Diacetone Glucose: A Closer Look at an Understated Chemical
Historical Development
Diacetone glucose didn’t show up on the chemist’s radar by accident. As far back as the early 20th century, organic chemists played with glucose derivatives aiming to improve their utility in both industrial and pharmaceutical settings. Back then, cane sugar seemed to rule the world, but it took some hard work and luck for researchers to expose some hidden gems among its relatives. Diacetone glucose emerged as one of those oddballs—something different made by protecting certain positions on the glucose molecule, changing its nature and sparking new possibilities for use. Laboratories in France and Germany led the charge before the compound jumped to the other side of the Atlantic. By the 1940s, chemists recognized diacetone glucose as more than a curiosity. It had real value, opening up opportunities for further chemical tweaks and downstream applications. From academic journals to industrial synthesis, this sugar-based chemical steadily earned its stripes.
Product Overview
Diacetone glucose belongs to a family of specialty chemicals known as protected sugars. The basic idea: tweak a glucose molecule so some functional groups get masked, making it both stable and flexible for chemical synthesis. That shift in structure can open doors that plain old glucose doesn’t. It shows up as a white crystalline powder, not clumpy or sticky, and it blends easily with other powders. Manufacturing facilities package it in moisture-resistant drums, keeping it away from the kinds of humidity that cause caking or clumping. The compound brings a steady hand to chemical reactions, holding up under conditions that might degrade less sturdy sugars.
Physical & Chemical Properties
In practice, chemists trust diacetone glucose for its predictable physical and chemical profile. The powder flows easily—no wrangling clumps or fussing with sticky residues. Its melting point sits in the moderate range, not so low as to pose handling issues, not high enough to require special equipment. Its solubility plays a big role too: water brings it into solution fast, and it won’t crash out unexpectedly during a reaction. As for the molecule itself, the diacetone groups on the C1 and C2 positions lock down the reactive ends of glucose, shielding them from casual attack. That means better stability and more control during chemical elaborations. Its refractive index and specific rotation give quality control labs reliable numbers for identity and purity checks. Color and odor stick close to “clean”—another bonus for food and pharma manufacturing.
Technical Specifications & Labeling
Reputable producers establish clear specifications. Labs rely on details like assay (purity, usually 98% or higher), moisture content (under 0.5%), melting point (160-165°C), and heavy metal limits—because nobody wants to risk patient safety with trace contaminants. Labels on sacks and drums reflect more than just chemical names: they carry batch numbers, hazard designations, storage advice, and date of manufacture. Some regulations require harmonized system (HS) codes and emergency contact info, especially for cross-border shipments.
Preparation Method
Chemists don’t dig diacetone glucose out of the ground—they build it. The common way begins with plain glucose. Reacting it with acetone, often in the presence of an acid catalyst like sulfuric acid, selectively protects the hydroxyl groups to give the diacetone derivative. The challenge: hit the right temperature and acidity to avoid unwanted byproducts, keeping yields high and contaminants low. After reaction, purification steps—crystallization, filtration, and drying—bring the compound up to standard. This well-behaved, crystalline solid can then move to warehouse shelves or be sent straight into further chemical conversions.
Chemical Reactions & Modifications
The main attraction with diacetone glucose sits in its versatility. Those locked-down sites—the diacetone groups—keep part of the molecule protected, leaving the other positions open for selective reactions. In synthetic chemistry, this kind of control means all sorts of functional group installations become possible without side-reactions ruining the party. Chemists use diacetone glucose as a starting material for glycosylation, grafting on new functional groups one at a time. The protected molecule handles strong acids and bases more easily than raw glucose, standing up to aggressive conditions that otherwise break sugar chemistry wide open. When the reaction reaches its end, a simple acid hydrolysis step removes the protecting groups, leaving modified glucose derivatives ready for new duties.
Synonyms & Product Names
Diacetone glucose carries alternate names across textbooks and supplier lists. Some chemists call it 1,2:5,6-Di-O-isopropylidene-α-D-glucofuranose. Others shorten it to DAG or use trade names registered by major chemical houses. These aliases pop up in patents or process documentation, creating a small amount of confusion for newcomers.
