Diallylamine: The Chemical with Many Faces
Tracing Diallylamine Through Its History
Diallylamine didn’t take the same glamorous path as some better-known chemicals. Its story started in early twentieth-century labs, where researchers tinkered with simple amine compounds to drive the expansion of synthetic chemistry. During those decades, diallylamine found its first real purpose as a building block that fit well in the fast-growing plastics and pharmaceuticals sectors. By the postwar years, synthetic techniques improved and opened broader use across specialty chemicals, water treatment agents, and even crop protection. My own work in academic labs exposed me to its adaptability: teachers would point to diallylamine while explaining how small changes in a molecule set off a cascade of new industrial possibilities.
Product Overview: More Than a Simple Amine
Diallylamine holds a very practical appeal. It stands out because its structure brings two reactive allyl groups attached to a single amine nitrogen. This gives it surprising versatility. In the real world, diallylamine gets attention as both a base material for polymers and as a key ingredient in specialty organic syntheses. Far from just another bulk commodity, it acts as a springboard for creating tailor-made compounds found in ion-exchange resins and certain pharmaceutical intermediates. Scientists value how easily it links up with other molecules and how much control they get over its reactivity.
Physical and Chemical Properties: What Makes It Tick
Diallylamine comes across as a colorless to light yellow liquid at room temperature. Its boiling point is relatively low compared to long-chain amines, and it gives off an ammonia-like odor that sticks in memory after a day in the lab. It dissolves reasonably well in most common organic solvents and moderately in water, a trait that lets chemists blend it into process streams with less fuss. The molecule stands out because each nitrogen carries two allyl groups—these carbon chains crank up its reactivity in classic substitution and addition reactions. The amine itself leans toward basicity, making it handy in manipulating acid-base balances during syntheses.
Technical Specs and Real-World Labeling
Labeling isn’t just about slapping a name on a drum. Chemicals like diallylamine show why clear documentation matters. It takes a pure product with a defined assay, low water content, and a guarantee that no unstable byproducts hitch a ride. In my experience, trace impurities, especially peroxide-formers, can cause process headaches or even safety incidents, so quality testing and mindful storage protocols aren’t optional extras. Regulatory frameworks require hazard warnings, flammability markers, and pictograms that workers can see at a glance. There’s a practical side: good operator training reduces risks from accidental exposure or mislabeling.
The Art and Grit of Preparation
Chemists learned to make diallylamine by channeling the right blend of ingenuity and patience. The mainstay method builds it through reaction of acrylonitrile with ammonia, followed by catalytic hydrogenation. At scale, this process draws on well-honed engineering to keep yields up and waste down, with metal catalysts that deliver selectivity. Early attempts led to a patchwork of byproducts, but tweaks over decades allowed cleaner conversion and safer containment. The hydrogenation step brings its own set of safety rules, since working with flammable gases and active metal catalysts doesn’t make for an idle afternoon. At the bench scale, it’s a dozen moving parts that all have to balance—timing, pressure, temperature, and purity of feedstocks.
Chemical Reactions and Creative Modifications
Diallylamine earns its stripes by how easily it gets pulled into larger molecules. Classic examples include polymerization, where it brings flexibility and resistance to finished materials, and nucleophilic substitution, where the amine’s basicity and the double bonds let it participate in a wide array of syntheses. I’ve seen researchers push the boundaries by grafting diallylamine onto resins, making new ion-exchange materials tailored to grab hold of specific metals or toxins. Other labs jump at its ease of quaternization, driving the formation of quaternary ammonium salts used in antimicrobial agents and water treatment chemicals. The allyl groups act like handles—easy to grab and pull the molecule into new shapes.
Synonyms and Naming Conventions
Chemicals like diallylamine get saddled with a laundry list of names that vary by industry and region. Some old-timers refer to it as N-2-propenylamine, emphasizing the dual latch of allyl chains, while others stick to abbreviations like DAA. On any given week, a chemist could run into synonyms such as diprop-2-enylamine or diallyl amine depending on the paperwork. Navigating these names in real time serves as a reminder that communication errors can cause supply chain hiccups or, worse, dangerous mix-ups in the plant.
