Diazoaminobenzene: Past, Present, and What Comes Next
Historical Development
Few chemical compounds have attracted as much early curiosity as diazoaminobenzene. In the late 1800s, researchers in Europe explored the strange world of azo compounds, driven by a hunt for new colors and the makings of basic organic chemistry. Wilhelm Borsche first prepared diazoaminobenzene in 1895, paving the way for questions about its structure and how stable these molecules could be. Chemists soon realized they had stumbled upon more than a curiosity. They saw a versatile platform for making bigger dyes and laid the groundwork for understanding diazo chemistry. I remember digging through worn-out lab notebooks during my time as a student—one could see scientists doggedly refining methods, drawn by the surprises hidden in what seemed a simple yellow powder. Work on this compound shaped approaches to organic synthesis, even in today’s advanced labs.
Product Overview
Diazoaminobenzene now goes by several names in catalogs and handbooks: benzene, 1,2,3-triazene; DAB; and the simpler “yellow benzene.” Some refer to it as diazoaminobenzol, borrowing from old nomenclature. Labs buy it heavy in crystalline form or as a reagent for further chemical reactions. Its persistence in chemical supply lists reflects its unique reactivity: this is not a casual compound, but one built to unlock deeper transformations, particularly in dye-building and research.
Physical & Chemical Properties
Taking a close look at diazoaminobenzene, one finds it as a light yellow, sometimes orange-tinted solid, distinctive by its bitter almond smell. It melts around 125°C, but won’t dissolve much in water. Try ethanol or ether, and it goes into solution far more easily. I’ve seen how quickly pure samples react when mishandled—contact with acids or strong light triggers visible decomposition. With the formula C12H11N3, its molecular weight checks in around 197.24 g/mol. Technically, the compound holds together by a chain of nitrogen atoms linking two phenyl rings, a setup eager for all manner of chemical adventures. That triazene linkage is the source of both its usefulness and the reason for its careful handling requirements.
Technical Specifications & Labeling
Commercial batches of diazoaminobenzene come with strict quality demands. Labs require a purity often greater than 98%, measured through melting point checks and chromatographic tests. Organizing storage is not just about dry boxes and amber bottles—safety data sheets insist on segregation from acids or reducing agents. The right labeling makes a difference. One glance at a bottle on the shelf tells you: “I am unstable, keep away from open flames, avoid shocks.” Industry standard labels mention “H300 (Fatal if swallowed),” “H410 (Very toxic to aquatic life),” and other emergency codes. Such warnings sound alarmist, but seeing a poorly stored bottle degrade over months will convince anyone of the need for accurate, detailed labeling. That extra line or two on the safety sheet can prevent an accident months down the line.
Preparation Method
Time-tested syntheses rely on diazotization of aniline in acidic solution, followed by addition of a second aniline molecule. Fill a beaker with cold hydrochloric acid and dissolve sodium nitrite, add aniline dropwise and keep everything near ice-cold, or you risk a runaway. As soon as the diazonium salt forms, more aniline goes in—here, speed and steady stirring prevent local overheating. After a short wait, diazoaminobenzene crashes out as yellow crystals, ready for washing and purification through recrystallization. I’ve watched experienced chemists frown over slight temperature changes or pH shifts, knowing yield drops or unwanted isomers might show up with the smallest error. History records endless tweaks to these recipes as people search for improved safety, higher purity, and reproducibility.
Chemical Reactions & Modifications
Diazoaminobenzene acts as a stepping stone for complicated molecules. Chemists exploit its triazene chain to make new aryl derivatives, especially through rearrangements like the Wallach reaction—where rearrangement leads to substituted aminoazobenzenes. Under acidic conditions or UV light, breakdown into aniline and diazonium salts comes easy. That tendency explains its use in diazotization studies, serving as a model substrate for student labs and advanced synthesis projects. My first experience involved trying to trick it into coupling with active aromatic compounds and hoping for vibrant colors—results ranged from spectacular to a bricked mess requiring hours of cleaning. Industry teams use it to launch other nitrogen-rich scaffolds or in some cases to stabilize short-lived intermediates during multi-step organic syntheses.