Safety & Operational Standards
Worker safety comes first with all powdered chemicals. Diacetone glucose doesn’t tend to trigger violent reactions, but it still needs respectful handling. Dust collection, good ventilation, gloves, and eye shields all show up in plant safety protocols. Accidentally eating or breathing in the dust isn’t advised, since the long-term effects haven’t been completely studied. Storage means cool, dry, and away from incompatible substances (strong oxidizers ruin the compound and cause safety headaches). Modern plants follow standards like ISO, GHS labeling for hazards, and regular air monitoring, ensuring both product purity and worker health are maintained.
Application Area
Chemists and formulators value diacetone glucose across plenty of industries. In pharmaceuticals, it serves as both starting material and intermediate, supporting the manufacture of complex sugars, antibiotics, and fine specialty chemicals. Food scientists leverage its properties for research into carbohydrate metabolism and the synthesis of artificial sweeteners or sugar substitutes. The cosmetic industry uses related protected sugars in formulations where stability counts—a touch of diacetone glucose can help keep some active ingredients from breaking down too soon. Technologists in polymer and adhesive development also reach for this compound when working on water-soluble polymers and glue systems. In the research lab, it’s a workhorse for testing new reaction pathways, thanks to its firm but flexible scaffold.
Research & Development
Universities and industrial labs haven’t run out of ideas for diacetone glucose derivatives. Ongoing studies look at analogues for new drug candidates, enzyme inhibitors, and specialty additives. With every tweak—changing the protecting groups or targeting new reaction sites—chemists try to coax new bioactivity or improved reactivity out of the molecule. Some efforts push toward better food additives with improved texture or shelf life. Others seek new catalysts for organic synthesis, leveraging the stability of the diacetone glucose skeleton. Research into renewable feedstocks and green chemistry has brought interest in sugar-based molecules, and diacetone glucose fits the bill as a building block from non-petroleum sources.
Toxicity Research
Toxicologists keep diacetone glucose on their radar. Animal studies and cellular screening point to low acute toxicity, especially compared to solvents or heavy metal additives. Risk assessments focus on both worker exposure and end-user safety, mainly in food and pharma applications. Unlike some sugar derivatives that trigger weird metabolic effects, diacetone glucose aligns closely with the safety profile of other protected sugars, but long-term studies continue to confirm or refine guidance. Environmental fate matters too—the compound breaks down by hydrolysis and microbial action, though persistence in water and soil can vary depending on local conditions and concentrations.
Future Prospects
Diacetone glucose isn’t stuck in a static market. Demand tracks upward in line with research on renewable chemicals, biodegradable polymers, and non-traditional sweeteners. New patent filings suggest a healthy appetite for improved methods of synthesis, greener catalysts, and innovative uses in pharma and biotechnology. Some researchers speculate about potential antiviral properties, while others chase after advanced composite materials. As sugar chemistry shifts from commodity to specialty status, diacetone glucose stands out as a molecule with staying power amid a fast-changing landscape.
What is Diacetone Glucose used for?
Everyday Uses Behind the Lab Name
Diacetone glucose doesn’t sound like something you’d come across in daily life, but, truth is, it shapes a lot of products we often take for granted. Chemists know it as a modified sugar, tweaked to work as a solvent and stabilizer across several industries. I once saw it listed in an ingredient catalogue for specialty paints and wondered why a sugar would end up in coatings at all. Turns out, companies turn to it for specific reasons that highlight the close connection between chemistry and the stuff we use at home or work.
How Companies Rely on Diacetone Glucose
You’ll find diacetone glucose on the ingredient list for certain paints and coatings. Manufacturers choose it not just to thin out paints, but also to help pigments mix evenly and stay suspended longer. That means no odd clumping in the paint can, no streaks when you roll it on the wall, and a finish that actually looks the way it should after drying. This has a ripple effect: contractors can finish jobs faster without goopy leftovers, and homeowners end up with neater-looking walls.
I’ve spoken with a few staffers at industrial cleaning supply shops, and they pointed out another use. Diacetone glucose gets added to some cleaning solutions, making tricky stains easier to dissolve. It doesn’t react aggressively like some traditional solvents, so workers cleaning machinery or lab benches avoid dealing with harsh fumes. That’s a tangible health benefit for folks putting in long hours in production plants.
