Dibenzyldichlorosilane: A Commentary on its Place in Modern Chemistry
Historical Development
Dibenzyldichlorosilane isn’t a household name, but folks working in silicon chemistry have crossed its path more often than they might admit. Like many organosilicon compounds, dibenzyldichlorosilane traces its roots back to the post-war boom in synthetic chemistry. The mid-20th century brought a frenzy of curiosity about how silicon could be shaped into new molecules, especially after the surprising versatility of silicon-based polymers emerged. Dibenzyldichlorosilane came around as researchers hunted for building blocks that would help steer the direction of growing industries—the plastics field, for one, but also electronics, coatings, and specialty intermediates. Back then, much of the excitement sprang from the ability to swap out methyl or phenyl groups for new organic branches, like benzyl, to pin down differences in reactivity and stability. This wasn’t just academic tinkering—modifications like this laid the groundwork for entire families of materials found in high-value industrial, medical, and even consumer settings now.
Product Overview
With two benzyl groups attached to silicon, and two more slots occupied by chlorine atoms, dibenzyldichlorosilane steps onto the scene as a key intermediate, not an end in itself. Its value lands in how readily it hands off its chlorines in exchange for functional groups that open up new reaction possibilities. The structure gives chemists a bit of flexibility; those benzyl moieties, with their stable yet modifiable nature, can drop into a chemical reaction with predictable consequences. In my bench work years ago, I found the compound reliable in synthesis protocols that called for cleaner reaction channels than bulkier, multi-chlorinated silanes could offer. Compared to older standbys like dimethyldichlorosilane, the presence of benzyl groups affects both handling and product outcomes—traits you can’t get just by tinkering with methyl or ethyl substituents.
Physical & Chemical Properties
Dibenzyldichlorosilane flows as a colorless or nearly colorless liquid, usually with a detectable, pungent odor, not unlike many organosilicon chlorides. The molecular weight tips well over 300 grams per mole—a hefty load for a molecule this small by carbon count, but that comes from the benzyl rings and the silicon center. Anyone who has transferred it between containers remembers its tendency to fume gently in moist air. Water contacts the chlorosilanes and produces hydrogen chloride gas and silanols in quick order; it’s a telltale sign that you’re working with a moisture-sensitive chemical. Its solubility in common organic solvents is broad, especially in aromatic and non-polar media, making it practical for multi-step organic syntheses.
Technical Specifications & Labeling
Chemical labels for organosilanes like dibenzyldichlorosilane must stress the hazards linked to corrosivity and reactivity with water. Industry standards require clear hazard pictograms and GHS-compliant statements. Researchers used to learn the hard way that poorly sealed bottles led to degraded, useless chemicals and a lingering whiff of hydrochloric acid in storerooms. These days, people rely on consistent purity metrics for advanced synthesis, so suppliers provide rigorous analyses such as NMR and GC-MS data. This kind of detail gives purchasers confidence in scaling syntheses or using the chemical in sensitive downstream applications.
Preparation Method
People generally make dibenzyldichlorosilane by reacting benzyl chloride with silicon tetrachloride under Friedel–Crafts conditions, usually catalyzed by an aluminum-based Lewis acid. There’s a dance of stoichiometry to get the two benzyl groups to substitute correctly. If conditions go off—too hot, too fast, or too little catalyst—the product mixture slides toward undesired side products like mono- or tribenzyl substituted silanes. In my earlier lab days, keeping water out of the reaction chamber was critical, not just for yield, but to avoid vigorous side reactions. As synthetic routes become greener, people tinker with milder conditions or recyclable catalysts, aiming to keep waste and energy demands manageable.
Chemical Reactions & Modifications
The reactivity of dibenzyldichlorosilane centers on its twin chlorine atoms. Nucleophilic substitution transforms these chlorides into silanols, alkoxides, or amino derivatives, each bringing new behaviors. Chemists prize this versatility for linking up organic and inorganic frameworks, essentially building the backbone for hybrid materials. The benzyl side chains survive moderate reaction conditions, but harsher treatment or catalytic hydrogenation can convert or strip them if needed. Over time, I saw teams experiment with integrating dibenzyldichlorosilane into crosslinking resins or as a precursor for functionalized siloxanes, aiming for new molecules with targeted flexibility or strength.
