Diethylstilbestrol: The Rise, Fall, and Rethinking of a Synthetic Estrogen
Historical Development
Diethylstilbestrol, often called DES, found its roots in the late 1930s following a breakthrough by British scientists Charles Dodds and colleagues. Their aim was clear: craft an affordable, potent synthetic alternative to natural estrogens at a time when extracting hormones like estrone from animal urine was neither practical nor efficient. Early studies promised much—by the 1940s, DES hit the medical market as a treatment for a range of hormone-linked problems, from menopausal symptoms to the prevention of miscarriages. Pharmaceutical makers promoted it broadly, and for decades, doctors assured patients of its safety without suspicion that this was an illusion built on limited evidence and trust in novelty. By the 1970s, everything collapsed when links between DES exposure in pregnancy and rare cancers in daughters came to light, shattering faith in what once looked like a medical triumph.
Product Overview
DES is a nonsteroidal synthetic estrogen. Unlike naturally occurring estrogens, DES’s structure skips the steroid nucleus, making it cheaper to produce and easier to modify for industrial and scientific uses. Back in its heyday, DES appeared across tablets, topical creams, and injectables, touching everything from women’s health clinics to livestock feedlots. Its convenience and relatively long shelf life set it apart from older hormone therapies. The U.S. Food and Drug Administration approved DES for pregnancy, menopause, and breast cancer therapy—opening the doors to mass prescription, which by the 1950s, meant millions received DES without second thought. The broad spectrum of uses painted DES as a miracle solution long before its faults became headline news.
Physical & Chemical Properties
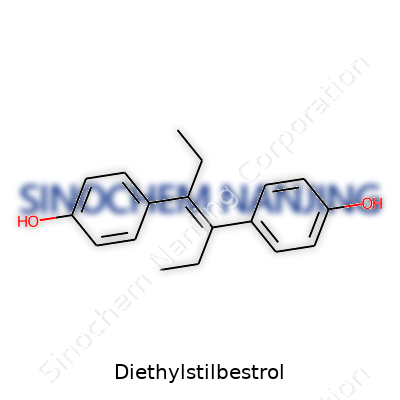
On the lab bench, diethylstilbestrol appears as a pale, crystalline powder, odorless and stable under normal room conditions. Chemists record its melting point between 168 and 173°C, and it dissolves with ease in ethanol, acetone, and chloroform, but much less so in water. The molecular core carries two phenolic rings linked by an ethylene bridge, with two ethyl groups tacked on for good measure, giving DES a chemical formula of C18H20O2. Its potency as an estrogen mimicker comes from the way these rings imitate the binding of natural estrogens to hormone receptors. This physical resilience partly explains why DES lingers so long in both human tissue and the environment.
Technical Specifications & Labeling
Pharmaceutical-grade DES once demanded strict purity, usually not falling below 99%. Tablets most often delivered doses in micrograms or milligrams, with color-coded tablets and vials to flag strengths. Chemical suppliers continue to provide DES for research, strictly labeling containers with hazard codes (GHS), batch records, and safety information as required by OSHA and REACH. Product sheets outline melting point, solubility, storage temperatures (preferably cool and dry), recommended PPE during handling, and disposal instructions—piecing together a level of transparency that was sorely lacking in DES’s early years as a consumer product.
Preparation Method
Manufacturing DES depends on a multi-step organic synthesis process that starts from benzaldehyde. Through a condensation reaction, usually with diethyl ketone and a base, chemists form the central ethylene bridge of the molecule. Purification follows, often using recrystallization or chromatography to reach high purity. Modern labs aim for tighter quality controls and environmental management to reduce byproduct pollution. Despite the simplicity of the reaction from a chemistry perspective, the environmental persistence and bioactivity call for careful oversight.