Hidden Helper in Printing and Adhesives
In commercial printing, print techs trust diacetone glucose to keep inks consistent. Printers don’t want ink smears or sticky build-up on their rollers. A small measure of this chemical helps ensure prints come out crisp batch after batch. In adhesives, the story’s similar. Today’s glues, especially specialty adhesives, use diacetone glucose to strike a balance between stickiness and spreadability. This keeps packaging intact during hot summers and cold shipment routes.
Safety and Trust: What Matters Most
I’ve met parents worried about new chemicals in consumer goods. They don’t want unknown risks. It matters to check that substances like diacetone glucose meet health and environmental standards. Companies that comply with global regulations—REACH in Europe, EPA guidelines in the U.S.—show that these additives have a recognized safety profile. The studies available point out low toxicity for both people and most forms of wildlife at the amounts used in products.
Alternatives and a Greener Tomorrow
Some innovators look for plant-based or biodegradable replacements. Behaving responsibly as a manufacturer doesn’t stop at just choosing “safer” chemicals; it involves tracking end-of-life impact as well. I’ve seen university projects focus on developing bio-alternatives to solvents like diacetone glucose, aiming to cut down on persistent waste without sacrificing performance. If companies keep investing in this kind of progress, everyone—from factory workers to consumers—stands to benefit, and our landfills and waterways stay a bit cleaner.
A Practical Ingredient with Big Impact
Whether you’re painting a room, running an offset press, or keeping a hospital lab spotless, diacetone glucose plays its part behind the scenes. It’s a story of chemistry working for people, not just in factories but in everyday tasks—sometimes hidden in plain sight but always making things a little easier, a little safer, and a bit more reliable for all of us.
Is Diacetone Glucose safe for human consumption?
What Exactly Is Diacetone Glucose?
Diacetone glucose doesn’t ring the same bell as ascorbic acid or citric acid. It belongs to the family of compounds formed by modifying glucose. Food chemists sometimes use such derivatives to help with texture, stability, or preservation. Seeing an unfamiliar ingredient on a label can cause concern, especially if its name sounds more like something from a lab than a kitchen. I’ve learned people react strongly to chemical-sounding names, often expecting the worst before digging a little deeper.
Is It Poisonous, Or Not?
Most of us don’t have the time to scan scientific journals every time new ingredients pop up in our food. A name like diacetone glucose can kick up doubts, yet the safety of an ingredient boils down to two things: how our bodies process it and the evidence around its use. Diacetone glucose hasn’t been flagged by agencies like the FDA or EFSA as dangerous or toxic. It doesn’t break down into anything known to be hazardous. The structure stays close to that of glucose, which the body recognizes and handles. That’s not a sweeping green light, but it gives some reassurance for those concerned about surprises after eating processed foods.
How Much Is Too Much?
It’s easy to forget that even kitchen staples can cause trouble in wild quantities—sugar, salt, water. With unfamiliar ingredients, the question isn’t just what it is, but how much gets used. Food manufacturers add diacetone glucose in tiny amounts, usually as part of a blend to fine-tune consistency or extend shelf life. Long-term studies on enormous doses don’t exist, likely because no one expects people to eat tablespoons of the stuff. The lack of high-profile scandals or recalls over its use tells a story in itself; if there were problems, they’d be splashed across headlines.
Why Safety Conversations Matter
Mistrust grows quickly when food labels read like chemistry textbooks. It’s become clear to me that transparency is the missing piece. Many folks in my circle, especially those who shop with allergies or children in mind, want companies to share more information in plain language. A culture of secrecy or tech jargon tends to backfire. If someone sees “diacetone glucose” on a snack label, they should find real answers from official sources—not just the opinion mill online. The more companies and agencies invest in outreach, the safer and more confident people feel about their choices.
What To Look Out For
Not all chemical relatives act alike. Some are harmless, others not so much. Just as regular sugar and artificial sweeteners have wildly different effects, glucose derivatives can land all over the safety spectrum. The fact that diacetone glucose escapes the usual red-flag lists doesn't mean it's above scrutiny. Every new ingredient deserves routine checks and clear, up-to-date reviews by organizations with nothing to gain from sales numbers. Keeping the public looped in with readable, honest safety summaries wouldn't just put minds at ease, it would cut down on confusion and distrust.