Staying Safe: Handling and Operational Standards
Anyone working around diallylamine feels the reality of chemical safety. It isn’t something left to chance. The compound carries moderate toxicity, and even short-term skin or eye contact leads to irritation. Inhalation can set off respiratory distress. From my own years at the lab bench, I remember the need to check ventilation, double-seal storage drums, and suit up in gloves and goggles. Fire risks come into play, as diallylamine carries a flash point low enough to invite trouble without proper precautions. Regulatory agencies, from OSHA to EU’s ECHA, step in to hammer home safe limits for exposure, labeling, and disposal. Meeting these standards means building a workplace where everyone gets home in one piece after every shift.
Applications Across Sectors
It surprises some to learn diallylamine doesn’t just stay locked in a lab cabinet. It breaks into daily life through water treatment chemicals, where it helps pull out heavy metals and dyes from waste streams. Crop scientists use diallylamine-derived products in certain herbicide formulations to boost both activity and selectivity. Brush up against the pharmaceutical world, and you’ll find it showing up as a reactive site in the early stages of drug synthesis. On the industrial front, major producers fold it into resins and adhesives, capitalizing on its dual-purpose reactivity and solubility. In the world of specialty polymers, researchers design new materials with diallylamine to chase after stronger, more chemically resistant coatings and membranes.
Innovation and Ongoing Research
Every so often, a paper comes out that pushes diallylamine into fresh ground. Some teams focus on making greener synthesis routes, experimenting with catalysts that avoid toxic byproducts or lower operating temperatures. Others chase after new bio-compatible polymers, spinning diallylamine into medical devices and diagnostic systems. After working with graduate students who investigated novel water purification gels, I’ve seen firsthand how minor tweaks at the molecular level kick off big improvements in trapping pollutants. Across the globe, research groups compare notes on improved storage stability, better quaternization methods, and more efficient hydrogenation steps. Industry watches closely as even small process breakthroughs can slash costs or lift product performance.
Digging Into Toxicity
No one shrugs off chemical safety anymore, and that’s clear in how much energy research teams pour into toxicity testing. Diallylamine shows acute toxicity at higher concentrations; even mild exposure risks skin and respiratory irritation. Researchers have nailed down safe handling levels through repeated animal and cell culture studies, which laid the groundwork for current exposure limits. The ongoing work now drills into chronic effects, seeking to understand any potential bioaccumulation, genetic impacts, or longer-term carcinogenic risks. Proper workplace monitoring—air sampling, health surveillance, and waste audit—translates those findings into day-to-day operations. Each study adds another layer between industry users and potential harm.
Looking Ahead: The Future of Diallylamine
Prospects for diallylamine feel anything but stagnant. Environmental pressures push chemists to revisit old preparation steps and redesign the process for greater safety and less waste. New markets in advanced water purification, medical diagnostics, and functional polymers stand ready to pull in higher-purity or customized forms. My prediction, based on experience with evolving chemical supply chains, is that advances in catalysis and digital process controls can lead to safer, more efficient ways to make and use diallylamine at scale. Add to that a rising demand for versatile, adaptable chemical building blocks, and diallylamine is sure to keep spinning out new value in both the lab and the real world.
What is Diallylamine used for?
What Diallylamine Does in the Real World
Diallylamine often doesn’t get headlines, but anyone who’s spent time around a chemical plant or worked in materials engineering knows how often these smaller molecules drive bigger changes. It’s a clear, colorless liquid that gives off a sharp, amine smell and brings a set of practical skills to the table. Its main draw comes from two things: reactivity and flexibility. With two allyl groups and an amine, it can fit into all sorts of chemical reactions. That puts it on the map for folks making polymers, water treatment chemicals, and agricultural products.
Agriculture: Where Diallylamine Steps Up
Out in the fields, farmers might not realize diallylamine helped make some of their tools or even their fertilizers. Manufacturers use it to create plant growth regulators and certain herbicides. These chemicals need building blocks that react the right way in processing. Diallylamine fits well because it creates compounds that protect seeds or support plant health—helping grow food and keep weeds in check. Decades of use have backed up its value, although it’s important to handle it safely, as improper exposure can irritate skin and lungs.
