Dichloroisocyanuric Acid: Science, Safety, and the Future
Historical Development and Importance
Dichloroisocyanuric Acid has found a long-standing place in the world of disinfectants and water treatment. Its roots go back to the early 20th century, a time when chemists looked for alternatives to the bulky, hard-to-handle forms of chlorine that were common in sanitation. My work with municipal water systems in small towns made me appreciate the challenges faced before stable chlorine products became available. It’s hard to undervalue the shift this compound represented; chemicals like Dichloroisocyanuric Acid allowed towns without strict infrastructure to keep water safe for drinking and swimming. These shifts in availability and reliability meant fewer people got sick from something as basic as drinking water, and trust in public systems grew stronger.
What Sets Dichloroisocyanuric Acid Apart
Most people buying pool shock or a jar of cleaner probably don’t think deeply about the powder or tablet in their hands. Dichloroisocyanuric Acid stands out for its stability and ease of use, making it a favorite for both home users and industrial facilities. This chemical shows up as a white crystalline powder, and it dissolves relatively fast, breaking down to release chlorine just when it’s needed—no more, no less. Handling the raw material gave me an appreciation for its compact efficiency. It carries high “available chlorine” content by weight, something impossible to accomplish with simple bleach or liquid forms. This matters more than ever with modern logistics, storage limits, and price pressures.
Physical & Chemical Properties Matter
Dichloroisocyanuric Acid offers a pleasant surprise to anyone obsessed with industrial hygiene. Light and stable at room temperature, this compound doesn’t create headaches during transport or storage. Unlike elemental chlorine, which wants to escape in dangerous clouds or react with whatever gets close, these powdery granules stay put until water unlocks their punch. I remember field calls after floods: emergency teams looked for solutions that wouldn’t degrade in a hot, damp warehouse. Dichloroisocyanuric Acid didn’t disappoint. Its slow vapor loss and resilience boosted confidence during crisis management. In terms of solubility and reactivity, being able to control dose levels saves time and money in places running on skinny budgets and old pipes.
Preparation and Chemical Reactions—Lab Bench to Factory Floor
Manufacturers typically prepare Dichloroisocyanuric Acid by chlorinating cyanuric acid under carefully controlled conditions. This might sound like textbook chemistry, but I’ve seen firsthand the trade-offs that come with scaling up: temperature swings, moisture hiccups, and the risk of making too many chlorinated byproducts. Each time the process gets tweaked, small gains in yield or purity make operations more efficient. Downstream, this translates into a more predictable product for water plant operators and sanitation workers. In handling and mixing, Dichloroisocyanuric Acid reacts sharply with acids and some organic materials—sometimes producing dangerous chlorine gas. Consistent labeling and robust ventilation protocols keep these risks in check, but human error remains a challenge.
Synonyms and Label Confusion
This compound shows up on shelves and shipping manifests under many names: sodium dichloroisocyanurate, NaDCC, and even shorthand versions that swap “acid” for “salt” depending on the form. It’s easy to get turned around if you aren’t paying close attention. I’ve had conversations with new colleagues who only knew the “pool” version and didn’t connect it with the same cleaner in hospital use. To avoid mishaps, regulators have tightened rules on labeling. Modern packaging explains usage, cross-references synonyms, and flags hazards in straightforward language. But experience shows that confusion still crops up, especially in cross-border shipments.
Safety and Operational Standards—Lessons from Experience
From the plant floor to the janitor’s closet, everyone benefits from clear guidelines on handling Dichloroisocyanuric Acid. Splash a concentrated solution on your hands and you’ll regret it—skin dries and gets irritated quickly. I’ve worked in facilities where a simple misstep led to coughing fits or burning eyes. Many workplaces now require gloves and goggles, and the trend leans toward premeasured tablets or sachets instead of loose powder. Emergency protocols exist for a reason, and it’s smart to keep calcium gluconate and eyewash stations handy wherever concentrated chemicals are stored. Storage practices demand attention too; keeping this chemical dry and away from incompatible materials like acids reduces the odds of unwanted reactions or releases.