Practical Advice
If you’ve got stomach issues, food allergies, or shop for kids, treating unfamiliar ingredients with a healthy curiosity beats either panic or blind trust. Look up credible sources, check for agency approval, and watch for new research. Stay skeptical of wild claims on either extreme. If something ever causes strange symptoms, take the packaging and your questions straight to a healthcare provider. Most of the time, ingredients like diacetone glucose remain behind-the-scenes helpers, hardly noticed by the average eater. That said, everyone deserves to know exactly what’s in their meal.
What are the main applications of Diacetone Glucose?
Cleaning, Coating, and Color: The Clear Uses
Diacetone glucose sounds like something you might find in a chemistry class, but in reality, it finds its way into products most of us use without ever noticing the label. My work with small manufacturers in the paints and coatings world helped me spot how often this ingredient shows up when people need better performance from their products.
In paints and varnishes, diacetone glucose acts as a coalescent agent, meaning it helps the particles in a mixture come together smoothly. I’ve seen workshops swear by this chemical for water-based paints that need smoothness and longevity without the harsh smell of strong solvents. Since environmental rules keep pushing for “greener” chemicals, this is a solid swap from traditional, harsher additives. The science checks out — diacetone glucose leaves a lower VOC (volatile organic compound) footprint than many older options, which means less harm to people and the planet.
Adding Shine and Lasting Power to Inks
Printing inks lean heavily on chemicals that prevent clumping and streaks. Diacetone glucose plays a supporting role here by acting as a modifier that balances drying time and lets color spread evenly across paper or packaging. I worked with a small print shop in my city that moved to water-based inks and saw their waste go down. By using diacetone glucose, their prints came out crisper and they managed to curb some of the strong smells that made working conditions tough.
Helping Cosmetics Do Their Job
Personal care companies search high and low for hydrating yet stable chemical blends for foundations, eye shadows, and creams. Diacetone glucose pops up in ingredient lists for its ability to boost shelf life and help pigments dissolve better. Some major brands began using it as part of the shift away from controversial preservatives and solvents. Dermatological studies show its mildness — a win for anyone with sensitive skin. My own experience talking with formulators in the beauty sector is that they keep an eye on any ingredient that solves multiple challenges at once. Here, diacetone glucose has proved reliable.
Industrial and Household Applications
Outside of flashy consumer products, you’ll find diacetone glucose behind the scenes in cleaning products and adhesives. Detergent makers value it for solvency — it breaks up greasy stains without much fuss. I remember testing out sample cleaning products in a lab that included this ingredient; results showed it cut through oily residue faster than traditional formulas that depended on heavier, less environmentally-friendly chemicals.
In adhesives, diacetone glucose works to improve flexibility and consistency, especially in products aimed at craftwork or packaging. The glue held better on odd surfaces after tweaks with this additive. Here, the switch gave a small packaging firm the edge it needed to beat out a bigger competitor.
Challenges and Solutions
Despite these benefits, some manufacturers face cost barriers. Diacetone glucose isn’t always the cheapest option on the market. I’ve watched purchasing managers wrestle with budgets, knowing they could get safer, higher-quality results, but needing to justify a higher price tag. Companies could consider pooled purchasing with industry peers or strike deals with chemical suppliers for longer-term contracts. This strategy isn’t new, but it keeps safer chemicals in the mix even for small-scale users.
As regulations on chemical safety keep evolving, tracking the origin and purity of additives like diacetone glucose stands out as crucial. Regular lab testing — and full ingredient transparency — helps keep trust up, especially for personal care and food-related applications.
How is Diacetone Glucose different from regular glucose?
Understanding the Real Difference
At first glance, diacetone glucose might sound like a fancy twist on the simple glucose most of us have heard about since grade school. In truth, these two substances play entirely different roles in science and industry. Regular glucose shows up everywhere: blood tests, breakfast foods, even the ingredients list on a box of energy bars. It’s the sugar our bodies run on—one molecule and nothing added.
Diacetone glucose comes from chemical tweaks to plain glucose. Chemists transform it by attaching certain groups—acetone groups, not what you’d find in the nail polish remover, but something similar in structure. These changes completely flip its personality. The new structure no longer acts as a powerhouse for living things. You won’t see it fueling cells or giving a quick energy bump to athletes. Instead, it pops up in specialty manufacturing and laboratory settings, and it stops acting like food altogether.
