Dibutyltin Dilaurate: From Industry Cornerstone to Future Challenge
A History Written in Chemistry Labs and Factories
Dibutyltin dilaurate started turning heads in the chemical industry decades ago. Workers in paint shops, vinyl plants, and plastics manufacturing all recognized its signature name, even if only from a warning sticker or label on a drum. Before the widespread shift toward greener chemistry, companies relied on this compound to move reactions along—a catalyst that coaxed polymers and urethanes into the forms that hold our wires, strengthen our cars, and build the cushions in our chairs. Chemists filed patents, swapped synthesis tips, and debated tweaks to the manufacturing process. The product’s value came from its knack for speeding up curing times and improving flow, and people chasing productivity paid attention. Newsletters and journals from the 1970s and onwards trace a clear path of adoption: each sector cited dibutyltin dilaurate for making production lines run smoother.
Looking Closely at the Compound
This substance wears a mouthful of a name, but most folks in facilities just call it DBTDL. What matters for those outside a chemistry set? It comes as a pale yellow, oily liquid, with a slight odor and a tendency to stick to anything it touches. The chemical formula, for those keeping score, sums up why it binds well and reacts easily: its tin at the center serves as a handshake point between organic chains and other industrial molecules. It dissolves in organic solvents, resists water, and doesn’t evaporate quickly under factory conditions—traits that protect a process from unexpected shutdowns. I’ve seen it poured straight from the drum: the liquid gleams, thick and ready, making a factory smell just a bit sharper.
Technical Details and the Shelf
The packaging says a lot: hazard symbols, handling notes, tin content, and a long list of product grades depending on purity and application. Labeling regulations from the EU and US mandate special language on toxicity and environmental threats, which led to more than one safety audit in places I’ve worked. It’s not a material for the home hobbyist, and training sessions pitch its dangers head-on. In factories, workers wear gloves, goggles, and sometimes respirators, especially when heating it in bulk reactors. Technical documentation gets thick: melting points, flash points, and shelf life are spelled out in detail.
How Production Shapes Use
Plants that make dibutyltin dilaurate start with lauric acid and dibutyltin oxide or dibutyltin dichloride. Most production lines use a direct reaction method, controlled by careful quantities and temperature schedules. If something veers off—wrong ratio, unexpected surge—byproducts rear up, and waste piles on. The process, while not simple, rewards those who respect careful dosing and timing. Clean reactors and smart feeds keep the batch on track and yields high. In my visits to chemical plants, veteran operators often tell new hires this: “You can’t rush a good batch.”
Tinkering with Chemistry
Chemists have spent years fiddling with structural tweaks to dibutyltin dilaurate and exploring its reactivity. Swap out a chain here, add another organic moiety there, and the properties shift—better stability, more solubility, or less environmental harm. The catalysis reactions matter for polyurethane foaming and silicone curing, plus PVC stabilization. In pilot labs, researchers look for substitutes or blend the compound with co-catalysts to lower health risks and keep performance up. Patents stack up for these tweaks, most aiming to harness the core benefits while sidestepping sticky regulatory issues.
It Responds to Many Names
Ask a chemist, and you hear dibutyltin laurate. In other documents, it runs as DBTDL or the CAS number 77-58-7. In specialty shops, someone may shout a trade name from a catalog. Regardless, seasoned shop foremen and technical managers quickly recognize it as “that tin catalyst in the yellow drum.”
Staying Safe is Non-Negotiable
Factory workers and lab techs get regular reminders that DBTDL brings health risks. Skin irritation, respiratory danger, and worries about chronic exposure show up in training slide decks. The EU classified dibutyltin compounds as reproductive toxins and flagged them for environmental threats—especially in aquatic settings. I’ve been in safety meetings where the MSDS sheets get handed out like candy, and every seasoned worker has a story about a near miss—spills, fumes, or a malfunctioning drum pump. Local air and water standards keep drifting closer to zero tolerance, and companies scramble to keep up. A single incident can shut down an entire operation pending a safety review.