Industrial Polymers and Resins
Think about the pipes under a street or a coating that resists corrosion—diallylamine probably hides in their origin stories. It’s a basic ingredient for creating special polymers called polyelectrolytes. These help control how water flows and settles in treatment plants, and they improve oil recovery. My uncle worked at a water treatment facility and he always pointed out how important these chemicals are for clearing out all the gunk you don’t want in your drinking water.
Diallylamine also triggers reactions in resin manufacturing. It works well for toughening up coatings and adhesives. Industries use those coatings in packaging, electronics, and even some consumer products. It’s not the star of the show, but everything runs smoother and lasts longer because of small molecules like this one.
Pharmaceuticals and Laboratory Work
Chemists in pharma and biotech sometimes turn to diallylamine when they need to synthesize new molecules in a lab. Its structure gives room for creative chemistry, letting researchers build advanced drugs, diagnostic compounds, or dyes. Experienced chemists know to respect its reactivity, keeping ventilation up and safety equipment close by.
Sticking to Safety and Accountability
Sitting on the safety committee for a small industrial plant, I saw how chemicals like diallylamine require careful handling. Overuse or spills lead to health problems and environmental damage. Regulations set by agencies like the EPA and OSHA step in for a reason. Training, transparent labeling, and new storage systems keep the workplace safer and the supply chain accountable. These protections matter even if the public barely hears the name diallylamine.
Looking Ahead: Smarter Chemistry and New Uses
Some folks in green chemistry circles are pushing for smarter, safer ways to use diallylamine. They want less waste and lower health risks. That means new catalysts, better reaction controls, and possibly even bio-based alternatives. Responsible companies will invest in monitoring, better containers, and, where possible, safer substitutes. As new regulations arrive and sustainability grows more crucial, diallylamine’s role may shift or shrink—but its value right now speaks for itself across several industries.
What are the safety precautions when handling Diallylamine?
Understanding Diallylamine’s Risks in the Workplace
Diallylamine doesn’t show up in household conversations, but facilities and labs using it know how quickly it can cause trouble for anyone who gets careless. Its chemical nature brings more than an unpleasant odor; it attacks skin, respiratory systems, and eyes with surprising intensity. Unlike more familiar solvents, diallylamine doesn’t give much warning before it causes problems. Inhaling even small amounts burns the nose and throat and leaves behind a dry cough that lingers. People who work with it say it feels like it sticks to their clothes, finding its way to exposed wrists or necks if they don’t layer on the right PPE.
PPE: Gloves, Goggles, and More Than Wishful Thinking
Wearing nitrile gloves, snug-fitting goggles, and a decent lab coat can feel like overkill some days. The trouble starts when folks convince themselves that splashing happens to someone else. Diallylamine soaks through thin latex and weakens those single-use gloves people grab out of habit. With this chemical, gloves act as shields, but not all shields hold up. Nitrile gives a solid line of defense, and thicker styles help limit exposure during weighing or pouring. Once splashed, skin absorbs diallylamine faster than you realize.
Safety goggles don’t just protect against direct contact; they stop vapors from sneaking up. Eye-wash stations and emergency showers need to stay clear and ready, and nobody who’s seen what diallylamine can do skips testing one now and then. When some workers stopped checking the water pressure on those stations, one unlucky spill meant a trip to the hospital. It serves as a reminder: take the 30 seconds to make sure emergency equipment actually works.
Ventilation is Not Optional
Trying to cut corners on ventilation ends badly with diallylamine. It doesn’t take much vapor for symptoms to show up—and fume hoods draw the vapors away before they drift into the room. People sometimes crack a window and hope for the best, but that approach invites health problems, especially in small labs or rooms without airflow. Using a professionally installed hood makes a difference. Daily fume hood checks and regular filter replacements prevent high exposure, especially if the hood sees frequent use.
Spill Plans That Actually Work
Spills tend to happen at the worst possible times. Having an actual plan in place helps people react instead of freeze up. Absorbent pads, neutralizing agents, and clear walkways matter more than paperwork when the clock starts ticking. One worker recalls knocking over a beaker before lunch—having a padded bench mat stopped diallylamine from spreading to the floor. It turned a nerve-wracking cleanup into something manageable. Keeping the spill kit nearby and restocked keeps panic out of the equation.
