Diethylaminocyanide: A Deep Dive into Chemistry’s Unsung Workhorse
Finding Diethylaminocyanide in the Annals of Chemistry
Every chemist runs into molecules that don’t make the headlines but quietly hold entire industrial workflows together. Diethylaminocyanide, known to folks in the lab as DEAC, has hovered at the fringe of mainstream chemical fame ever since its discovery in the early twentieth century. Early records show researchers poking around cyanides, seeking new reagents that could shape organic frameworks in ways simple cyanide ions could not. Diethylaminocyanide emerged as a handy variant, more manageable than its infamous relatives, and quickly gained a place in the organic chemist’s toolkit. Despite its relatively low profile, it has shown remarkable staying power, around long enough to shape generations of synthetic work.
Sizing Up the Chemical and Physical Realities
Lifting the lid off a sample bottle of this chemical, you'll notice a faintly fishy, almond-like smell. In the flask, diethylaminocyanide usually appears as a clear, colorless to pale yellow liquid. Its molecular structure centers on a cyanide group bound to a nitrogen atom, which is itself connected to two ethyl groups. The result is a compound with a modest boiling point, not wild like diethylamine but noticeably higher than hydrogen cyanide. It dissolves readily in most organic solvents but shies away from water. Stability is good under cool, dry storage, but exposure to moisture and open air eats away at its potency, making careful handling important. It’s not the most volatile member of the cyanide club, but you respect it the same way a carpenter respects a sharp chisel—one slip, and you’ve got problems.
Technical Details and How Labels Count
Pick up a commercial container, and the label tends to sport the universal language of chemists: structural formula, molecular weight, and any hazard pictograms that spell out the substance’s risky side. Safety data always features heavily. Regulatory rules demand labels for storage temperature, recommended shelf life, and protective measures needed during transport or storage. These details carry weight in the field, often making the difference between a productive experiment and an emergency room visit. Labels also highlight the need for reliable suppliers; even subtle impurities can alter reaction outcomes, especially in industries where product consistency keeps profit margins alive.
Making the Stuff: A Story of Synthetic Craftsmanship
Producing diethylaminocyanide takes a practiced hand. Many prep routes show up in the published literature, but one of the classics involves bubbling hydrogen cyanide into a mixture of diethylamine and an activator under anhydrous, low-temperature conditions. Another popular method starts with diethylammonium chloride, then brings in sodium cyanide. From personal experience, reactions demand strict temperature and moisture control. Small laboratory scale-ups can turn ugly quickly—cyanide’s toxic bite doesn’t forgive sloppy technique. Industrial producers rely on closed-loop systems for containment, generating higher yields and tighter quality control. Chemists keep close tabs on side products, since even minor variations can tailspin downstream syntheses.
How Diethylaminocyanide Moves in Reaction Space
The chemistry behind diethylaminocyanide is where real magic happens. Its reactivity focuses strongly on the active cyanide group, which engages with electrophiles and other reagents to build up new chemical bonds. It’s a go-to synthon for introducing the N-cyanoethyl group—a precursor moiety that unlocks chains of subsequent chemical transformations. Organic chemists have come to lean on DEAC, especially in pharmaceutical research, to assemble unusual heterocycles and push boundaries in medicinal chemistry. Once you start modifying it or tacking it onto other frameworks, doors open to more complex, often more bioactive, molecules.
Different Names, Same Role
In the world of chemical jargon, diethylaminocyanide shows up under a tangle of synonyms. N,N-Diethylcyanamino, N-Cyano-N,N-diethylamine, and others all map to the same core structure. Differences in naming conventions often trace to country of origin or subtle quirks in how various research groups like to describe functional groups. This mishmash can confuse newcomers, especially when reading older literature or patents that stick to obscure terminology. Sticking to the official IUPAC name in research and documentation clears up a lot of muddle down the line.
Following the Rules: Handling and Safety in Practice
Anyone who’s logged hours around cyanides knows respect, not fear, is the way forward. Handling diethylaminocyanide means donning gloves resistant to organics, keeping good ventilation, and double-checking that fume hoods actually work. Training for spill control and rapid response to accidental exposure stays top-of-mind in both academic and industrial settings. The industry blueprint includes regular training, easy access to antidotes where available, and clear protocols that treat “almost an accident” as a learning opportunity. Consistent enforcement and a culture that values careful work outrank any equipment investment alone. On the storage front, dry, cool, and sealed environments slow decomposition and cut down on dangerous byproducts like diethylamine and hydrogen cyanide gas.
