Dichlorophenyl Isocyanate: Insight, Progress, and Prospects
Historical Development
People working in chemical research have explored the phenyl isocyanate family since the early twentieth century. After discovering how isocyanates react with other organic compounds, chemists started searching for reactive analogues with unique behavior in industrial settings. Dichlorophenyl isocyanate emerged from these pursuits, thanks to its distinctive reactivity caused by chlorine atoms snugged onto the phenyl ring. During the boom in plastics and pesticide industries in the mid-1900s, demand for building block molecules like this soared. Research journals across Europe and the United States published investigations showing that dichlorinated isocyanates could anchor themselves in new polymer backbones, leading to critical innovations in specialty coatings and tough resins. The market for this compound expanded through the years not by accident but from hard lessons learned in both lab accidents and commercial breakthroughs, as safety standards and handling protocols toughened after early mishaps. This chemical’s story isn’t just about scientific curiosity—it’s about finding solutions as technology changed society’s expectations on durability, performance, and environmental responsibility.
Product Overview
Dichlorophenyl isocyanate stands out as an organic compound used mainly in advanced manufacturing. You find it as a colorless or slightly yellowish crystalline solid or powder, depending on the grade and purification method. Industry often uses it for synthesizing specialty polyurethanes, unique crop protection agents, and even medical intermediates. This compound owes its popularity to its unmatched ability to graft isocyanate groups onto aromatic frameworks, broadening the toolbox for resins, adhesives, and surface treatments. Research-driven manufacturers tout its ability to introduce reactive chlorine sites, which allow downstream producers to selectively tweak final products. Cost has always played a role in its wider adoption; over time, economic synthesis routes and increased global supply helped drop prices and made it viable even for mid-sized enterprises. While large-scale players dominate procurement, niche chemical formulators increasingly order custom batches tailored for specific end-market needs.
Physical & Chemical Properties
Anyone working around dichlorophenyl isocyanate tends to take careful notes—this is not a run-of-the-mill commodity chemical. It comes with a sharp, acrid odor that signals danger for unprotected handlers. Its melting point hovers near 50°C, making it stable enough for bulk shipments but still sensitive to heat or friction. Solubility leans toward organic solvents like toluene, xylene, and dichloromethane, meaning aqueous cleanups don’t help much after a spill. Its structure features two chlorine atoms bonded directly to the aromatic ring, sandwiched opposite the isocyanate group. This substitution pattern draws electron density away from the ring, altering both its nucleophilicity and reactivity compared to other aryl isocyanates. As a result, reactions proceed faster under milder conditions, which helps when you need large batches on tight schedules—the sort of efficiency chemical engineers appreciate. In the lab, this compound goes from shelf-stable solid to highly volatile when heated, sending toxic fumes airside if not carefully controlled.
Technical Specifications & Labeling
Bulk suppliers grade dichlorophenyl isocyanate based on purity, moisture content, and residual chlorinated aromatics. For synthesis work, you want assay values above 99% and moisture below 0.1%, since water triggers hazardous decomposition. The best suppliers package each drum or bottle with the UN number, complete GHS pictograms, and precise shipping class—failure to follow international labeling rules nets serious fines from customs and workplace authorities. Labels warn about respiratory hazards and urge double-gloving; chemical-resistant aprons see frequent use in responsible facilities. Technical data sheets provide spectral analysis (usually IR and NMR), melting range, and storage guidelines, helping chemists match each lot to their planned applications. Internally, manufacturing audits check label integrity, sample traceability, and shelf-life monitoring—a missing label or faded code often brings work to a halt until re-verification.
Preparation Method
Synthesizing dichlorophenyl isocyanate requires not only technical skill but also experience with isocyanate chemistry. Commercially, the go-to method uses phosgenation of dichloroaniline derivatives in an anhydrous medium. Operators add liquid phosgene slowly under inert gas, with careful temperature control to keep side reactions in check. Once the reaction completes, a controlled distillation recovers the purified isocyanate, usually under vacuum to suppress decomposition. Smaller batch chemists sometimes turn to diphosgene as a less hazardous alternative, accepting lower yields for safer handling. Raw material sourcing, particularly high-purity dichloroaniline, sets the tone for the overall quality and price—suppliers with solid technical backgrounds know shortcuts backfire easily. The phosgenation step remains hazardous, so plants run closed-loop systems, with exhaustive air scrubbing and pressure monitoring. No one in a modern lab attempts this without proper training, since the risk of runaway reactions or gas exposure never quite disappears.