Synonyms & Product Names
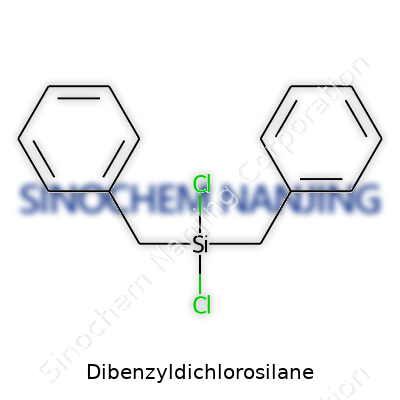
Dibenzyldichlorosilane sometimes passes under different names, like dichlorodibenzylsilane or silane, dibenzyldichloro-. These synonyms appear in catalogs and academic papers. In industry, product names may reflect supplier brands but usually stick close to the IUPAC or common variants, helping avoid confusion in ordering or compliance. If you refer to structure alone, the formula C14H14Cl2Si distinguishes it among organosilanes.
Safety & Operational Standards
Careless handling of dibenzyldichlorosilane leads to accidents—burns, toxic fume exposure, and sometimes costly spills. Safety starts with keeping water and acids far from storage and work areas. Proper PPE—gloves, goggles, and laboratory coats—matters no less today than it did back in the days of clunky glassware and limited fume hoods. Modern labs use vented enclosures and sealed apparatus, but real safety comes from respect for the chemical’s volatility and the speed of its reactions with air moisture. Regulations across the US, Europe, and mainland Asia require careful waste management and reporting on quantities stored or transported.
Application Area
The typical uses for dibenzyldichlorosilane stay tightly wound around specialty polymer synthesis, silicon-based coupling chemistry, and intermediate step transformations. Materials researchers might select dibenzyldichlorosilane where certain organic functionalities hang off the silicon atom, offering a path to advanced coatings, adhesives, or even drug delivery vehicles. In electronics, organosilanes with customized reactivity contribute to surface treatments or insulation materials. As industries call for more tailored materials, the demand for specialty intermediates like dibenzyldichlorosilane persists, even if production volumes never rival commodity silicones or standard chlorosilanes.
Research & Development
R&D involving dibenzyldichlorosilane often seeks practical improvements: greener synthetic routes, catalyst recovery, safer byproduct management, and broader scope in reactions. The challenge remains to swap out expensive, corrosive catalysts for more benign alternatives, all while maintaining high selectivity and yield. Literature over the last decade shows steady interest in using dibenzyl-derived silanes in making well-defined oligomers and polymers, particularly in advanced composites. Universities and industrial labs alike value silanes that balance reactivity with manageable safety profiles, but costs and sourcing logistics can hamper mainstream adoption outside core specialty fields.
Toxicity Research
Toxicological profiles for dibenzyldichlorosilane and other similar chlorosilanes paint a cautionary picture. Inhalation or skin exposure creates acute hazards—irritation, chemical burns, and respiratory problems. Animal studies reveal cytotoxic and possibly mutagenic effects if exposure levels climb beyond laboratory-safe limits. Chronic effects remain less well-defined, as use patterns rarely entail long-term low-level exposure. Labs and regulatory bodies alike take a conservative approach: fume capture, containment, and strict adherence to exposure limits are routine. Modern research continues to monitor metabolic breakdown and persistence, since accidental releases or spills can have significant downstream effects on both human health and local environments.
Future Prospects
With the surge in demand for specialty materials and advanced silicon-based technologies, dibenzyldichlorosilane holds its ground as a stepping stone rather than a celebrity molecule. Many new composite materials and hybrid organosilicon products grow from advances in functional silane intermediates. Silicon-based drug carriers, smart surface coatings, and flexible electronics all trace development roots to precursors like dibenzyldichlorosilane. The green chemistry movement nudges companies to seek safer, less toxic reagents; some researchers pursue silanes generated under solvent-free or catalytic conditions. Future breakthroughs likely hinge on innovation in both technical and regulatory spheres, balancing performance and sustainability as the world leans further into high-tech and resource-responsible manufacturing.