Chemical Reactions & Modifications
DES reacts readily with strong acids and bases, losing its activity if hydrolyzed or oxidized under harsh conditions. Chemists sometimes tweak the ethyl side arms or swap functional groups to probe new analogues for research: something I’ve seen used mostly in the search for better anti-cancer drugs or alternative hormonal therapies. Modifying the phenolic rings can change receptor binding, while retaining estrogen-like activity. Each tweak, though, comes with the baggage of toxicity-testing—no shortcut exists for learning how even a small molecular adjustment might translate to real-world risks or benefits.
Synonyms & Product Names
DES appears under many names: Stilbestrol, Stilboestrol, and the legal chemical moniker diethylstilbestrol all describe the exact same compound. Drugstores once sold it under brand names including Stilbestrol, Distilbène, and Estrosyn. In research circles, the search for references often turns up registry numbers like CAS 56-53-1 and shorthand like “DES,” but no matter the label, the story beneath the name stays the same—a chemical once trusted for safety, later shown to leave a trail of harm.
Safety & Operational Standards
Safety guidance on DES leaves no room for shortcuts. Even minimal handling calls for gloves, lab coats, and eye protection; spills prompt immediate cleanup and complete decontamination of surfaces to keep particles from entering the respiratory tract or contaminating groundwater. Storage away from direct sunlight and in sealed, labeled containers is the rule in any research setting. Any waste containing DES demands hazardous disposal, never the regular trash or sink. People working in labs today get mandatory training and reminders of DES’s legacy to avoid compounding its past harms.
Application Area
At the peak of its use, DES pervaded both human and veterinary medicine. Doctors prescribed it for high-risk pregnancies, delayed puberty, estrogen deficiency, and the suppression of lactation, believing it prevented miscarriage and fostered healthy pregnancies in “problem” cases. Agricultural practices looked to DES to speed up cattle growth and improve feed efficiency, with little reflection on the hormone’s ecological fallout. Now, DES’s legacy continues only in research—studies on endocrine disruption and cancer biology, instruction in pharmacy and chemistry programs, and toxicology. The hard-earned lesson: even the most promising treatments deserve scrutiny before widespread adoption.
Research & Development
Research around DES never faded, but its focus changed. Decades of epidemiological studies have mapped out the cancer risks for the so-called “DES daughters” and “DES sons”—showing clear spikes in clear cell adenocarcinoma, reproductive tract abnormalities, and fertility issues. Laboratory studies keep exploring how DES changes gene expression, cell signaling, and immune functions—a rich field for those investigating hormone-driven cancers and endocrine disruptors. DES also continues as a benchmark in toxicology, giving researchers a model compound to test the effects of synthetic estrogens on both wildlife and human cell lines.
Toxicity Research
Extensive studies reveal the dark side of exposure—one dose in utero can change a life trajectory. Research shows that fetal exposure causes rare cancers of the vagina and cervix, urogenital abnormalities, infertility, and increased miscarriage rates in both generations directly exposed and their offspring. Animal experiments confirm similar patterns: reproductive system malformations, lowered sperm counts, and hormone imbalances. When I studied toxicology, no other pharmaceutical stood out as the “textbook” example of unforeseen medical disaster like DES did. Over the years, these findings have shaped not just regulatory standards for pharmaceuticals, but sparked new regulatory categories for endocrine-disrupting chemicals found in plastics, pesticides, and more.
Future Prospects
DES shaped the world’s cautionary approach to hormone drugs and persistent chemicals. Regulatory agencies now demand extensive, multi-generational studies before approving similar compounds. The future of DES mostly means research into long-term, transgenerational effects—how one exposure might carry risk all the way down to grandchildren. Ongoing work also looks at remediation strategies for soils and waters contaminated with hormone disruptors. Lessons drawn from the DES catastrophe have informed safer, targeted therapies for menopause and cancer, and encouraged vigilance about new pharmaceuticals. Science looks for ways to detect, neutralize, and prevent exposure in ways few could have imagined back in the 1950s: this vigilance is the enduring legacy of DES’s complicated past.
What is Diethylstilbestrol used for?
Understanding The History Of Diethylstilbestrol
Diethylstilbestrol, or DES, once stood as a beacon of hope in medical circles. Doctors started using it in the 1940s, believing it could help prevent miscarriages and pregnancy complications. In those days, medical advice leaned on trust and new discoveries often jumped from theory to practice faster than they do today. No one could predict the storm brewing beneath early excitement.
