Dichlorotetrafluoroethane: Balancing Legacy, Science, and Responsibility
Tracing the Roots and Realities of Dichlorotetrafluoroethane
Dichlorotetrafluoroethane, known to chemists and industry workers mostly by the shorthand CFC-114 or R-114, has seen its fortunes rise and fall with the growing awareness of how chemicals interact with the environment. Emerging in the early decades of the Refrigeration Age as a supposedly clean replacement for dangerous or toxic refrigerants, this compound caught on quickly during the postwar economic expansion. Chlorofluorocarbons (CFCs) like this one found their way into air conditioners, aerosol propellants, and even as a blowing agent in foam production. Engineers, driven by optimism and the need for more comfort and convenience, kept finding uses for such chemicals without foreseeing the complex reactions that play out in the atmosphere.
Everyday Chemistry Behind Dichlorotetrafluoroethane
CFC-114, with its formula C2Cl2F4, gives off no odor and cannot be seen. It boils well below room temperature and does not burn in air, which creates fewer workplace hazards than more volatile refrigerants or propellants from earlier times. Its stability, which allowed manufacturers to move and store it with fewer explosions or leaks, ultimately led to bigger problems. As this molecule scattered through the world’s appliances and containers, its slow breakdown let it spiral skyward into the upper atmosphere, where high-energy sunlight nudged chlorine atoms free. These atoms go on to steal oxygen atoms from ozone, cutting open a layer that life on Earth depends on. The chemical reliability that once made this molecule valuable showed its double edge.
What Goes on the Label and the Lab Table
Product names offer no shortage of confusion, with synonyms like Freon-114, Refrigerant 114, and even obscure identifiers like UN1958 appearing in regulations or industry manuals. Reading some of the old technical brochures, you’ll see claims of purity and pressure ratings, with warnings about storing this refrigerant in steel tanks out of sunlight or away from strong acids and bases. Labels in various countries reference boiling points around 4°C, vapor pressures that can rupture containers at higher temperatures, and an unwavering demand for sealed systems designed for stable pressures and easy gas handling. Mechanics and researchers who grew up in the industry remember sweating over pressure gauges and checking for leaks with foaming agents, knowing even minor escapes meant not just loss of expensive material but direct environmental harm.
Crafting, Reacting, and Rebuilding a Molecule
Manufacturers turn dichlorotetrafluoroethane into a reality by reacting simple precursors such as carbon tetrachloride and hydrofluoric acid under the watchful eye of catalysts and strict temperature controls. The chemistry isn’t terribly fancy—halogen exchange offers a fairly direct way to swap in the fluorine atoms for the hydrogens or chlorines that sit on simpler molecules. What makes it tricky is that side products, like other CFCs or even perfluorocarbons, need to be kept in check. Once formed, this compound doesn’t go away easily. Sunlight in the lower atmosphere leaves it untouched for decades. Chemists have shown that ultraviolet light split this molecule only in the stratosphere. There, every single atom of chlorine freed up can start a chain reaction whittling down ozone. Scientists such as Mario Molina and Sherwood Rowland pieced together this destructive loop, triggering a global move to shut down production and limit emissions.
Applications and the Spillover of Innovation
CFC-114’s story never stopped with just air conditioners or spray cans. It powered military refrigerators at polar research stations and even helped push medicine into fine mists for inhalers early on. Wartime engineers found it indispensable for certain nuclear reactor operations, where its chemical inertness kept it from eating away sensitive metals. Aerospace contractors valued it for cooling, stymied by the tradeoff between fire risk and chemical stability. I remember old mechanical shops where cans labeled “Freon 114” were standard toolkit fare, used for everything from cleaning electronics to spotting leaks. The short-term victories—longer-lasting appliances, safer propellants, and lower maintenance costs—gave way to long-term reflection.