Where the Molecule Works: Application Hotspots
Lab veterans can point to places where diethylaminocyanide shined when few other reagents cut it. In drug development, the molecule helps chemists build up intricate nitrogen-containing scaffolds. The so-called cyanamide method gave new leads in the hunt for antiviral and anticancer drugs. Some specialty polymers depend on its well-behaved nucleophilic properties to produce tough, flexible, or chemically resistant chains. Color chemistry even tested DEAC in making certain dyes, though competition from larger-scale cyanide sources eventually won out. On a smaller scale, research in material science and electronics poked at this compound for applications in semiconductors, though the expense and risk often left it a specialty tool rather than a daily staple.
Pushing the Boundaries: Research and Forward-Thinking Development
What keeps diethylaminocyanide interesting is the sense that unexplored ground remains. Researchers keep plugging away at better ways to harness its reactive cyanide, aiming for cleaner, more select synthetic steps. As the demand for tailored pharmaceuticals rises, so does the need for reagents with pinpoint selectivity. Methodologies using this cyanide source regularly surface in academic journals. Most folks hope for procedures that cut down on toxic waste and reduce the handling risks. Greener solvents, continuous flow processes, and clever catalysis often get trialed with DEAC in the starring role. Every incremental improvement in yield, selectivity, or safety counts, because slim profit margins in chemical development turn these gains into real savings over time.
Toxicity and the Human Factor
Nobody in the lab takes the hazard symbols on a bottle of diethylaminocyanide as decoration. Its key threat springs from the same chemistry that gives all cyanides their punch: it blocks cytochrome oxidase, an enzyme vital for cellular respiration, starving tissues of oxygen. In larger doses, inhalation or skin contact spells big trouble, and even small mishaps require immediate response. Over the decades, animal toxicity studies shaped the current safety guidelines. Chronic exposure studies, while limited, pushed organizations to mandate strict protective gear and protocol enforcement. Stories exchanged in break rooms often revolve around close calls, and these serve as living reminders for new generations. Broader adoption of closed-system technologies offers hope for dialing down direct exposure and carrying risk even lower.
Looking Beyond Today: Where Does Diethylaminocyanide Go from Here?
In chemistry’s ever-evolving landscape, diethylaminocyanide may never claim the limelight, yet it still shapes countless critical projects. Future advances point toward applications in next-generation pharmaceuticals where difficult nitrogen insertions continue to stump researchers. Sustainable manufacturing aims for less reliance on harsh solvents and more efficient reaction cascades, showcasing this reagent’s potential for adaptation. Pressure never lets up for better safety protocols and environmentally sound disposal plans. With industry moving toward automation and digital control, the next wave of chemists will likely manage this molecule from behind a digital dashboard, lowering risk and raising throughput along the way. What always grounds progress is a firm grasp of both the rewards and the responsibilities that come with handling reagents like diethylaminocyanide. Chemistry may change, but the need for respect, precision, and careful stewardship never fades.
What is Diethylaminocyanide used for?
The Role and Reach of Diethylaminocyanide
Diethylaminocyanide doesn’t get much attention outside chemistry circles. Anyone who’s spent time in a lab knows the long list of chemicals that look like tongue twisters and serve as matchmakers, glue, or stepping stones for bigger, splashier molecules. Diethylaminocyanide falls squarely in the category of “building block.” It shows up mostly in organic synthesis, helping chemists piece together compounds that push medicine, materials, and technology forward.
I remember jitters working with it as a graduate student. You respect a molecule robust enough to add a cyano group—a carbon and nitrogen trio—onto other molecules, especially since the cyano group can act as a launchpad for even more chemistry down the line. This compound packs a punch not because of fanfare or sharp color, but because it does its job effectively.
Digging into its Main Uses
Researchers use diethylaminocyanide for the creation of so-called “heterocycles”—the ring structures that turn up everywhere from antibiotics to smartphone screens. It gives chemists a way to introduce complexity fast. Its main talent centers on donating that cyanide group in controlled ways, opening doors to molecules with medical promise.
