Diethyl Chlorophosphonate: Unpacking Its Role in Science and Industry
Tracing the Roots: Historical Development
Diethyl chlorophosphonate started making its mark in the chemical world decades ago. The compound, which belongs to the group of organophosphorus chemicals, came into broader use after World War II, right as industrial chemistry began shaping up for mass production of new synthetics. Early synthetic chemistry depended on reagents like phosphorus trichloride, and as chemists got ambitious about functional group transformations, diethyl chlorophosphonate provided a valuable link between basic phosphorus chemistry and new applications. Its presence in scientific literature often signals advances in pesticide design, flame retardant research, or even medicinal chemistry, showing just how utility and risk often ride side by side in chemical innovation.
Getting Acquainted: Product Overview
What strikes me about diethyl chlorophosphonate is that it walks a line between being a backbone intermediate and a tool for targeted reactions. It’s not a household commodity, but in the hands of skilled chemists, it transforms how we build up molecules. Researchers reach for it mainly to introduce phosphorus-containing groups into other chemicals. Such actions ripple into products ranging from nerve agents to pharmaceuticals, stark reminders that breakthroughs carry consequences – and bonuses – well beyond lab benches.
Core Details: Physical and Chemical Properties
This liquid carries a distinct odor and offers few surprises to those familiar with organophosphorus compounds. At room temperature, it remains stable, though contact with moisture triggers slow hydrolysis, yielding hydrochloric acid and diethyl phosphite. Over the years, workers noticed its tendency to irritate skin and lungs, which probably pushed early safety protocols for handling chemicals like this. Its reactivity toward nucleophiles opens doors for chemists but flags hazards for the unwary. Since it’s heavier than water, any accidental spill or improper handling in a lab or manufacturing site poses practical challenges for containment and remediation.
Digging Into Details: Technical Specifications and Labeling
In the lab, chemists look for purity by gas chromatography or NMR. Those familiar with handling legal or controlled chemicals know that labeling rules demand clear signaling of toxicity, so bottles almost always broadcast prominent hazard warnings. Safety data sheets dwell on necessary storage – cool, dry, well-ventilated spaces, separate from acids and oxidizers. Anyone with experience handling such chemicals recognizes those precautions are not just bureaucratic red tape—they result from hard-earned lessons.
Method Behind the Matter: Preparation Methods
The classic method for preparing diethyl chlorophosphonate involves the reaction of diethyl phosphite with thionyl chloride or phosphorus pentachloride. Both methods have quirks: Thionyl chloride releases sulfur dioxide gas, which means preparations demand serious ventilation. Phosphorus pentachloride works cleaner, but in a large batch, it generates heat, so careful temperature control becomes critical. I recall a colleague once describing a runaway reaction that filled the hood with choking gas; safety systems managed the risk, but nobody forgot that day.
Beyond Basics: Chemical Reactions and Modifications
Diethyl chlorophosphonate, for all its hazards, grants chemists power to form phosphonate esters, which are popular in medicinal chemistry for their ability to mimic natural phosphate groups. Reacting with alcohols, amines, and water unlocks a series of transformations, allowing functionalization that’s tough to do by other means. These reactions underpin everything from prodrug design to specialty flame retardants. Each new reaction scheme invites innovation, though always drawing a sharp line that demands robust safety oversight.
Known by Many Names: Synonyms and Alternate Product Names
In chemical catalogs, diethyl chlorophosphonate appears as Diethoxyphosphoryl chloride, Chlorophosphonic acid diethyl ester, and sometimes simply as Chlorophosphonic acid, diethyl ester. For research professionals, keeping straight the handful of names avoids confusion—mix-ups in even small-scale synthetic work can lead to ruined experiments or worse, accidents.
Line in the Sand: Safety and Operational Standards
Safety with diethyl chlorophosphonate has evolved thanks to both regulator pressure and in-house company rules. Labs require full PPE, fume hoods, eyewash stations, and tight accountability on inventory. Waste disposal must follow hazardous chemical protocols, often incinerated offsite by specialty firms. Decades of chemical safety training show that even experts never underestimate the risks—most accidents tie back to lapses in established procedures or inadequate emergency planning. Steady improvement in response plans and transparent incident reporting represents real progress here. In my career, I’ve watched industry and academia shift from casual caution to detailed, drill-heavy training for compounds just like this one.