Weighing Safety and Acceptable Risks
Learning from the slow spread of knowledge on CFC safety, those in charge of standards realized relying on chemical inactivity can mask major risks. Early on, users shrugged off its low flammability and acute toxicity, focusing on the fact that spills didn’t lead to explosions or asphyxiation. The real risk lay in slow, invisible movements: trace releases from leaking tubes, landfill coffins leaking into groundwater, and forgotten cylinders venting decades after use. Modern standards demand tight handling, with storage rooms bearing ventilators, personal protective gear for those handling pressurized tanks, and strict checks on recovery and recycling. Manuals now spell out the deadly impact on the ozone hole, backstopped by laws that push for responsible collection and destruction, not just workplace safety.
Digging Into the Science: Toxicity and Human Health
We have known for a long time that dichlorotetrafluoroethane does little harm in small, short exposures, thanks to its chemical stability and low reactivity at ground level. People who worked daily with refrigerants in garages or factories, though, chased headaches or spells of dizziness if a valve burst or a hose failed. Flooding an enclosed space with even these “safe” gases pushes out oxygen, leaving the risk of simple suffocation. Chronic studies, where animals or even unlucky volunteers got exposed to higher doses, highlighted slow buildups in fatty tissues and the chance for organ damage. The indirect toll, through atmospheric damage and increased radiation exposure, proved even more insidious. Researchers tracked rates of skin cancer, crop failure, and ecological loss, all back to the thinning ozone and increased ultraviolet exposure resulting partly from compounds like this one.
New Paths and Unfinished Business
As global treaties like the Montreal Protocol cut back on CFC-114 production, the hunt for cleaner replacements hit overdrive. Hydrofluorocarbons (HFCs), hydrochlorofluorocarbons (HCFCs), and still newer blends stepped in to fill gaps. They bring complications—climate warming, tricky waste disposal, and new types of leaks. Research labs haven’t stopped testing molecular tweaks or exploring non-chemical solutions, including advanced heat pumps or alternative cooling cycles powered by magnetism or water. Studies keep adjusting toxicity models and exposure limits, thanks to findings that long-lasting molecules can appear unexpectedly in wild places—from mountaintop air to deep-sea silt. Environmental chemists, engineers, and regulators now work together (sometimes uneasily) to balance public comfort with planetary risk. Tools for measuring and destroying leftover dichlorotetrafluoroethane have grown more precise, but old stockpiles and buried waste threaten to come back at us for years. Engineers who once trusted in the “miracle molecule” realize that chemistry never forgets and rarely forgives careless uses, making the ongoing debate over legacy chemicals such as this one a lesson in both humility and hope.
What are the primary uses of Dichlorotetrafluoroethane?
Where It Shows Up in Everyday Life
Dichlorotetrafluoroethane, often called CFC-114, popped up as a go-to solution in industries searching for a reliable, non-flammable, and stable chemical. I remember the first time I read about CFCs in old appliance manuals—they’ve touched more lives than most people realize. This compound made a splash thanks to its performance as a refrigerant, especially when households across the world started demanding reliable refrigerators and air conditioners that didn’t catch fire or break down under pressure.
Refrigeration and Cooling
The real workhorse role for dichlorotetrafluoroethane landed in the world of cooling. As modern life brought more air conditioners, commercial refrigeration, and even large-scale chillers, CFC-114 filled those coils for decades. One reason for its popularity was its chemical stability and low toxicity compared to the early alternatives. The substance let manufacturers push for more efficient refrigerators, freezers, and industrial coolers before anyone threw out concerns about climate impact.
Propellant in Aerosols
Spray cans revolutionized packaging, bringing canned shaving cream, cooking sprays, and even bug sprays to store shelves. CFC-114 helped make these products better. Its stability and ability to turn liquids into fine mists without causing fires made it attractive for consumer and industrial aerosols. In factories and workshops, workers could rely on consistent results without worrying about explosions or harmful fumes during use.
Foam Blowing Agent
Foam insulations and packaging materials owe a lot to dichlorotetrafluoroethane. Insulation panels, flotation devices, and commercial packaging improved noticeably when this chemical joined production lines as a blowing agent. Companies produced lighter, more effective panels and shipping containers. Looking around older houses, I can almost guarantee that some attic or walls keep warmth inside because CFC-114 helped puff up that insulation years ago.