In pharmaceutical labs, this opens a faster route to medications. Think antiviral drugs or compounds with anti-cancer properties: many owe their beginnings to clever intermediates, and diethylaminocyanide can help make those intermediates affordably and quickly. In industry, it brings efficiency to specialty chemical syntheses, where fine margins and strict purity rules matter.
Hazards and Responsible Handling
As any chemist will tell you, neat reactions come with serious risks. Diethylaminocyanide releases fumes with a strong odor, and it’s toxic if mishandled. Anyone using it works behind fume hoods, wearing gloves, goggles, and lab coats—safety comes before speed. A story I still recall involves a bottle uncapped in an undergraduate teaching lab; instructors quickly cleared the room. No harm done, but a reminder of why safety protocols never get skipped.
Proper disposal remains a sticking point. Unlike household waste, used diethylaminocyanide and its by-products can’t go down a drain. Labs rely on waste contractors with expertise in handling hazardous materials, ensuring environmental impact stays minimal. Oversight programs demand records, audits, and can shut down research if storage or use falls short of regulations.
Moving Forward: Better Use and Safer Chemistry
Chemical manufacturers persistently search for safer and greener ways to deliver the same results, sometimes swapping out old reagents for newer, less hazardous options. Green chemistry pushes companies and universities to rethink traditional methods, favoring reactions with lower toxicity and fewer nasty leftovers. Diethylaminocyanide sticks around because it gets the job done, yet many labs now ask whether a less risky alternative could fill the same role.
Looking at the bigger picture, the lesson with diethylaminocyanide extends well beyond the bottle. Every advance in chemistry brings responsibility: safety for lab workers, the wider community, and the planet. As new materials and medicines emerge, that balance of progress and caution shapes the future of science, one reagent at a time.
What are the safety precautions when handling Diethylaminocyanide?
The Real Dangers Behind the Name
Diethylaminocyanide doesn’t show up in many kitchens or garages, but anyone who’s stepped into a synthetic chemistry lab will recognize it. This chemical, used for certain organic reactions, comes with more baggage than just an unpronounceable name. It releases toxic gases when it touches water or acids, and it irritates the skin, eyes, and respiratory tract. I’ve seen a few corners cut with hazardous chemicals in academic labs, and it always ends with regret. Once, a spill sent a coworker scrambling for medical attention—a reminder nobody needs twice.
Personal Protective Equipment: Not Optional
While some might rush through setting up a reaction, gloves, goggles, and a lab coat matter with diethylaminocyanide. Nitrile gloves hold up well against most organic solvents—you want more than a thin latex barrier. Good splash goggles beat those old safety glasses. More than once, surprise splashes happen, especially if the reaction heats up or foams. Long sleeves and closed shoes should be second nature for any chemist.
Ventilation or Bust
There’s a certain false confidence that can settle in after years of mixing weird chemicals, but diethylaminocyanide fumes are sneaky. They build up faster than you notice. Fume hoods act like a second pair of lungs. Even for quick transfers, I learned the hard way it isn’t worth “just this once” exposure. Chronic exposure to cyanide compounds leads to everything from headaches to lasting nerve damage.
Storage: Less Is More
Diethylaminocyanide doesn’t belong on a crowded open shelf. It breaks down with moisture and can catch fire with the right trigger. Air-tight bottles kept in a cool, dry spot away from acids or bases work best. Think about spill control, too—pad the storage cabinet with enough absorbent material to soak up a leak. Labeling should list hazard classes in bold, not just some scrawled abbreviation. Good records prevent panicked confusion if a bottle cracks or goes missing.
Dealing with Spills and Exposure
No one forgets the slap of cold fear after a chemical spill. I saw someone freeze up when their glove split open and liquid touched their skin. Fast action reduces harm: rinse and remove contaminated clothing, then keep rinsing. Eye splashes mean heading right to an eyewash station for a fifteen-minute rinse, even if you’re in a rush. Training and posted emergency charts save critical minutes. Small spills soak up with pads—then the waste goes straight to a hazardous disposal bag, never the regular trash.
Disposal: Never Down the Sink
Pouring cyanide-containing waste down a drain breaks environmental laws and endangers others. Licensed hazardous waste disposal companies deal with diethylaminocyanide residues safely. I’ve seen labs fined, and it isn’t pretty or cheap. Check your local laws; some places send compliance officers frequently. Making waste logs part of the experiment notebook keeps everyone honest.