Application Areas
From disinfecting swimming pools and drinking water to sanitizing medical instruments, Dichloroisocyanuric Acid has a broad reach. I’ve seen the switch from regular bleach to this chlorine donor bring big benefits in public restrooms, cutting down on storage space and lowering spill risk. Hospitals, schools, and disaster zones all rely on the ability to supply fast, effective disinfection where infrastructure may be limited or intermittent. On farm visits, livestock handlers explained how they use this chemical in cleaning routines, valuing the long shelf-life and predictable action. In a world where pandemics and clean water shortages keep appearing in headlines, having stable, portable, and powerful disinfectants remains non-negotiable.
Current Research and Development—What’s Next?
Lab teams and chemical engineers still look for ways to refine Dichloroisocyanuric Acid production, cut costs, and tackle the challenges of environmental residue. Efforts to reduce chlorinated byproduct formation deserve note; these byproducts present major concerns in both wastewater discharge and treated drinking supplies. Fresh research draws links between extended, low-level exposures and the rise of disinfectant-resistant microbes, pushing industry and regulators to rethink standards. In my conversations with university chemists, I hear optimism that clever catalysis and greener solvents could reduce waste and energy demand. Field trials with slow-release formulations and combined-action cleaners also point to new markets.
Toxicity Research—What We Really Know
No one should treat high-dose exposure to any chlorine compound lightly. Toxicology studies in humans and animals consistently warn about the effects of swallowing, inhaling, or repeated skin contact with Dichloroisocyanuric Acid in concentrated form. Human experience matches the science: headaches, eye irritation, nausea, or even more severe respiratory problems show up in reported cases where ventilation or training slipped. Water treatment professionals understand the need to balance raw power with careful measurement. On the other side, public health gains from controlled use far outweigh the risks when proper safety steps are followed. Independent review committees keep tabs on toxicities, ensuring guidelines stay current and shaped by real-world incidents, not just laboratory data.
Looking Ahead—Future Prospects
Dichloroisocyanuric Acid has not reached the end of its utility. Interest keeps growing as water access, hygiene, and public health remain high on national agendas. Upgrades in manufacturing, a sharper focus on environmental impacts, and smarter packaging could keep this compound at the frontline of sanitation for decades. Ventures combine Dichloroisocyanuric Acid with other agents for broad-spectrum cleaning and lower environmental residue. In the rush to find new answers to old problems, the challenge lies in making chemistry work for people and the places they live—not just for profit or convenience. My time in public health tells me that the ability to deliver clean water with minimal risk counts as one of modern society’s greatest achievements, and this humble compound plays a crucial part in that story.
What is Dichloroisocyanuric Acid used for?
What’s Behind the Label?
Every pool owner recognizes that sharp, chlorine-clean smell. Most people point the finger at bleach, but that’s not always the real culprit. Dichloroisocyanuric acid, or “dichlor,” works as a core ingredient in many household cleaners, pool shock treatments, and sanitizing tablets. This white, crystalline powder packs strong punch thanks to its ability to release chlorine efficiently. Chlorine isn’t just one flavor—compounds like dichlor give us cleaner water across towns, schools, and clinics.
More Than Just “Pool Shock”
People often link dichlor only to backyard swimming pools. Those familiar blue pucks and granules get tossed in the water because they dissolve steadily and fight off bacteria. Hospitals and first responders also trust dichlor—in diluted solutions, it kills tricky bacteria and viruses on surfaces and equipment. Outside, municipal water treatment plants often tap dichlor to disinfect drinking water in emergencies. It doesn’t stay in the spotlight, but its work happens in the background for millions of people every day.
Chasing Cleanliness, Facing Risks
Too little sanitation spells danger, but overusing strong chemicals can spark its own problems. Nobody wants a dose of chlorine gas indoors, nor skin irritation from careless handling. One summer, I learned first-hand that tossing chlorine tablets into a humid shed creates a caustic, cough-inducing air. Instructions matter. Proper storage keeps families, pets, and pollinators safe around pools and cleaning closets. The American Chemistry Council points out that accidents mostly stem from lack of label reading or mixing with other cleaners.
