions. For phase-transfer, extractions, and some kinds of distillation, it serves well. Beyond the standard, researchers have looked at modifying its structure, usually aiming at new ether families, but those studies rarely break into mainstream use.
Where Diisoamyl Ether Finds Its Purpose
Ask a handful of industry chemists where they see diisoamyl ether at work, and you’ll hear about its mainstay role as a solvent—especially for tasks that need efficiency and a bit of selectivity. In the lab, its non-miscibility with water allows for classic liquid-liquid extraction processes. Extracting precious metals from ore leachates, removing contaminants from reaction blends—the list stretches as long as imagination and process chemistry allow. In some countries, it’s popped up for use in organic syntheses, chromatography, and even fragrance applications, given its unique aroma and volatility. Deeper in the industrial world, specialty applications have tempted researchers, from resin manufacture to fuel additives. Still, nothing so far has pulled it widely outside its comfort zone as a stalwart solvent and extraction medium.
Safety, Compliance, and Working Realities
No story about an ether fits without a word or two on dangers. Flammable liquids can flip from benign to disaster lurking on any bench. Diisoamyl ether—like most ethers—demands respect for its low flash point. Labs keep it in metal or amber glass, tucked away from sunlight and kept dry. Over time, the real sneaky risk surfaces—peroxide formation. Ethers can quietly oxidize, especially near air and light, forming compounds that detonate with a spark or bump. Old instructors drilled routines into us: test aged bottles, never evaporate to dryness, and track storage times. Handling spills takes more than a mop—proper ventilation, spark-free cleanup tools, and never forgetting its volatility or ability to travel vapor-wise to pilot lights. Regulations expect such caution, and firms that slip up usually learn fast from insurance agents or factory inspectors.
From the Lab to the Frontier: What Research Overlooks and Finds
Every new substance tempts researchers with the lure of forgotten uses or overlooked quirks, and diisoamyl ether is no exception. Recent years have seen studies probing its environmental fate, looking at breakdown rates and chances for bioaccumulation, sparked by tougher environmental rules and the green chemistry movement. Not much turns up in terms of toxicity—animal studies show typical patterns for low-molecular-weight ethers, with narcotic effects at high doses and limited chronic exposure studies. This gap stands out—no one wants to spark a regulatory scramble after decades of routine use. New solvent guides now talk up greener, safer alternatives, and diisoamyl ether lands somewhere in the middle, neither friendliest nor most hazardous. Researchers dabble in modifying its core, nudging its properties to match modern needs—more stability, less flammability, higher selectivity. Some forays into using diisoamyl ether in nanomaterials, separation science, and even bioprocessing keep appearing in specialty journals, hinting at futures that might materialize.
Toxicity and Environmental Threads
Toxicity remains the card few want to play. Acute exposure risks mirror those of other simple ethers—dizziness, headaches, and in high enough vapor, unconsciousness. Chronic exposure studies in humans remain thin on the ground. A handful of animal trials suggest low acute toxicity, but no one’s wasting time splashing the stuff around. Skin contact causes dryness and mild irritation, nothing out of line with its class. Environmental impact boils down to volatility and breakdown; diisoamyl ether evaporates fast, and light plus oxygen seem to degrade it with time. There’s some concern over water contamination near manufacturing sites, and environmental chemists keep watch for any signal in bioassays. So far, nobody’s flagged widespread risk, but it pays to keep an eye out as usage patterns change.
Tomorrow’s Prospects: More Than Routine
Industries stick with diisoamyl ether because it keeps working and fits existing processes with little fuss. Green chemistry pressures continue to mount, with calls for solvent recycling, safer alternatives, and tighter waste controls. Some speculate diisoamyl ether’s structure offers routes to modify its profile—perhaps building a molecule that mixes easier with water, or resists peroxide build-up. Advances in catalysis could streamline its preparation, making production cheaper or less risky. Demand for better separations in pharmaceuticals and materials science might keep diisoamyl ether relevant, at least for research and niche use. If history hints at anything, compounds with a balance of stubborn reliability and quiet adaptability don’t vanish overnight. Still, any future depends on keeping it practical and safe—less about being dazzling than about showing up and doing its job, year after year.
What is Diisoamyl Ether used for?