Improving Safety Culture
Even seasoned chemists make mistakes, especially after long days or late nights. Regular safety walkthroughs, clear signage, and honest reporting of near-misses keep dangers in check. I’ve worked in a few labs that built friendly competition around safety—awarding pizza for proactive suggestions. Small changes mean fewer accidents over time. Sharing hard-learned lessons builds trust and keeps everyone focused on going home in one piece.
Stay sharp, speak up, and don’t let routine invite risk.What is the chemical formula of Diethylaminocyanide?
The Structure and Composition of Diethylaminocyanide
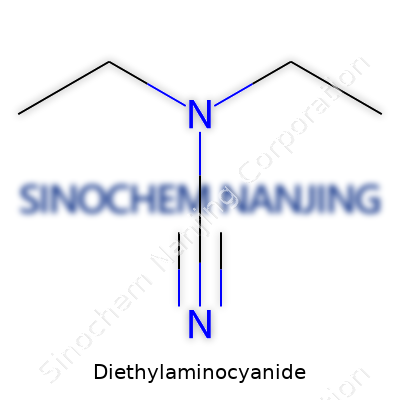
Few compounds grab the attention of organic chemists and pharmaceutical researchers like diethylaminocyanide. The chemical formula for diethylaminocyanide is C5H10N2. This small and unassuming molecule opens the door to a broad variety of synthetic possibilities. Chemists recognize it by its smell and its reactivity. It’s made up of five carbon atoms, ten hydrogens, and two nitrogens, and its structure involves a cyanide group (-CN) linked to a diethylamino group (-N(C2H5)2). That pairing gives it a unique fingerprint for laboratory work.
The Real-World Usefulness of Diethylaminocyanide
I’ve seen diethylaminocyanide used in synthetic labs where there’s an urgent need to build new molecules. The –CN group often serves as a starting point for other chemical reactions. It’s involved in forming heterocycles, pharmaceuticals, and dyes. Heterocycles give rise to several drugs and crop protection agents. In the right chemical company, the presence of the cyanide group allows for the formation of new carbon-nitrogen bonds, which helps create new structures not found anywhere else.
In practice, the value comes not just from what the molecule looks like on paper but from the doors it opens. Once someone working in synthesis gets their hands on diethylaminocyanide, the molecule helps push projects forward—whether aiming for a more potent medication or searching for a brighter, more stable pigment.
Safety Matters and Handling Concerns
Working with this compound calls for careful treatment. Both diethylamine and cyanides give off toxic vapors and come with a risk of poisoning. The best chemists won’t open a container of diethylaminocyanide without good ventilation or a working fume hood. Skin contact and inhalation need to be avoided, since the cyanide can be absorbed rapidly. Even students in university lab settings learn to respect its hazards early on, because those warnings carry real consequences.
Safe handling practices extend to storage too. It reacts with acids and strong oxidizing agents and must be kept away from incompatible substances. Spills or leaks require quick, coordinated clean-up using protective gear, with everything else in the lab treated as contaminated until proven safe.
Addressing the Availability and Ethical Questions
Diethylaminocyanide is not available on drug-store shelves, and for good reason. Because it contains a cyanide group, regulatory agencies pay attention to who uses it and for what purposes. It shows up mostly in well-monitored industrial or academic settings handled by trained staff. Anyone with practical experience in chemical supply knows that ordering, storing, or moving any cyanide-containing compound means paperwork, accountability, and a clear chain of custody.
There’s always debate about how to balance open science and public protection. On one hand, molecules like diethylaminocyanide help researchers find answers to diseases that have stumped doctors for decades. On the other, they demand respect and caution to prevent harm, intentional or accidental. Responsible usage by those with the right experience, under strong oversight, keeps society safe and allows science to advance.
Looking Ahead: Solutions and Best Practices
For those responsible for using or managing chemicals with such hazards, training matters as much as technology. Clear procedures, well-marked storage, and frequent drills help prevent mistakes. In my lab days, peer checks before any work with cyanides cut down on human error. Manufacturers invest in packaging that alerts you to danger at a glance, and chemical suppliers keep thorough records of sales.