Synonyms & Product Names
Search for diazoaminobenzene, and you will meet plenty of aliases beyond the ones mentioned earlier. Triazene, 1,2-diphenyl-, Yellow Benzene, and Benzene, 1,2,3-triazene crop up in labeling systems. Its registry in chemical catalogs spans hundreds of suppliers, usually accompanied by CAS Number 136-35-6. There’s a certain comfort in seeing the same handful of trade names and structures, no matter which global supplier you start from—the chemical world protects clarity through shared vocabulary, even for foundational compounds like this.
Safety & Operational Standards
Workplace safety around diazoaminobenzene takes more than a pair of gloves. Dust inhalation, accidental skin contact, or spills threaten serious health effects. Labs enforce fume hood work as standard practice. GHS labels come with pictograms warning of acute toxicity. The toxicity is not just theoretical: animal studies and accident reports in chemical plants underscore the risks of careless handling. I once witnessed a technician ignore recommended protocol—just a brush with contaminated gloves left reddened skin and required urgent washing. Such incidents underline the importance of constant training on chemical hazards, routine audits of procedure, and a culture where “just this once” is not tolerated.
Application Area
You don’t meet diazoaminobenzene in everyday products, but dig beneath the surface in organic labs or pigment factories, and you’ll see its imprint. Synthetic dye manufacturers use it as a precursor for a range of azo compounds, turning what starts as yellow powder into everything from red lake pigments to base dyes for textile fibers. Analytical labs select it for controlled proteolysis or as a nitrosating agent. There are research settings where its ability to mask or release nitrogen groups unlocks more sensitive compound development. Its utility in the field of molecular probes should not be overlooked, either. Experience has shown that researchers build on such legacy compounds, finding new application twists as analytical gear grows more sensitive and environmental controls get tighter.
Research & Development
Behind the scenes, research teams continue to test the boundaries of what diazoaminobenzene can do. Advances in computational chemistry help map out its reactivity puzzles, while green chemists explore safer, less toxic synthesis routes. There is ongoing study into stabilization tricks—adding polymer support, dispersing in safe matrices, or embedding in silica—to cut handling risks. My peers share stories of automation systems, robotic platforms that meter out micrograms under fully enclosed conditions, a far cry from the open-bench work of the last century. New generations of students tackle projects connecting diazoaminobenzene chemistry with modern photonics, or enzyme-mimicking catalysis—it’s a compound that refuses to become obsolete.
Toxicity Research
Toxicological studies leave no question about the dangers of careless exposure. Short-term studies reveal it irritates skin and eyes, while chronic exposure threatens the blood and nervous system. Some past papers linked metabolites to possible carcinogenicity, raising concerns about long-term handling. Environmental chemists document how accidental spills or careless discharge harm aquatic systems, as the compound resists easy biodegradation. Practical safety needs more than numbers on a sheet: it calls for education and ongoing vigilance in lab design and training. Emergency response teams prepare for diazoaminobenzene incidents with full-scale drills, treating every vial as a real-world hazard.
Future Prospects
Few compounds balance history and potential as sharply as diazoaminobenzene. Given its role as a versatile precursor, synthetic chemists look for ways to make it safer, less polluting, and easier to handle. Research may focus on encapsulation methods or finding milder alternatives for core reactions, cutting down on side products and toxic releases. With advances in computational modeling and real-time spectroscopy, teams search for new modifications, hoping to uncover dyes, sensors, and pharmaceutical ingredients built on this chemistry. I expect the days of hand-mixed batches in open flasks to keep fading, replaced by automated microfluidic synthesis and real-time hazard monitoring. Even in a world of high-tech chemistry, the lessons from the past—respect for risk, obsession with purity, curiosity about structure—stay relevant with diazoaminobenzene at the bench.
What is Diazoaminobenzene used for?
Diazoaminobenzene: Not Just a Chemical Name
Some chemicals hide in the background, doing the work no one talks about at dinner parties. Diazoaminobenzene fits that role. It comes to mind from my undergraduate chemistry days, where it often showed up in experiments most folks outside the lab might never even hear about. I still remember its bright yellow color in a test tube, as if its look tried to warn us about handling it with care. This stuff goes beyond just a complicated word; it’s about what researchers do with it and why it matters in labs and industry alike.
Main Uses of Diazoaminobenzene
Diazoaminobenzene finds a place in organic synthesis. Chemists don’t just use it for fun—I’ve seen it become a building block in making other chemicals. Its role in preparing dyes stands out. Way before synthetic dyes filled store shelves, color came from natural sources. Fast forward, and diazoaminobenzene helped shape the range of colors we see in textiles today. Textile production shifted into high gear because of chemicals like this. The industry still leans on these compounds. After being converted, diazoaminobenzene produces azo dyes, famous for their vibrant shades and lasting power on fabrics and leathers.