What Diisoamyl Ether Means Beyond the Lab
Diisoamyl ether doesn’t pop up in everyday conversation, but it finds its way into some places that deserve more attention. Think of it as one of those chemicals that the people behind the curtains rely on — always working, rarely getting credit. It works as a solvent with a job to do in chemical synthesis, extraction, and even analysis. The stuff has staying power because it does what other solvents can’t, thanks to its low water solubility and high hydrophobicity. That makes it perfect to pull out organic compounds from a mixture, like gold miners panning for flecks in muddy water.
Shaping Modern Industry
Chemical laboratories and plants put diisoamyl ether to work as a separation tool for extracting organic molecules. In my own years dealing with extractions, I ran up against plenty of solvents, but few separated organic and inorganic layers as cleanly as diisoamyl ether. In the pharmaceutical world, where the right extraction means the difference between a clean product and a mess, this stuff counts. Even small changes in solvent quality can cause major headaches down the line, so people notice when diisoamyl ether shows up in the bottle.
Manufacturers reach for it in places where purity matters. From synthesizing specialty flavors for food to making certain agrochemicals, diisoamyl ether pulls its weight as a process solvent, not just an afterthought. When researchers need to conquer stubborn mixtures and improve yields in reaction work-ups, this chemical often gets a spot in the protocol.
Need for Safety and Environmental Awareness
Reliance comes with responsibility. Diisoamyl ether brings fire risks because of its low flash point — I’ve seen enough near misses in fume hoods to respect how flammable it is. Mishandling open bottles or careless storage doesn’t just threaten equipment; it endangers anyone nearby. In a world still waking up to the importance of occupational health, many people underestimate the cumulative toll of chronic solvent exposure. The sharp, sweet odor serves as a warning, but not everyone pays attention until headaches or worse set in. Proper training, ventilation, and storage cut down on avoidable injuries.
Besides the human angle, there’s the land and water where this stuff can end up. Industrial waste streams sometimes leave traces behind if disposal routines get sloppy. Nobody wants persistent, volatile organics leaking into soil. More companies need to upgrade their waste management and invest in recovery setups that minimize environmental fallout. Regulatory oversight and routine audits can help. Some labs and factories already invest in solvent recycling. Yes, it takes extra work, but the relief on both the conscience and the bottom line grows apparent once less waste rolls out the door.
Room for Smarter Use
Diisoamyl ether earns its place in chemistry, but like most chemicals with a strong profile, its story doesn’t stop with its utility. People in chemistry can push further by tightening up how they use and reuse solvents, looking for greener alternatives, and actually listening to those safety talks. Cleaner protocols and better stewardship add more value than just cutting costs — they keep labs humming, people healthy, and the ground clean for those coming up next.
What is the chemical formula of Diisoamyl Ether?
Digging into Chemistry Basics
Diisoamyl ether pops up mostly in labs and specialty manufacturing, and not everyone has heard of it. Even among chemistry majors, plenty of folks can rattle off the formula for sodium chloride without blinking, but ask about diisoamyl ether and you’ll see more hesitation. So, what is it? To put it plainly, the chemical formula for diisoamyl ether is C10H22O. That tells us the molecule contains ten carbon atoms, twenty-two hydrogen atoms, and one oxygen atom. This structure puts it among the family of ethers, notable for their oxygen “bridge” linking two carbon chains.
Knowing What’s Inside Matters
From firsthand experience, back in my organic chemistry days, knowing the formula often meant the difference between a successful reaction and hours of wasted time. Diisoamyl ether, with its structure, ends up being pretty nonpolar, which makes it good for separating or extracting oily components in mixtures. This single oxygen atom handles the bulk of the reactivity, and the rest of the molecule helps it mix with certain chemicals and not others. So much of applied chemistry depends on those small differences — one missing hydrogen or carbon, and suddenly the behavior changes.
Practical Uses Show Why the Details Count
In the real world, workers use diisoamyl ether mostly as a solvent. Its main job? Pulling out certain chemicals during processing, especially in fine chemical manufacturing and refineries. A chemist in an industrial plant cares about those atoms because the slightest mix-up in the formula can lead to totally different results. Take another ether with only one extra carbon. The boiling point climbs, and it doesn’t extract the same set of ingredients. If you’ve handled chemical extractions, you know that every new solvent calls for double-checking the safety data sheets. Diisoamyl ether, thanks to its carbon-rich structure, floats on water and often gets chosen for nonpolar compounds that don’t dissolve in alcohols or other common solvents.