People in health and safety circles continue working to find alternatives where possible, but for now, diethylaminocyanide remains an important building block. Using it well means knowing both its promise and its risks, and supporting those working with it by giving them the best training, tools, and oversight.
How should Diethylaminocyanide be stored?
Packing Dangerous Chemicals: Not a Game
Diethylaminocyanide pops up in research labs more than most people realize. Its uses range from creating pharmaceuticals to serving as a reagent in organic synthesis. The catch? It’s not something you can leave sitting on a shelf without a second thought. From personal experience handling other sensitive compounds, it becomes clear that small mistakes lead to big messes. It only takes a minor misstep—improper container, wrong storage condition—for hazardous chemicals to turn into safety nightmares.
Volatility Isn’t Just About the Stock Market
This chemical stands out for its tendency to decompose and give off dangerous gases. Consider the story of a research assistant who cracked open an old bottle—just touching the cap led to a puff of smoke and a trip to the emergency shower. Not storing diethylaminocyanide in a tightly sealed, chemical-resistant container is asking for trouble. Glass or high-grade polyethylene are usually trusted because metal can corrode or trigger reactions. Reusing old or mystery containers doesn’t cut it; labels matter, and so does peace of mind for staff walking through that storeroom.
Keep Cool, Stay Safe
Temperature control isn’t just a “nice to have.” Diethylaminocyanide starts breaking down above room temperature, and that breakdown isn’t friendly. Heat speeds up the risk of dangerous byproducts. A fridge designed for hazardous chemicals—never food and drinks—works best. Cold storage helps maintain stability, and lets everyone breathe a little easier. From firsthand lab work, one notices labs with strict cold-chain storage cut down on incidents and thrown-out chemicals. Room temperature storage fails more often than folks admit.
Separation Prevents Chain Reactions
Accidents rarely come from one mistake—they pile up. Stashing diethylaminocyanide beside acids, oxidizers, or even open bottles of alcohol? Tempting fate. If a bottle leaks, incompatible substances may react together, and chaos follows. A good chemical inventory system avoids surprises. I’ve witnessed the overtime hours lost when chemists had to sift through cabinets, searching for misplaced bottles. Dedicated shelving, clear signage, and keeping reactive and flammable items apart save more than just money—they save lives.
Information Isn’t Optional
No chemical takes care of itself. Anyone handling diethylaminocyanide should know what they’re getting into. That means up-to-date Safety Data Sheets, regular training, and real-world drills. It isn’t enough to tick a box. After seeing colleagues caught off guard by outdated instructions, I know that good information and practice reduce panic and error. Emergencies test what you really know, not just what’s written in a binder.
Accountability Makes the Difference
Trusting that everyone will respect storage rules is not enough. Labs with checklists and supervisors following up run safer workspaces. Disposal can’t be an afterthought either—expired or spilled diethylaminocyanide isn’t just inconvenient, it’s hazardous waste that can’t just be poured down a drain. Clear protocols and trained disposal technicians keep risks managed.
Building a Better Chemical Culture
Chemical safety isn’t flashy, but experience shows it draws a clear line between routine science and disaster. Diethylaminocyanide deserves careful storage: sealed container, cold and dry, away from incompatible chemicals, with informed hands nearby. This isn’t bureaucracy—it’s responsible science, rooted in keeping everyone safe.
What is the CAS number of Diethylaminocyanide?
What Makes a CAS Number So Important?
The chemical world runs on precision. If you’ve ever worked in a lab, or dealt with chemical inventories, you’ll know that guessing isn’t allowed. Chemical Abstracts Service, or CAS, assigns unique numbers to substances so scientists, suppliers, and regulators can speak the same language. Diethylaminocyanide, a compound used in organic synthesis, carries the CAS number 140-29-4. With thousands of compounds sharing similar names or common formulas, the CAS number avoids confusion that can lead to serious safety mistakes or misidentifications in reports and publications.
Personal Experience: Data Matters in the Real World
Years ago, I worked on a project cataloging reagents in a university lab. Without CAS numbers, identifying bottles collecting dust in storage proved challenging, especially when labels wore off or got written in shorthand. Cross-checking with authoritative lists helped us grab each compound’s real identity. For something like diethylaminocyanide, with a structure that can get confused with other cyanide-based amines, that number 140-29-4 draws a line between safety and disaster. Mix-ups in the lab go beyond just inconvenience — with toxic and reactive substances, a mistake costs more than just money.