Why Do Factories Cling to It?
Despite regulatory headaches and toxicity concerns, dibutyltin dilaurate still anchors production in polyurethane, silicone sealants, adhesives, and PVC. Its ability to speed up reactions, drive crosslinking, and add reliability makes it tough to drop. Some industries run on tight margins and can’t risk line stoppages from incomplete cures or inconsistent gels. Replacement candidates pop up, but few deliver the same mix of speed, quality, and price per ton. Every research team I’ve met in this arena faces the same riddle: how to break free of an old standby without breaking production budgets or sacrificing product life.
Looking Ahead: New Problems and Possible Solutions
Tougher restrictions push research both inside companies and at universities. Green chemistry aims to replace tin-based catalysts with organocatalysts or biobased alternatives. Some labs have raised performance with zinc, bismuth, or even enzyme-based solutions, yet transitioning entire supply chains brings pain. Costs matter. Change takes more than swapping out a drum at the end of the line—testing, new equipment, and workforce retraining all pile up. On the policy side, tighter labeling and more transparent reporting force companies to face up to the risks they’ve tolerated for decades. Some smaller firms have shifted away entirely, risking higher costs for peace of mind. Larger companies hedge their bets—dual system lines, test batches, endless data collection. I’ve watched teams debate policy versus practice, weighing headline risks against day-to-day reality.
Ongoing Research and Real-World Results
Anyone who’s dug into scientific journals sees steady publication about DBTDL toxicity and environmental persistence. Chronic exposure studies on rats, aquatic risk assessments, cellular response investigations—they pile up year after year. Data confirms tin compounds break down slowly, travel in water, and end up in living tissue. The science sounds the alarm, and it reaches the ears of regulators and consumer advocates. Surveys and environmental tests shape policy, but on the factory floor, action often lags. Industry partnerships with universities push experimental alternatives, hoping for that elusive blend of cost, safety, and efficiency. Right now, the best solutions walk a tightrope—awareness, careful controls, and a slow pivot to replacements, all while meeting demand for widely-used, well-understood materials.
What is Dibutyltin Dilaurate used for?
Foundations in Manufacturing
Dibutyltin dilaurate pops up all over the manufacturing world, especially among anyone who’s handled polyurethane foams or silicone sealants. This compound acts as a powerful catalyst for speeding up reactions. Factories producing flexible foams for furniture or insulation depend on it to get the foaming right. Without this compound, the reaction would drag, energy use would go up, and costs would pile on. I used to work in a construction supply shop where tubes of silicone sealant flew off the shelves—most wouldn’t set properly unless formulators included the right catalysts. If you ever tried to patch a bathroom tile with old sealant that wouldn’t cure, you’ve seen what a missing catalyst does.
Paints, Coatings, and More
Companies making paints, varnishes, and surface coatings add dibutyltin dilaurate to the mix for smoother application and sturdy finishes. It helps bind ingredients and brings about chemical reactions on schedule, which means the customer can paint one day and move furniture the next. A painter told me how faster-drying paints transformed his daily routine—less downtime meant he could squeeze in another job before the sun set. Speed isn’t the only benefit: the finish holds up better against chips and peeling since the cross-linking happens in a controlled way.
Everyday Uses and Hidden Risks
Beyond industry, this chemical sometimes ends up in adhesives, gaskets, and flexible rubber pieces because it helps with curing and setting. But these convenience gains carry a cost. Researchers from the European Chemicals Agency flagged dibutyltin dilaurate for its toxicity. Routine factory use can release vapors, which, if inhaled or touched in large amounts, hurts people’s lungs and skin. Some countries have started watching its use more closely, especially in consumer products ranging from electronics to shoes.
Safer Working Practices and Alternatives
Experience gives plenty of reasons not to overlook safety. In a small factory I visited, workers set up new ventilation systems after a co-worker developed respiratory symptoms linked to repeated exposure. Masks and gloves are non-negotiable now on those shop floors. State regulators can support change by holding companies accountable and providing up-to-date guidance for safe handling.