Why It Matters: Chemistry in Everyday Life
This difference looks small on paper, but it tells a big story. Chemistry shapes stuff we use daily in ways we hardly notice. Diacetone glucose, for instance, gets a seat at the table not because of its sweetness, but because of how it interacts with other chemicals. It serves as a solvent in some paints and coatings, helping dissolve substances that plain water or alcohols struggle with. Its stability under different conditions stands out: diacetone glucose resists breaking down, which makes it suitable for uses that regular glucose could never handle.
I once worked in a small business refinishing furniture. Our supplier swapped a finishing solvent to a formula containing diacetone glucose. The crew noticed the paint dried better and gave a smoother appearance. The trick hadn’t come from a new machine or luck—it stemmed from a lab inventing a compound off a simple sugar. This kind of real-world outcome sticks with you, especially if you’re used to seeing chemistry as just something for scientists in lab coats.
Safety and Health Considerations
People hear “glucose” and sometimes assume safety follows. This isn’t true for diacetone glucose. While glucose is safe in food, diacetone glucose gets handled with gloves and goggles. Swallowing it wouldn’t end with a sugar rush. Safety data sheets flag it as an irritant. This point matters for consumers and workers alike—a name that sounds familiar can lead to careless handling.
European regulators and agencies like OSHA in the US keep tabs on chemicals for a reason. The rise of synthetic compounds—some based on innocuous building blocks—makes regulation essential. As industries continue to move towards safer, more sustainable materials, tracking the health effects of compounds like diacetone glucose needs ongoing attention, not just in factories but in downstream products, too.
Moving Forward: Better Communication and Alternatives
Making sense of names in chemistry remains tricky for non-specialists. Bringing more transparency to labeling—especially in paints, coatings, and lab supplies—helps workers recognize the chemicals they handle. Schools and training programs benefit from teaching the distinction between a sugar you eat and one you shouldn’t touch without protection. For manufacturers, there’s room for greener alternatives. Some research groups explore bio-based solvents with lower toxicity, hoping to swap out compounds like diacetone glucose with options that blend safer handling and efficiency.
The right knowledge and clear information keep both workers and end-users safe. In a world built on chemistry, this is a lesson worth repeating.
Where can I purchase Diacetone Glucose?
Why the Search for Diacetone Glucose Matters
Searching for a place to buy diacetone glucose sends a signal about the bigger world of specialty chemicals. Handmade perfumers, academic labs, and research teams know how tough it can feel to source trusted ingredients. Any time you deal with rare or not-so-household chemicals, safety and trust beat everything. I’ve seen academics get stuck for weeks because regulatory hurdles slow down deliveries, or worse, because suppliers cut corners with purity.
There’s not exactly a grocery aisle for diacetone glucose. Its uses pop up in chemical synthesis, analytical chemistry, and select manufacturing processes. If you’re on the hunt for pure, research-grade material, it’s easy to fall down an online rabbit hole packed with confusing claims and questionable vendors.
Spotting Reliable Sources
The most dependable way involves big-name chemical suppliers. Companies like Sigma-Aldrich, Fisher Scientific, and TCI America dominate this world for good reason. They publish full certificates of analysis for every batch. Their product descriptions give you breakdowns by lot, covering potential contaminants and handling instructions. You need to register as a business or an accredited lab, not just a curious individual. Beyond verifying your credentials, these suppliers issue invoices, tracking, and receipts for compliance purposes.
Buying from laboratory suppliers means you get exactly what’s on the label. Every interaction involves paperwork that matters for audits and ensures you’re not skirting regulations. Shipping will ask for a physical address and might prompt additional paperwork on your end. If you’re not part of a registered institution or legitimate business, expect refusals. It’s not about gatekeeping – it’s how the chemical industry tackles public safety and liability head-on.
Red Flags on the Internet
You’ll run across global business-to-business sites – Alibaba comes to mind fast. They promise bulk orders with little oversight, but risk piles up. Consistency, purity, and authenticity don’t always make the cut. Some sellers fudge their paperwork. Orders could vanish into thin air, or worse, arrive with substances you can’t verify. Local import regulations might flag your delivery, inviting investigation or penalties.