Other Industrial Applications
Labs and specialty manufacturers sometimes picked this compound for chemical processing and cleaning. High-tech industries—even aerospace—found it useful for precision cleaning parts where residues from other chemicals couldn’t be risked. While far less common than refrigeration or aerosols, these niche uses pushed technology forward and supported critical research.
Environmental Impact and the Shift Away
Science doesn’t stand still. Researchers in the 1970s and 1980s discovered CFC-114 and similar chemicals ate away at the ozone layer. The risk pushed regulators and companies to phase out these products under agreements like the Montreal Protocol. Most refrigerators and aerosol cans today use alternatives, but older equipment might still contain CFC-114.
Ways Forward
Handling legacy systems and old foam insulation takes care. Proper disposal matters—a lesson every eco-conscious contractor and technician learns quickly. Safe collection and destruction keep leftover CFC-114 from escaping. For new products, chemists favor refrigerants and propellants with lower ozone and climate risks. These changes cost time and money up front, but public health and safe air deserve that investment.
Trust and Responsibility
Looking back, most people used CFC-114 with the best technology and knowledge available. Science learned more, and society made changes, showing that good stewardship grows from honest mistakes and responsible action. Staying informed and choosing safer alternatives keeps communities and the planet healthier—I’ve seen first-hand how much progress happens when people work together on solutions.
Is Dichlorotetrafluoroethane harmful to the environment?
A Chemical Hidden in Plain Sight
Dichlorotetrafluoroethane, known by its short name CFC-114, might sound like something out of a chemistry book most folks never crack open. In reality, this compound played a huge role in keeping food cold and air conditioning running for decades. As a person who worked in facilities management, I spent years around equipment that relied on this and similar chemicals. Most folks wouldn't think a refrigerant could cause global trouble, but the legacy of these substances teaches otherwise.
From Sky to Sea: Where CFC-114 Lingers
CFC-114 belongs to the family of chlorofluorocarbons, and that family shaped more than industries — it shaped the sky. What I saw on maintenance logs across buildings from schools to grocery stores matched what scientists reported: leaks and vents releasing refrigerants straight into the air. Once there, CFC-114 sticks around for nearly 200 years. Longer than any of us will be here. That’s plenty of time to drift to the upper atmosphere, break apart under sunlight, and release chlorine atoms that chew through ozone molecules.
Ozone keeps dangerous ultraviolet rays in check. Many policy changes, including the Montreal Protocol, put strict controls on CFCs decades ago because ozone holes started showing up above Antarctica. Studies from NASA and the European Environment Agency report that ozone loss peaked in the 1990s, but we’re still cleaning up that mess today. Some regions deal with more skin cancer and weakened immune systems linked to extra UV rays, not to mention plant and animal harm.
Not Just an Ozone Issue
People talk a lot about ozone, but CFC-114 packs another punch. This stuff traps heat in the atmosphere. Its global warming potential reaches over 8,700 times that of carbon dioxide over a century. It doesn’t just vanish, either. Commercial buildings, old refrigerators, and scrap yards leak CFC-114 and similar chemicals each time a seal fails. In my early days hauling old chillers, workers shrugged at the gas escaping; few realized those leaks would linger in the air, stacking up with every careless dump.
Factoring in Solutions
Most countries outlawed the fresh production of CFC-114, but what’s done is done. Millions of units packed with legacy chemicals still operate worldwide. In the waste industry, I learned that proper reclamation and destruction methods can trap or destroy CFC-114 before it escapes. Non-profit programs and manufacturer take-back efforts help, but results run uneven, especially in low-income settings where technical skill and equipment lag.
Alternatives like HFCs and newer blends roll out, and while some fix the ozone problem, others keep warming the climate. Watching change on the ground, I found that strong enforcement, public education, and better incentives for proper disposal matter just as much as fancy chemical research. Only with steady teamwork from industry, regulators, and folks doing the actual work can we chip away at the mess CFC-114 made — and dodge repeats with new chemicals down the line.
What safety precautions should be taken when handling Dichlorotetrafluoroethane?