Chemical Reactions & Modifications
Dichlorophenyl isocyanate reacts vigorously with nucleophilic compounds, creating polyurethanes, ureas, and carbamates prized for specialized performance. The two chlorine atoms on the ring don’t just sit idle—they enable further modifications post-synthesis, allowing downstream chemists to tune electronic properties or add protective groups. In my own experience, introducing this isocyanate into co-polymer systems grants remarkable resistance to solvents and mechanical stress, a property often overlooked until you find your test coupons outlasting the competition in harsh industrial trials. The compound participates in step-growth polymerizations, curing reactions with diols and diamines, and even serves as a precursor for more exotic, halogen-rich intermediates. Organic synthesis textbooks highlight its spot in forming heterocycles and advanced drug scaffolds, though scale-up needs strict contamination checks, as residual byproducts can destroy batch-to-batch reproducibility.
Synonyms & Product Names
On order forms and safety data sheets, you spot a variety of trade and systematic names for dichlorophenyl isocyanate. Common aliases include 2,4-dichlorophenyl isocyanate, DCPI, or simply dichlorinated phenyl isocyanate. Some suppliers brand their version as “Isochlor,” but savvy procurement staff check CAS numbers—errors here often mean procurement delays or, worse, the wrong material delivered. Regional regulations sometimes favor alternate spellings, leading to confusion for multinational teams who rely on harmonized product codes and documentation. Long-serving chemists keep lists of synonyms on hand, a habit I picked up the hard way after a mislabeled drum showed up during a rush project in the late 2000s.
Safety & Operational Standards
Dichlorophenyl isocyanate leaves little room for safety corner-cutting. Inhalation of its vapors causes severe respiratory irritation and, over time, can sensitize or asthma. Recommended gear includes air-purifying respirators, chemical splash goggles, and gloves built for aggressive solvents and sharp solids. Handling protocols keep the compound away from open water or amines outside closed containers, since exothermic reactions can escalate quickly. Industry relies on detailed process hazard analyses before allowing operators to open any vessel—experienced plant managers enforce written procedures, monitoring systems, and emergency scrubbers running at all hours. I recall a time in a startup pilot plant when a line leak almost went unnoticed; only a vigilant technician’s nose caught the scent before it grew into a work-stopping incident. Regular training and live drills become routine, not only to check the boxes, but because real accidents reinforce how unforgiving these chemicals can be.
Application Area
Specialized industries look to dichlorophenyl isocyanate for tasks where ordinary isocyanates come up short. Manufacturers producing advanced polyurethanes depend on it to add chlorinated features for fire resistance, solvent tolerance, and long-term flexibility, essential in fields like transportation and aerospace. Agricultural chemists prize it as a starting point for active ingredients in pesticides, where its chlorine content and isocyanate function can combine for unique biological effects. Laboratories investigating new medical devices use it to anchor surface coatings that stand up to repeated sterilization cycles. Even in electronics, where insulation and encapsulation require exceptional chemical resistance, engineers rely on the reliability this molecule brings. Small businesses face hurdles acquiring it in the right purity and quantity, but that hasn’t stopped innovation in coatings or specialty adhesives—demand for these remains steady wherever performance, not price, drives purchases.
Research & Development
R&D teams still find untapped potential in dichlorophenyl isocyanate. Academic groups examine its reactivity as a building block for new pharmaceuticals, where the dichloro substitution pattern affects both biological activity and metabolic stability. Formulation scientists dig into co-polymerization mechanics, searching for ways to build lighter, tougher materials for high-stress environments like automotive manufacturing. Industry consortia fund multi-year projects, often at the interface between universities and corporate labs, trying to harness its versatile chemistry for environmental cleanup materials or next-generation sensor coatings. I’ve seen the payoff in collaborative projects where new analytical techniques unlock improved control over the distribution of chlorine atoms, sharpening the performance edge in adhesives or elastomers. Researchers keep the creativity coming, motivated by competitive grants as well as the promise of higher-value products that meet rising regulatory and customer expectations for sustainability, toxics reduction, and product life extension.
