Dihydroxyaluminum Aminoacetate: A Closer Look at a Pharmaceutical Mainstay
Historical Development
The story behind dihydroxyaluminum aminoacetate reaches back to the search for safer, more effective antacid treatments in the mid-twentieth century. Doctors and pharmacists wanted an aluminum compound that could buffer stomach acid without the problems associated with older agents. Before dihydroxyaluminum aminoacetate entered the clinic, treatments often relied on sodium-based antacids or simple aluminum hydroxide, both carrying their own baggage. Sodium-based antacids could disrupt heart health and blood pressure. Simpler aluminum salts raised concerns over constipation and phosphate binding. Through a bit of trial and error in pharmaceutical labs, chemists developed this amino acid-derivatized aluminum compound, which featured gentler action and broader safety. It quickly found a home in chewable tablets and suspensions, quietly building its reputation through decades of use.
Product Overview
This molecule blends an aluminum base with glycine, a naturally occurring amino acid. Most pills on the market show it as a white or off-white, tasteless powder pressed into tablets or mixed into oral suspensions. Simple as it seems, every pharmacist knows its value in providing reliable acid buffering. Drug makers favor it not just for its effect on acid but also for its chemical stability and compatibility with other antacids or digestive agents like magnesium carbonate. The compact size and dependable shelf life set it apart from many alternatives, making it a workhorse in GI clinics.
Physical & Chemical Properties
This compound typically arrives as a free-flowing white powder, not especially sticky and without an obvious odor. Its solubility stands out: while modest in water, it dissolves a bit more in acidic environments, giving it the slow-acting, sustained neutralization that patients appreciate during a reflux attack. Each gram contains about 12% available aluminum. The presence of glycine alters its crystal structure, reducing the astringency and abrasive mouthfeel found with simple aluminum hydroxide. It holds up well in both tablets and liquids, resisting breakdown at room temperature. This stability helps keep label claims meaningful, even with months of storage on pharmacy shelves.
Technical Specifications & Labeling
Manufacturers set strict technical parameters for this compound. Minimum available aluminum content and glycine ratio must stay within narrow limits on every batch. Moisture must stay below a given threshold to prevent caking and to maintain easy blending into finished tablets. Drug labels spell out the elemental content, total weight per dose, and warnings about potential aluminum accumulation in patients with kidney issues. Reputable firms back up their labels with certification for each lot, listing test methods for purity, loss on drying, and freedom from heavy metal impurities. In my own work, I’ve seen how clear technical specs reassure doctors and patients worried about what really goes into their medicine.
Preparation Method
The main preparation involves a reaction between aluminum hydroxide and glycine in water, kept at a controlled pH. The solution forms a white precipitate—dihydroxyaluminum aminoacetate itself—which gets filtered, washed, and dried at low temperatures. Each step matters: excess heat can break down the product, reducing its buffering ability. These days, most plants use stainless steel reactors and automated controls, minimizing exposure to airborne contaminants. Every batch undergoes close testing to confirm that the final product remains free from unreacted starting materials and that key properties stay within limits. Methods have evolved to use less water and create less waste, which helps with cost and environmental impact.
Chemical Reactions & Modifications
Even though its main job happens as an antacid, this compound doesn’t stand still in chemical development. Some research outfits push boundaries by altering the glycine-to-aluminum ratio or by swapping glycine for other amino acids, hoping to fine-tune the acidic response for new therapeutic candidates. Certain modifications can shift the dissolution rate or reduce the likelihood of side reactions in the gut. Labs also study its behavior mixed with other gastrointestinal agents, such as simethicone or magnesium hydroxide, tracking whether the blend holds together chemically and physically. These tweaks may not always make the news, but they shape everything from formulation stability to patient comfort.
Synonyms & Product Names
Pharmacists recognize this compound under several names, depending on the region. You’ll find labels listing “Dihydroxyaluminum Glycinate,” “Aminoglucinic Aluminum,” or “Aluminoglicinato” in European and Latin American makets. Occasionally, product inserts use the shorthand “DHAA” or “Algenic acid.” Despite the naming variety, the core product shares chemical identity and expected properties, with regulatory agencies tying approval to clear specifications for each synonym. I’ve learned the hard way that confusion sometimes arises as a result—especially when patients import medicine from abroad—so using trusted reference resources becomes key to sorting out the names.