The Fallout From Medical Use
Women who took DES during pregnancy learned much later that this compound did not deliver the protection they were promised. Their daughters, instead of benefiting, saw increased risks for a rare vaginal cancer, clear cell adenocarcinoma, and other reproductive tract issues. Sons didn't escape untouched, experiencing a higher rate of testicular problems and other health concerns. The human stories emerging from families dealing with the aftermath of DES use speak louder than any data table. Generations paid a price for what science then saw as a harmless fix.
Other Purposes And The Shift In Perspective
Back before the full range of harms came to light, drug companies also marketed DES for menopausal symptoms, certain cancers, and to stunt growth in very young girls. Farmers used it to bulk up livestock, thinking it safe and effective. Hospitals and clinics knew much less about the long-term consequences of hormone interference. My own experience in health libraries showed me how once-popular treatments can, if unchecked, become sources of regret rather than pride.
Why DES Matters As A Case Study
Many people, especially those born after the early 1970s, learned about DES only during college biology classes or through families affected by the scandal. DES taught both the scientific and medical community about the need for rigorous long-term studies before assuming safety. It shapes how drugs get evaluated and monitored today. I remember reading about the DES tragedies, realizing that every “miracle drug” undergoes lessons that could hurt real people. It's not just a footnote in an old textbook.
Current Perspective On Usage
Doctors no longer prescribe DES for people or animals. The evidence against it stands strong and clear. Regulators pulled approvals and drug guides issue prominent warnings. Despite the ban, the legacy lives on in people known as DES daughters and sons, many of whom seek specialized screening for conditions that remain higher risks because of their mothers’ exposure.
Addressing Ongoing Health Concerns
Healthcare providers now flag anyone with a DES exposure history early and push for honest, clear discussion about risks. People with links to DES in their family need more than reassurance—they want clinics to recognize symptoms early and avoid missing red flags. Cancer societies and women’s health groups push for targeted education. No one wants a repeat of a story where hope turned sour for so many families. Up-to-date, compassionate care should guide every conversation about DES and history’s lessons about trusting “safe” solutions.
Building Better Drug Safety Today
Trust in medicine grows stronger through accountability and transparency. Regulatory bodies test new drugs harder than ever before. Patients—especially those with experience of earlier medical disasters—ask sharper questions and expect clear answers. What happened with DES reminds us that real harm sits behind statistics. Each pill or treatment reaches a living person, and that truth sets medicine’s clearest guidepost.
What are the side effects of Diethylstilbestrol?
History Leaves a Mark
Diethylstilbestrol, better known as DES, has a story tangled with lessons we should not ignore. Doctors once handed it out to millions of women, mostly from the 1940s through the 1970s, aiming to prevent miscarriages and pregnancy complications. The science at that time didn’t reveal the full list of problems DES created. Looking back, the experiences of those exposed—mothers and their children—show how faith in medicine can swing toward regret.
Clear Health Risks
Women who took DES during pregnancy saw their risk of developing certain cancers rise. Studies found a link to clear cell adenocarcinoma of the vagina and cervix. This rare cancer rarely hit women not exposed to DES, and it forced doctors to rethink how they viewed risks tied to hormone treatments. The connection between DES and breast cancer also became plain. Research backed up stories from women—pointing out a stronger likelihood of breast tumors years after their first dose.
Impact Across Generations
What stands out about DES goes beyond the original patients. The daughters of women who took the drug, often called "DES Daughters," deal with a set of problems much different from those faced by their mothers. Reports tell of higher chances of vaginal and cervical abnormalities. For some women, everyday life now involves regular monitoring and biopsies to rule out cancer or keep an eye on unusual growths that wouldn’t have turned up without DES exposure.
Infertility also weighs heavy on many DES Daughters. A study from the National Cancer Institute tracked these women and saw higher rates of miscarriage, premature birth, and ectopic pregnancy. While people often talk about advances in reproductive medicine, these stories often leave out those who still pay for yesterday’s mistakes.