Toxicity Research
Studies on dichlorophenyl isocyanate’s toxicity paint a mixed picture—short-term exposure data shows strong irritancy and respiratory effects, while animal studies flag risks of chronic toxicity and carcinogenicity in unregulated environments. Regulatory watchdogs in Europe and North America strictly control its transport, labeling, and workplace exposure limits. In practical terms, workplaces install active ventilation, local leak detectors, and medical surveillance for staff handling these chemicals regularly. Medical research teams monitor for delayed-onset asthma and hypersensitivity among exposed workers, a lesson learned after decades of similar compounds triggering workplace health crises. Policy teams routinely revisit exposure limits as new animal test data emerges and as high-throughput screening uncovers deeper links between chronic exposure and subtle health impacts. In my own experience, cooperation between environmental health scientists and operations staff helps identify risk early—well before health symptoms force costly interventions or losing skilled workers becomes a real threat.
Future Prospects
Looking ahead, dichlorophenyl isocyanate could play a bigger role in both green chemistry and advanced manufacturing if the industry tackles key hurdles on safety and lifecycle impacts. Efforts focus on developing less hazardous synthesis processes, better containment, and next-generation PPE tailored for isocyanate work. In materials science, demand rises for high-performance plastics and coatings that meet new regulations on flame retardancy and environmental persistence—a space where chlorinated isocyanates shine. Ongoing research could deliver derivatives that decompose safely or that recycle more easily, closing the loop on chemical use. Advances in automation and digital monitoring shrink the risks linked to operator error or process upsets, nudging standards higher across the industry. As competition and regulation push for safer, greener alternatives, companies investing in innovation and best practices have the upper hand—reaping financial and reputational rewards for protecting workers and the planet.
What are the common applications of Dichlorophenyl Isocyanate?
The Versatile Role in Polymer Production
Dichlorophenyl isocyanate isn’t a household name, but anyone who has worn water-resistant shoes or relied on hard plastics has brushed up against it in daily life. This compound is a core building block for certain polymers, especially in the production of specialty polyurethanes. Its molecular structure gives finished products more resistance to weather, abrasion, and heat. Manufacturers turn to dichlorophenyl isocyanate to craft polyurethane coatings and foams that stand up to rough conditions, helping extend the useful life of consumer goods and industrial components alike.
Protecting Surfaces with Industrial Coatings
Factories that build bridges, ships, and machinery look for coatings that fend off rust, chemicals, and sunlight. Dichlorophenyl isocyanate forms the backbone of these industrial coatings. Its ability to help create tight, protective barriers ensures that infrastructure doesn’t wear out quickly. The coatings dry into tough films that hang on even in harsh environments. I’ve seen bridge repair projects where specialty coatings with isocyanate chemistry kept steelwork rust-free for years, saving cities hefty maintenance funds.
Making Advanced Adhesives Stick
From building cars to assembling electronics, strong adhesives play a big role in modern manufacturing. Dichlorophenyl isocyanate delivers the crosslinking power that makes certain adhesives bond tightly and last longer. Original equipment manufacturers often lean on this chemistry for applications where glue failure isn’t an option, like attaching windshields or assembling electronics meant to survive rough handling. I’ve spoken with engineers in the automotive sector who appreciate these adhesives for their consistent performance under heat and mechanical stress.
Specialty Uses in Pharmaceuticals and Agrochemicals
Beyond coatings and adhesives, dichlorophenyl isocyanate shows up at the start of more complex chemical syntheses. Certain pharmaceutical compounds draw from isocyanate chemistry to join molecular pieces together. Researchers value its reactivity for crafting intermediates that lead to novel drug candidates. Likewise, the agricultural industry sometimes uses this compound to help make crop protection agents more stable and effective. Though not used as an active ingredient, it shapes the production process for products that help farmers fight pests and disease.
