Diammonium Hydrogen Arsenate: Insight into Its Science, Use, and Future
Historical Development
Diammonium hydrogen arsenate traces its origins to an age marked by rapid industrial expansion and a quest for better understanding of minerals. Chemists of the late nineteenth century, curious about arsenic chemistry, started isolating this salt for analytical work and for developing insecticides that could curtail crop losses. Over time, knowledge around its dangers shifted applications from agricultural experiments to more controlled laboratory settings. Early on, arsenic compounds gained a sinister reputation for their toxic properties, yet scientific rigor continued to push further research, offering a reminder that discovery often grows from a need to solve pressing challenges—crop blights, pests, or refining analytical methods.
Product Overview
Diammonium hydrogen arsenate comes in crystalline or powder form, blending the sharp presence of ammonia with arsenic's clinical edge. Commonly referred by different names in scientific circles—diammonium arsenate(V), ammonium arsenate dibasic—this compound stands as a staple in chemical collections, particularly where precise arsenic solutions or calibration standards are required. It may not boast a spread of commercial brands like household cleaners or kitchen staples, but its presence in chemical catalogs and lab inventories remains steady. Even with its limitations stemming from toxicity and safety, this salt carries a trace of the era when chemistry classrooms ran with open bottles and bold exploration.
Physical & Chemical Properties
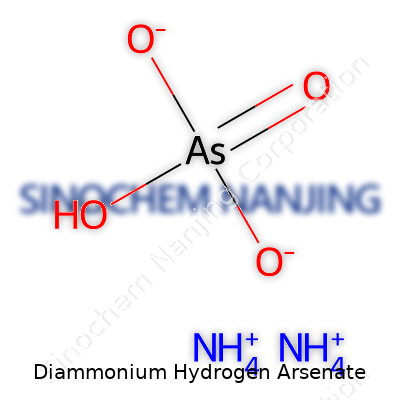
At its core, diammonium hydrogen arsenate holds the formula (NH4)2HAsO4, sporting colorless crystals that readily dissolve in water. Anyone working near it knows the faint ammonia smell and appreciates its stability under dry storage. It doesn’t boil; instead, it decomposes under high heat, throwing off arsenic oxides and ammonia gas. With a molecular weight around 230.05 g/mol, this compound lies heavier than many ammonium salts and packs arsenic in a way that sparks both interest and caution. As a moderately strong base (thanks to the ammonium ions), it reacts easily with acids to form triammonium arsenate or with strong bases to give insoluble arsenate salts.
Technical Specifications & Labeling
Manufacturers and laboratory suppliers label this compound with clear warnings due to its acute toxicity. Purity levels stretch up to analytical grade, with packaging including the UN number 1556 and hazard diamond indicating toxic substance status. Tech datasheets lay out acceptable impurity thresholds, water content, and recommended storage in airtight containers at room temperature, away from light and sources of ignition. Precise CAS registration (number 7784-39-8) shows on every bottle, while required safety phrases urge the use of gloves, goggles, and fume hoods. No reputable supplier skirts these practices, as exposure leads directly to health emergencies that regulatory audits don’t take lightly.
Preparation Method
Making diammonium hydrogen arsenate follows a straightforward route: dissolve arsenic acid (H3AsO4) in water, slowly neutralize with concentrated ammonia solution until the right pH tips the balance toward the dibasic salt. Once the reaction mixture cools, crystals begin to form, separating out by filtration and drying. Strict protocols limit ammonia vapors from getting into the workspace, and diligent chemists monitor the pH and temperature, ensuring maximum yield without pushing side reactions. This process has stayed almost unchanged for decades, illustrating how some methods require more careful oversight than radical innovation.
Chemical Reactions & Modifications
Diammonium hydrogen arsenate doesn’t sit idle in the lab. Under strong heat, it breaks down, releasing gases that force researchers to run these operations within fume extraction units. Mixing with strong acids can convert it back to arsenic acid, while reacting with alkali earth salts yields insoluble metal arsenates used in geochemical or forensic testing. Scientists tweak the ammonium-ion ratio to reset crystallization, and add chelating agents or competing anions to study arsenate behavior in environmental matrices. These reactions show how arsenic can pivot between mobility and fixation, linking lab chemistry to concerns about groundwater pollution and arsenic remediation efforts.