Where It Matters: Application Area
Life sciences, material engineering, and the armaments sector each find value in diethyl chlorophosphonate. Organic synthesis labs use the compound to make alkyl phosphonates, which can slip into drug prototypes or agricultural chemicals. Flame retardant makers also use phosphorus intermediates as building blocks for safer, more stable product lines. Unfortunately, its inclusion in chemical weapons programs, especially nerve agents, means researchers face export controls and end-use monitoring. Legitimate research faces extra hurdles, yet those steps are crucial, given the stakes.
Keeping Up: Research and Development
Ongoing research aims to find safer routes to these valuable phosphorus intermediates. Newer synthetic strategies target higher yields with fewer byproducts, focusing on green chemistry and worker safety. Analytical chemists build better detection and leak monitoring methods, hoping to spot problems before they bloom into incidents. Collaborative projects between university labs and industry partners sometimes unlock fresh approaches—safer reagents, milder reaction conditions, more responsible life-cycle management. These changes arrive slowly, but real improvements start with that focus on reducing exposure and environmental burden.
Understanding the Risks: Toxicity Research
Organophosphorus compounds like diethyl chlorophosphonate sit under scrutiny because of their acute toxicity potential. Inhalation or direct contact leads to respiratory irritation, skin burns, and eye damage. Long-term studies link these compounds to neurotoxicity. Based on what we know, even modest doses build up organ damage over time. Regulatory limits reflect this—no surprises for those accustomed to working with potent reagents, but the barrier for new researchers remains high. Animal studies led to strict occupational exposure limits; only thoroughly trained staff are cleared to handle such chemistry. My own exposure to the literature stressed just how broad the impact can be—not just acute poisonings, but subtle metabolic and neurological changes worth worrying about in the long run.
On the Horizon: Future Prospects
Chemists keep searching for ways to sidestep hazards without losing synthetic flexibility. Some teams experiment with alternative phosphorus donors that produce fewer toxic byproducts. Others push for automated handling and improved ventilation. Environmental scientists study degradation pathways and focus on remediation strategies for accidental releases. Digital monitoring—sensors that track concentration in air or effluent streams—helps in spotting leaks before they reach dangerous levels. The push for sustainability will likely drive further innovation, transforming how high-risk chemicals are handled across the entire supply chain. Meanwhile, robust oversight and clear communication remain the best shields against both human and environmental risk, and as the years roll forward, those lessons need revisiting just as much as the technical details themselves.
What is Diethyl Chlorophosphonate used for?
Used in Complex Chemistry, Beyond the Lab Bench
Step into a synthetic chemistry lab, and you’ll run into some strange names. Diethyl chlorophosphonate shows up often. This chemical looks simple on paper, with a structure that carries both chlorine and phosphorus. In practice, it packs much more punch than its unassuming name suggests. It’s the kind of ingredient hard to swap out, especially when making things that demand precision like pharmaceuticals, crop protection agents, or flame retardants.
The Real Reason It Gets Attention: Making Bonds That Last
Phosphorus often crops up in bioactive molecules. Diethyl chlorophosphonate makes it possible to introduce phosphorus into new compounds. Once it hits the mixing stage, it reacts with alcohols or amines, leading to sturdy phosphorus-containing bonds. This isn’t just an academic detail—many antiviral medications and pesticides couldn’t exist without phosphonate or phosphonate ester groups. That’s where this chemical plays a key role. Just ask any medicinal chemist finishing a complicated synthesis.
Crafting Products That Touch Everyday Life
Pharmaceuticals grab headlines, but everyday items also depend on advanced chemical ingredients. Take flame retardants in electronics or furniture. Many work by using phosphorus chemistry to slow or stop fires. Some start with diethyl chlorophosphonate. Without it, consumer goods might become less safe. I've spent hours reading about electronics safety, and flame retardants often leaned on phosphorus-based molecules that require chemistry like this.