Beyond dyes, researchers depend on diazoaminobenzene to study how reactions work. In academic labs, undergrads learn about azo coupling reactions, and diazoaminobenzene often sits right at the center. Try explaining to a student why a color suddenly appears or disappears without talking about this compound. In my teaching days, we depended on this reaction as a simple demonstration. It gave students a visual language to understand more abstract reaction pathways.
Challenges in Handling and Environmental Impact
No story about chemicals skips over safety. Growing up in the 90s, safety gear often took a backseat to curiosity. As I later discovered, accidental exposure to diazoaminobenzene can lead to headaches and dizziness. Researchers have noted it might act as a carcinogen, pushing institutions to limit its use. Factories must plan carefully: one mistake could harm workers or spill into the water supply. Disposal becomes tricky, as you don’t just toss this down a drain. Strict protocols keep it under control, but mistakes still happen.
Environmental concerns go hand in hand with industrial progress. Azo compounds, in general, sometimes break down into toxic substances. In rivers near textile plants overseas, scientists have found traces of these chemicals, and concern rises for both animal life and people using that water. Following strict laws and turning to alternative dye methods could ease these tensions. Natural dyes and better filtration systems start making sense, but they cost money. For companies with tight margins, switching takes leadership and perseverance—traits often in short supply. Still, there’s growing pressure for cleaner chemistry, coming from both buyers and advocacy groups.
Moving Forward with Chemistry
Diazoaminobenzene might never land on a celebrity top 10 list, but it holds a seat in the front row of modern chemistry labs and industrial dye plants. With new research, safer alternatives and greener methods beckon on the horizon. Researchers, workers, and investors have to balance progress with responsibility, remembering stories from labs gone by. Like so much in chemistry, the ability to innovate comes with the need to protect both people and the planet.
What are the storage requirements for Diazoaminobenzene?
Minding the Details of Chemical Safety
In labs where synthetic dyes or research chemicals are routine, Diazoaminobenzene catches attention not just for its yellow appearance but for the risks it carries. Folks run into trouble when giving only half their attention to safe storage, thinking old habits can cover the job. That mindset often turns minor mistakes into dangerous situations. Working with chemicals like Diazoaminobenzene drove home how everyday choices contribute to lab safety — or risk turning a laboratory into a hazard zone.
Why Temperature and Light Are Not Small Matters
Diazoaminobenzene doesn’t handle heat well. Temperatures above room temperature cause decomposition. Once, someone stacked a box of it near a window; sunlight pried the box open and the room filled with a strong odor as the compound slowly broke down. The lesson? Sheltering this chemical in a cool, shaded spot means the difference between peace of mind and a headache — both figuratively and literally. Chemical manufacturers set a recommended range for a reason: 2-8°C. This keeps energetic reactions to a minimum. Nobody enjoys mopping up after an avoidable accident just because a bottle spent too long on a sunny shelf.
Dry and Airtight: More Than Just Precaution
Humidity lurks in the background and quietly ruins entire batches. In my early days, I stored Diazoaminobenzene beside a sink. The humidity crept in, causing caking and loss of quality. Moisture triggers slow decomposition, releasing potentially harmful gases. Airtight, moisture-proof containers aren’t optional; they’re non-negotiable. Glass jars with tight-sealing lids or specialist safety bottles continue to deliver best results. If labels fade, or if lids rest ajar, the risk creeps up like a silent visitor.
Choosing the Right Storage Partners
Mixing Diazoaminobenzene with oxidizers or acids ends poorly. Once, a coworker thought saving shelf space mattered more than a proper separation strategy. Containers sat side-by-side. An accidental spill set off a chain reaction, handing the safety officer a mountain of paperwork. Good chemical housekeeping means dedicated shelves, color codes, and clear separation. No one wants flammable materials and explosives sleeping next to each other.
Why Personal Vigilance Beats a Written Rulebook
Guidelines list the dos and don'ts, but real safety grows from a sense of ownership. Sensors for temperature, humidity, and access provide oversight, but nothing beats the impact of a committed team. I keep a dedicated checklist on the wall near every storage room. Nothing fancy — just a tracking sheet for inspection, signed daily. Every ignored checklist line is a gamble with safety. Masks and gloves aren’t just for show, either. Contact or inhalation of breakdown products means irritation or worse. Fume hoods replace open benches for weighing and transferring.