What is Dibenzyldichlorosilane used for?
Understanding Its Real-World Uses
Dibenzyldichlorosilane doesn’t make headlines, but folks in chemistry labs and factories know its value. The compound steps up when chemists look for a way to link silicon atoms with organic groups. Think of it as a bridge between worlds—helping build molecules that do more than just sit on a shelf. The basic structure includes silicon with two benzyl groups and two chlorine atoms. This setup gives it special qualities, making it handy for a bunch of applications—from crafting custom materials to fine-tuning electronics.
Key Roles in Silicone Chemistry
Years ago, during a stint at a specialty polymers lab, I watched how Dibenzyldichlorosilane changed the game for silicone synthesis. The compound reacts with other chemicals to open doors for making modified silicone polymers. These tailored silicones show up in sealants, medical devices, and coatings—products folks handle every single day without thinking twice about what’s inside.
Chemists use Dibenzyldichlorosilane as an intermediate, adding specific organic groups to the silicon backbone. This doesn’t just create strong bonds; it gives the finished material a certain flexibility or resistance to specific chemicals. Tech companies draw on these tweaks to hit tight performance targets, especially in industries where everything counts—like aerospace or high-performance engineering.
Helping Electronics Grow Smarter
Electronic materials keep getting smaller and more precise. Dibenzyldichlorosilane helps set those stages by acting as a precursor for silicon-based layers used in microelectronics. These layers buffer and protect sensitive circuits from water and faulty electrical signals. The demand for faster, more reliable electronics keeps growing, pulling more weight onto materials like this one.
This ability to attach organic links to silicon allows for greater customization in how electronic parts behave. From my experience, engineers want options on the molecular level. That kind of versatility lets companies push devices to run cooler, last longer, or resist harsh conditions.
Challenges and Safety Considerations
Handling chemicals like Dibenzyldichlorosilane calls for care. The chlorine atoms in its structure make it reactive—good for making things, but a risk if something spills or leaks. Strict protocols guide how labs and plants store and process it. Workers rely on proper ventilation, gloves, and training. Incidents still happen. Public health agencies and EPA keep watch over manufacturing processes to limit exposure and environmental release.
Looking for Safer Paths Forward
The future of specialty chemicals includes cleaner processes and improved personal safety. Companies can explore ways to capture waste before it hits the drain or adapt the chemistry to cut down on hazardous byproducts. Open communication and transparency, from factories down to the consumer level, matter too. In my opinion, demand for better safety and environmental performance isn’t going anywhere.
Final Thoughts
Dibenzyldichlorosilane might sound obscure, but its impact shapes many pieces of modern life, from the nurse’s office to the smartphone in your pocket. Understanding where it fits keeps industries moving ahead—while reminding everyone to balance innovation with responsibility and safety.
What is the chemical formula of Dibenzyldichlorosilane?
A Closer Look at an Under-the-Radar Compound
Dibenzyldichlorosilane doesn’t grab headlines, but its formula—C14H14Cl2Si—carries more importance than most people realize. This compound gets used in laboratories and industry for building bigger molecules, shaping research into better materials, and pushing forward pharmaceutical work. That may sound like a lot for one chemical, but every letter and number in C14H14Cl2Si spells out a tool for scientists on the hunt for new ideas.
Breaking Down the Structure
Each molecule comes packed with fourteen carbons, fourteen hydrogens, two chlorines, and a single silicon atom. The “dibenzyl” part means two benzyl groups (that’s a phenyl ring attached to a CH2 group) are bonded to the silicon atom. The “dichloro” hints at two chlorines also stuck to the silicon. Silicon in the middle, benzyls on each side, and chlorines filling in the empty spots. The way these groups sit together matters for both chemistry and practical work. As someone who’s seen plenty of glassware cloud up from improper handling of Si-compounds, keeping a close eye on the chemistry of these pieces stops a lot of headaches.