Synonyms & Product Names
Technical literature rarely settles for one name, adding to the confusion for anyone tracking this compound across chemical indexes. Some papers call it diammonium arsenate, others opt for ammonium arsenate dibasic. European pharmacists once listed it as "arsenate d'ammonium" in old formularies. Cheminformatics software files it under registry numbers rather than trade names. The diversity in synonyms may seem like an arcane quirk, but for safety data sheets and regulatory filings, the CAS number bridges language and regional naming habits, reminding buyers and lab techs that what matters is substance, not the label.
Safety & Operational Standards
Arsenates haunt toxicology textbooks, and diammonium hydrogen arsenate fits squarely in restricted material lists worldwide. Regulations require full PPE—lab coats, nitrile gloves, splash-proof goggles—and strict documentation down to the gram. Any accidental spill triggers immediate containment and reporting, as arsenic traces persist on surfaces and resist casual cleaning. Medical surveillance programs monitor those with occupational exposure, and instructors warn new students against taking even minor shortcuts. Waste from experiments goes to licensed hazardous disposal channels, not down the drain, turning what could be routine chemistry into a disciplined, safety-driven exercise.
Application Area
You don’t spot diammonium hydrogen arsenate in toy kits or common goods. Most use stays in scientific research: setting up arsenic reference standards, calibrating analytical instruments, and exploring arsenate’s interaction with biological and geological materials. Soil chemists, for instance, use it to trace arsenic’s movement through clay and sand, linking lab results to real-world contamination challenges. In older studies, it propped up toxicology models for metabolic research, but new restrictions now steer its use toward tightly regulated protocols, often as part of multidisciplinary projects in environmental science or public health.
Research & Development
Growing concern over arsenic pollution fuels constant R&D around this compound. Studies focus on everything from stabilization techniques—using diammonium hydrogen arsenate to mimic natural arsenate in soil processes—to developing more sensitive ways to detect parts per billion levels in water. Researchers assess how phosphate-arsenate substitutions affect enzyme systems, and gather data on arsenic mobility under changing pH, redox potential, and microbial activity. The push for greener chemistry nudges teams to design new methods that either minimize the need for such toxic controls or at least handle them in closed-loop, waste-minimizing setups.
Toxicity Research
Diammonium hydrogen arsenate impresses with its acute and chronic toxicity. Swallowing even small amounts produces classic arsenic poisoning symptoms: stomach pain, vomiting, confusion, and long-term organ damage. Animal studies probe its mechanism, focusing on disruption of cellular respiration and enzyme inhibition. Toxicologists measure how it accumulates in organs, latches onto proteins, and triggers both immediate and delayed health risks. Medical literature links arsenic exposure to cancer risks, skin lesions, and vascular damage, anchoring safety standards in grim statistics collected over generations. Emergency response guides hammer home that any exposure, no matter how brief, deserves serious attention and medical follow-up.
Future Prospects
The chemical industry won’t erase its dependence on arsenic compounds overnight, thanks to their role in reference standards, calibration solutions, and special analytical workflows. Despite the hazards, scientists continue refining protocols for safer handling, detection, and remediation. Innovations in nano-filtration and bioremediation promise to cut risks for water systems and people living near contaminated sites. Stricter regulations and growing global awareness around arsenic toxicity put pressure on suppliers and researchers to look for alternatives, invest in closed-system reactors, and support green chemistry approaches. Still, diammonium hydrogen arsenate stays on as a critical reference for environmental and analytical work, acting as both a tool and a warning from chemistry’s complex past.
What is Diammonium Hydrogen Arsenate used for?
A Closer Look at Diammonium Hydrogen Arsenate
Diammonium hydrogen arsenate sounds like one of those chemicals you only encounter in textbooks, but its real-world impact stretches beyond the theoretical. In the lab, chemists often turn to this compound for various analytical applications. Its main draw stems from its arsenic content, which brings its own set of challenges and responsibilities.