Building Safety Habits Into Routine
Proper storage for Diazoaminobenzene isn’t about following rote instructions; it's about understanding the risks and building better habits. Every overlooked shortcut echoes louder in a chemical storeroom. A temperature log, a correctly labeled bottle, and a habit of checking seals each day keep problems at bay. Factoring in training and regular audits goes further than any single policy. Talk about lessons learned spreads faster than notices on a bulletin board. Years in the lab taught me that awareness and responsibility are stronger shields than any lock on a cabinet.
Is Diazoaminobenzene hazardous to health?
What It Is and Where People Encounter It
Walk into a chemistry lab, and you might find something called diazoaminobenzene. Sometimes, it pops up as a yellow crystalline powder. Labs use it for dye preparation and as an intermediate in organic synthesis. Outside the lab, people rarely cross paths with it. But the risks don’t stop at the lab door, especially for workers handling the stuff every day.
The Risks That Matter
Scientists figured out early that diazoaminobenzene causes skin and eye irritation. Just touching it can lead to redness and discomfort. Inhaling its dust triggers coughing and headaches. Folks working with it — often chemists or chemical industry staff — face these problems if proper safety measures aren’t in place.
Studies, particularly from animal testing, show bigger dangers. Researchers saw that long-term exposure could affect the liver and kidneys. Some older reports offered hints about possible links to tumors, though the evidence in people stays thin. That kind of uncertainty should make anyone pay attention.
Why This Is a Big Deal
When I ran chemicals through high school or undergraduate courses, basic safety got drilled into our heads. Gloves, goggles, ventilation — they weren’t just for show. Even small slips with compounds like diazoaminobenzene matter. In my time working in a chemical storeroom, I saw firsthand how careless storage or spills could create a real mess, and sometimes those incidents left coworkers in the health clinic. Less hazardous chemicals still sent folks home with rashes or persistent coughs. Diazoaminobenzene stands a couple of notches higher on the risk ladder, even if it’s nowhere near the most toxic stuff out there.
What Science and Regulatory Agencies Say
Agencies such as the International Agency for Research on Cancer (IARC) keep a close eye on compounds like this. Diazoaminobenzene hasn’t seen wide commercial use, so big population studies are limited. The few available studies push for caution, and many chemical catalogs mark it as “hazardous.” OSHA has set exposure guidelines, requiring proper fume hoods and personal protection around the chemical. Europe’s REACH regulations push companies to minimize use and swap in safer alternatives if possible.
What People Can Actually Do
Training matters more than protocols on paper. Regular safety reviews help remind everyone how to handle things right. Chemical companies now require workers to use protective gear, install proper ventilation, and isolate processes that generate dust or fumes.
If you’re in a research setting, don’t cut corners on storage. Even students just starting out need this knowledge. It’s not uncommon for universities to phase out diazoaminobenzene in favor of compounds with lower toxicity, but sometimes research still calls for it. In those cases, keep spill kits reachable and follow up-to-date disposal guidelines.
People outside the lab want transparency about what’s near their neighborhoods or water supply. Communities often feel uneasy if universities or companies store old chemicals. Clear communication helps avoid bigger problems by showing a plan is in place for safe use and disposal.
Looking Ahead
Industry pushes forward with safer chemicals all the time. Chemical manufacturers can keep investing in green chemistry. If small-time labs or businesses feel unsure about their stockrooms, consultation with a chemical safety expert can prevent tomorrow’s news story about an avoidable accident or exposure.
Chemicals like diazoaminobenzene don’t belong in everyday life. With the right respect and practices, the dangers stay manageable. Ignoring the known risks only leads to unnecessary problems, and that’s something no researcher or community wants to read about next in the headlines.
What is the chemical formula of Diazoaminobenzene?
Understanding the Chemistry Behind Diazoaminobenzene
Diazoaminobenzene isn’t a name you hear around the kitchen table, but it reveals something fascinating about how chemical structure shapes function. Its formula, C12H11N3, spells out the mix: twelve carbons, eleven hydrogens, and three nitrogens. In plain terms, this combination packs together a benzene ring with a chain of nitrogen atoms—something that lets the compound fit into all sorts of applications, both in science classrooms and research labs. The unique structure means this compound acts as a bridge between basic organic chemistry and applied material science.