Real Uses, Real Impact
Lab workers often reach for dibenzyldichlorosilane during the process of making siloxane polymers, or when they need to modify surfaces for better performance (think electronics coatings or medical devices). A handful of well-placed molecules can change how water beads up on a surface or give an edge to a microchip process. Silanes like this one have been woven into the technical fabrics of synthetic chemistry, where their reactivity allows for new material development, and sometimes paves the way for better biocompatibility in implants.
Handling this compound can't be done without respect for its reactivity. Exposure to water can make it release hydrochloric acid, which does a real number on skin and eyes. In my personal experience, if you aren’t wearing goggles and gloves, one splash can be memorable for all the wrong reasons. It’s important to have proper training and a safe space to work, plus chemical-resistant gear. Safe storage in dry, inert environments helps everyone walk out at the end of the day without surprise injuries or ruined experiments.
Pushing for Safer and Smarter Chemistry
The formula for dibenzyldichlorosilane doesn’t change, but the ways people use it do. Cleaner production methods using less toxic reagents, closed systems that limit emissions, and better waste handling can lower risks. Knowledge is key, too; newer chemists benefit from experienced voices stressing the value of slow, methodical work. Making time for routine safety drills goes a long way. Technology can help, but nothing beats an atmosphere where folks look out for each other and double-check one another’s set-ups.
New applications pop up in fields like electronics and biomaterials, and each new use kicks off questions about health and safety. Reviewing best practices, keeping up with recent studies on toxicity, and challenging manufacturers to label their materials honestly provides a safety net for workers and consumers. Trying to substitute less hazardous compounds when performance allows can make a real difference, and it takes a community to keep standards high.
How should Dibenzyldichlorosilane be stored?
Behind the Label: What Makes Storage So Important?
Dibenzyldichlorosilane doesn’t sound like something most folks would keep in their garage. It’s a chemical often found in labs and certain industrial processes, but just because it isn’t a household name doesn’t mean those handling it can afford to cut corners. There’s a reason for that. One wrong move with a compound like this, and you’re looking at risks to health, ruined research, or even serious workplace accidents. My years in a small research lab taught me fast that chemical safety isn’t about ticking boxes. It’s about protecting people, the work, and the environment right now, every day.
Why Moisture Changes Everything
Out of all the things to worry about, water probably tops the list for anyone storing dibenzyldichlorosilane. This compound reacts—pretty fiercely—with moisture. It doesn’t just degrade quietly. It lets off hydrochloric acid gas, and that’s harmful for both people’s lungs and expensive equipment around it. Trust me, the smell alone from a botched experiment once stuck in our lab for weeks. So, the smartest first line of defense? Keep the stuff bone-dry. Airtight containers made of tough glass or stainless steel work well. Forget about the plastic bottles from the hardware store. Over time, even tiny leaks ruin everything.
Low Temperatures: A Hard-Learned Lesson
Many chemicals sit on open shelves. Dibenzyldichlorosilane shouldn’t. A cool, dark cabinet works best. Direct sunlight and warm rooms speed up chemical reactions, and not the good kind. Temperatures around 2–8°C keep it stable. Most labs use a fridge designed just for chemicals. It costs more, but stealing space in your office minifridge isn’t safe for anyone, especially with lunch packed in next to reactive compounds.
The Case for Good Labels and Shared Accountability
Some see labeling as tedious, but a clear label can stop a big mistake. Write the date, hazards, and anyone’s name who uses it. Back when I worked with a rotating team, we all agreed to double-check labels before every use. It sounds basic, but after hearing about accidents at other labs from mixed-up containers, our rule didn’t seem so obsessive.
Emergency Gear—Not Just Decoration
Storing dibenzyldichlorosilane safely doesn’t stop with the right container and fridge. Emergency showers, eyewash stations, gloves, goggles, and fume hoods must stay ready and available. Nobody expects an accident. But putting the right gear within reach takes chaos out of panic moments. The one time a colleague splashed a much milder chemical near his eye, that emergency eyewash bottle worked because someone cared enough to check it each month.
Smart Storage: A Matter of Integrity
A single oversight risks more than one person’s shift. The smart storage of dibenzyldichlorosilane shows a team pays attention—to health, to science, to those counting on their work. Clear rules, the right equipment, and a culture where everyone takes responsibility for making sure dangers stay inside their containers—that’s not wishful thinking. It’s just good science, and it reflects real-world care for people and progress.