Safety & Operational Standards
Safety draws enormous attention in every stage of this compound’s life cycle. Manufacturers stick to GMP-certified processes, regularly audited by regulators who test for both elemental impurity and microbial contamination. Handling the raw powder requires basic dust controls to avoid inhalation. Packaging lines use sealed systems and controlled humidity, reducing the risk of caking before the drug reaches patients. On the clinical side, prescribers monitor high-risk patients—especially those with impaired kidney function—since aluminum clearance drops sharply in renal failure. Reliable adverse event tracking systems offer another safeguard, helping spot trends and keep doctors and patients informed about the risks of long-term or high-dose use.
Application Area
Most commonly, this aluminum compound finds its use in antacid tablets, both as a solo active ingredient and in blends with other acid-neutralizing agents. It soothes symptoms of heartburn, peptic ulcers, and esophagitis, sparing patients the discomfort of rapid pH changes. Because of its modest reactivity and milder effect on intestinal transit, many clinicians recommend it to patients who can’t tolerate magnesium-containing products. Outside the GI clinic, this compound sometimes serves in veterinary practice, especially for acid-related disorders in small animals. Some investigators have poked at its use in topical wound care, but most mainstream application sticks to gastrointestinal health.
Research & Development
The R&D focus keeps shifting with emerging needs. Drug formulation specialists look for ways to pack more drug into smaller tablets, tweak the flavorings, and speed up disintegration without sacrificing shelf life. One persistent development challenge is refining the molecular structure to balance acid neutralization with minimized aluminum absorption. As regulatory guidance evolves, companies invest in new analytical methods to precisely measure aluminum release and to guarantee absence of risky impurities like lead or mercury. A few research teams have started looking at nanostructured forms, which promise faster action or targeted delivery. Most published advances turn up in pharmaceutical conference proceedings before they hit the mass market.
Toxicity Research
Long-term toxicity work underpins every antacid class. The aluminum content, while helpful in the gut, raises red flags in the context of impaired kidney function or unusually high doses over weeks or months. Ongoing studies track aluminum build-up and look for subtle effects on bone health, central nervous system, and mineral metabolism. Dedicated research investigated whether chronic use contributes to dementia risk or worsens phosphate depletion, especially in elderly patients. So far, mainstream clinical studies suggest this compound compares favorably to similar aluminum-containing drugs, provided total daily aluminum intake stays within limits. Patient education and real-world surveillance matter as much as the original toxicity testing.
Future Prospects
The pharmaceutical field always seeks to improve patient comfort, safety, and adherence to therapy. Future research could help doctors deliver more targeted antacid therapy using personalized drug blends and improved excipients, perhaps combining dihydroxyaluminum aminoacetate with newer agents that work faster or last longer. Safe, low-aluminum formulations will stay a research focus, reflecting public concern about heavy metal exposure. As health systems in emerging economies grow, the need for reliable, cost-effective antacid products keeps this compound in the spotlight. Sustainability pressures may drive production methods to produce less waste and use greener energy sources. Even with fancier antacids on the shelf, demand holds steady for dependable medicines with known track records, and dihydroxyaluminum aminoacetate clearly fits that niche.
What is Dihydroxyaluminum Aminoacetate used for?
A Closer Look at a Trusted Antacid
Dihydroxyaluminum aminoacetate pops up in many households as a quiet ingredient most folks would have trouble pronouncing, but its job is pretty straightforward—helping calm acid troubles in the stomach. Doctors and pharmacists offer it to people with complaints of heartburn, sour stomach, or an overfull feeling after meals. I remember my own grandfather, who grew up in a time when heavy, greasy meals were almost a way of life, swearing by a white bottle in his medicine cabinet marked for antacids. He might not have known the long chemical name, but he counted on it to settle his gut after stews and pies.