Men didn’t escape the fallout either. DES Sons faced genital abnormalities, such as undescended testicles or cysts on the epididymis. Evidence points to possible lower sperm counts and an increased risk of testicular cancer. Living with these health risks means many never get simple answers about their futures.
Mental Toll and Awareness
Beyond the strictly medical risks, DES forced families to live with years of anxiety about future cancers and birth complications. Growing up, I remember my aunt talking about doctors asking her about “unusual hormone treatments” after her first miscarriage. She never realized it traced back to a tiny pill her mother took decades earlier. That family memory underlines why communication matters, not just from doctor to patient but also across generations.
Staying Honest and Finding Solutions
No one can unwind what happened, but we can keep the lessons in sight. Doctors today track people exposed to DES through special registries and research studies. They share guidelines for screening, especially for cancers and reproductive health issues tied to DES. Honest conversations help. People with a family history of DES exposure need doctors who listen and consider these unique risks.
DES will always carry a heavy legacy. Modern science grows from uncomfortable truths like this. By boosting awareness, supporting long-term studies, and making sure patients understand their own histories, we let the past guide better choices for the future.
Is Diethylstilbestrol still available or approved for use?
History Leaves Scars
Diethylstilbestrol, known as DES, once carried hope for women and doctors. People trusted it to help prevent miscarriages, and drug companies handed out prescriptions without second thoughts. Then studies in the 1970s showed a link between DES and rare vaginal cancers showing up in daughters exposed before birth. Legacies like that don’t fade.
No Longer Approved for Human Use
Current FDA rules tell a clear story. DES no longer holds approval for any use in humans in the United States. That means no doctor with a license can legally write a prescription for DES to help with any condition in people. Europe, Australia, and most countries with strong health regulation took the same path. Now, seeing DES for sale in a human pharmacy anywhere in those countries should set off alarm bells.
Animal Drug Loophole: Still Around, But Changing
DES didn’t disappear overnight, though. Feedlots once used it to fatten up cattle more quickly. The goal: boost profits, put more steak on the table. This practice also ended decades ago. Worries about the same cancers in people turned up evidence that eating animals treated with DES brought its own health risks. As scientists learned more, regulators demanded that ranchers stop using DES in animal feed.
A few countries in Asia and parts of Africa have shown weaker enforcement or slower change. Even there, pressure from scientists and international agencies keeps pushing countries to close the door on hormone use in meat production. For those who travel or do business overseas, knowing the source of your food helps cut exposure.
Health Consequences Linger
Families affected by DES never really escape it. Daughters born to women who took DES face greater risks for certain cancers and infertility issues. The science turned opinion fast. Personal stories stay painful—questions about pregnancy, young people facing serious diagnoses, family records haunted by a drug that doctors once said was safe.
Clinicians still screen these patients for related health issues. Even now, guidelines for the care of sons and daughters of people given DES exist in major clinics. So DES may not show up on the shelf, but its legacy stays active in exam rooms and family histories.
The Role of Trust and Lessons Learned
Every drug tells a story about trust and the costs of mistakes. Regulatory agencies like the FDA now demand more, not less, proof before saying yes. Doctors grew warier about “miracle preventatives” during pregnancy. Society remembers DES as one of the biggest lessons in safety oversight.
Fact-checking and science-based regulation matter. Fast cures steal headlines, but hasty treatments carry a price. Anyone worried about hidden drug residues, whether in older family members or their own medicine, can always check with a pharmacist or doctor. Speaking up protects personal health—and sometimes shapes the rules for everyone else.
Solutions: Keep the Lessons Alive
Transparency and independent science protect public health far more than quick fixes. Drug approval processes must remain tough, no matter how much pressure comes from business or fear. Telling the real stories of drugs like DES builds public awareness and trust in health decisions. Everyone—scientists, regulators, and families—plays a part in that ongoing work.
Can Diethylstilbestrol cause cancer or other long-term health issues?