Risks and Handling: Not One for Amateurs
One fact stands out. Diethyl chlorophosphonate isn’t something to handle lightly. The same reactivity that makes it useful gives it a sharp, biting nature. It irritates the skin, eyes, and lungs. Strong chemical smells aren’t just uncomfortable—they signal potential danger. So, before chemistry workers even begin, they suit up with goggles, gloves, and full ventilation. As a student, I remember professors stressing respect for reagents like this. Safety data sheets weren’t some bureaucratic paper trail—they spelled out steps to prevent tragic lab accidents.
Limiting Unintended Impact: Waste Treatment and Disposal
Every lab and factory faces a challenge. How to prevent strong chemicals from reaching people or wildlife outside? Diethyl chlorophosphonate requires special waste management. High temperatures and strong bases break it down safely. Letting even small amounts slip into water would have nasty effects. Press coverage about chemical leaks taught me the difference between careful handling and careless shortcuts.
Alternative Pathways: Greener Chemistry in the Future
Some chemists work on producing the same molecules by milder methods, hoping to steer away from sharper reagents. That means designing reactions that either don’t use diethyl chlorophosphonate or replace it with less hazardous cousins. It takes patience, research funding, and bold ideas, since you can’t just swap one chemical for another without unwanted surprises. Still, biochemistry and green chemistry labs push for safer ways forward, with regulators encouraging this shift whenever possible.
Accountability and Trust Matter
Chemicals like diethyl chlorophosphonate live in the gap between breakthrough and risk. The people making, using, and monitoring it have to stay sharp, sharing what they know and looking ahead to safety. Tighter rules and better transparency shape how it gets used. For anyone using a phone, taking medicine, or sitting on a couch, those behind-the-scenes decisions really do matter. Knowledge and responsibility walk hand in hand, especially with chemistry that affects lives beyond the lab.
What precautions should be taken when handling Diethyl Chlorophosphonate?
Respecting the Risks
Diethyl Chlorophosphonate belongs to a group of chemicals that demand respect and care from those who handle them. Years spent working in labs and communicating with researchers taught me one thing—complacency around chemicals causes trouble. This compound packs danger on two fronts: it delivers toxic fumes and burns through tissues on contact. An accidental splash can bring severe pain, and just one whiff of vapors leads to coughing and choking. The stories from hospital beds aren’t worth the shortcut.
Understanding Proper Protection
Stepping into a workspace with Diethyl Chlorophosphonate means putting on more than a pair of gloves. Chemical splash goggles, face shields, and lab coats that don’t leave any skin uncovered become essentials. Folks who want to keep their hands safe choose thick, nitrile gloves and double up if possible. No one wants to end the day with a chemical burn, least of all from something this unforgiving.
Engineering Controls Make a Difference
Most mistakes I’ve seen happen where shortcuts replace proper setups. A fume hood pulls dangerous vapors away from faces and stops fumes drifting through the air. Trying to pour or transfer this chemical outside of a vented area risks breathing in poisonous phosphorus oxides. Just thinking about cleaning up a spill in an open room raises hairs on the back of my neck. Spills, even small ones, need containment—spill kits with proper neutralizers can prevent damage and injury fast.
Storage Demands Careful Attention
Diethyl Chlorophosphonate breaks down when exposed to moisture and heat. Stashing bottles on a crowded shelf runs the risk of leaks, corrosion, and surprise reactions. A dry, cool cabinet marked for corrosives, away from bases, strong oxidizers, and water sources, never failed me. Each bottle gets a clear label, and older stocks stay separated from fresh supplies. The trouble starts small and grows quickly when chemicals hide in the wrong place.
Preparation Changes Everything
Safety plans are only plans until people train for them. Everyone who steps into the lab must know where to find eyewash stations, showers, and exits. I remember a drill where a scientist fumbled for an eyewash after a splash. Practicing response routines drilled the location into memory—after that, hands moved without thinking. Quick access sets apart minor accidents from lasting injury.
Waste Disposal Shouldn’t Be an Afterthought
No one enjoys handling chemical waste, but leaving even a little behind can cause trouble for the next worker. Residues from Diethyl Chlorophosphonate need sealing in dedicated containers and must never rush down the drain. Certified waste disposal removes the dangers, and inspections keep everyone honest. I’ve seen the cost of improper disposal paid in hospital bills and lost work.