Environmental and Safety Considerations
Working with dichlorophenyl isocyanate brings some real-world challenges. The compound can irritate eyes, skin, and lungs, making proper precautions a must for anyone handling it. I remember a chemical plant that trained every new worker on protective gear and emergency procedures before they started handling any isocyanates at all. Regulations in the US and EU require strict controls on emissions and disposal, pushing companies to capture any releases and minimize waste. As health and environmental awareness grows, ongoing research seeks safer alternatives and better ways to recycle chemical byproducts.
The Path Forward
Demand for materials that last longer, perform better, and leave less environmental impact keeps growing. Scientists and engineers keep tweaking production processes to squeeze more durability and utility out of less raw material. Some newer coatings and adhesives rely on greener chemical routes, lowering their toxicity and ecological footprint. It’s not easy breaking habits in large-scale manufacturing, but the move toward safer, more responsible use of isocyanates marks a step forward for everyone involved—from the plant worker to the end user.
What are the safety precautions when handling Dichlorophenyl Isocyanate?
Danger Lurks in the Lab
Experience in the chemical industry teaches a simple lesson: respect chemicals, especially the tough ones like dichlorophenyl isocyanate. This compound isn’t friendly. With its choking fumes and harsh effects on skin and lungs, nobody wants to face the aftermath of a careless spill. Lung irritation and severe reactions catch even seasoned workers off guard. I have seen colleagues struggle with coughing fits or rashes because they skipped a glove or mask one busy morning. A safety culture builds from those moments.
Personal Protective Equipment: Not Optional
Full coverage gear feels bulky and sometimes slows down work, yet the stakes are too high to cut corners. A sturdy pair of nitrile gloves, a snug lab coat, chemical goggles, and a face shield form a real barrier between the body and toxic dust or splash. For tasks that could stir up vapors, a tight-fitting respirator fitted with the right filter stops dangerous gases from sneaking into lungs. Lab shoes ought to cover the whole foot – sneaker toes won’t shield against a spill.
Ventilation Takes Priority
Bringing large volumes of isocyanates into a poorly ventilated room asks for trouble. Even small amounts evaporate and hang in the air, ready to irritate throats or eyes. Proper fume hoods or exhaust systems pull these vapors away from noses and skin. I once worked in a lab running on a shoestring without an updated fume hood. Productivity came at a cost: more coughing, more complaints. Once we fixed airflow, breathing felt easier, and focus returned.
Spill Control: Planning Saves Pain
Spills never announce themselves. They just happen. Having a cleanup kit ready turns disaster into inconvenience. Absorbent pads, neutralizers, and airtight waste containers belong within arm’s reach, not buried in a back room. Clear steps posted near workstations guide panicked minds during stressful moments. My habit before every big experiment: check the kit and review the spill plan. Over time, preparation smooths out responses and helps everyone stay safer.
Training Translates to Survival
A crash course on hazards seems dull until muscles remember what to do. I learned the value of yearly refreshers after seeing a rookie freeze during a simulated emergency. Quick actions, such as flushing affected skin and eyes or getting to fresh air, come only through practice. Supervisors need to invest in more than paperwork – real drills and hands-on walkthroughs make the difference if things go south.
Storage and Waste: Out of Sight, Not Out of Mind
Something as volatile as dichlorophenyl isocyanate belongs in a sealed, labeled container, far from sunlight and damp spots. Using dedicated cabinets, away from acids and bases, stops runaway reactions from starting. Any waste, even soiled gloves or wipes, counts as hazardous and requires secure bins built for safe transit off-site. At my last workplace, we switched to locked drum disposal following a near-miss with a leaky trash bag—lesson learned the hard way.
Caring Means Watching Out for Each Other
Working with tough chemicals becomes safer when teams look out for one another. Quick reminders, double checks before opening containers, and honest reporting of accidents build trust. Keeping eyes open and helping each other gear up creates the sort of workplace where everyone walks out healthy at the end of the shift.
What is the chemical formula and structure of Dichlorophenyl Isocyanate?