Manufacturers and researchers haven’t stood still. New catalysts, including tin-free options, have started to replace dibutyltin dilaurate in some products. These alternatives aim to match that effectiveness without leaving the same risks behind. Progress takes time, though, and finding the right balance between productivity and health requires careful study and real-world testing.
A Product With Pros and Cons
Dibutyltin dilaurate has shaped how modern industry works, keeping lines moving in everything from foams and adhesives to coatings used in construction and interior design. The health risks mean buyers and workers need to stay informed. People deserve to know what’s in the air they breathe and the products they handle, so companies should keep updating labels and safety sheets. Keeping workplaces safe has to go hand in hand with innovation.
Is Dibutyltin Dilaurate hazardous to health?
Understanding Daily Exposure
Dibutyltin dilaurate gets used in factories for making plastics, paints, and adhesives. Its name doesn’t ring a bell for most people, but plenty of us come into contact with products touched by it. Workers breathe in fumes or get it on their skin during production. Use at home is rare. That’s little comfort for folks who work around it, or for those curious about long-term risks from products touching food or skin.
Why Health Concerns Continue
After years of study, researchers have seen enough to worry about the health impact. Breathing in dusts or vapors can irritate noses, throats, and lungs. Repeated skin contact sometimes leads to rashes. If too much slips past protections, the body pays a higher price—laboratory results link dibutyltin dilaurate to damage in liver, kidneys, and immune systems. Some animal studies even suggest that reproductive harm is a real possibility.
Old workplace stories never disappear. A friend once told me about headaches and sore hands by the end of long shifts at a plant using chemical additives like this one. He didn’t think much of it—until colleagues started comparing notes about breathing trouble and skin flare-ups. Nobody at the plant wanted to raise a fuss and risk their jobs. That sense of worry still echoes through the industrial sector. Factories don’t often advertise chemical hazards unless forced.
Regulations and Gaps
Government agencies have noticed. The European Union added dibutyltin dilaurate to the list of substances whose use must be restricted in consumer goods. Formulations for children’s products and items in close contact with skin face especially strict controls. Safety standards in the United States tend to be laxer, but agencies like OSHA offer guidelines for workplace exposure. It’s not as though the chemical can just be tossed into anything and shipped to market.
Still, laws only go so far. Oversight in practice depends on companies playing by the rules. Some fly under the radar, hoping nobody notices lax ventilation or old, leaky storage drums. Until inspections catch violations, or workers speak up, gaps stay open. Even with harder rules on paper, enforcement can slide.
Consumer Responsibility and Industry Changes
For folks outside the factory gates, risk drops off fast. Yet nobody wants to deal with allergic reactions from touching a rubber handle or breathing in fumes from freshly painted walls. Reading product labels and safety sheets builds awareness, but those documents can be tricky to find and tough to understand.
Alternatives do exist. Some manufacturers have begun switching to less toxic stabilizers in plastics or paint, often because consumers and advocacy groups demanded fewer health risks. Pressing companies for transparency keeps the conversation alive. Large businesses rarely budge toward safer chemicals without public pressure or clear economic reasons.
Practical Steps Moving Forward
Education matters most. Workers handling chemicals like dibutyltin dilaurate ought to have real training and better protective equipment. Reporting symptoms early helps push companies and regulators to take necessary action. For the rest of us, asking tough questions about household products nudges producers toward safer, clearer labeling.
Dibutyltin dilaurate won’t vanish overnight. Its health risks remain strongest for those on the production line. Greater transparency, stronger enforcement, and research on replacements—these build safer workplaces and reduce health worries over time.
How should Dibutyltin Dilaurate be stored?
Everyday Storage Choices Matter
Too often, folks think storing a specialty chemical means putting it on a back shelf and forgetting about it. With dibutyltin dilaurate, that choice can mean much more. This stuff isn’t harmless kitchen vinegar—it’s a powerful organotin compound. I once worked in an adhesives factory that used it as a catalyst for polyurethane products. One careless decision—leaving a cracked lid on a drum—led to a stressful cleanup and a lot of worried faces. The risks push workers and facility managers to treat it far more cautiously than common solvents.