Why This Compound Works
In the simplest terms, dihydroxyaluminum aminoacetate acts like a chemical sponge for stomach acid. The aluminum in this compound reacts with hydrochloric acid in the gut, neutralizing it, and the glycine part helps buffer and soothe. For people leaning over the bathroom sink after a night of pizza and spicy wings, relief from the burning comes thanks to this reaction. Statistics show nearly 60 million Americans experience heartburn at least once a month, according to the American College of Gastroenterology. Products with this compound soften that burden for millions—just as it did for my own relatives. It’s remarkable how easily many of us take for granted a settled stomach, until discomfort hits.
Beyond Heartburn: Other Uses
Some patients battling ulcers or gastritis also find comfort from dihydroxyaluminum aminoacetate. Doctors add it to the treatment plan because the gentler action makes it less likely to cause constipation compared to many other aluminum antacids. Not every antacid is created equal; some people deal with bad side effects. This compound feels like a bridge—offering relief without doubling down on new problems.
Safety and Concerns
Like anything taken regularly, there are reasons to be careful. Overusing aluminum-based antacids can cause troubles, including changes in bone health or phosphate levels, especially in people with kidney impairment. Flashbacks to my own pharmacy days bring up customers who asked if daily antacid use was fine. The lesson is steady: relief from heartburn should not overshadow the need to talk to a healthcare professional if these symptoms become part of daily life. Chronic acid problems sometimes mean something bigger is happening, not just spicy food.
Looking for Better Relief: New Developments and Simple Solutions
Scientists keep tweaking old favorites to help folks avoid building up too much aluminum or calcium. Combinations with magnesium or newer formulas with less risk are gradually showing up. Even so, nothing replaces common sense. Regular use of any medication—even a trusty old antacid—calls for paying attention to diet, reducing late-night snacks, cutting back on triggers like coffee, and taking time to talk to a doctor. My loved ones learned to balance quick fixes with these daily adjustments, and many others could benefit from that same approach.
A Simple Pill with a Big Role
Life brings enough discomfort. Having something safe and reliable for stomach acid makes a difference for people who want simple relief that works. Dihydroxyaluminum aminoacetate doesn’t grab headlines, but it quietly supports the daily routines of millions, just like it did in my own family’s kitchen.
What are the possible side effects of Dihydroxyaluminum Aminoacetate?
Why People Use Dihydroxyaluminum Aminoacetate
Dihydroxyaluminum aminoacetate often pops up in antacid tablets and liquid formulas at the pharmacy. Most folks reach for it hoping to settle an upset stomach or dial down that burning feeling after a rough meal. The compound does the job by neutralizing excess stomach acid, offering fast relief for conditions like heartburn or indigestion.
The Not-So-Pleasant Trade-Offs
As useful as it can be, dihydroxyaluminum aminoacetate can bring its own share of issues to the table. On a personal note, I remember a family member picking up an over-the-counter bottle after a heavy dinner, sure it would smooth things over. By the next morning, she found herself dealing with constipation. This isn’t rare—aluminum-based antacids, including this one, tend to slow down the digestive tract. Some users may only notice mild changes, while others can get quite uncomfortable, especially with regular use.
Bloating can follow, making clothes feel a bit tighter than usual. Nausea or stomach cramps sometimes show up, too. More sensitive individuals might notice a chalky taste in the mouth, or even mild vomiting if they’re unlucky. Data from multiple reports highlight constipation and bloating as the most common gripes, especially among older adults and anyone who needs to limit their fluid intake or who struggles to stay hydrated. The aluminum content can interfere with how the kidneys handle waste, so anyone with kidney issues should be careful.
Less Common, More Serious Issues
The risk doesn’t stop with the gut. Anyone using antacids with dihydroxyaluminum aminoacetate over a long period risks higher levels of aluminum in the blood. In rare cases, this can build up, especially for those with weakened kidneys, leading to muscle weakness, bone pain, or even problems with memory and focus. This kind of toxicity doesn’t just go away with a glass of water, and it demands medical attention. High aluminum levels threaten bone health and have been linked to neurological issues. Also, these antacids can mess with absorption of other medicines, from antibiotics to iron supplements, so timing doses or talking with a healthcare provider makes sense before starting regular use.