Safety Ties Into Knowing the Formula
Understanding chemical formulas doesn’t just help with the math. It plays a big part in working safely. Diisoamyl ether can form peroxides, which, given enough time or exposure to air, become explosion hazards. People need to know exactly what’s in their containers, how it reacts, and how long it’s been stored. In one industrial lab I worked with, a colleague made the mistake of treating an ether the same as every other liquid in the storeroom, storing it in a clear bottle under a sunny window. They discovered a layer of crystals at the bottom. That was a close call avoided only because a more experienced chemist caught the mistake and recognized the risk from the formula.
Improving Awareness and Handling
Today, information about chemical formulas is just a click away, but carelessness still creates problems. Sharing clear, dependable data and making chemical safety training routine both help. Using better labeling and chemical tracking software also reduces the chance of dangerous mishaps. For students and seasoned chemists alike, double-checking a chemical’s structure before use remains a simple way to protect people and keep the science moving forward.
Is Diisoamyl Ether hazardous or toxic?
Understanding the Substance Beyond the Lab Bench
Diisoamyl ether pops up in specialty labs and industry settings, especially where chemists look for a way to separate or extract things that don’t mix well with water. Its fruity smell and low boiling point might seem harmless, but the real story sits in how people work with it and what risks actually come up in real-world use.
Safety Starts with How It Behaves
Diisoamyl ether shares a reputation among ethers: it’s flammable, volatile, and can form explosive peroxides over time. Stories from the lab echo warnings on data sheets—storing it for months can lead to precarious peroxide buildup, especially if bottles are left half-empty. I remember colleagues testing for peroxides before using older supplies, just in case. Ether container explosions, though rare, still make folks triple-check before opening anything that’s been forgotten in the back of a cabinet.
Health Risks from Smell to Skin
A whiff of diisoamyl ether travels fast across a lab, sometimes triggering headaches or dizziness. The fruity aroma feels misleading; it’s easy to forget there’s a risk of irritation if someone breathes in concentrated vapors for too long. Direct skin contact often means redness or itchiness, especially for those with sensitive skin. If it splashes into the eyes, it stings badly and needs immediate washing to avoid real trouble.
Puts Pressure on Good Ventilation and PPE
Busy technicians often work behind fume hoods and don gloves—not because they’re overly cautious but because past slip-ups have taught some tough lessons. Even with training, folks can get careless. A cavalier approach to ether spills and poor housekeeping multiplies the risk. Years spent in university labs made me appreciate strict habits: labeling old bottles with the date opened, never skipping daily hood checks, and keeping solvents as cold as possible to slow evaporation.
Environmental Impact Ties Back to Human Habits
Any spills end up not just in the lab but in drains, which can raise bigger flags for water systems and aquatic life. Diisoamyl ether doesn’t easily dissolve, so it floats and lingers if not handled right. Waste programs often drive home the point: solvents belong in special bins, and regular audits keep that workflow running. My own stint helping with hazardous waste disposal taught me that skimping on procedure always costs more in the end—ergonomic shortcuts only fuel fires, literally.
Clear Steps to Reduce Hazards
People using diisoamyl ether need more than just warnings. Training should include peroxide testing and clear protocols for what to do with aged stock. Supply storage should keep sunlight out, limit oxygen, and stick to small containers if possible. Community colleges and research facilities teaching chemistry can help build safer habits early on.
Diisoamyl ether isn’t the most toxic thing on a chemist’s shelf, but it can become dangerous through inattention. Learning from history and staying up-to-date with disposal methods and storage keeps accidents away. Science always comes with a dose of risk, but respect and routine cut that risk way down.
How should Diisoamyl Ether be stored?
Risks You Should Not Ignore
Diisoamyl ether is a clear liquid, often used as a solvent or in chemical syntheses. It has that distinct smell you don’t forget, sort of fruity and faintly pungent. But behind the aroma, there’s a real risk sitting in any lab or facility using it. Fires, explosions, and health problems follow sloppy storage. I once walked into a chemical stockroom during college and caught a whiff. The container’s lid looked loose; a lab-mate shrugged it off. We all learned quickly after a safety lecture that carelessness around these solvents could cost someone more than a ruined experiment.