Understanding the Risks
Dichlorotetrafluoroethane finds a place in many industrial settings, often as a refrigerant or blowing agent. The chemical serves a purpose, but respect for its power keeps people out of trouble. My first encounter with refrigeration work taught me fast: this isn’t stuff you experiment with. When inhaled, exposure does more than just irritate; it can leave a person dizzy or short of breath. Long-term or repeated exposure threatens more, with potential effects on the heart and the nervous system. It’s not just about what happens in the moment—long-term safety rides on what you do each step of the way.
Personal Protective Equipment Saves More Than Just Skin
I’ve seen mechanics shrug off gear, saying goggles “fog up” or gloves slow them down. The right gloves make a difference, and good ventilation never seems like a big deal until the vapors show up in your chest. The Centers for Disease Control and Prevention say proper PPE cuts risk sharply. This chemical calls for goggles that seal to your face and gloves rated for chemical protection. A simple dust mask won’t help; a cartridge respirator suited for organic vapors stands between you and a hospital visit. Even one splash or inhalation changes your outlook fast. In the end, PPE feels like a small price against the cost of a mistake.
Ventilation: Your Frontline Defense
Small spaces trap fumes quickly. An open window works better than nothing, but mechanical ventilation pushes air out where it belongs. Installing exhaust fans above workstations keeps air moving and drops the odds of respiratory problems. On days when things feel stuffy or there’s a hint of a chemical odor, you know the ventilation has slipped. Air monitors that pick up on dangerous concentrations don’t cost much compared to hospital bills or shutdowns after accidents. I remember an old shop foreman who never turned on the fans “to save a few bucks”—his team suffered every winter. Few shortcuts backfire as quickly.
Proper Storage and Labeling
Leaving chemicals unlabeled or piling containers in warm, cramped corners opens the door to leaks and confusion. Store this compound in tightly sealed, clearly marked containers and keep everything away from open flames, heat sources, or direct sunlight. Shelves should stay well away from staff gathering spots or break areas. In my own work, a clear labeling system and up-to-date safety sheets hung by the entrance have saved headaches more than once. The right labels and a no-excuses attitude on cleanup stop small problems from turning into emergencies.
Training Matters; Complacency Hurts
People enter this line of work every day believing rules slow them down. Proper training goes beyond handing someone a material safety data sheet. Regular drills, hands-on demonstrations, and real stories about accidents give safety meaning. Companies that put real effort into training see fewer incidents and respond faster when things go wrong. Keeping lines open between workers and supervisors, encouraging questions, and never rushing through directions pay off in lives, not just statistics.
What Happens Next
Technology changes, and alternatives to dangerous chemicals keep popping up, but nothing replaces vigilance. The push for safer alternatives to Dichlorotetrafluoroethane gains steam every year, but people still need to handle the real thing for now. Simple habits, clear communication, and a crew unwilling to cut corners—these outrun fancy equipment every time. In my experience, a team that works together and listens to lessons from the past stands up to any hazard, chemical or otherwise.
What are the storage requirements for Dichlorotetrafluoroethane?
Why Proper Storage Matters
Dichlorotetrafluoroethane, known to many as a refrigerant or as a chemical used in industrial cleaning, comes with risks if stored without planning. Breathing in its fumes or letting it leak near heat sources can cause serious health and fire hazards. Years spent working with refrigerants have taught me that even a small mistake in handling or storage can mean heavy fines, ruined stock, and health scares you remember long after.
Temperature and Ventilation
This chemical stands out for its volatility. Keep it cool and away from sunlight. Store it in a spot where the temperature stays steady, ideally below 50°C. Fluctuating heat can push vapor pressure up, which increases the risk of leaks or ruptures. Avoid cramming it into warm supply rooms or stashing it close to boilers and break areas.
A well-ventilated storage area makes all the difference. While it barely smells, undetected vapors displace oxygen and put workers at risk if the space is poorly aired. I’ve seen shops thrive on simple routines, like cracking a window or installing a low-cost exhaust fan to keep air moving and keep workers safe.