The Story of Diethylstilbestrol
Doctors started giving Diethylstilbestrol (DES) to pregnant women in the mid-20th century because it was supposed to prevent miscarriages and other complications. Well over a million women around the world took this synthetic estrogen. People trusted the science of the day. The results turned out to be far from what was promised.
Cancer Risk Runs Across Generations
By the 1970s, clear signs pointed to trouble. Women whose mothers took DES while pregnant carried a higher risk of a rare vaginal cancer called clear cell adenocarcinoma. This wasn’t the kind of bump in risk that gets lost in statistics. It showed up in young women and girls who normally wouldn’t face such a disease. For mothers, breast cancer rates landed higher than those who never took the drug. Researchers pin this on the way DES can mess with hormone regulation, making normal tissue more likely to turn cancerous after enough time passes.
The Damage Doesn’t Stop at Cancer
Cancer’s not the only shadow hanging over DES-exposed families. Women exposed in the womb deal with more problems when trying to get pregnant. Some face infertility, miscarriages, or an increased chance of early labor. Others have structural changes in the uterus or cervix, which doctors call "T-shaped uterus" or similar terms, and this can make full-term pregnancy harder. Men exposed before birth also carry a higher risk of genital abnormalities and possibly testicular cancer. It’s a reminder that chemicals fiddling with hormones can leave a mark before a baby even meets the world.
Lessons I’ve Learned Watching Medical Stories Unfold
Stories like DES cut through the confidence people sometimes have about medical innovation. Reading about DES made me more aware of the ripple effects that can start with one decision. A medication that seems helpful at the start can sow the seeds for problems that only show up decades later. The ripple can even hit people who never took the drug but happened to be in the womb when decisions were made. I’ve seen families deal with the shock of cancer diagnoses tied to a pill taken long ago. That shock turns into frustration and mistrust when people find out the harms were suspected well before the prescriptions stopped.
How to Respond and Move Forward
DES taught a hard lesson about the dangers of skipping long-term research. Regulators learned to demand more safety data, especially with drugs that shape hormones or target pregnant women. For anyone who knows their family faced DES exposure, staying in touch with a doctor matters. Screening for signs of cancer or reproductive trouble helps catch problems early.
We need more than just better research rules. Open conversation between patients and healthcare providers about the history of drugs like DES keeps these stories from fading. Clinics can help by pushing patients to know their health history. As a society, putting patient safety ahead of convenience or quick solutions builds the trust that medicine depends on.
My experience teaches that real safety comes from listening—to patients, to stories from the past, and to scientific caution. The legacy of DES reaches far beyond what anyone expected, and that’s why paying attention to history matters every day.
Who should avoid taking Diethylstilbestrol?
The Hidden Hazards of a Synthetic Hormone
Many years back, doctors prescribed Diethylstilbestrol, better known as DES, to millions of women. The drug had big promises: support healthy pregnancies and manage certain cancers. Those promises fell apart. Research caught up, showing DES caused more harm than good—risk skyrocketed not only for the women who took it but also for their children and even grandchildren.
Pregnant Women: A Group at Great Risk
DES seemed safe on the surface, but there’s a dark side to its history. Pregnant women who received DES often did so to avoid miscarriages or complications. The cost turned out steep. Studies in JAMA and the New England Journal of Medicine show those women faced higher rates of breast cancer. That alone says enough. No expecting mother deserves to trade a hopeful pregnancy for a future filled with anxiety and health scares. Women considering any hormonal drug during pregnancy owe it to themselves to confirm it’s not DES lurking in the prescription bottle.
Children, Grandchildren—Families Caught in the Net
The damage didn’t stop at one generation. Sons and daughters exposed to DES in the womb, the so-called “DES daughters” and “DES sons,” live with increased risks of rare cancers, fertility struggles, and complications not normally seen. Many daughters developed a rare clear cell vaginal and cervical cancer, discovered in young adulthood. Sons reported issues like non-cancerous growths in the testicles and changes in the structure of their reproductive tracts. The weight of that legacy still shows up in medical journals and family histories long after the drug left the pharmacy shelves.