Why Diethylaminocyanide Gets Noticed
Diethylaminocyanide shows up in research labs, especially in organic and medicinal chemistry. Chemists value it for forming carbon-nitrogen bonds, which underpin drug development and materials science. The world depends on researchers getting their compounds right, as the wrong substance derails months of work. Some manufacturers and reagent suppliers use CAS numbers in digital catalogs so buyers never second-guess what they’re getting. That unique identifier gives scientists and safety managers a simple way to track inventory, prepare material safety data sheets, and meet regulatory rules.
Fact-Driven Challenges and Solutions
Confusion between chemical names still happens. Diethylaminocyanide sometimes turns up under alternative names or misspellings in industry catalogs. From a regulatory angle, the U.S. EPA, European Chemicals Agency, and others use CAS numbers to enforce handling rules and environmental restrictions. Processes become streamlined and loopholes close when products get correctly listed by CAS numbers.
Technology offers some hope. Inventory apps now include direct scanning from manufacturer barcodes tied to CAS numbers, dramatically dropping the chance of errors. Labs that invest in robust training and digital inventory management see fewer chemical mix-ups and better compliance during audits.
Supporting Safety and Transparency
Proper use of the CAS number for diethylaminocyanide, 140-29-4, demonstrates a commitment to scientific rigor and safety. Regulatory agencies and independent watchdogs rely on these numbers to identify and track hazardous chemicals moving through supply chains. Researchers, educators, and industry professionals can pull up hazard data, safe handling instructions, and regulatory info in seconds, cutting down on communication gaps.
I’ve seen new lab staff struggle until they get comfortable searching and referencing compounds by CAS numbers. A small investment in education goes a long way, reducing incidents where the wrong chemical gets used or stored improperly. CAS numbers, in the right hands, prevent accidents and foster an environment built on facts.
| Names | |
| Preferred IUPAC name | N,N-Diethylethanamide |
| Other names |
N,N-Diethylcyanamide N-Cyano-N,N-diethylamine Cyanodiethylamine |
| Pronunciation | /daɪˌɛθɪlaɪˌmiːnoʊsaɪəˈnaɪd/ |
| Identifiers | |
| CAS Number | 100-86-7 |
| Beilstein Reference | 1201183 |
| ChEBI | CHEBI:51409 |
| ChEMBL | CHEMBL378053 |
| ChemSpider | 13616711 |
| DrugBank | DB08548 |
| ECHA InfoCard | 05bd778b-ff62-4c57-8170-c5e85eae3d6e |
| EC Number | 203-708-2 |
| Gmelin Reference | 8187 |
| KEGG | C18816 |
| MeSH | Diethylaminocyanide |
| PubChem CID | 12218 |
| RTECS number | NJ3150000 |
| UNII | B4GMY5M69B |
| UN number | UN2382 |
| CompTox Dashboard (EPA) | DTXSID0044360 |
| Properties | |
| Chemical formula | C5H10N2 |
| Molar mass | 98.15 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | fishy |
| Density | 0.89 g/cm3 |
| Solubility in water | Insoluble |
| log P | 1.02 |
| Vapor pressure | 0.7 mmHg (20 °C) |
| Acidity (pKa) | 16.1 |
| Basicity (pKb) | 3.30 |
| Magnetic susceptibility (χ) | -41.0 x 10^-6 cm^3/mol |
| Refractive index (nD) | 1.459 |
| Dipole moment | 4.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4467 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P332+P313, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | 3-3-2 |
| Flash point | 35 °C |
| Autoignition temperature | 403 °C |
| Explosive limits | Lower: 2.1%, Upper: 10.5% |
| Lethal dose or concentration | LD50 (oral, rat): 380 mg/kg |
| LD50 (median dose) | LD50 (median dose): 190 mg/kg (oral, rat) |
| NIOSH | KW3675000 |
| PEL (Permissible) | PEL (Permissible exposure limit) for Diethylaminocyanide: Not established |
| REL (Recommended) | 0.07 mg/m³ |
| Related compounds | |
| Related compounds |
Ethyl cyanide Cyanogen bromide Cyanamide Dimethylaminocyanide |