Why Look Twice at Diazoaminobenzene?
The value of diazoaminobenzene goes beyond textbook learning. Over the years, researchers have used it to make dyes, especially long before mass-market synthetics emerged. The backbone structure—a benzene ring with two nitrogen groups—gives those dyes unusual stability and colorfastness. It’s no secret that chemistry students get their first taste of real synthesis by making compounds like this. You pour in aniline, treat it with nitrous acid, and watch a yellow powder settle out—that’s the stuff. Lab hands quickly learn the importance of safety, since the same nitrogen chains making diazoaminobenzene useful also create unpredictable risks.
From Synthesis to Safety: The Conversation We Need
Living through the trial and error of student labs, a strong memory sticks: the importance of respect, not just for chemicals, but for their legacy. Diazoaminobenzene formed part of early twentieth-century life as industrial chemists raced to create new pigments and materials. Yet with time comes perspective, and so the focus has turned to health. N-nitroso compounds (created as by-products in some diazoaminobenzene reactions) link back to worrisome findings in toxicology studies. Safety guidelines now insist on protective gear and good fume control. These updates weren’t always in place, a reminder that progress in labs can sometimes run ahead of thoughtful review.
Real-World Decisions: Knowledge and Responsibility
In the broader chemical industry, diazoaminobenzene stands as a fork in the road. Innovations in dye technology shifted attention to safer, greener molecules. Regulators took note, and research now weighs the environmental cost of legacy chemicals like this one. Teachers, too, have steered students toward experiments with lower risk, knowing that science education works best where curiosity and caution walk hand in hand.
The take-home lesson carries weight outside the lab. Chemicals shaped the twentieth century, improving daily life and industry, but the cost sometimes came out later—in health issues and pollution. Chemists, educators, and product makers have started moving with more care, driven by clear-eyed assessments and a culture of openness about risk. Solutions range from greener chemistry approaches, finding alternatives for pigments, and building stronger regulations that treat safety as an investment, not a burden.
The Ongoing Dialogue Between Science and Society
Trust in chemistry gets built when researchers, educators, and regulators pay close attention to how compounds like diazoaminobenzene work and how they’re managed. The formula C12H11N3 stands for more than atoms and bonds—it’s a reminder that human progress depends on honest, ongoing conversations about what we create and how we handle it.
How should Diazoaminobenzene be handled during disposal?
The Hidden Risks in Chemical Labs
Diazoaminobenzene looks like a harmless yellow powder, but beneath that mild appearance lies a set of risks that go beyond what most folks expect from run-of-the-mill chemicals. Anyone who's spent long hours working in a research lab with this compound can tell stories about the nervous respect it commands among professionals. This isn't your everyday solvent or salt. Overlooking basic procedures with diazoaminobenzene is playing with fire, sometimes quite literally.
Not Just a Lab Problem
Few people outside the chemistry world recognize the dangers tied to improper disposal. Throughout the years, I’ve seen how a “toss it and forget it” attitude can lead to dangerously close calls. Diazoaminobenzene breaks down unpredictably, especially if it heats up, dries out, or reacts with strong acids. Nasty things like toxic fumes or, in worst cases, violent explosions become a real threat. If you’ve ever wondered why some labs keep outdated or unused chemicals locked up so tightly, this is exactly why.
Disposal Is Everyone’s Responsibility
Lab safety doesn’t stop at gloves and goggles. Something as basic as thoughtful disposal separates decent labs from excellent ones, and it keeps people out of emergency rooms. In my career, I’ve seen teams lean on guidance from the Environmental Protection Agency and other health bodies. These aren’t meaningless hoops — they have roots in real, sometimes tragic, accidents.
As a starting point, diazoaminobenzene should never be dumped in the drain, regular trash, or left out to “air out.” Instead, responsible disposal means transferring any leftover powder or contaminated materials into airtight, clearly labeled hazardous waste containers. This slows decomposition and stops the spread of harmful dust or vapors. Labeling everything clearly keeps sanitation workers or custodians from getting a nasty surprise. Every university and research center has a chemical waste program for a good reason. Regular chemical pickups by licensed professionals remain the gold standard.
Fresh Solutions and Smarter Mindsets
Following disposal rules is a baseline, but innovators find ways to tackle the root causes. For instance, some labs have designed on-site neutralization systems that break down diazoaminobenzene under temperature-controlled, contained conditions. These methods reduce the volume of dangerous waste heading out into the world, and they offer better oversight. The extra effort may seem like a headache, but real safety and sustainability don’t grow from shortcuts.