Building a Culture of Respect
Diethyl Chlorophosphonate works as a powerful tool in the right hands, but care and knowledge make it safe. Mistakes shrink when people respect the compound and follow the rules every time. Protective gear, good training, and clear safety routines stand together to prevent the worst. The best labs build a culture where safety means looking after yourself and the next person at the bench.
What is the chemical formula and structure of Diethyl Chlorophosphonate?
Chemical Formula and Structure
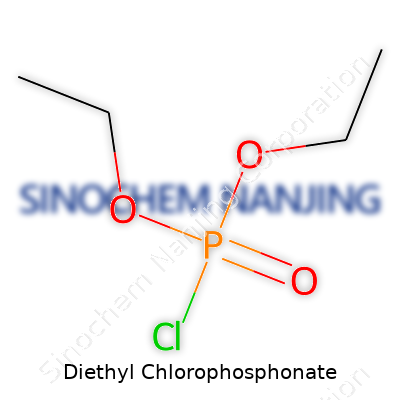
I find chemistry becomes more approachable when a compound’s identity and layout are clear. Diethyl chlorophosphonate carries the formula C4H10ClO2P. If you break things down, it fuses the ethyl group’s flexibility with the clout of phosphate chemistry. At the core sits a phosphorus atom. It forms bonds with two ethoxy groups, a double-bonded oxygen, and a chlorine atom. Chemists draw it like this: Cl–P(=O)(OC2H5)2. The two ethyl branches stick out from the phosphorus, chlorine balances the electronegativity, and the double-bonded oxygen gives it that punchy reactivity chemists rely on.
Structurally, you see a tetrahedral arrangement around phosphorus. Two oxygen atoms link to ethyl chains (–OC2H5). One oxygen binds doubly (P=O), stabilizing the structure. The single chlorine atom leaves fewer electrons on phosphorus, making it more open to attack—useful in chemical synthesis. Its SMILES representation, CCO[P](=O)(Cl)OCC, gives another angle for those juggling molecular modelling.
Why Diethyl Chlorophosphonate Matters
This compound plays a real workhorse role in organic chemistry. I remember prepping for a synthesis at the lab bench: nothing quite matched the efficiency of diethyl chlorophosphonate when it came to making phosphonates through the Michaelis-Arbuzov reaction. The reagent’s balanced reactivity lets chemists tweak molecules, whether building new drugs, crafting custom insecticides, or putting together flame retardants. The world leans hard on intermediates like these.
Economic demand lines up with research. Statista and industry reports show strong interest in chemical markets using phosphonates, including pharma and materials science. Transparency about origin and purity also matters: suspiciously sourced material disrupts scientific and commercial supply chains. The European Chemicals Agency maintains close tabs, flagging hazardous properties and highlighting proper handling since toxicity concerns surround compounds with phosphorus-chlorine bonds.
Health and Safety Concerns
Anyone who has handled diethyl chlorophosphonate knows its hazards aren’t hypothetical. Chlorinated organophosphorus compounds often release noxious fumes and can burn on skin contact. Direct exposure risks eye and respiratory damage, and improper waste treatment creates long-term environmental contamination. A research report in the “Journal of Occupational Medicine” described multiple incidents where exposure outpaced protection. Personal experience says even experienced techs must stay vigilant—basic goggles and gloves sometimes won’t cut it; fume hoods, full PPE, and smart storage turn into standard, not luxury.
Legislation reflects this. The EPA’s chemical regulatory framework tags organophosphorus agents as “chemicals of interest,” requiring robust control and disposal plans. Failing to respect these guidelines threatens both worker health and licensing.
Looking for Better Ways Forward
Safer chemistry is gaining momentum. Green chemistry circles talk about moving toward alternative reagents. Instead of direct chlorinated phosphorus compounds, labs are trialling milder agents or solvent-free reactions. Experiences in educational settings show students increasingly learning these techniques, swapping out hazardous chemicals for less toxic options wherever possible.
One practical step for facilities: invest in engineering controls and automation that limit human contact. Another: continuous training with case studies so that every chemist on the bench knows why protocols matter, not just what they are. That’s the approach keeping both labs productive and workers safe as phosphonate chemistry keeps powering innovation.
How should Diethyl Chlorophosphonate be stored?