People With a History of Thrombosis
Anyone who has had blood clots or stroke sits near the top of the avoid list. DES raises the odds of another clot forming. A 2020 article in “Drug Safety” broke down how estrogen-like drugs make the blood thicker and a lot more likely to clump together. One bad clot can mean a stroke, a heart attack, or something that strips independence away faster than anyone would like to think. Doctors learned to be skeptical of giving estrogenic drugs to anyone with a clotting problem, and DES should land straight in the “never” pile for this group.
People Fighting Cancer—Or Trying to Avoid It
Some older cancer treatments tossed DES into the mix, mostly for prostate cancer. Researchers found alternatives that work better and with fewer lifelong risks. For anyone facing cancer, the risk of secondary cancers from DES stacks on top of the original diagnosis. The American Cancer Society dropped DES from its recommendation lists for good reason.
Anyone Allergic or Sensitive to Estrogen-Like Medications
People with bad reactions to estrogenic drugs—think hives, swelling, or severe headaches—shouldn’t touch DES. Doctors often spot reactions after just a few doses. That makes the choice simple: pick something safer. Alternatives exist for nearly every use case that originally called for DES.
Best Medicine: Clear Communication and Solid Alternatives
Doctors and pharmacists need to talk openly with patients about drug histories. Electronic records help, but self-advocacy saves lives, too. No prescription should slip past an informed patient. Safer, time-tested therapies replace DES today—there’s no real excuse for mixing up old and new drugs. Trust built between patient and provider goes a long way. I’ve watched friends change their medication after open talks with their doctors, swapping risky drugs for safer choices and sleeping better for it.
| Names | |
| Preferred IUPAC name | 4,4'-(prop-1-ene-1,2-diyl)diphenol |
| Other names |
DES Stilbestrol Stilboestrol Hexestrol Stilbestrolum Stilbestrol-Estradiol |
| Pronunciation | /daɪˌiːθəlstɪlˈbɛstrɒl/ |
| Identifiers | |
| CAS Number | 56-53-1 |
| Beilstein Reference | 1462076 |
| ChEBI | CHEBI:4485 |
| ChEMBL | CHEMBL49 |
| ChemSpider | 5826 |
| DrugBank | DB00255 |
| ECHA InfoCard | 17-211-610-3 |
| EC Number | 200-175-5 |
| Gmelin Reference | 78572 |
| KEGG | C07081 |
| MeSH | D004052 |
| PubChem CID | 448537 |
| RTECS number | KW2975000 |
| UNII | YL5FZ2Y5U1 |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID8020203 |
| Properties | |
| Chemical formula | C18H20O2 |
| Molar mass | 268.363 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.2 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 3.74 |
| Vapor pressure | 6.48E-09 mmHg at 25°C |
| Acidity (pKa) | 10.28 |
| Basicity (pKb) | 10.70 |
| Magnetic susceptibility (χ) | -73.7e-6 cm³/mol |
| Refractive index (nD) | 1.618 |
| Viscosity | Viscous liquid |
| Dipole moment | 4.37 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 510 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –161.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7254 kJ/mol |
| Pharmacology | |
| ATC code | G03CB02 |
| Hazards | |
| Main hazards | Carcinogenic, reproductive toxin, endocrine disruptor, harmful if swallowed or inhaled. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS08 |
| Signal word | Danger |
| Hazard statements | H350: May cause cancer. |
| Precautionary statements | P201, P202, P281, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0 Health=2, Flammability=2, Instability=0 |
| Flash point | 131°C |
| Autoignition temperature | 444°C |
| Lethal dose or concentration | LD50 (oral, rat): 68 mg/kg |
| LD50 (median dose) | LD50 68 mg/kg (oral, rat) |
| NIOSH | ST3500000 |
| PEL (Permissible) | PEL: 0.1 mg/m³ |
| REL (Recommended) | 0.1 mg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Hexestrol Benzestrol Dienestrol Methestrol |