Global Health Impact
Travel to regions fighting cholera outbreaks, and you find powdered dichlor pouches getting mixed into buckets and wells. Groups like WHO and UNICEF distribute it in emergencies because it keeps water from becoming a silent killer. Some brands even market portable tablets for camping or backpacking—a pocket-sized way to make river water drinkable. Not every country can afford complex filtration, so the right disinfectant at the right moment saves thousands of lives each year.
Letting Chemicals Work for Us
I’ve seen families argue that “natural” cleaning beats all, but after a norovirus outbreak at my nephew’s daycare, everyone became instant converts to sanitizing sprays and thorough cleaning. The Environmental Protection Agency recognizes dichlor for knocking out a wide array of pathogens, and regular audits track how much residue remains in drinking water. Responsible companies adapt dosing instructions based on new research, and health agencies confirm safe levels through rigorous monitoring.
Better Handling, Smarter Rules
Label clarity makes the biggest difference—bold hazard warnings, storage tips, and mixing instructions cut down on misunderstandings. Large bulk containers need extra tamper-proofing, especially in facilities with children or pets in the area. The CDC provides online guidance about chemical safety and cleaning after floods or sewage leaks, turning what once felt mysterious into doable steps for anyone.
Looking Ahead
Science keeps hunting for more sustainable disinfectants. For now, dichloroisocyanuric acid remains a key player in keeping public spaces, drinking water, and home environments sanitary. Respecting this tool, instead of fearing it or getting careless, lets everyone benefit from its life-saving potential. Good hygiene often looks invisible—but the evidence shows up in healthier communities and fewer outbreaks.
Is Dichloroisocyanuric Acid safe for human health?
What Is Dichloroisocyanuric Acid?
Dichloroisocyanuric acid pops up a lot in talk about clean water and sanitation. It shows up in pool chemicals, disinfectants, and even some emergency water purification tablets. Its job: wiping out bacteria and viruses. I’ve seen it used in campsites, swimming pools, and even schools, all with the aim of keeping water safe to touch and drink. So it makes sense people wonder how much risk it brings.
Everyday Exposure Explained
Most folks encounter this chemical through pool water, surface cleaning, or during hiking trips where quick water disinfection is a big deal. In my own experience, dropping one of those tablets in a cloudy bottle while camping made me appreciate fast disinfection. The instructions always suggest careful dosing – that small difference between clean and chemical soup. It’s the right dose that kills germs but doesn’t make you sick.
Scientists have put dichloroisocyanuric acid through a lot of tests. In pools, studies show that the low concentrations used for routine water treatment haven’t been linked to major health problems for adults or kids. The World Health Organization reviewed the research and gave a thumbs-up for these low-level uses, stressing the importance of following directions closely.
Why Dose Really Matters
Problems show up when someone swallows or mixes up highly concentrated products. Acute poisoning is rare, yet it happens because of poor labeling, lack of training, or pure curiosity – especially in unsupervised kids. The main symptoms look like nausea, vomiting, or irritation in the mouth and throat. Most cases clear up with medical care, but really severe incidents ask for hospital support.
Breathing in the powder or the fumes after mixing it—like if cleaning in a small, unventilated bathroom—can trigger asthma symptoms, coughing, or eye irritation. During my years as a lifeguard, we learned to store and dose pool chemicals with good ventilation and gloves. Safety rules make the difference, not just for staff but for swimmers who never think about what keeps pool water blue, balanced, and germ-free.
Long-Term Questions and Trustworthy Guidelines
So far, no evidence shows that daily exposure through treated water creates long-term health issues. Long-term studies are limited, but nothing has waved major red flags. Still, the unpredictability of improper use – like dumping pool shock into a fish pond or backyard well – can end up poisoning animals, tipping environmental balance, or burning skin. That’s why following the guidelines from health agencies becomes critical.
Taking Care with Chemical Products
Training, clear instructions, and responsible storage reduce the odds of accidents. Manufacturers print guidelines in big letters, but it doesn’t hurt to ask questions or look up more information if something seems unclear. The safest route goes beyond the fine print: storing chemicals out of reach, using the right amount, and never mixing with other cleansers. I remember a neighbor who unknowingly mixed two cleaning tablets, setting off an overwhelming cloud – luckily, quick action and open windows saved the day.