Staying Ahead of the Problem
Taking a close look at labels and understanding what’s inside really helps. Many folks trust over-the-counter products, but doctors and pharmacists stay busy fielding questions about safe use. If someone already deals with kidney disease, heart failure, or follows a sodium-restricted diet, a safer approach means picking a different kind of antacid, often one without aluminum or magnesium.
Drinking enough water, moving around regularly, and eating foods with plenty of fiber offer gentle relief from the constipation that follows aluminum-based antacids. Anyone who notices unusual symptoms—persistent muscle aches, weakness, confusion, or changes in bowel habits—should speak up and seek advice. Keeping track of every medication and supplement in use helps avoid those hidden drug interactions that can cause trouble down the line.
Looking for Alternatives
Some people find relief using calcium carbonate antacids or non-pharmacological approaches, like cutting back on spicy foods and late-night snacks. Non-drug methods don’t cause the same risk of constipation or aluminum build-up, and can sometimes manage symptoms just as well for mild heartburn.
Making informed decisions about what goes into the medicine cabinet, paying attention to how the body reacts, and checking in with a professional lays the groundwork for safer relief. No one wants stomach pain—but no one wants worse side effects either.
How should Dihydroxyaluminum Aminoacetate be taken or administered?
Getting Real With It
Dihydroxyaluminum aminoacetate doesn’t turn up at dinner-table conversations, but for many people struggling with heartburn and indigestion, this antacid absolutely matters. I remember my uncle grabbing for his trusty bottle of chewable tablets right after Sunday lunch, swearing by the fast relief. As he taught me, knowing how to use stomach remedies turns a dinner from miserable to manageable.
Real Instructions You Need to Know
Nothing turns a handy drug into a health risk faster than guessing the directions. Read the label, even if you think you’ve seen it all before. The instructions give the amount, how often to take it, and whether your stomach should be empty or full. For Dihydroxyaluminum aminoacetate, most brands suggest tablets or liquid, each needing its own approach. Chew the tablets well. Don’t swallow them like painkillers or you lose the effect. Liquid should be measured—not eyeballed. No one needs a “that looks about right” moment with medicine.
Timing and Food: Not Just Details
Many antacids work best either before symptoms start or when they first appear. After slogging through a week of upset guts, I realized late-night snacks bring pain that doesn’t back down. Taking Dihydroxyaluminum aminoacetate after a heavy, greasy meal gave the quickest relief. Still, I learned the hard way that guzzling this stuff all day instead of changing eating habits doesn’t get long-term results. The package and your pharmacist know the score—follow their schedules.
Who Should Really Think Twice
Easy access can be deceiving. People with kidney trouble can’t handle extra aluminum in their system. My neighbor told me she learned after a scare at the clinic—her doctor said too much antacid stacked up and spelled new medical problems. If you notice constipation or black stools, it isn’t time for a new brand, it’s time to call a doctor. No relief after two weeks? More of the same won’t fix the root problem. Sometimes a persistent stomach ache means there’s something bigger going on.
Interactions: Some Facts Matter More Than Labels
Sharing a medicine cabinet brings its own drama. Some drugs just don’t mix, especially if you’re juggling chronic meds or antibiotics. Dihydroxyaluminum aminoacetate soaks up some medications, making them less useful. Take other tablets either a couple hours before or after. Make it part of your daily rhythm—turn reminders on your phone if that helps, or stick notes to the fridge. Messing up this timing cheats your main medication of its job.
What Makes a Difference? Trust and Good Habits
Pharmacists aren’t just price checkers behind the counter. I’ve walked into a store with a list of questions and left knowing exactly what to watch for with Dihydroxyaluminum aminoacetate. If something feels off, don’t guess. Trust your instincts, and trust the people with training who’ve seen every mistake before. Too many folks keep quiet and hope for the best. Open your mouth, ask the hard questions, pay attention to your body’s signals. Relief is important, but health lasts longer.
Are there any drug interactions with Dihydroxyaluminum Aminoacetate?
What This Antacid Really Does
Dihydroxyaluminum aminoacetate offers relief for folks dealing with indigestion, sour stomach, or heartburn. Like other aluminum-based antacids, it works by neutralizing stomach acid fast. I remember reaching for antacids during nights spent with too much spicy food or too much coffee, thinking little of potential snags with my daily prescriptions. That’s where some trouble can start.