Laboratory Applications and Testing
Research labs sometimes call on diammonium hydrogen arsenate when they need a reliable arsenate source. For instance, it's common in calibration processes, especially in environmental labs that monitor arsenic levels in soil or water. Calibration ensures scientific instruments spit out numbers researchers can trust, and accuracy matters most when public health hangs in the balance. When I trained as a lab tech, we set up controls using standards like these to triple-check that drinking water fell under legal arsenic limits.
Techniques like spectroscopy or titration require well-characterized chemicals. Diammonium hydrogen arsenate forms predictable, stable solutions, so scientists get repeatable results. Reliable standards support quality control at drinking water plants and industrial waste sites.
Warnings from Experience and History
Dealing with arsenic-based compounds comes with a weighty obligation. Arsenic toxicity is well documented: long-term exposure can trigger cancer, cardiovascular disease, and developmental issues. At the lab bench, strict procedures rule. Carelessness here doesn’t only threaten safety inside the building—the legacy of poorly handled arsenic stretches back through abandoned mine sites and poisoned groundwater aquifers.
It’s easy to forget how widespread arsenic contamination has been, from 20th-century pesticides to natural deposits leaching up through wells. Spending time in communities struggling with tainted water drives home the point. Standards like diammonium hydrogen arsenate give scientists the tools to track, contain, and, with effort, reverse this legacy—if they use them responsibly.
Industry, Research, and the Changing Landscape
Most outside of analytical chemistry never see diammonium hydrogen arsenate up close. It does have niche uses in chemical research for exploring new treatment methods or in developing reference materials for arsenic speciation. The compound’s role has faded somewhat in industrial settings, as regulations and public awareness around arsenic hazards have tightened.
Green chemistry trends push for safer alternatives and stricter waste protocols. Labs adopt cleaner standards, double down on protective equipment, and update training. Safety data gets shared more transparently, and experienced staff share stories—sometimes cautionary tales—of what happens when corners get cut.
The Bigger Picture: Responsibility and Progress
Diammonium hydrogen arsenate sits squarely in the world of scientific responsibility. Proper use safeguards public health, and misuse spells disaster. This compound reminds us why scientific rigor matters, and why society watches over the shoulders of those who work in chemistry.
To lower the potential for harm, clear training, modern equipment, and ongoing oversight act as basic guardrails. Whether it’s double-bagging waste or logging each use, details form the backbone of safe chemical handling. As technology and awareness grow, so does the potential to keep research effective and risk as low as possible.
Is Diammonium Hydrogen Arsenate toxic or hazardous?
Why This Chemical Matters
Growing up near an industrial plant, I always wondered about the long words stamped on shipping labels and train cars. Diammonium hydrogen arsenate isn’t a familiar term at the family dinner table, but for some communities and workplaces, its risks hang heavy.
What’s Lurking in Diammonium Hydrogen Arsenate?
Diammonium hydrogen arsenate is a salt that includes arsenic, a naturally occurring element known for its toxicity. It can end up in fertilizers, insecticides, wood preservatives, or lab environments. While the chemical itself carries a technical name, it brings a pile of real-life consequences.
Arsenic has a long history of being both useful and deadly. At the molecular level, it slides easily into body systems, sometimes getting past basic safety routines. Exposure—even in tiny amounts—can eventually catch up to you. This isn’t fearmongering. The International Agency for Research on Cancer has labeled many arsenic compounds as carcinogenic. Consuming or inhaling dust from this compound may invite health problems ranging from skin irritation to organ damage, and, over time, cancers of the lung, bladder, skin, and more.
Workplace Exposure Isn’t Just a Hypothetical
No one starts their shift with the hope of getting a toxic dose of arsenic. In reality, folks who handle chemicals like diammonium hydrogen arsenate aren’t just risking an annoying rash. Their health can suffer years down the road, especially if workplaces cut corners or skim over training.
The CDC says that even short-term exposure can cause sore throat, stomach pain, or nausea. Long-term, the story turns darker with kidney problems, nerve disorders, or cancer. I met a floor supervisor who developed nerve issues after a decade around arsenic salts and—back then—no one handed out respirators.