Chemical Formula and Structure
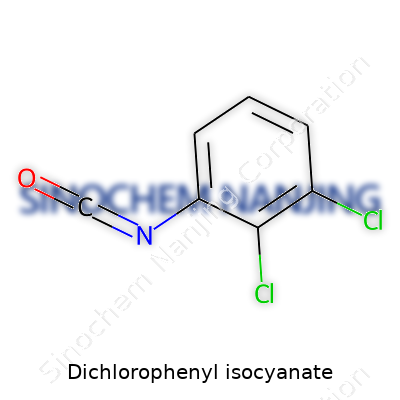
Dichlorophenyl isocyanate draws attention in laboratories because of its unique arrangement of atoms. Its formula, C7H3Cl2NO, tells a story of a benzene ring carrying two chlorine atoms and a reactive isocyanate group. There are different ways to substitute chlorine atoms on the ring, but researchers and manufacturers most often talk about 2,4-dichlorophenyl isocyanate.
The molecule features a benzene backbone, which most chemists encounter early in their studies. The isocyanate group (–N=C=O) connects directly to the ring. Chlorines take up spots at the second and fourth positions. Drawn out on paper, the layout shows the isocyanate group extending from the first carbon, with chlorines branching off from neighbors. This arrangement doesn’t just look neat—it shifts how the molecule behaves in a reaction, particularly in the presence of nucleophiles like alcohols and amines.
Why This Chemical Matters
Dichlorophenyl isocyanate holds a firm place in the world of organic synthesis. It plays a key part in manufacturing specialty polyurethanes and protective coatings. The two chlorines pull electron density away from the ring, making the isocyanate group even more hungry for reaction than in unsubstituted phenyl isocyanate. In my own lab days, I remember how a small tweak to a phenyl ring, like adding a couple of chlorines, could flip a molecule’s reactivity upside down.
This compound poses risks. Isocyanates irritate eyes and respiratory tracts, and the dichloro version often cuts through gloves faster than you'd expect. Safety demands respect. Several global agencies flag isocyanates as hazards, with tight exposure limits in the workplace. Once, during a summer research stint, I saw a bottle with its cap left a bit loose. The vapor didn’t just smell odd—it sent everyone scrambling for fresh air, reminding us how even trace leaks matter.
Environmental and Health Considerations
Dichlorophenyl isocyanate doesn't break down quickly in the environment. Its stability comes from the benzene ring, which resists microbial attack. If released, it lingers in soil and water, raising concerns over time. Workers handling drums or buckets of this substance risk chronic exposure, which can lead to asthma or other long-term problems. I’ve talked with industrial hygienists who always stress monitoring for airborne isocyanates, not just in visible spills.
Disposal creates another headache. This isn’t something you can pour down a drain or toss in a landfill. Specialized incineration keeps reaction byproducts from harming downstream ecosystems. Community groups living near manufacturing zones track these rules closely, since mistakes can have consequences for years down the line.
Path Forward
Safer practices start with informed workers. Companies now train staff to seal containers, use personal protective equipment, and manage spills without delay. Engineering controls, such as fume hoods and scrubbers, often run nonstop in plants working with isocyanates. Governments increasingly mandate real-time air monitoring in plants, pushing the industry toward transparency. Substitution remains tough, since few other compounds bring it all together—chemical reactivity, cost, and performance—like dichlorophenyl isocyanate. Still, researchers chase alternatives, hoping for a less toxic building block that delivers similar results in finished products.
Every bottle or drum of dichlorophenyl isocyanate tells an old chemistry story—one of reactivity, risk, and the long slog toward stewardship. Even for folks outside the lab or factory, the choices made with this compound touch daily life, from paints on a wall to equipment in a hospital. Asking smart questions about its journey and impact leads, one step at a time, toward safer science.
How should Dichlorophenyl Isocyanate be stored and transported?
Storing Chemicals: Lessons From Experience
Dichlorophenyl Isocyanate often turns up on chemical safety lists for good reason. Having spent years walking through midsize warehouse floors—sometimes with a clipboard, sometimes not—I’ve seen what happens when storage misses the mark. Sometimes, folks forget that these substances carry more weight than just a line in a material safety data sheet. One missed label, one cracked container, and suddenly you are dealing with more than just a mess. For anyone used to handling chemicals, the habits built in the storage room matter as much as those in the lab.