Seeking Solutions
Manufacturers could make sachets for single-use applications to cut down on measuring errors. School programs could cover safe water purifier use, just like fire safety drills. Local pools and gyms might post simple info sheets for families, not just employees. Healthcare workers need up-to-date instructions for what to do in accidental exposures and a way to report incidents. Everyone can take a little more time reading labels – it’s such a small step with a big payoff.
How should Dichloroisocyanuric Acid be stored?
A Chemical That Deserves Caution
Dichloroisocyanuric acid often pops up in places where cleanliness is not just a preference but a requirement. Pools, water treatment setups, even some disinfectant products include it. People might think of it as just another cleaning agent, but this compound can be hazardous if ignored or put away carelessly. Too many stories exist about workers recalling a sharp smell in the back room or noticing gummed-up packaging on the shelf. This stuff releases a strong chlorine odor, and left unchecked, it can become both a health and fire risk.
Why Dry and Sealed Matters More Than Most Realize
Anyone who’s returned home with soggy bags of groceries knows there’s a big difference between dry storage and damp surroundings. For Dichloroisocyanuric acid, even brief brushes with moisture can start chemical changes. The moment humidity or splashes touch it, chlorine gas may form. Chlorine is no friend to lungs or eyes. Moisture doesn’t just ruin the product’s usefulness by clumping it up; it causes danger for anyone nearby. Using an airtight container isn’t a detail—it keeps the chemical from reacting and keeps emergencies at bay.
Keep It Cool and Out of the Sun
Many think warehouse lights and closed windows mean products are safe from the sun’s effects. Heat complicates storage. Some warehouses get warm in summer, especially if boxes sit near windows or vents. Heat speeds up chemical reactions. This can increase pressure in containers or lead to leaking fumes. Storing Dichloroisocyanuric acid in a shaded, cool section of the building keeps temperature shifts in check. A little bit of planning saves plenty of headaches down the road.
Separate from Combustibles and Acids
Look at any material data sheet, and there’s a clear warning to keep Dichloroisocyanuric acid far from anything flammable or acidic. I’ve seen storerooms where pool cleaner jugs sit near mops doused in ammonia or shelves lined with paint thinner. This is a recipe for disaster. Add just a dash of acid, and the chemical throws off toxic gases almost instantly. Even organic material, like paper or sawdust, can ignite from accidental mixing, leading to warehouse fires. Responsible storage keeps incompatible chemicals on different shelves, away from materials that catch fire easily.
Labels and Training—No Substitutes Here
It’s one thing to keep a chemical on the right shelf; it's another for every worker to know what’s inside those containers. Clear, durable labels help, especially after a shift change or in busy supply rooms. Labels won’t stop a spill, but they reduce confusion if a problem pops up. Training matters even more. People remember real-life stories from co-workers or safety seminars longer than they do bullet points from a manual. Quick, regular reminders on safe handling do more to protect people—and property—than any rulebook stuck in a drawer.
Common Sense and Respect for the Chemical
Dichloroisocyanuric acid can serve well in its intended roles. Approaching it with care and preparation, not guesswork, makes all the difference. I’ve seen workplaces thrive where safety routines are treated as everyday tasks rather than last-minute checklists. The price of cutting corners is almost always too steep—lost product, damaged health, unnecessary risks. With proper storage, regular training, and honest communication, dangers shrink and the chemical sticks to its job: making water safer, not the storeroom more dangerous.
What are the proper handling and usage precautions for Dichloroisocyanuric Acid?
Understanding What’s at Stake
Many people know dichloroisocyanuric acid as the chemical packed into pool disinfectant tablets or used as a sanitizer in food processing. On a casual glance, it seems pretty straightforward—white, granular, strong-smelling chlorine. But anyone who’s worked with it, even briefly, understands its punch. Problems show up when the handling gets sloppy, whether in a pool shed or industrial warehouse.