Handling Something This Nasty
Anyone who has worked in a decent lab knows that not every chemical deserves the same shelf. Diethyl Chlorophosphonate proves that point right away. It stings your nose, burns your skin, and it doesn't forgive sloppy storage. Keeping this bottle anywhere near water, heat, or just flammable stuff spells out trouble. It can start a fire or turn an accidental dribble into toxic fumes that nobody wants to breathe.
Locking Down the Basics
A dry, cool place does more than meet some regulation. Chemicals like this love to react, especially if moisture leaks in. I remember a time a bottle not unlike this one sweated under a loose seal — we caught it fast, but it etched a lesson into everyone there. So, use tightly sealed containers, and keep the bottle upright. Never just tuck it in with acids or bases. One small mix-up led to a shelf evacuation in my grad school days, and that's an afternoon you never wish to repeat.
Proper ventilation turns a risky storage area into something manageable. I've seen storerooms with vents rigged higher than the city permits demanded, and nobody ever regretted the extra effort. Vapors build up before you even know, so aim for constant airflow. Steel or polyethylene cabinets marked for poisons or corrosives serve the purpose much better than home plastic bins. Good signage stops confusion and leaves no excuse for careless hands reaching in the dark.
Risk Sits Right Outside the Door
The real worry isn’t just about spills. This stuff reacts with water — and pipes always leak sooner or later. Keeping distance from any plumbing or cleaning area prevents emergency calls. Smart labs put all high-risk bottles together behind locked doors, building in fire suppression and spill kits within arm’s reach. That roll of absorbent pads once stopped a small puddle from becoming an all-out disaster. Keeping those tools close never slows you down; it only protects everyone on the team.
People Over Paperwork
Rules make sense, but people following them make the difference. Every chemist, tech, and custodian needs real training. Watching a video won't help in that split-second when you knock something over. Instructors walking new staff through the actual bottles, talking face-to-face about mistakes we've all seen, keeps safety fresh. Labeling each bottle with the date, hazard symbols, and contact information makes sure nobody is left guessing.
Getting Ahead of Emergencies
Spill containment goes beyond one of those wishful "spill trays." Double containment — a bin within a bin — slows leaks. Emergency eyewash stations and showers shouldn't collect dust. I once watched a quick-eyed postdoc push a panicked intern right under the water after an exposure. She knew her training and it saved time, skin, and possibly someone's sight. Having that lifesaving equipment as close as possible makes a difference when seconds count.
Responsibility Travels Home
These harsh chemicals remind me that nothing replaces respect for the bottle. It isn’t enough to tag a rulebook to the wall. Checking containers each week, keeping logs for every use, and making sure no bottle sits forgotten — that earns trust from coworkers and keeps your conscience clear. If you ever feel lazy around something like Diethyl Chlorophosphonate, take a breath and remember: you take that risk home on your shoes. Let’s make sure nobody has to learn that the hard way.
What are the health and safety hazards associated with Diethyl Chlorophosphonate?
Real Risks, No Room for Error
Diethyl chlorophosphonate doesn’t come up often at the dinner table, but people using it in labs or industrial settings face big risks. This chemical goes well beyond the typical irritant. Those who handle it know the burn it brings to skin, eyes, or the lining of the nose. Even a small spill can send sharp, choking fumes into the air. I remember working in a lab where a tiny bottle cracked, and the stinging odor alone sent us scrambling for fresh air and our safety gear.
What Makes It So Dangerous
Phosphorus compounds can lay waste to tissues. Diethyl chlorophosphonate, in liquid or vapor form, hits harder than simple acids or bases. On contact, it wrecks proteins and fats in skin, causing blisters and deep burns. Inhalation risks turn serious fast—one breath of the fumes can choke off airways, inflame lungs, even lead to chemical pneumonia.
The Scariest Part: Long-Term Exposure
Prolonged exposure brings subtler damage. Chronic low-level inhalation may weaken the nervous system. Workers report everything from chronic coughs to memory lapses and poor coordination. The data is pretty clear: regular contact damages nerves, especially if the job skips proper ventilation or ignores protective protocols. The chemical has shown signs of toxicity to internal organs over long periods in animal studies. Long-term low-dose studies in workers could bring more data, but companies aren’t always eager to gather this kind of information.