Flammability and Volatility: Always a Concern
Diisoamyl ether catches fire fast. The flash point sits low, making it easy for vapors to ignite from a spark, static, or even a warm light bulb too close. I’ve seen enough fire drill mishaps to take flammable storage seriously. This chemical belongs in a flame-proof, dedicated safety cabinet, away from heat sources. You might want to toss it on a general storage shelf, but vapors find their way out sometimes, mixing in with the room air. Once, I saw a cabinet door left open in another lab, and a static zap set off a tiny firework show—that burned a notebook to ash and cleared out the building.
Tight Seals Outperform Any Quick Fix
Containers should fit tight, and caps or seals must do their job. Sitting Diisoamyl ether in a bottle with a chipped rim or cracked plastic invites trouble. The liquid forms dangerous peroxides as it reacts with air over time. These build up, forming solids around the cap. Unscrewing that cap after a few months can trigger an explosion. You learn respect for chemical storage by handling just one bottle that’s sweated out some crystals near the lid.
Sunlight and Heat Don’t Help Anyone
Don’t store this stuff near windows, radiators, or in sunrooms. Sunlight breaks down the ether, making it degrade faster. Heat speeds up evaporation and increases internal pressure, risking leaks or ruptures. Back in my research days, one grad student left a solvent rack too near a window. The afternoon sun warped a bottle, and the resulting fumes lingered in the hallways all week.
Labeling Isn’t Busywork
Every bottle should have a clear, dated label. This lets you spot the old stock and toss anything past its use-by date. Proper labeling also means you know what you’re reaching for—more than once, I watched people confuse similar bottles, risking a bad chemical mix.
Storing with Awareness: A Responsibility
Storing diisoamyl ether safely comes down to knowledge and discipline. Current safety data tells you which cabinet you’re after—explosion-proof, ventilated, and far from acids or oxidizers. Inspections every month cut the risk of surprise peroxide build-up. Some labs keep small, fresh stocks on hand, splitting large orders among teams to avoid long-term storage. Local policies often require disposal or testing for peroxides once the chemical gets past six months.
Better Habits Save More Than Products
No shortcut or excuse justifies dodging basic care. Chain-of-custody logs, regular safety checks, and training sessions help everyone respect the risks. Diisoamyl ether needs more than a shelf in the corner; it demands respect for people’s lives, lab investments, and health. Look after these things—the difference between “just another bottle” and a major emergency rides on small daily habits.
What are the physical properties of Diisoamyl Ether?
An Unfamiliar Liquid in the Lab
Not everyone has handled bottles labeled “Diisoamyl Ether” in a research lab. My first run-in with it brought an unexpected mix of curiosity and caution. It’s clear from the outset—this isn’t your everyday ether. Diisoamyl Ether, known in chemical circles for its unique structure, is a colorless liquid that carries a sweet, slightly fruity aroma. That scent sometimes lingers long after the bottle’s been closed, much to the chagrin of anyone without a fondness for chemistry’s weirder smells.
Evaporation and Flammability: Not to Be Ignored
One’s nose isn’t the only thing it sneaks up on. Exposure in open air doesn’t last long, thanks to its fairly low boiling point—right around 160 °C. Don’t let the number fool you; compared to many solvents, it still turns to vapor easily enough to demand respect. On a dry summer day, a spill disappears faster than you might expect. This quick evaporation comes with a downside: extreme flammability. Diisoamyl Ether catches fire with little provocation, so it absolutely belongs far from open flames or heat sources. I’ve seen more than one student learn that lesson the hard way.
Density and Solubility: Like Oil in the Water
Pour Diisoamyl Ether into water, and you’ll spot droplets float to the top. This ether’s density sits around 0.77 g/cm³ at room temperature—noticeably lighter than water. In practical terms, the two don’t mix. You find yourself dealing with two distinct phases. That separation helps if you’re extracting organic compounds, since the ether brings many stubborn molecules along for the ride out of aqueous solutions. At the same time, it tells you storage and cleanup aren’t as simple as rinsing everything down the drain.