Teaching people why this all matters builds an extra safety net. Training sessions have real power to sink in the message that skipping steps with chemicals creates risk for everyone: researchers, waste handlers, and the wider community. Just handing over a set of instructions isn’t enough. Story-sharing about real-life near-misses and keeping honest about mistakes gets the point across. A safety culture grows from shared responsibility, and it sticks around longest when fresh experiences help shape the rules.
Personal Experience: No Room for Complacency
I still remember the first time my lab ran a routine inventory, uncovering an old, mislabeled jar tucked away in the back of a drawer. Turns out it was a small batch of diazoaminobenzene, left since an early project. Nobody wanted to move it until we called in hazardous material experts. That moment hammered home how old chemical ghosts can linger, and why strict protocols are not just bureaucracy but a real shield.
Respect for chemicals like diazoaminobenzene isn’t about paranoia; it’s about valuing people’s lives, workplaces, and the environment around us. The right habits and some honest sharing of practical stories trump clever shortcuts every time.
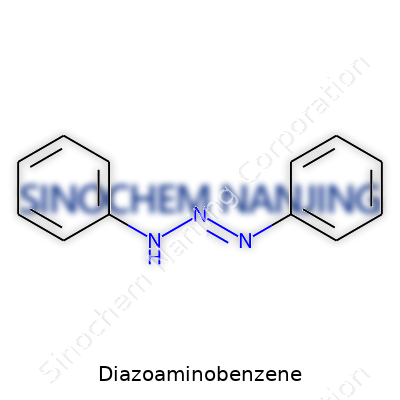
| Names | |
| Preferred IUPAC name | 1-Phenyldiazene-1-amine |
| Other names |
Diazobenzeneamine 4-Phenylazoaniline N,N-Diphenyldiazeneamine N-Phenylbenzenediazonium amine Yellow Tartrazine |
| Pronunciation | /daɪˌæzoʊˌæməˈnoʊˌbɛnziːn/ |
| Identifiers | |
| CAS Number | 136-35-6 |
| Beilstein Reference | 1207932 |
| ChEBI | CHEBI:28545 |
| ChEMBL | CHEMBL15800 |
| ChemSpider | 8660 |
| DrugBank | DB04257 |
| ECHA InfoCard | 100.007.278 |
| EC Number | 200-194-9 |
| Gmelin Reference | 5778 |
| KEGG | C06432 |
| MeSH | D003972 |
| PubChem CID | 69241 |
| RTECS number | DJ8925000 |
| UNII | 6P9RDW0V38 |
| UN number | UN2586 |
| CompTox Dashboard (EPA) | DTXSID4020772 |
| Properties | |
| Chemical formula | C12H11N3 |
| Molar mass | 198.23 g/mol |
| Appearance | Orange-yellow crystalline powder |
| Odor | Odorless |
| Density | 1.18 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.97 |
| Vapor pressure | 0.0000715 mmHg (25°C) |
| Acidity (pKa) | 2.79 |
| Basicity (pKb) | 6.71 |
| Magnetic susceptibility (χ) | \(-0.67 \times 10^{-6}\) cm³/mol |
| Refractive index (nD) | 1.642 |
| Viscosity | 2.16 mPa·s (at 158 °C) |
| Dipole moment | 2.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 144.3 J K⁻¹ mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 79.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -44 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | D03AX02 |
| Hazards | |
| Main hazards | Explosive; harmful if swallowed, inhaled, or absorbed through skin; irritant to skin, eyes, and respiratory tract. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS01,GHS07 |
| Signal word | Warning |
| Hazard statements | H302 + H351 + H319 + H411 |
| Precautionary statements | P261, P264, P270, P272, P280, P302+P352, P308+P313, P363, P405, P501 |
| Flash point | 113°C |
| Autoignition temperature | 130 °C |
| Lethal dose or concentration | LD50 oral rat 1800 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2000 mg/kg (oral, rat) |
| NIOSH | DZB |
| PEL (Permissible) | 1 mg/m³ |
| REL (Recommended) | 0.05 mg/L |
| IDLH (Immediate danger) | 50 mg/m3 |
| Related compounds | |
| Related compounds |
Aniline Azo compounds |