What safety precautions are needed when handling Dibenzyldichlorosilane?
What Dibenzyldichlorosilane Teaches About Lab Safety
Handling dibenzyldichlorosilane offers a clear reminder that chemistry’s beauty comes with hazards you can’t ignore. In the past, I’ve worked with similar chlorosilane compounds, and the wrong move always brought me straight back to the basics of protective gear, steady procedures, and solid teamwork. No clever shortcuts helped—only respect for the risks and attention to detail kept us out of trouble.
Understanding the Real Dangers
This chemical reacts fast with water, releasing hydrochloric acid and a mess of fumes. My lab shoes have seen a splash or two, and prompt rinsing kept my skin safe, but the stinging burn of HCl vapor sticks with me as a warning. Your skin doesn’t forgive. Eyes can’t tough it out. Even a noseful—well, ask anyone who’s had it happen, you don’t forget. Goggle lines on my face after long days remind me PPE is never optional.
Major chemical suppliers and OSHA call dibenzyldichlorosilane corrosive and irritating to eyes, skin, and the respiratory tract. A quick search on PubChem or its safety data sheet paints the same picture: gloves, lab coats, splash shields, goggles, never open bottles outside a real fume hood. In dry air, it might seem less threatening, but don’t drop your guard. One careless moment with moisture and you get hydrochloric acid clouds fast.
Solid Steps Keep You Safe
I remember my mentor insisting on triple-checking PPE and doing a “dummy run” with just bottles of water. That routine never felt like overkill. Nitrile gloves, thick enough to resist splashes, always went on before touching bottles. Respirators? Rare in some labs, but standard any time the fume hood had hiccups.
Working with a partner always made sense. You need a second set of eyes—the friend watching you pipet, measuring, or pouring. If anything splashed, someone had the eye wash open, the shower running, or the spill kit unzipped before I could blink. Solid communication saved time, and maybe more than that.
Storage gets overlooked. I’ve seen jars tucked behind acids or left open on a crowded bench, which never ended well. Silanes like this stay only in tightly sealed, labeled containers, out of direct sunlight and moisture, away from anything that could cause a reaction. Never back in the shared fridge where food sits. In some labs, we set up a dedicated spot, with extra signage just to remind people to think before reaching.
Cultivating Sensible Behaviors
Safety depends on habits, not just memorized rules. For every training I sat through, it was real-life stories from colleagues that changed how I did things. Cleanup matters as much as setup. Dirty gloves or unmarked waste can create new hazards—chlorosilanes react in unpredictable ways with old rags or chemical trash. Attack spills with neutralizing agents meant for acids, ventilate the area, and always log the incident, so others can learn from it.
The solutions boil down to respect for the compound, support from the team, and never letting routine turn into mindlessness. If you notice someone skipping safety gear or rushing, speak up. If protocols seem out of date, push to review them. Every bottle of dibenzyldichlorosilane brings the same truth: nobody’s too experienced to get careless, and nobody’s immune from the consequences of a lazy moment.
What is the molecular weight of Dibenzyldichlorosilane?
The Number: 297.2 g/mol
Dibenzyldichlorosilane packs a molecular weight of 297.2 grams per mole. The structure carries two benzyl groups, two chlorine atoms, and silicon holding the center. Its chemical formula reads C14H14Cl2Si. That number might look like another line in a table, but accuracy in this value builds the foundation for good science—especially in synthesis and material design.
Why This Number Stands Out
In the lab, measuring by the gram demands attention to every digit in molecular weight. Planning to react dibenzyldichlorosilane with another compound? The stoichiometry tells you exactly how much to measure to avoid waste, danger, or outright failed reactions. A miscalculation leads to excess reagents, wasted money, or at worst, an unpredictable mix that spoils a whole batch. From my own bench days, even a small deviation in measurement compounds fast—costing more money than most budgets can handle.
Implications Outside the Academic World
Scale up to industrial settings, mistakes with molecular weight turn serious. One kilogram of dibenzyldichlorosilane holds thousands of dollars in precursors and downstream products. Overusing or underusing the compound means missed deadlines and lost revenue. Anyone working with specialty chemicals, like those used to craft coatings for electronics or build silicon-based polymers, relies on this calculation. Silicon chemistry does not forgive sloppiness.