The Real Risks Behind Poor Storage
Dibutyltin dilaurate hits the lungs and skin hard. Inhalation or repeated contact can irritate, sometimes causing lasting problems. That risk isn’t just from handling during production. Improper storage makes leaks and contaminated work surfaces more likely. Just a little moisture or air can degrade purity and increase dangerous byproducts. I remember a warehouse supervisor who ignored recommended storage temperatures. That mistake led to a sticky residue in the containers, ruining a week’s output and costing thousands. This isn’t about regulatory checklists, but about health and staying in business.
Solid Storage Practices Save Trouble
It doesn’t take elaborate systems to reduce risk. A stable, well-ventilated room, far from heat, direct sunlight, and acids, works fine. Low humidity is key. Metal shelves might seem sturdy, but they can cause unnecessary reactions if the container leaks. Chemical compatibility charts help staff avoid misplacing a container among oxidizers. I never store dibutyltin dilaurate near peroxide-rich supplies, no matter how short the shelf cycle.
Original packaging stands out for a reason. It’s designed to resist corrosion and accidental vapor leaks. Glass works well for small amounts, but suppliers often use high-density polyethylene drums for bulk storage. Containers need tight sealing. Don’t trust a loose cap, especially after repeated opening. I once forgot to double-check a lid after a delivery; even a short lapse let enough fumes loose that I felt the chemical’s sharp, headache-inducing smell the next morning.
Training Protects People, Not Just Product
No storage method beats a well-trained workforce. Most accidents I’ve seen on the shop floor were less about equipment and more about rushed, half-remembered routines. Annual refreshers, clear labels, and regular leak checks do more than any label or datasheet on a forgotten shelf. In one crew I trained, we added a color-coded storage system and conducted monthly walkthroughs. Spill rates dropped, and so did health issues. Simple steps like these build a culture of responsibility, not just compliance.
Solutions Need Accountability
Real solutions rely on checklists, but also on everyday accountability. If your facility can't keep dibutyltin dilaurate in a cool, isolated spot away from food, water, and potential combustibles, you’re not just risking fines—you're putting lives at risk. Inventory tracking shouldn’t only happen during audits. Day-to-day walkthroughs, reliable secondary containment (trays, not cardboard boxes), and secure locks on cabinets make a difference.
In my experience, the places that treat chemical storage as a living process avoid nasty surprises. Dibutyltin dilaurate isn’t forgiving. Safe storage isn’t an afterthought; it's daily discipline that keeps products pure and people out of harm’s way.
What are the handling precautions for Dibutyltin Dilaurate?
Why Handling Matters
Dibutyltin dilaurate finds work in everything from making plastics to curing certain paints and sealants. People who use it every day know how strong it can be. It isn’t just a harmless helper in manufacturing—there’s enough evidence about its effects on breathing, skin, and the environment to make anyone pause. Over the years, health experts and regulatory agencies have tagged dibutyltin dilaurate as a substance needing real caution. For anyone working with it, safety steps make all the difference.
The Risks Everyone Should Respect
If you’ve ever opened a drum of this stuff without gloves or a mask, you probably caught that sharp smell and maybe felt your skin burn after a splash. Repeated contact can set off allergies or start chronic skin irritation. Breathing in its vapor stings the nose and throat, and a strong dose really can knock you back. Some studies show links to organ problems after long-term exposure, and animal research has pointed out reproductive risks. Losing respect for these hazards can cost your health, or even your life.
Tough Lessons on Proper Storage and Cleanup
One mistake: leaving containers open or stored in the sun. Heat and light speed up decomposition, which can result in toxic gases or spoil the product entirely. I saw one shop suffer a nasty mess when rainwater leaked into an open barrel—the reaction nearly cleared the floor. Sharing these stories in toolbox talks sticks with new crew members more than dry rules ever could.