Tackling the Problem Starts With Straight Talk
The solution doesn’t sit in pouring funds into generic “safety initiatives.” Protections must include safe handling protocols, high-quality PPE (not makeshift masks from the break room), and up-to-date training programs. When companies try to dodge regulation or cut budgets, workers pay the price. The truth surfaces in communities near plants too, where runoff and spills poison water or soil.
Keeping exposure low isn’t just good policy. It’s a moral issue. OSHA and international agencies set legal limits for exposure—the number stands at a tiny 0.01 mg/m³ in the United States. But limits do little unless enforced on the job. Clear hazard communication is just as important: every label, sign, and onboarding packet should explain the risks in plain language, so everyone—no matter their experience—gets the message.
Communities Shouldn’t Be Left Guessing
Testing local water and soil for arsenic contamination isn’t something to ignore. Several towns across the United States discovered chronically high arsenic levels in their drinking water—often years after industry set up shop nearby. Parents shouldn’t wonder if the tap water will harm their kids. Regulators, scientists, and local groups all shape the broader response.
Diammonium hydrogen arsenate demands honesty, vigilance, and a sense that real lives count—the lives in labs, in shops, in the neighborhoods next door.
What is the chemical formula of Diammonium Hydrogen Arsenate?
The Chemical Formula
Diammonium hydrogen arsenate uses the formula (NH4)2HAsO4. In day-to-day lab work, anyone handling this compound should recognize these letters and numbers as more than just a label. This formula spells out two ammonium groups balanced with a hydrogen arsenate ion. Understanding the pieces matters because you never want surprises in a chemical storage area or at a workbench.
Health Risks and Real Experience
Workplaces in mining, environmental science, or chemistry labs rarely teach you about these compounds with enough weight. I remember a quieter moment working with a similar arsenic-based substance. My supervisor ran me through the toxic dangers—one waft of dust in the air, and your body feels the damage fast. The same risk lives in diammonium hydrogen arsenate, since arsenic is one of those infamous elements you never want in your bloodstream. Arsenic compounds have long histories in both industry and unwelcome environmental impact. The Centers for Disease Control and Prevention highlights how arsenic exposure leads to skin damage, cancer risks, circulatory issues, and more, which brings a real sense of respect to something as simple as a white powder in a bottle.
Applications in Agriculture and Science
People may run into diammonium hydrogen arsenate in research settings or specialty industries, especially where selective pesticides come into play. Historically, compounds related to this formula propped up as pesticides before stricter health rules stopped their general use. Researchers also turn to it for some chemical syntheses or as a source of arsenic and phosphate in controlled tests. These specialized uses become tricky, because once the chemicals leave the lab, the risk to wildlife, water, and families moves with them. Drinking water tainted with traces of arsenic compounds creates long-term health hazards, which communities around the globe know firsthand.
Safe Handling and Clear Communication
Anyone working with diammonium hydrogen arsenate deserves training that goes beyond memorizing formulas. Safety Data Sheets get read, but nothing beats real walkthroughs on using gloves, proper storage in sealed, labeled containers, and the right ventilation procedures. If chemical hygiene takes a day off, all it takes is a tiny spill for things to go wrong—these arsenic compounds do not offer second chances. Communication plays a big role. I always remind younger lab workers to speak up if they notice anything strange, because silence keeps accidents hidden.
Room for Safer Alternatives
Modern agriculture and laboratory testing look for safer ways forward. A shift away from arsenic-based compounds already takes shape, especially in food production and pest management. There’s a push to phase out old stock and teach anyone handling legacy chemicals about their dangers. Water monitoring technologies are also catching up. Fast, affordable test kits help communities detect arsenic contamination long before it hits the crisis point.
Understanding the formula (NH4)2HAsO4 opens the door to curiosity, but that curiosity does not replace a healthy respect for the risks involved when handling arsenic.How should Diammonium Hydrogen Arsenate be stored and handled safely?