Safety and Regulation
The weight also impacts labeling, shipping, and compliance. Dangerous goods rules in many countries—like the U.S. Department of Transportation or the European Chemicals Agency—trace amounts and limits based on both weight and chemical identity. Chlorine atoms bring their own hazards. Regulatory teams check molecular weights while filling out safety documentation or confirming the right packaging for transport. Relying on incorrect numbers risks fines—the kind that cut deep into operational budgets—or confiscated shipments at ports.
The Path to Reliable Values
Mistakes in published molecular weights sow confusion. Researchers sometimes trust online sources or outdated catalogs. For my lab work, I learned long ago that digging up peer-reviewed articles, supplier data sheets, or checking calculations using the atomic masses gives peace of mind. It sounds simple but double-checking each mass—carbon, hydrogen, silicon, and chlorine—remains one of those steps that saves hours and headaches later.
Building Routines for Better Chemistry
Taking the time to get things right beats repeating work or cleaning up messes. Trust grows between team members who see careful calculation as a sign of respect, not bureaucracy. If the plan is to use dibenzyldichlorosilane for new materials or as a building block in advanced electronics, a molecular weight of 297.2 g/mol isn’t just a figure in a database—it’s a cornerstone for innovation. While it’s tempting to cut corners, the experienced chemist knows every digit counts.
Tools and Solutions on Hand
Digital scales, reliable resource libraries, and well-tested software help take human error out of the equation. Sharing updated information across labs and suppliers lowers the risks. Some teams develop simple checklists to verify weights every time they try a new protocol. Good habits like these travel as far as your data does. Where digital and manual converge, real quality starts.
| Names | |
| Preferred IUPAC name | dichloro(dibenzyl)silane |
| Other names |
Dibenzyl dichlorosilane Benzyldichlorosilylbenzene Dichlorobis(phenylmethyl)silane |
| Pronunciation | /daɪˌbɛn.zɪl.daɪˌklɔː.roʊˈsaɪ.leɪn/ |
| Identifiers | |
| CAS Number | [821-23-8] |
| Beilstein Reference | 3919613 |
| ChEBI | CHEBI:33325 |
| ChEMBL | CHEMBL3114035 |
| ChemSpider | 157853 |
| DrugBank | DB14671 |
| ECHA InfoCard | ECHA InfoCard: 100.032.997 |
| EC Number | 205-861-2 |
| Gmelin Reference | 78048 |
| KEGG | C19307 |
| MeSH | D008167 |
| PubChem CID | 66068 |
| RTECS number | VS9450000 |
| UNII | W10X2S1I8D |
| UN number | 1760 |
| CompTox Dashboard (EPA) | DTXSID4061451 |
| Properties | |
| Chemical formula | C14H14Cl2Si |
| Molar mass | 295.24 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Aromatic |
| Density | 1.14 g/mL at 25 °C (lit.) |
| Solubility in water | Decomposes |
| log P | 2.9 |
| Vapor pressure | 0.01 mmHg (25°C) |
| Acidity (pKa) | 13.1 |
| Magnetic susceptibility (χ) | -58.0e-6 cm³/mol |
| Refractive index (nD) | 1.603 |
| Viscosity | 1.74 mm2/s (25 °C) |
| Dipole moment | 2.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 489.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | No ATC code |
| Hazards | |
| Main hazards | Causes severe skin burns and eye damage. Causes serious eye damage. Toxic if inhaled. Reacts violently with water. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS06, GHS05 |
| Signal word | Danger |
| Hazard statements | H302, H314 |
| Precautionary statements | P261, P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 85 °C |
| Autoignition temperature | Autoignition temperature: 500 °C |
| LD50 (median dose) | LD50 (oral, rat): 1800 mg/kg |
| NIOSH | NA |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 ppm |
| Related compounds | |
| Related compounds |
Benzyltrichlorosilane Benzylmethyldichlorosilane Dibenzylsilane Dimethyldichlorosilane Phenyltrichlorosilane |