Splashy, dusty workplaces make it far too easy to track chemical residue onto break room tables or into raw materials. Simple habits can help: use dedicated gloves, coveralls, and eye protection, and wash hands before eating or rubbing your eyes. Change out of work clothes before leaving the plant. These steps shield not just workers, but also their families back home.
Why Air Quality and Ventilation Demand Respect
I learned plenty about the value of decent ventilation after feeling those tell-tale headaches at the end of a shift. Scrubbing the air, using strong hoods, and running fans doesn’t just protect workers. It prevents fires and keeps the smell from bothering people outside the factory, which is key for staying in good standing with neighbors and local regulators. Tech teams need to check the systems often and fix leaks or blockages right away. Never wait for a safety audit to act.
The Right Training Delivers Results
Workplaces that invest in hands-on training see fewer accidents. It’s worth drilling the basics every few months: how to open a drum without splashing, which respirator fits best, and how to read those hazardous materials labels. Quick response in emergencies saves lives, so have spill kits, eyewash stations, and clean-up supplies where they’re easy to spot. Regular drills help create muscle memory, which means people react calmly under stress. Training also gets people talking about problems before they turn into injuries.
Real Solutions: Substitution and Monitoring
Some companies already look for less toxic alternatives, or change their recipes so that less dibutyltin dilaurate gets used. It’s a smart move for long-term health and keeps regulators satisfied. For workplaces that aren’t ready to switch, regular medical checks and air sampling catch trouble early, sometimes before workers even notice symptoms. The best companies don’t just meet the rules—they set the bar higher.
Experience shows that safe practices keep everyone healthy and keep jobs running. Clear warnings, daily habits, and solid support from managers make a dangerous chemical manageable for those who depend on it.
Can Dibutyltin Dilaurate be shipped internationally?
The Chemical on the Move
Dibutyltin dilaurate pops up across industries, especially in making polyurethane foams, silicones, and certain plastics. Many don't realize how tightly this compound ties countries together through raw material trade. Moving it from one place to another, though, isn’t just about packing barrels and loading trucks. Every shipper faces a maze of regulations, paperwork, and changing rules. I’ve watched folks in the chemical business pour countless hours into compliance just to avoid fines and container seizures.
International Shipping Regulations
The world treats chemicals with suspicion, and for good reason. Dibutyltin dilaurate comes with hazards. Handling or inhaling its vapors causes health problems—respiratory issues, skin irritation, and more. Countries set up strict controls so that this chemical doesn’t wind up somewhere unsafe or in the wrong hands.
Regulations stack up. The United Nations’ Globally Harmonized System assigns this chemical hazard codes. The U.S. includes it in the Toxic Substances Control Act. Europe covers it with the REACH regulation. Asia and South America mark their own import-export conditions. Every shipment demands proof that the sender and recipient follow best practices. If a document goes missing or the packaging leaks, border agents can seize the cargo or refuse entry. I’ve seen shipments stuck in limbo for months just because the label didn’t show both the chemical name and the safety phrases.
Environmental Concerns
Dibutyltin dilaurate impacts more than just the workers handling barrels. It enters water systems during manufacturing and leaks. Research links organotin compounds to environmental damage, especially harming aquatic life. Some governments, worried about pollution, keep tighter watch over shipping and storage. Japan, for example, reduced imports after environmental reviews, and European ports demand certified disposal plans for any spills.
Real-World Shipping Hurdles
Hard lessons come quick for anyone new to international shipping. Customs departments want safety data sheets, precise labeling, and proof that both sender and receiver have licenses. Some airlines avoid chemicals like this altogether, forcing shipments to take slower sea routes. There’s nothing more frustrating than customers waiting on products that sit on a dock because someone didn’t double-check requirements. I've spent late nights on the phone with freight forwarders, just trying to figure out why a shipment vanished into regulatory black holes.