Physical Containment and Labeling
Heavy-duty steel cylinders mark the best choice for storing dichlorotetrafluoroethane. Make sure each tank sits upright and secure. Chains or racks work well to stop them from falling over. I’ve seen fires start from tanks rolling and striking each other, which makes anchoring them a step nobody can skip.
Every container needs a clear label. I can't count how many times someone in a warehouse has said, “I thought it was just compressed air.” A clear label with content, hazard signs, and emergency contact info can spare a business from disaster. Safety Data Sheets, or SDS, should sit right nearby for quick look-up.
Keep Away from Incompatible Materials
Some chemicals do not play well with dichlorotetrafluoroethane. Strong alkalis, alkaline earth metals, or burning magnesium and aluminum can trigger violent reactions. It pays to organize storage spaces by chemical class. Even the most stretched facilities manage better when common sense rules the layout, keeping flammables and oxidizers well apart.
Emergency Planning and Training
Written procedures and regular staff training make the biggest difference. Fire drills, spill plans, and first aid refreshers create an environment where anyone on site knows what to do. Even on hectic days, if storage safety becomes routine, the chances of slip-ups go way down.
Leak detection systems—whether basic alarms or more elaborate monitoring—alert staff at the earliest sign of trouble. Prompt action stops a minor problem from turning major.
Long-Term Considerations
Regulations shift frequently. Be ready to review chemical storage policies at least once a year. I check updates from OSHA and the EPA here in the States; other countries issue their own clear guidelines. Compliance doesn’t just avoid fines, it builds trust with staff and customers.
Dichlorotetrafluoroethane brings value for refrigeration and cleaning, but only where storage matches safety. Skipping precautions bites back hard in fines, downtime, or health scares, so regular review and plain communication make every workplace more resilient.
Are there any alternatives to Dichlorotetrafluoroethane for industrial applications?
Why Stick With Old Refrigerants?
People in manufacturing, HVAC, and chemical processing know Dichlorotetrafluoroethane by its trade names, but to most folks it’s just another part of cooling systems or foam blowing agents. It rarely grabs headlines, yet regulations and environmental concerns put this chemical in the spotlight. The ozone depletion and global warming numbers aren’t small; if you look at the Global Warming Potential (GWP), Dichlorotetrafluoroethane carries a heavy load compared to what’s now possible. The Montreal Protocol and its updates weren’t just about picking on one chemical—they came from real science and experience. Nitrous oxide and carbon dioxide from cars get more attention, but these refrigerants quietly do more damage per molecule than a full highway at rush hour.
Alternatives With Fewer Regrets
Having worked on retrofitting chillers in an aging office park, I’ve seen how new refrigerants perform. Hydrofluorocarbons such as HFC-134a became popular as drop-in replacements because they skipped the chlorine atom, so they didn’t shred ozone overhead. Some technicians balked at the change, just out of habit. It turns out, though, these substitutes still trap heat effectively in the atmosphere. The climate impact may sit lower on the list than Dichlorotetrafluoroethane, but it’s no lasting fix.
Hydrofluoroolefins (HFOs) deserve a closer look. They break down faster in the air and keep their GWP low. Chemours markets HFO-1234yf and it’s now found in most newer car air conditioners. Honeywell’s HFO-1234ze is landing in supermarkets and even industrial chillers. Changing to HFOs took some adjustment with handling and equipment upgrades, but the science supports their positive impact. These switches also lead to better worker safety, since HFOs don’t linger in a workspace like older refrigerants.
Not Just About New Chemicals
Some facilities skip fluorinated refrigerants altogether and pick natural alternatives. Carbon dioxide (CO₂, R-744) shows up in large supermarket freezers. The technology used to be too pricey and complicated, but better compressors and controls fixed that. Email chains between plant engineers swapped stories about first trying CO₂: pressure spikes made everyone nervous. With good training and new safety valves, these problems faded.
Ammonia (NH₃, R-717) gets a bad rap for being pungent and hazardous if released, but it cools efficiently, never hurts the ozone layer, and has no real climate downside. Plenty of food processors rely on ammonia—my cousin’s dairy plant has used it for decades. They run safety drills, and their response time beats most fire departments when alarms go off. Regular maintenance beats chemical risk in this business, no matter what refrigerant you choose.