Training That Sticks
Reading an instruction manual rarely sticks the way live practice does. Giving staff the chance to run through emergency drills pays off. Even seasoned techs benefit from refreshing their memory about what to do for skin or eye exposure. Bringing real examples—burn stories, evacuation missteps, bits of personal history—does more to drive home the importance of each safety step than a dozen warning labels ever could.
Why Diallylamine Safety Isn’t Just Red Tape
Ignoring the risks tied to diallylamine invites disaster. In this line of work, the small stuff—like changing gloves before and after handling, checking the hood's draw, keeping exits clear—adds up. Professional pride means trusting protocols and respecting firsthand stories, not just ticking through a checklist. For those who see colleagues go home healthy after decades on the job, these precautions never feel optional—they simply feel like the right way to do things.
What is the chemical formula of Diallylamine?
Understanding Diallylamine: More Than Just a Formula
Diallylamine shows up on the radar of folks in chemistry, industry, and environmental circles. Its chemical formula, C6H11N, carries some weight. You’ll find two allyl groups and one amine group behind that name, which ties right into what gives this compound its character and punch. If you’ve spent time in a teaching lab or a manufacturing plant, you spot straight away that simple formulas don’t just tell you what’s inside—they hint at how substances act, how safe they are, and how much trouble they can cause if things go wrong.
Why Chemical Formulas Matter
I remember as a young technician, getting that “aha” moment: the structure behind the letters meant everything. C6H11N—six carbons, eleven hydrogens, one nitrogen. It tells you more than a name on a label. The nitrogen sitting with the two allyl groups means this amine won’t behave like basic ammonia or aniline. Diallylamine brings unique reactivity, lending itself to making polymers, drugs, and specialized chemicals.
Industry uses C6H11N for water treatment, pesticide manufacturing, and in crafting ion-exchange resins. That little tweak in structure turns a simple amine into something with practical bite. If you’ve ever thought nothing fascinating goes on in a bag of fertilizer, think again: diallylamine often plays a backstage part.
Risks and Responsibility
Not every laboratory chemical pushes the same boundaries on safety. Diallylamine gives off a strong fishy odor—a clear warning. Its low boiling point and high flammability raise the stakes for storage and handling. Mishaps won’t just set off the smoke alarm: concentrated vapors can irritate the lungs or skin.
Some years back, I watched a co-worker learn the hard way. A careless splash led to an emergency rinse and some tough questions from a supervisor. That morning made chemical literacy real for everyone. It’s tempting to treat every colorless liquid the same, but the formula C6H11N flashes a reminder: each arrangement of atoms means unique risks.
Pathways Toward Safer Use
Real-world experience points to a problem with how diallylamine is sometimes handled on shop floors or in small labs. Many mishaps crop up because the person holding the bottle never saw past the chemical code on the label. Standard chemical training covers the basics, but it helps to dig deeper—showing real consequences and talking through real events.
Better labeling, stronger training, and easy-to-read data sheets beef up safety. Chemical educators can invite students to decode formulas, not just memorize them, so folks recognize the stakes behind a label or a spill. Learning the formula of diallylamine isn’t rote homework—it’s a reminder that real chemistry relies on respect, attention, and teamwork.
Looking Ahead
C6H11N won’t make daily headlines, but it’s part of a bigger story about how compounds shape our daily lives. Diallylamine bridges the gap between textbook chemistry and real-world hazard. Recognizing that short formula on the side of a tank gives everyone a head start—fewer accidents, cleaner processes, and maybe a few more “aha” moments for the next generation of hands-on chemists.
How should Diallylamine be stored?
Understanding Diallylamine’s Risks
Diallylamine brings both opportunity and risk to any workspace. This colorless liquid, recognized by its fishy, ammonia-like smell, plays a part in chemical synthesis for agriculture, pharmaceuticals, and polymers. But with benefits come hazards. Diallylamine burns easily and forms toxic fumes. Exposure may turn into headaches, respiratory trouble, or worse. Back in college, a friend referred to diallylamine as “liquid risk,” and that always stuck with me. One knock or spill, and the whole lab scrambled. That anxious energy points to a lesson: chemicals aren’t just stored—they’re respected.