Small leaks have sparked evacuations. Improper stacking has damaged drums. These risks travel with every shipment of Dichlorophenyl Isocyanate. Left out of proper containers, this compound reacts to air and moisture, releasing fumes that irritate eyes, skin, and lungs. Storage starts with the basics: tight sealing, clear labeling, and keeping the chemical dry, cool, and out of sunlight. Locking it in a ventilated, temperature-controlled space away from acids, bases, and water cuts down the chances of a bad surprise.
Transportation: Bringing Practical Sense to Protocol
Moving Dichlorophenyl Isocyanate from site to site isn't a simple hand-off. Chemical transport regulations ask for more than just a sturdy box. In my time arranging shipments—sometimes to rural plants, sometimes urban labs—I watched the best drivers double-check seals, paperwork, and even the route out of habit, not just protocol. Unreliable packaging or missed hazard markings led to shipping delays, fines, and stress for everyone involved.
Transport vehicles ought to use containment that stands up to jostling and temperature swings. Clean, dry conditions inside the truck make a difference—as does space that separates incompatible materials. Regulatory bodies, like the US Department of Transportation, wrap rules around this chemical for good reason. Leaks during transit can quickly become emergencies. Drivers who know their load and handle “what if” situations with cool heads help avoid disaster, but employers should back them up with training and solid incident plans.
Solutions Built on Care, Not Just Compliance
Layering safety goes beyond following a checklist. Regular, hands-on inspections spot early warning signs, like a container leaking at the seam. Clear and visible signage, reliable ventilation in storage rooms, and prompt clean-up tools aren’t just checkboxes—they’re how people go home healthy each day. Coordinating with emergency services and keeping antidotes or neutralizers nearby tells everyone that health takes priority.
Training changes how teams approach storage and shipping. Running drills—actual, sweaty practice, not just reading manuals—turns safe handling into muscle memory. Digital inventory can help track where and when chemicals move, sparing headaches during audits or actual emergencies. If there’s one thing decades around hazardous materials teaches, it’s the value of an involved, informed team over lone experts or passive compliance. Experience carries more weight than the thickest binder of regulations.
Storing and moving Dichlorophenyl Isocyanate safely boils down to a simple promise: care for workers, communities, and the integrity of every shipment. Practical safeguards—built on lessons learned, not just lessons written—form the strongest defense.
What are the potential health hazards of exposure to Dichlorophenyl Isocyanate?
Understanding the Risks
People who manage chemicals at work or study them in labs usually take material safety data sheets seriously, but some names—long, tough ones like Dichlorophenyl Isocyanate—can get overlooked in the jumble of acronyms and other similar-sounding substances. This one isn’t something you’d want to get careless with. Dichlorophenyl Isocyanate, used to make certain coatings, foams, and adhesives, packs a health punch most folks might not realize, and ignoring safety rules brings real dangers into the picture.
What Happens During Exposure?
I once spent a humid summer month in a small manufacturing plant, where a leaking container sent out a sharp, chemical bitterness. Before anyone pinpointed the problem, eyes were streaming, a few workers coughed relentlessly, and there was a debate over whether to shut down for the rest of the day. That acute irritation—burning eyes, pounding headaches, scratchy throats—that's classic isocyanate exposure. The isocyanate group in Dichlorophenyl Isocyanate can irritate the skin, eyes, and especially the lungs, even if the dose seems low. Short-term symptoms might seem like a bad bout of allergies, but long-term, it can get a whole lot worse.
Chronic Health Problems Aren’t Just for “Other People”
Breathing in this chemical over months or years often sets up workers for asthma-like reactions that stick. It hits some sooner than others—a bit of genetic hard luck, maybe, or just a heavier hand with the glue—but once the body starts fighting back, those wheezes and short breaths show up with every little exposure. Sensitization can happen faster than most expect. The immune system, once dialed in to this chemical, launches a real battle every time, and standard asthma inhalers won’t fix chemical sensitivity.