Solutions and Safer Practices
Reducing headaches starts with choosing experienced freight partners. Look for companies that handle hazardous materials every day. Every team moving the shipment should train on chemical safety and documentation, from warehouse staff to truck drivers. Regularly update compliance files by checking government advisories and new trade deals. Many businesses build relationships with regulatory consultants who monitor laws in every market they supply.
Digital records make a huge difference. Electronic manifests, automated reminders for license renewals, and live tracking calm some worries. Well-run companies treat compliance work as an unbroken chain, not just a box to tick. They check packaging for leaks, double up on documentation, and talk to local regulators ahead of time. Following these habits cuts shipping risks, but vigilance never stops. One government’s ban or a sudden change in classification can halt trade overnight.
Looking Forward
Traders and manufacturers relying on dibutyltin dilaurate should keep one eye on the law and another on new green chemistry trends. The push for safer alternatives never sleeps, and stricter laws could appear with little warning. Those willing to adapt will keep products moving and stay out of regulatory trouble.
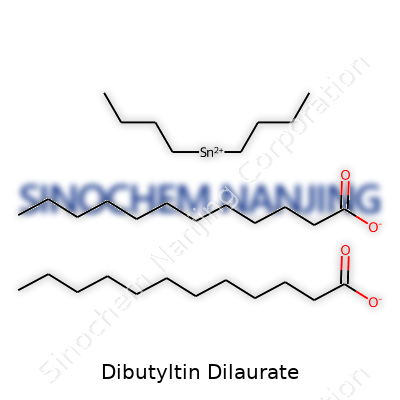
| Names | |
| Preferred IUPAC name | dibutylbis(dodecanoyloxy)stannane |
| Other names |
Butanoic acid, tin(2+) salt Butylstannoic acid, dibutyl ester DBTL Dibutylbis(dodecanoyloxy)stannane Dibutyl tin dilaurate Dibutyltin laurate Stannane, dibutyldilauroxy- Tin, dibutyldilauroxy- |
| Pronunciation | /daɪˈbjuːtɪl.tɪn daɪˈlɔːr.eɪt/ |
| Identifiers | |
| CAS Number | 77-58-7 |
| Beilstein Reference | 1711073 |
| ChEBI | CHEBI:60084 |
| ChEMBL | CHEMBL278723 |
| ChemSpider | 2032182 |
| DrugBank | DB15662 |
| ECHA InfoCard | 100.044.211 |
| EC Number | 201-039-8 |
| Gmelin Reference | 56359 |
| KEGG | C18666 |
| MeSH | D008937 |
| PubChem CID | 30357 |
| RTECS number | WN0280000 |
| UNII | 6CB0M38846 |
| UN number | UN2922 |
| Properties | |
| Chemical formula | C32H64O4Sn |
| Molar mass | 631.56 g/mol |
| Appearance | Clear yellow liquid |
| Odor | slight characteristic odor |
| Density | 1.066 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 4.7 |
| Vapor pressure | 0.00007 mmHg (25°C) |
| Basicity (pKb) | 2.7 |
| Magnetic susceptibility (χ) | -64.0e-6 cm³/mol |
| Refractive index (nD) | 1.479 |
| Viscosity | Viscosity: 15 mPa·s (25 °C) |
| Dipole moment | 2.32 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 987.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | D11AX10 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May damage fertility or the unborn child. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H317, H319, H360, H372, H411 |
| Precautionary statements | P201, P202, P261, P264, P270, P271, P272, P280, P302+P352, P304+P340, P305+P351+P338, P308+P311, P321, P333+P313, P362+P364, P391, P405, P501 |
| NFPA 704 (fire diamond) | 2-1-1-Ask |
| Flash point | 107°C |
| Autoignition temperature | 343°C |
| Lethal dose or concentration | LD50 Oral Rat 175 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 207 mg/kg |
| NIOSH | WA2625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Dibutyltin Dilaurate: "0.1 mg/m³ (as tin, OSHA Ceiling) |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: 25 mg/m³ |
| Related compounds | |
| Related compounds |
Stannous octoate Triphenyltin chloride Tributyltin oxide Tetra-n-butyl orthotitanate |