Why Caution Matters Around Diammonium Hydrogen Arsenate
Diammonium hydrogen arsenate shows up on safety data sheets with more hazard symbols than most people want to see. If someone worked in an academic lab, they’d recognize the quick scramble for gloves and goggles when a jar with “arsenate” rolls out. This chemical brings real risks to people and the environment if it gets out, mostly because arsenic compounds hurt the liver, kidneys, and nervous system, and don’t just break down or disappear. So, handling it with slack routines just doesn’t make sense for anyone looking to keep themselves and coworkers safe.
Storage Practices That Actually Work
Years in shared laboratories teach hard lessons. Storing dangerous chemicals under the sink or in a mislabeled plastic tub didn’t work ten years ago, and it’s a recipe for emergency room visits today. Diammonium hydrogen arsenate belongs in a cool, dry, well-ventilated space, kept away from acids and foods. Locked cabinets with a sturdy label and a list of authorized users lower the chance of accidental use. Metal shelving or secure secondary containers stop spillage disasters. Every container stays sealed tight, checked for cracks, and never sits next to incompatible substances. Regular audits keep everyone honest, especially if the group changes often or the storage area doubles for different chemicals.
Protecting People First
Experienced techs share advice early: double up on gloves, put on full-length lab coats, and slap on goggles every single time. No matter how familiar someone is with the substance, a moment of carelessness around this powder means a rush for decontamination. Key facts float up from injury logs—a tiny amount absorbed through skin or inhaled dust can cause acute symptoms after a single exposure. Face masks with true filtration (not the dust blockers from a hardware store) keep fine particles out. Skin contact calls for immediate washing with soap and water, and spills get covered with damp paper towels, not swept or brushed.
The Right Response if Something Goes Wrong
Small spills can feel daunting, but the playbook says stop, contain, and call for trained help. Anyone in the area steps out while the right cleanup gear comes in—think gloves, goggles, and proper disposal waste bags. Colleagues shouldn’t rely on gut feeling; university and workplace policies spell out procedures, and ignoring them often leads to fines or worse. If dust clouds rise or anyone breathes it in, head outside and call for medical review, just in case. Even seasoned chemists avoid the lone-wolf mistake; two sets of eyes protect against oversight under pressure.
Solutions for Safer Workplaces
Many accidents happen from guesswork. That’s why frequent training sessions save more than just company reputation. Eye-catching signs, up-to-date safety data sheets, and regular drills catch mistakes before they turn into emergencies. Labs can put together a buddy system, so no one faces high-risk tasks alone. Sharing stories—near misses and wins—keeps safety at the front of people’s minds, not just as a clipboard checklist.
Keeping Communities Safe
Disposal has its own set of rules. Pouring leftovers down the drain puts local water at risk and stacks up legal trouble faster than most realize. Handing over waste to certified disposal services costs more, but it beats unknown health issues surfacing down the line. People who live near industrial or academic labs pay attention to chemical management for a reason. Trust grows with clear communication—letting neighbors and regulators know the playbook for handling incidents, so no one is left guessing in a crisis.
What are the potential health effects of exposure to Diammonium Hydrogen Arsenate?
Understanding Where the Real Dangers Lie
Diammonium hydrogen arsenate isn’t some new chemical out of a sci-fi movie; it’s been around a long time in labs and industry. Most people won’t run into it while grocery shopping, but it sometimes pops up in agriculture, glass-making, and pest control. The real risk appears because this compound contains arsenic—a word that probably rings alarm bells for anyone with even basic science class memories.
Short-Term and Long-Term Health Effects
Arsenic’s reputation for toxicity isn’t hype. Breathe in even a little dust from diammonium hydrogen arsenate, and the lungs start feeling the burn. Coughing, chest pain, and throat irritation show up quickly. If someone swallows this stuff? That’s worse—nausea, vomiting, stomach pain, and diarrhea hit hard. I’ve never experienced this first-hand, but emergency room stats make it clear: people who’ve ingested arsenic compounds can get really sick, really fast, in a way that often leads to organ failure without quick medical care.
Ongoing contact with even small amounts over weeks or months leaves nastier footprints. Skin might change color or develop rough, wart-like growths—something dermatologists have noted repeatedly from people living near contaminated water. Kidneys and liver quietly build up damage, which may not scream out until the harm is deep. The lungs and heart also take hits over time, though folks may chalk early warning signs up to “getting old” or a persistent cough.