Immediate First Aid Can Make a Difference
You don’t need to be a chemist to understand the value of fast action. If skin meets this compound, flush with water for twenty minutes—don't just run it under the tap for a second. If you breathe it in, get to fresh air and seek medical help—not just rest. Eyes need an eye-wash station, not a quick splash. People who don’t take these steps usually remember, and not in a good way.
The Limits of Protective Gear
Standard gloves and goggles aren’t enough here. Only thick nitrile or butyl rubber gloves block the chemical. Face shields and lab coats cut exposure, but the most important safeguard is a working fume hood. Labs that treat hoods like storage closets take needless chances. Ventilation isn’t just another box to check; it stands out as the frontline defense.
How Workplaces Can Step Up
Workplaces can cut risks by enforcing tougher training, honest risk communication, and strict equipment checks. Refresher training saves lives, especially for new hires unfamiliar with less common chemicals. Storage should never mix diethyl chlorophosphonate with water or alcohols; those combinations spark violent reactions, possible fires, or toxic smoke. Labeling every container with the right hazard warnings isn’t optional, it’s basic decency.
Better Regulation, Better Protection
Authorities like OSHA and the EPA offer some guidance, but enforcement always lags behind best practice. More oversight and real-world inspections, along with consistent updates to chemical safety data sheets, would boost confidence. Worker whistleblower protections matter too—people need to feel safe speaking up if shortcuts creep in.
Knowledge Means Real Safety
Those who work around diethyl chlorophosphonate can’t afford complacency. One missed step—one bit of bad advice—can shape a life forever. For all the talk of personal responsibility, safe environments grow from solid information and collective action. Experience, facts, and open communication help keep everyone standing, safe, and looking out for each other.
| Names | |
| Other names |
Diethyl phosphorochloridate Chlorophosphoric acid diethyl ester Phosphonic chloride, diethyl ester Phosphonochloridic acid, diethyl ester Diethylphosphoryl chloride |
| Pronunciation | /daɪˌɛθaɪl klɔːrəˈfɒsfəneɪt/ |
| Identifiers | |
| CAS Number | '2523-68-8' |
| Beilstein Reference | 1638733 |
| ChEBI | CHEBI:132679 |
| ChEMBL | CHEMBL504553 |
| ChemSpider | 13718621 |
| DrugBank | DB08385 |
| ECHA InfoCard | 03e6e242-9d83-4bba-a4db-6ff99cf0a8ab |
| EC Number | 203-652-6 |
| Gmelin Reference | 82055 |
| KEGG | C18621 |
| MeSH | D017977 |
| PubChem CID | 79211 |
| RTECS number | TB5950000 |
| UNII | 2U36NZZ5C3 |
| UN number | UN2037 |
| Properties | |
| Chemical formula | C4H10ClO2P |
| Molar mass | 170.56 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Pungent |
| Density | 1.319 g/mL at 25 °C |
| Solubility in water | soluble |
| log P | 0.79 |
| Vapor pressure | 0.4 mmHg (20 °C) |
| Acidity (pKa) | 1.62 |
| Basicity (pKb) | 1.74 |
| Magnetic susceptibility (χ) | -43.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.424 |
| Viscosity | 1.669 mPa·s (20°C) |
| Dipole moment | 3.33 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 324.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -675.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1538.8 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS07 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301, H311, H314, H331 |
| Precautionary statements | H302 + H312 + H332, H314, H411, P261, P264, P270, P271, P273, P280, P301 + P312, P302 + P352, P304 + P340, P305 + P351 + P338, P310, P311, P321, P330, P363, P391, P403 + P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 118 °C (244 °F; 391 K) |
| Autoignition temperature | 456 °C |
| Lethal dose or concentration | LD50 oral rat 25 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 75 mg/kg |
| NIOSH | SB8925000 |
| PEL (Permissible) | PEL (Permissible) for Diethyl Chlorophosphonate: Not established |
| REL (Recommended) | 3 mg/m³ |
| IDLH (Immediate danger) | IDLH: 5 ppm |
| Related compounds | |
| Related compounds |
Dimethyl chlorophosphonate Diethyl phosphite Diethyl phosphonate Ethyl dichlorophosphate Triethyl phosphite |