Real-World Steps for Safer Storage
Keeping diallylamine from causing harm starts with a simple plan: controlled access, cool spaces, protective containers, and constant vigilance. Fire risk tops the list, so find a storage spot far from open flames, sparks, or electrical switches. Think about how quickly vapors can ignite, then imagine losing control. Don’t place it near oxidizers or acids, which can create an even bigger problem.
Pick containers tough enough to handle leaks or corrosion. Stainless steel and certain plastics do the job better than glass, which gets brittle over time. On a particularly humid day, I've seen a glass bottle sweat and slip just enough to shatter. Using well-sealed drums or cans with tight-fitting lids offers peace of mind and protection.
The best temperature for diallylamine hovers below 25°C (about 77°F). Hot warehouses or poorly ventilated closets turn a safe setup into a hazard. A dry, well-ventilated chemical cabinet stands above leaving it on a general storage shelf. Strong airflow keeps fumes from building up, and it matters more than many realize. In my experience, opening a door without proper airflow invites a wall of fumes—not just a whiff. That stings, lingers, and can stick to your clothes.
Safety With People in Mind
Labels with hazard warnings and handling instructions deserve attention. Faded ink or handwritten scribbles won’t cut it. In a pinch, clear information saves lives. Safety Data Sheets (SDS) should stay nearby, easy to spot, since emergencies never give warnings. Spills, leaks, or fires require quick reflexes, not frantic searches. Providing spill kits with inert absorbents close to where diallylamine is stored gives workers a fighting chance if something goes wrong.
PPE—goggles, gloves, aprons—belongs near storage, not tucked far away. It’s easy to cut corners in the real world. When time gets tight, someone might reach for diallylamine bare-handed. Sharing stories about near-misses can drive home why those extra steps matter. A forgotten glove once led to chemical burns for someone I know. The pain stayed with him much longer than the lesson.
What Helps Most
Training transforms attitude more than any written checklist. Supervisors can walk staff through every step, not just hand out manuals. Drills test readiness for leaks or fires. Routine inspections catch small problems—cracked lids, shifting containers, clogged vents—before they turn into disasters.
Some workplaces use digital logs to track who takes or returns containers. That kind of recordkeeping builds accountability. Everyone knows where the chemical stands, who’s handled it, and how much remains. Leftover diallylamine requires responsible disposal, following all local rules. Dumping it down a drain or tossing containers in the trash doesn’t just break laws—it risks groundwater and the people who live nearby.
Treating diallylamine storage as a living, changing process protects workers, property, and the environment. Every decision adds up to a culture of safety—one with room for learning, improvement, and trust.
What are the health hazards associated with Diallylamine exposure?
Understanding What Diallylamine Brings Into the Workplace
Diallylamine shows up in facilities where folks make chemicals for rubber products, dyes, and water treatment solutions. It’s colorless, with an unmistakable fishy odor that grabs attention in labs and factories. Folks who work with diallylamine rarely have much choice in their exposure, and few realize just how quick its effects hit. Eyes burn, nose stings, and in a short time, a cough can shake loose from even the toughest worker.
Breathing Trouble and Skin Risks
Standing in the same room with diallylamine, breathing in its vapors, often sets off headaches and dizziness. It doesn’t take a neglected mask or missing exhaust fan for problems to start. Overexposure sometimes brings on nausea, and long shifts pull workers close to the edge where shallow breathing or chest tightness turn into something doctors can’t wave away. Years of studies point out that repeated exposure turns a mild irritant into a real threat for chronic bronchitis or asthma. Allergic reactions show up on skin as red, itchy rashes and swelling—sometimes bad enough to send people to the clinic. Even a few drops splashing on the skin, if not washed off right away, can peel or blister flesh. Chemical burns take their time to heal, leaving behind marks and sometimes lasting pain.
Pain Points for Eyes and Long-Term Exposure
Diallylamine stings the eyes with sharp efficiency. Rubbing them after accidental contact can make things worse, forcing a rush for the eyewash station. Blurred vision may come on fast, and untreated exposure can push someone toward serious eye damage or even permanent vision problems. I’ve seen folks miss warning signs and struggle with vision for weeks. That kind of injury isn’t something a pair of safety glasses fixes after the fact.