Viscosity and Everyday Handling
The viscosity of Diisoamyl Ether lands lower than olive oil but above the lightest solvents. It pours out quick, though not as fast as its more volatile cousins. Handling it in glassware, especially when precision matters, is less troublesome than wrangling runnier substances like diethyl ether. Yet, care found me wiping up messes from oily residues more than once. That slightly thicker consistency also means spills spread slower, buying a tiny bit more time for containment.
Why Physical Properties Matter in Real Work
There’s no substitute for understanding what a solvent offers before you pick up the bottle. Diisoamyl Ether’s distinctive boiling point and density can offer a cleaner separation than lighter ethers in some setups. Chemists in pharmaceuticals or fragrance creation sometimes reach for it, especially when less volatile options fail to pull the compounds they need. Fewer accidents happen when everyone in the room respects the fire risk, ventilates workspaces, and stores ethers under nitrogen or in tightly sealed containers away from light.
Choosing Storage and Disposal Wisely
Its physical quirks also demand smart disposal practices. Over time, ethers can form peroxides, raising the explosive danger. Regular testing for peroxides, prompt disposal of old stock, and using proper chemical waste channels keep trouble at bay. Knowledgeable lab techs and safety officers play a huge part here—one forgotten bottle tucked in the back of a cupboard has spelled disaster more than once.
Real Responsibility in the Chemistry World
Digging into a chemical’s physical traits goes far beyond passing safety quizzes. Each property tells a story—about volatility and risk, about how a chemical can help or harm, about how to keep workspaces safe. My experience with Diisoamyl Ether reminds me that the clear liquids in amber bottles can shape everything from the success of the day’s experiment to the safety of everyone on the team.
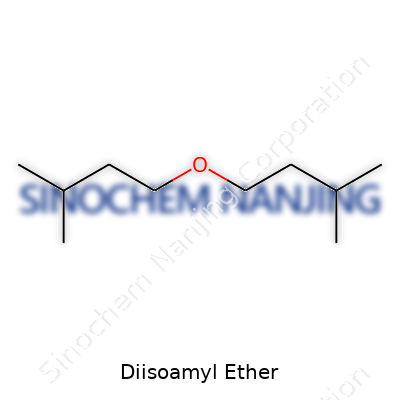
| Names | |
| Preferred IUPAC name | 3-methoxy-2,4-dimethylpentane |
| Other names |
Ethers,diisopentyl Diisopentyl ether Isoamyl ether |
| Pronunciation | /ˌdaɪˌaɪsoʊˈæmɪl ˈiːθər/ |
| Identifiers | |
| CAS Number | 2206-27-1 |
| Beilstein Reference | 1697552 |
| ChEBI | CHEBI:38312 |
| ChEMBL | CHEMBL137643 |
| ChemSpider | 15013 |
| DrugBank | DB14174 |
| ECHA InfoCard | 100.130.782 |
| EC Number | 203-561-1 |
| Gmelin Reference | 113093 |
| KEGG | C19661 |
| MeSH | D000384 |
| PubChem CID | 8097 |
| RTECS number | KI0175000 |
| UNII | H2R8Q37030 |
| UN number | UN1161 |
| Properties | |
| Chemical formula | C10H22O |
| Molar mass | 202.36 g/mol |
| Appearance | Clear colorless liquid |
| Odor | ether-like |
| Density | 0.783 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 3.68 |
| Vapor pressure | 0.74 mmHg (20 °C) |
| Acidity (pKa) | pKa ≈ 35 |
| Magnetic susceptibility (χ) | -77.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.410 |
| Viscosity | 1.98 cP (20°C) |
| Dipole moment | 0.63 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 314.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -393.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7213.3 kJ/mol |
| Pharmacology | |
| ATC code | Not assigned |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H304, H336, H411 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P370+P378 |
| NFPA 704 (fire diamond) | 1-3-2-0 |
| Flash point | 39 °C |
| Autoignition temperature | 370 °C |
| Explosive limits | 1.1–6.9% |
| Lethal dose or concentration | LD50 (oral, rat): 4,860 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat): 1,050 mg/kg |
| NIOSH | JN8575000 |
| PEL (Permissible) | PEL: 100 ppm |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | IDLH: 500 ppm |
| Related compounds | |
| Related compounds |
Diethyl ether Diisopropyl ether Methyl tert-butyl ether tert-Amyl ethyl ether |