There’s also a direct risk to the skin. Red, itchy rashes start as a nuisance but get worse with repeated contact. Cracked, painful skin can lead to infections. Eye exposure stings—anyone who’s had splash-back knows how badly these chemicals burn. Handling chemicals without full protection is tempting during busy shifts, but nobody should gamble with their own lungs and skin just to save a few minutes.
Long-Term Consequences Demand Real Solutions
Chronic lung issues tie in with higher healthcare bills, more sick days, nervous coworkers covering absent shifts, and families shouldering hidden burdens. I’ve seen folks lose their best jobs because of new sensitivities triggered by exposures they thought were “within limits.” These stories surface long after anybody remembers the safety talk given before lunch all those months ago.
Change starts by making personal protective equipment non-negotiable—gloves, goggles, snug-fitting masks, and working hoods need to stay on, no excuses. Managers and lab supervisors should keep fresh eye wash bottles stocked and train both new hires and long-timers through hands-on drills, not just mandatory slide shows. Regular air monitoring and reporting small leaks before they turn into emergencies matter more than hitting quotas. And anyone who smells something strange or feels the telltale burn should never shrug it off and just get back to work. Looking out for each other keeps everyone safer, every shift, every week.
Chemical safety rules sometimes feel like another layer of paperwork or a hassle to daily routines, but the price of ignoring them can pile up fast. Nobody signs up for long-term lung problems or painful skin issues just by showing up for a day’s pay. Being vigilant today prevents bad news down the line—not just for the worker, but for the family counting on them at home.
| Names | |
| Preferred IUPAC name | 1,3-dichloro-4-isocyanatobenzene |
| Other names |
NDI Naphthalene diisocyanate 1,5-Naphthalene diisocyanate |
| Pronunciation | /daɪˌklɔːrəˈfiːnɪl aɪsoʊˈsaɪəneɪt/ |
| Identifiers | |
| CAS Number | 28479-22-3 |
| Beilstein Reference | 613873 |
| ChEBI | CHEBI:134444 |
| ChEMBL | CHEMBL61382 |
| ChemSpider | 14807910 |
| DrugBank | DB08704 |
| ECHA InfoCard | 100.011.756 |
| EC Number | 202-779-5 |
| Gmelin Reference | 82827 |
| KEGG | C18602 |
| MeSH | D002604 |
| PubChem CID | 84951 |
| RTECS number | NL8925000 |
| UNII | 6I5VI1A411 |
| UN number | UN2206 |
| CompTox Dashboard (EPA) | DTXSID8045062 |
| Properties | |
| Chemical formula | C7H3Cl2NO |
| Molar mass | 202.03 g/mol |
| Appearance | White to pale yellow crystal or powder |
| Odor | Aromatic odor |
| Density | 1.44 g/cm3 |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 0.02 mmHg (25°C) |
| Acidity (pKa) | 23.5 |
| Basicity (pKb) | 11.93 |
| Magnetic susceptibility (χ) | -69.0e-6 cm³/mol |
| Refractive index (nD) | 1.571 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.95 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 356.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -95.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4478.8 kJ/mol |
| Pharmacology | |
| ATC code | D08AE17 |
| Hazards | |
| Main hazards | May cause allergy or asthma symptoms or breathing difficulties if inhaled. Causes serious eye irritation. Causes skin irritation. May cause an allergic skin reaction. Harmful if inhaled. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Causes serious eye irritation. May cause an allergic skin reaction. May cause allergy or asthma symptoms or breathing difficulties if inhaled. May cause respiratory irritation. Suspected of causing cancer. |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P304+P340, P305+P351+P338, P310, P321, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Flash point | 113°C |
| Autoignition temperature | 210°C |
| Lethal dose or concentration | LD₅₀ oral rat: 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2,000 mg/kg (rat, oral) |
| NIOSH | WM2450000 |
| PEL (Permissible) | 0.02 mg/m3 |
| REL (Recommended) | 0.005 ppm |
| IDLH (Immediate danger) | IDLH: 1 mg/m3 |
| Related compounds | |
| Related compounds |
Triphosgene Isocyanate Diisocyanate Phenyl isocyanate Toluene diisocyanate |