Medication Stacking: Where Things Get Tricky
Folks on several drugs at once know how important it feels to keep prescriptions organized. Dihydroxyaluminum aminoacetate stands as no exception when talking about drug interactions. Throw in something new, and suddenly a treatment plan can spin off course—often in ways that doctors and patients can dodge with a bit of extra info.
Common Drug Interactions and Real-World Issues
Taking this aluminum-based antacid too close to certain medications can block their absorption in the gut. For example, people using antibiotics like tetracycline or ciprofloxacin should pay close attention. Both meds depend on being absorbed in the stomach or small intestine. Once they bump into aluminum, a chemical reaction can make them nearly useless, leaving an infection untreated. Pharmacies often put warning labels on these meds for this reason.
Thyroid medications, such as levothyroxine, also run into problems. If someone takes their antacid around the same time as their thyroid pill, they might be chasing test results in circles with little progress. Proper spacing—at least two hours before or after—is more than a technical detail; it means daily pills do their job without interference.
For those living with kidney problems, extra caution steps in. Since the kidneys clear out aluminum, trouble can build up if the organ can’t do its work. Toxic levels can develop quietly over time. This risk touches anyone with chronic kidney disease or those on dialysis, a warning not to be ignored even if trouble seems distant.
Doctor Visits and Practical Conversations
Looking at this from personal experience, check-ins with doctors or pharmacists matter much more than most realize. Sharing every supplement and over-the-counter product helps them spot these invisible interactions early. Still, people forget, or sometimes don’t realize that an antacid can block a heart medicine or seizure drug. Work schedules or habits often get in the way of perfect timing, but knowing about a simple two-hour window adds a layer of control to a daily routine.
Steps for Safer Use
Pharmacists now more than ever double-check before dispensing prescriptions. Electronic medical records flag these conflicts automatically in many places. But the strongest partnership always comes from staying curious about everything put into the body—asking questions, reading labels, and making sure no pills get mixed just because they all come from the same pharmacy bag.
With so many medications and supplements around, taking a closer look at what goes down the hatch each day makes a world of difference. Asking your pharmacist about antacids or bringing up stomach trouble during a checkup doesn’t just clear up heartburn. It can keep important treatments running smoothly without any surprises down the road.
Is Dihydroxyaluminum Aminoacetate safe for long-term use?
The Role of Dihydroxyaluminum Aminoacetate
Dihydroxyaluminum aminoacetate slips into many medicine cabinets as an active ingredient in antacid tablets and suspensions. Chemists design it to buffer stomach acid, often providing some quick relief after a big meal. Plenty of people, including myself at one time with a stubborn case of heartburn, turn to these remedies a few days in a row to get through a tough patch. At first glance, products containing this compound seem as safe as anything on the pharmacy shelf, especially with clear instructions and FDA oversight. Still, the question about what happens with regular, long-term use needs more attention.
What Long-Term Use Looks Like
Using antacids for short discomfort is one story, but taking them every day builds a different picture. Research into long-term effects of aluminum-containing compounds raises reason for concern. Dihydroxyaluminum aminoacetate, like other aluminum-based antacids, can gradually contribute to higher aluminum burdens in the body. While our kidneys filter out a lot of this metal, people with kidney problems cannot handle that extra load very well. Too much aluminum in the body has been linked to bone disorders and neurological issues in sensitive groups, especially those with chronic kidney disease or elderly patients with multiple medications in daily routines.
Supporting Facts From Clinical Research
Several studies monitored patients who took these antacids every day over the course of months or even years. Researchers found that most healthy adults can process typical antacid doses without major problems, but they also noted that long-term use has led to aluminum accumulation in bones and brain tissue in rare cases. For reference, a 1996 review in the journal Drug Safety documented instances where patients developed osteomalacia (softening of bones) or even worsened cognitive decline when exposed to high doses over time. That may sound alarming, but the majority of healthy people see less risk, especially if use is only occasional.