The Real Hazards
This isn’t table salt. Dichloroisocyanuric acid carries serious health risks. Breathing its dust irritates the throat and lungs, sometimes triggering asthma-like symptoms in those with weak breathing. Skin contact can leave burns. Even more worrying, moisture sparks a quick, sharp chemical reaction—one reason it makes a great sanitizer, but also a hazard around spilled water or humidity. Mixed with other chemicals, especially acids or ammonia-based cleaners, things can go downhill fast: toxic gas, heat, sometimes even a small fire. Reports from the field tell the story—poor storage can ruin entire shipments, and ignoring label instructions has landed more than a few workers in the emergency room.
Common-Sense Protection Matters
Personal protection saves lives. Gloves—real chemical-resistant ones—keep skin out of harm's way. Safety goggles stop accidental splashes or dust from hitting your eyes. Working with dichloroisocyanuric acid in small, poorly ventilated spaces brings risk up fast. A basic dust mask or, better yet, a proper respirator, can save hours of coughing and long-term problems. It’s not about paranoia. It’s about respecting the chemical.
Storing and Using It the Right Way
A dry, cool, well-marked storage area shuts down a lot of risk right away. Keep containers off the floor, away from sunlight, and far from anything flammable or reactive. I’ve seen too many times where people think a broom closet is fine—until something leaks or spills and suddenly there’s choking smoke. Only store the needed amount. Seal all containers tightly after each use. Any dedicated training in chemical handling drills these rules—even in places that treat safety as an afterthought.
Read Every Label—Every Time
Labels are there for a reason. Each manufacturer might recommend slightly different concentrations or mixing instructions. One wrong move, like tossing the chemical directly into water without slowly dissolving and stirring, can give off a poisonous cloud. Never mix it with other cleaning products unless the instructions spell it out. Real accidents tend to start with rushing or skipping the basics. Always treat each batch like it’s the most dangerous thing in the room.
Keeping Emergency Plans Close
Accidents happen. Keep clean water and first-aid supplies nearby. Show coworkers or family where safety showers and eyewash stations are, just in case. Having local emergency numbers handy beats searching in a panic after something goes wrong. Training everyone who works near it cuts confusion and prevents small slip-ups from becoming disasters.
A Practical Mindset Goes a Long Way
Dichloroisocyanuric acid works exactly because it reacts so quickly. Harnessing that means staying alert and practicing safe habits every single time. For people in the field, safe habits become muscle memory—and that’s the difference between a routine job and a preventable injury.
Is Dichloroisocyanuric Acid effective as a disinfectant or sanitizer?
Real Use Shows Strong Results
In cleaning jobs, strong and reliable disinfectants go a long way. Dichloroisocyanuric acid stands out in pools, hospitals, and food processing because it kills bacteria, viruses, and even fungi. I have worked in public health, and it’s common to see this compound used for sanitizing water and surfaces. Pools worldwide rely on it because it breaks down into hypochlorous acid, which controls both bacteria and algae. This step has turned many swimming spots from unsafe to welcoming.
Science Backs Its Power
Lab research, reported by the World Health Organization, shows dichloroisocyanuric acid wipes out tough pathogens like E. coli, Salmonella, Staphylococcus, and even Norovirus. Schools, hospitals, and homes trust it not only for surfaces but also for water treatment tablets. In my area, many people keep emergency water purification tablets handy. In disaster situations—floods or storm fallout—clean water becomes rare, and these tablets offer peace of mind.
Easy to Store, Safe to Mix
Many cleaning agents bring storage headaches. Dichloroisocyanuric acid stays solid and stable for months, sometimes years, without losing power. My family keeps a tub of these chlorine-based tablets in our emergency kit. It doesn’t react wildly with other common cleaning agents and dissolves with no trace of grit, which sets it apart from cheaper powders and older liquid bleaches that leave layers behind. Schools and municipal buildings often choose it for this reason.
It’s Not All Smooth Sailing
As strong as this product cleans, real-world problems still pop up. Some folks ignore manufacturer directions, tossing in more tablets, hoping to get super-clean water or surfaces. That’s risky—too high a dose can irritate the skin, eyes, and lungs. I’ve seen facilities skip proper ventilation after treating with these solutions. Chlorine gas smells strong and can bother people, especially those with asthma or allergies.
There’s also the purity problem. Not every supplier tests for contaminants or watches for product breakdown in extreme heat or moisture. Counterfeit tablets sometimes enter markets in low-income regions, making results unpredictable and giving communities a false sense of safety.