Smaller online shops sometimes list rare chemicals, but “too good to be true” often means just that. Without a track record, third-party tests, or clear contact info, the risk outweighs the reward. Years ago, I watched a colleague land in a mess of customs nightmares because a cheap batch turned out mislabeled. Recovering sample money isn’t worth the administrative headache or hazard to anyone working with unknown substances.
Getting It Right and Staying Safe
Anyone needing diacetone glucose for sanctioned lab work, product development, or education should start conversations directly with established suppliers. If you’re in a university or industrial setting, reach out to your purchasing or procurement department. Long-term reliability always beats temporary savings or shortcuts.
Depending on your country, regulations may ask for permits, documentation about intended use, or proof you know the risks involved. Check these boxes before placing an order. Keep every document – from quotes to invoices to batch certificates – both for traceability and for health and safety audits.
If you hit dead ends sourcing from traditional vendors, contact local chemical distributors who might have broader access through their existing networks. Professional associations or trade groups sometimes keep private directories of reputable suppliers open to vetted members.
Trust, Documentation, and Best Practices
In specialty chemicals, nobody wins by cutting corners. Trust your supplier’s transparency. Insist on documentation, and don’t be shy about pestering for details. Chemists, researchers, and responsible makers know: every safe, legal, reliable purchase starts with asking the right questions and refusing the shortcuts that put people and projects at risk.
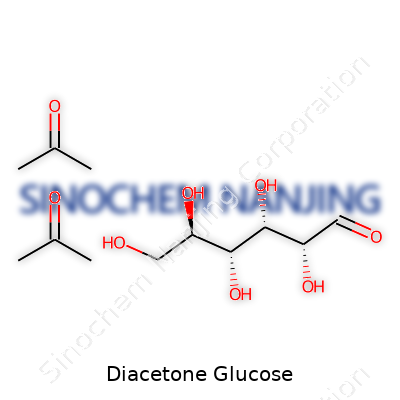
| Names | |
| Preferred IUPAC name | (1E,3R,4S,5S,6R)-3,4,5,6-tetrahydroxy-2-methyleneheptanal |
| Other names |
DAG 2-Isopropylidene-D-glucose |
| Pronunciation | /daɪˌæsɪˈtoʊn ˈɡluːkoʊs/ |
| Identifiers | |
| CAS Number | 7258-79-7 |
| Beilstein Reference | 2537485 |
| ChEBI | CHEBI:17827 |
| ChEMBL | CHEMBL2158886 |
| ChemSpider | 20278448 |
| DrugBank | DB14371 |
| ECHA InfoCard | 03d9b932-ea97-42e4-97e3-b2ad5244f534 |
| EC Number | 41035-61-8 |
| Gmelin Reference | 86323 |
| KEGG | C06707 |
| MeSH | D-glucose, 1-deoxy-1-(hydroxymethyl)-1-(1-methylethyl)-, (R*,R*)-(+-)- |
| PubChem CID | 12315850 |
| RTECS number | SL8650000 |
| UNII | 1W6HPC93H2 |
| UN number | 1321 |
| Properties | |
| Chemical formula | C12H22O7 |
| Molar mass | 226.25 g/mol |
| Appearance | White crystalline powder |
| Odor | odorless |
| Density | 1.12 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.4 |
| Vapor pressure | <0.01 mmHg (20°C) |
| Acidity (pKa) | 12.18 |
| Basicity (pKb) | pKb: 4.00 |
| Refractive index (nD) | 1.431 |
| Viscosity | 15-25 cP at 25°C |
| Dipole moment | 2.99 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 396.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1144.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3715.8 kJ/mol |
| Pharmacology | |
| ATC code | A11CC06 |
| Hazards | |
| GHS labelling | GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | No hazard statements. |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 144°C |
| Autoignition temperature | Autoignition temperature of Diacetone Glucose is 410°C |
| LD50 (median dose) | LD50 (median dose): >2000 mg/kg (rat, oral) |
| NIOSH | Not Listed |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Diacetone Glucose: Not established |
| REL (Recommended) | 0.03 |
| Related compounds | |
| Related compounds |
Dextrose Fructose Mannose Sorbitol Isomaltulose |