Recognizing Warning Signs
While looking at safety, it helps to keep an eye out for symptoms. People who use antacids containing dihydroxyaluminum aminoacetate for weeks or longer and notice muscle weakness, fatigue, or new confusion should talk with their doctor. Most doctors want to know about regular, even non-prescription, medication use since it can interact with other drugs like antibiotics or iron supplements. Over time, these interactions might limit how well your body absorbs other important nutrients.
Smart Use and Alternatives
Turning to dihydroxyaluminum aminoacetate once in a while brings little worry for most people. Long-term, daily use paints a different story. There’s wisdom in reevaluating why someone keeps reaching for antacids. Persistent stomach problems or frequent heartburn often call for a look at diet or stress, and sometimes a check-up with a healthcare provider. Alternatives can include other types of antacids, non-aluminum options, or medications like H2 blockers and proton-pump inhibitors under medical guidance. Sometimes, simple changes like cutting back on caffeine or spicy foods make as much difference as any pill.
Final Thoughts on Safety
Pharmacists and clinicians agree that one size rarely fits all when talking about medicine safety. Most folks tolerate dihydroxyaluminum aminoacetate well in occasional doses, but sticking with it long-term needs a conversation with someone who knows your health history. Staying informed, reading medication labels, and speaking up with questions help lower risks and keep your well-being on track.
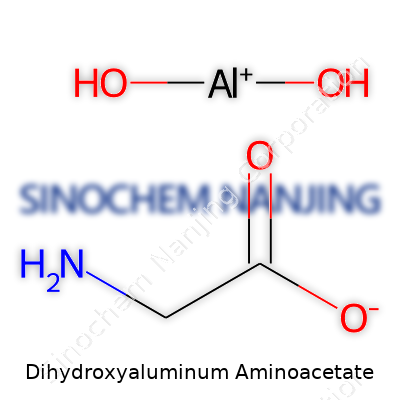
| Names | |
| Preferred IUPAC name | aluminium;2-aminoacetate;dihydrate |
| Other names |
Dihydroxyaluminum glycinate Aluminum, dihydroxy(aminoacetato)- Dihydroxy(aluminum) glycine |
| Pronunciation | /daɪˌhaɪdrɒk.si.əˈluː.mɪnəm əˌmiːnoʊ.əˈsiːteɪt/ |
| Identifiers | |
| CAS Number | 13682-92-3 |
| Beilstein Reference | 1462416 |
| ChEBI | CHEBI:31513 |
| ChEMBL | CHEMBL1201208 |
| ChemSpider | 11804 |
| DrugBank | DB14138 |
| ECHA InfoCard | 08f0b8be-8f0c-4911-a7d1-a6cf43ec99c0 |
| EC Number | 208-236-2 |
| Gmelin Reference | 84854 |
| KEGG | C13768 |
| MeSH | D000326 |
| PubChem CID | 166829 |
| RTECS number | WL6475000 |
| UNII | YK18F2UJ14 |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID6038121 |
| Properties | |
| Chemical formula | C2H6AlNO4 |
| Molar mass | 163.07 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 1.42 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | -2.63 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 5.0 |
| Basicity (pKb) | 8.2 |
| Magnetic susceptibility (χ) | -73.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.497 |
| Viscosity | Viscous liquid |
| Dipole moment | 5.27 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 173 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1512.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1265.6 kJ/mol |
| Pharmacology | |
| ATC code | A02AD05 |
| Hazards | |
| Main hazards | Irritating to eyes, respiratory system, and skin |
| GHS labelling | GHS labelling of Dihydroxyaluminum Aminoacetate: `"Not classified as hazardous according to GHS"` |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | Keep container tightly closed. Store in a cool, dry place. Avoid breathing dust. Wash thoroughly after handling. Use with adequate ventilation. Avoid contact with eyes, skin, and clothing. |
| Lethal dose or concentration | LD₅₀ oral (rat): 6200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 5 g/kg |
| NIOSH | DT2275000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5 mg(Al)/m3 |
| IDLH (Immediate danger) | Not Listed |
| Related compounds | |
| Related compounds |
Dihydroxyaluminum sodium carbonate Aluminum glycinate |