A Safer, Smarter Path Forward
The answer isn’t to turn away from dichloroisocyanuric acid. Instead, tougher quality checks and clear dose instructions should come standard. Governments need stronger oversight and public guides—short, clear posters in community centers and shelters would keep more people safe. Manufacturers can include color-change strips or simple test kits so users avoid over-chlorination.
Training staff in health, hospitality, and education means showing how much is enough and reminding them to check expiration dates. Investing in good storage spaces—dry and not sun-lit—will keep stockpiles ready for use in busy kitchens, hospitals, or after a flood.
What To Remember
My own work and home experience tell me dichloroisocyanuric acid gets the job done when trusted and used with care. It’s a powerful tool for health, public safety, and daily cleaning, but smart use matters as much as strong chemistry. Better handling, quality control, and honest instructions will make sure it rewards trust instead of risking health.
| Names | |
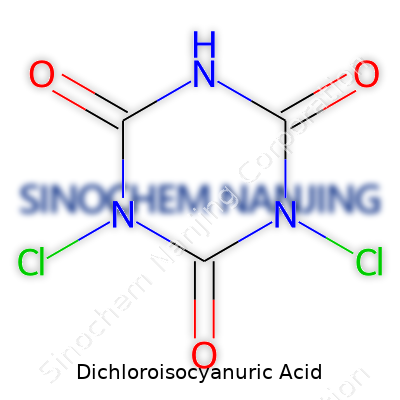
| Preferred IUPAC name | 1,3-Dichloro-1,3,5-triazinane-2,4,6-trione |
| Other names |
Troclosene Sodium dichloroisocyanurate Dichlor DCCA Sodium dichloro-s-triazinetrione |
| Pronunciation | /daɪˌklɔːroʊ.aɪsoʊ.saɪˈæn.jʊr.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 2893-78-9 |
| Beilstein Reference | 1767581 |
| ChEBI | CHEBI:38957 |
| ChEMBL | CHEMBL3980665 |
| ChemSpider | 70773 |
| DrugBank | DB11309 |
| ECHA InfoCard | 100.028.756 |
| EC Number | 220-767-7 |
| Gmelin Reference | 8079 |
| KEGG | C07052 |
| MeSH | Dichloroisocyanuric Acid MeSH: D004051 |
| PubChem CID | 60979 |
| RTECS number | JO1925000 |
| UNII | 2PND21DU91 |
| UN number | UN2465 |
| Properties | |
| Chemical formula | C3Cl2N3O3 |
| Molar mass | 197.94 g/mol |
| Appearance | White crystalline powder |
| Odor | Slight chlorine odor |
| Density | 1.1 g/cm³ |
| Solubility in water | 21 g/100 mL (25 °C) |
| log P | -0.8 |
| Vapor pressure | 0.0075 mmHg (25°C) |
| Acidity (pKa) | 1.3 |
| Basicity (pKb) | 12.7 |
| Magnetic susceptibility (χ) | -74.0e-6 cm³/mol |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 267 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -598.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -802.8 kJ/mol |
| Pharmacology | |
| ATC code | JAVEL |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes serious eye irritation, may cause respiratory irritation, harmful to aquatic life |
| GHS labelling | GHS05, GHS07, GHS09 |
| Pictograms | GHS05,GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H318, H335 |
| Precautionary statements | P261, P264, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P330, P337+P313, P362+P364, P391, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Autoignition temperature | The autoignition temperature of Dichloroisocyanuric Acid is "250°C". |
| Lethal dose or concentration | LD50 oral rat: 1,420 mg/kg |
| LD50 (median dose) | 770 mg/kg (rat, oral) |
| NIOSH | SW1730000 |
| PEL (Permissible) | PEL: 1 mg/m³ |
| REL (Recommended) | 0.2 mg/m3 |
| IDLH (Immediate danger) | IDLH: 20 mg/m³ |
| Related compounds | |
| Related compounds |
Chloroisocyanuric acids Trichloroisocyanuric acid Sodium dichloroisocyanurate Sodium hypochlorite Isocyanuric acid |