Inside the Body: Liver and Kidneys at Risk
While lungs take the first hit, organs like the liver and kidneys don’t escape untouched. Data has shown persistent chemical exposure sometimes throws off liver enzymes, causing fatigue and discomfort during what feels like a routine workday. Over time, these invisible injuries add up, leading to missed days, mounting medical costs, and stress at home no boss or colleague sees. The kidneys, responsible for clearing toxins, take a heavy load with diallylamine. Researchers in industrial medicine highlight how overexposure can set people up for years of health complications, far beyond the walls of the plant.
What Can Be Done—Practical Steps for Health and Safety
Labels flag diallylamine as a clear risk, but labels don’t catch spills or turn on fans. Protecting workers starts with simple habits—keeping gloves in good shape, replacing cartridge respirators before they fail, and making sure exhaust hoods pull air the right way. Training sessions led by experienced hands—folks who remember their own close calls—carry more weight than a warning on a faded wall poster. Proper storage in sealed containers, with spill kits close by, prevents most emergencies. Any person who works with diallylamine deserves access to regular medical check-ups focused on lungs, skin, and organ function. Fast action on early symptoms stops small exposures from growing into major health issues.
Living with the reality of diallylamine means facing risks head on, learning from those who have handled the stuff for decades, and demanding a safe work environment where every worker leaves as healthy as they walked in. Safety isn’t just about rules—it’s about people, their lives, and the respect owed to anyone doing difficult work.
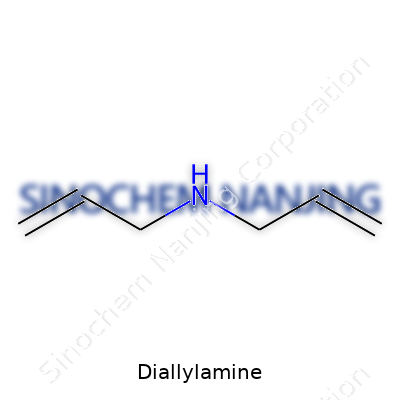
| Names | |
| Preferred IUPAC name | N-prop-2-enylprop-2-en-1-amine |
| Other names |
Di-2-propenylamine N,N-Diallylamine N-2-Propenyl-2-propen-1-amine Diallylamin Diallylamino |
| Pronunciation | /daɪˈælɪl.əˌmiːn/ |
| Identifiers | |
| CAS Number | 124-02-7 |
| Beilstein Reference | 606054 |
| ChEBI | CHEBI:51691 |
| ChEMBL | CHEMBL14246 |
| ChemSpider | 55836 |
| DrugBank | DB14174 |
| ECHA InfoCard | 03b6e24d-cd18-423d-ad51-72b81be178fc |
| EC Number | 203-815-1 |
| Gmelin Reference | 8227 |
| KEGG | C02573 |
| MeSH | Diallylamine.mesh |
| PubChem CID | 8007 |
| RTECS number | KL5600000 |
| UNII | 2POT1JLV19 |
| UN number | UN2215 |
| Properties | |
| Chemical formula | C6H11N |
| Molar mass | 85.15 g/mol |
| Appearance | Colorless to yellow liquid with an amine-like odor |
| Odor | Ammonia-like |
| Density | 0.789 g/mL at 25 °C |
| Solubility in water | Miscible |
| log P | 0.97 |
| Vapor pressure | 3.5 kPa (at 20 °C) |
| Acidity (pKa) | 10.0 |
| Basicity (pKb) | 2.84 |
| Magnetic susceptibility (χ) | -12.1×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.427 |
| Viscosity | 0.8 cP (20°C) |
| Dipole moment | 1.35 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 282.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -36.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4221 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D04AX |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05,GHS07 |
| Signal word | Danger |
| Precautionary statements | P210, P261, P280, P303+P361+P353, P304+P340, P305+P351+P338, P311, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | 49 °F |
| Autoignition temperature | 215 °C |
| Explosive limits | 1.8% - 10.1% |
| Lethal dose or concentration | LD50 oral rat 163 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 163 mg/kg |
| NIOSH | RX8575000 |
| PEL (Permissible) | Diallylamine: OSHA PEL 10 ppm (30 mg/m3) TWA |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Allylamine Methyldiallylamine Triallylamine |