Digging Deeper into Arsenic Poisoning
This isn’t just theory. Decades of research show a strong link between chronic arsenic exposure and several cancers, mostly of the skin, lungs, and bladder. Arsenic works at the cellular level, messing with DNA repair and immune function. Late effects might even show up in the next generation—studies on children in high-arsenic areas point to higher rates of neurological delays and birth defects.
It’s easy to forget about these risks in communities where industries use arsenic-based chemicals for years. I’ve talked with folks living near old smelting sites; some shrug off the risks since “it’s always been here.” But groundwater testing by public health teams often reveals contamination well above safety limits, leading to more frequent checks for cancers and organ disease.
Addressing the Problem and Keeping People Safe
Industry bodies in developed countries do require rigorous labeling and proper ventilation for anyone handling diammonium hydrogen arsenate. Gloves, masks, and well-maintained workspaces cut down on respiratory risks and skin contact. Routine blood and urine tests can catch problems in workers before larger health issues take root. Regular workplace training on handling chemicals has helped in some sectors; stories from older factory workers don’t always match up with these efforts, though, so continuing education matters.
For households that might find historical contamination in water or soil, water filters based on activated alumina or reverse osmosis work, but only if folks know to use them. Community education and regular public health outreach make a real difference—residents need clear information about symptoms, risks, and how to pressure authorities when testing uncovers unsafe levels.
The science on arsenic-based compounds doesn’t paint a pretty picture, but honest talk and practical education go further than scare tactics. Safe industrial practices and public health vigilance keep people out of doctors’ offices and give communities a fighting chance at better health.
| Names | |
| Preferred IUPAC name | Diammonium hydrogen arsenate |
| Other names |
Diammonium arsenate Diammonium hydrogenarsenate Arsenic acid diammonium salt |
| Pronunciation | /daɪˌæmˈoʊniəm ˈhaɪdrədʒən ɑːrˈsiːneɪt/ |
| Identifiers | |
| CAS Number | 7783-28-0 |
| Beilstein Reference | 136140 |
| ChEBI | CHEBI:63051 |
| ChEMBL | CHEMBL1201571 |
| ChemSpider | 22225 |
| DrugBank | DB11451 |
| ECHA InfoCard | 100.028.331 |
| EC Number | 232-067-2 |
| Gmelin Reference | 7847 |
| KEGG | C18734 |
| MeSH | D009032 |
| PubChem CID | 24963 |
| RTECS number | AG8750000 |
| UNII | 10U83L8J0G |
| UN number | UN1608 |
| Properties | |
| Chemical formula | (NH4)2HAsO4 |
| Molar mass | 210.03 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.44 g/cm³ |
| Solubility in water | Highly soluble |
| log P | -2.00 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 2.22 |
| Basicity (pKb) | 11.84 |
| Magnetic susceptibility (χ) | -66.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.525 |
| Dipole moment | 3.32 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 228.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1881.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | No data found |
| Pharmacology | |
| ATC code | V03AB38 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause cancer; causes damage to organs; very toxic to aquatic life |
| GHS labelling | GHS07, GHS08, GHS09, GHS06 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H332: Toxic if swallowed or if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P273-P280-P284-P301+P310-P302+P352-P304+P340-P305+P351+P338-P310 |
| NFPA 704 (fire diamond) | 2-3-0-✕ |
| Lethal dose or concentration | LD50 oral rat 155 mg/kg |
| LD50 (median dose) | LD50: 14 mg/kg (oral, rat) |
| NIOSH | GB5950000 |
| PEL (Permissible) | PEL: TWA 0.01 mg(As)/m³ |
| REL (Recommended) | 600 mg/m3 |
| IDLH (Immediate danger) | 5 mg As/m3 |
| Related compounds | |
| Related compounds |
Ammonium arsenate Ammonium dihydrogen arsenate Monopotassium arsenate Disodium hydrogen arsenate Monosodium dihydrogen arsenate |