Cleaner Choices Come With Tougher Decisions
No perfect answer exists in chemical replacements, and a move to greener options always comes with some headaches. Most big companies do lifecycle analysis now, tallying the cost of leaks, end-of-life disposal, and potential retrofits. For small businesses, the upfront bill matters most. Incentives help; when local governments chip in on chiller upgrades, adoption grows. The knowledge is out there—engineers compare equipment, regulators offer guidelines, and safety boards update their recommendations. The only thing left is to keep pushing for choices that don’t leave a mark on air, ozone, or ice caps. That takes resolve from boardrooms to plant floors.
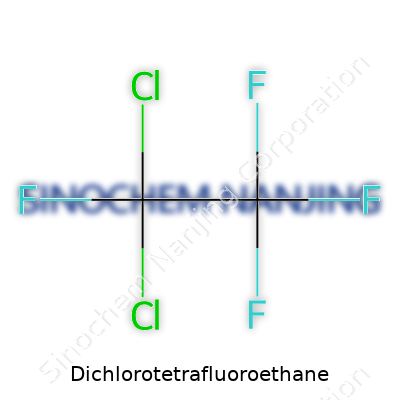
| Names | |
| Preferred IUPAC name | 1,1,2,2-Tetrafluoro-1,2-dichloroethane |
| Other names |
CFC-114 Freon 114 Refrigerant 114 R-114 1,2-Dichloro-1,1,2,2-tetrafluoroethane |
| Pronunciation | /daɪˌklɔːroʊˌtɛtrəˌflʊəroʊˈiːθeɪn/ |
| Identifiers | |
| CAS Number | '76-14-2' |
| Beilstein Reference | 1733733 |
| ChEBI | CHEBI:39097 |
| ChEMBL | CHEMBL2105961 |
| ChemSpider | 15306 |
| DrugBank | DB01370 |
| ECHA InfoCard | 03f93eaf-0973-43c5-96fc-ba8e42e3d2f2 |
| EC Number | 204-646-6 |
| Gmelin Reference | 6078 |
| KEGG | C06742 |
| MeSH | D002941 |
| PubChem CID | 6387 |
| RTECS number | KI1100000 |
| UNII | G5A3OM8Y7A |
| UN number | UN1022 |
| Properties | |
| Chemical formula | C2Cl2F4 |
| Molar mass | 170.922 g/mol |
| Appearance | Colorless liquefied gas |
| Odor | Faint ethereal odor |
| Density | 1.47 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.39 |
| Vapor pressure | 1420 mmHg (20°C) |
| Acidity (pKa) | >16.8 |
| Basicity (pKb) | 11.96 |
| Magnetic susceptibility (χ) | -6.43×10^-6 |
| Refractive index (nD) | 1.355 |
| Viscosity | 0.91 cP (25°C) |
| Dipole moment | 1.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 329.7 J·K⁻¹·mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –1034 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1622.6 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N01AB01 |
| Hazards | |
| Main hazards | Toxic if inhaled; may cause asphyxiation by displacement of air; may cause frostbite. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS04 |
| Signal word | Warning |
| Hazard statements | H220, H280 |
| Precautionary statements | P210, P273, P304+P340, P305+P351+P338, P403 |
| Autoignition temperature | 412°C |
| Explosive limits | Explosive limits: 7.5–15.5% |
| Lethal dose or concentration | Lethal Concentration (LC50) Inhalation - Rat - 16,900 ppm/4h |
| LD50 (median dose) | LD50 (median dose) of Dichlorotetrafluoroethane: >5000 mg/kg (rat, oral) |
| NIOSH | PB8775000 |
| PEL (Permissible) | 50 ppm |
| REL (Recommended) | 0.5 ppm (3.6 mg/m³) |
| IDLH (Immediate danger) | 2000 ppm |
| Related compounds | |
| Related compounds |
Chlorofluoroalkane Chlorotrifluoroethane Chloropentafluoroethane Chlorodifluoromethane |

