Decabromobiphenyl: Yesterday’s Solution, Today’s Concern
Tracing the Footsteps: Historical Development
Decabromobiphenyl first arrived on the scene in the 1970s. At that time, industries clamored for robust flame retardants, especially as plastics, electronics, and synthetic textiles flooded the market. Chemical companies got busy. They pitched decabromobiphenyl to furniture manufacturers, appliance companies, and building suppliers. Eyes fixated on the promise of fire protection, not the lingering questions that trailed behind about longevity or safety. For decades, people trusted that layer of chemical defense. Today, environmental agencies and health researchers see that early trust as misplaced, a reminder that safety rules often lag behind scientific discovery. There’s always a lesson in hindsight: don’t rush chemicals into everyday life without a closer look.
Get to Know the Compound: Product Overview
Decabromobiphenyl is all about structure—a pair of benzene rings locked together, each carrying a load of bromine atoms. It’s not subtle. Manufacturers counted on those ten bromines doing a lot of work, throttling fires by cutting off oxygen in the event of ignition. The pure stuff comes as a white to pale yellow powder, odorless and almost stubbornly insoluble in water. It doesn’t break down easily in nature, either. Shelf life never posed a problem for anyone involved—longevity almost became a selling point, at least for industry. Looking back, that permanence signals trouble. Once loose, decabromobiphenyl sticks around in soil, plants, animal tissue, and human bodies.
Physical & Chemical Properties That Matter
This molecule weighs in at about 961.2 g/mol, carrying its bromines like a badge. It only gives in to decomposition at temperatures above 300°C. In water, it barely dissolves, with a solubility close to zero—about 0.0001 mg/L at room temperature. The structure keeps it from moving around easily through the environment, but it’s happy to hitch a ride on soil particles or settle into sediment. Fatty tissues can soak it up, and it builds over time, especially in animals higher up the food chain. Heat resistance grabs most headlines, but stability poses new risks. The visible dust—fine, persistent—draws extra concern for workers.
Decoding the Specs: Technical Standards and Labeling
It doesn’t take an industry insider to understand that technical details set the bar for product quality. Material safety data sheets give away most of what you need: minimum purity around 97%, trace amounts of lighter brominated biphenyls, no heavy metals above strict limits. Regulations from places like the EU and US flag decabromobiphenyl as a substance of high concern—any movement, storage, or disposal requires transparent documentation and tight control. Labels have to shout warnings—chronic toxicity, likely persistence, bioaccumulation. Workers keep a close eye on those sheets, but the wider public usually misses these signals.
The Making of Decabromobiphenyl: Preparation Methods
Making decabromobiphenyl always meant reaching for elemental bromine and a biphenyl base. Large vessels, sometimes glass-lined to resist corrosion, handled biphenyl dissolved in solvents like carbon tetrachloride or dichlorobenzene. Bromine got pumped in under tight temperature control, sparking off a direct halogenation. Each reaction run pumped up the number of bromine atoms, stepwise, until all ten spaces got filled. Refining the final product called for multiple rounds of washing—first with water, then acids, sometimes solvents again—to peel away byproducts and free bromine residues. By the time plant operators got their hands on the end product, impurities stuck below industry thresholds, but process waste haunted nearby water and air.
Chemical Reactions and Tweaks
Chemists quickly realized that decabromobiphenyl stayed mostly inert under regular conditions. That helped when fire risks loomed, but hampered clean removal or breakdown. Some research groups tried debromination under intense UV light or catalytic conditions, hoping to cut the molecule down to something less persistent. High temperatures worked, but not gently—byproducts remained uncertain, some even more troublesome. Attempts to chemically modify decabromobiphenyl for easier disposal continue, but success in the lab doesn’t always scale up. There’s no simple undo button for a structure built to last.
Names Change, Molecule Remains: Synonyms and Product Names
No shortage of tags for this compound: Decabromodiphenyl, BDE-209, Dekabromobifenyl, Diphenyl decabromide, DBDP, and trade handle names litter product catalogs from decades past. Even though regulatory crackdowns pushed most companies to shift away, older commercial names still surface in international trade, legacy inventories, or mislabeled warehouse stock.
Staying Safe: Operational and Safety Rules
Years ago, plants often ran with little real protection. Dust masks, gloves, and basic vent hoods formed the mainline defense. These days, serious personal protective equipment—full respirators, chemical-resistant suits, industrial ventilation—get written into the job rules. Waste management stays strict: incineration with advanced scrubbers, never landfill, and certainly never down the drain. Health agencies recommend workers undergo regular blood tests to pick up early warning signs of exposure. Training covers chemical handling, spill response, emergency decontamination, and safe storage. Lapses in protocol can cost lives and livelihoods, both inside and outside the plant gates.
Everyday Uses: Application Area
Old building foam, electric cable insulation, car interiors, and some upholstery materials still contain decabromobiphenyl. Electrical engineers liked how it stopped fire in circuit boards. Upholsterers appreciated its invisible strength—one less thing to think about during furniture assembly. Plastic manufacturers felt shielded from fire code violations. Over time, strict bans closed the door on manufacture and use in many places, but imported goods, landfill waste, and stockpiled materials keep it circulating. People who never heard the name live with traces of it every day, whether in dust, food, or recycled foam.
Bridging Gaps: Research and Development
Once, R&D teams pushed hard to make production cleaner, yields higher, and byproducts lower-toxicity. The late 1990s brought new marching orders. Scientists now search for better, green alternatives, some based on minerals or new phosphorus chemistry. Engineers design filters and scrubbers to trap decabromobiphenyl at the source before it escapes. Analytical chemists improve detection in food, dust, and water, able to find parts per trillion where once only gross contamination caught notice. Teams test safer waste destruction—supercritical water oxidation, plasma incineration, or chemical reduction—hoping to shrink the toxic legacy. Each improvement requires investment and transparency, qualities sometimes missing from risk management conversations not so long ago.
Digging Into Harm: Toxicity Research
Scientists learned that small, regular exposure adds up in the body. Decabromobiphenyl acts like a stowaway, slipping into fat cells and hanging around for years. Animal studies on rodents point toward liver and thyroid disruption, with some trials hinting at neurodevelopmental effects in offspring. Wildlife samples often reveal levels higher upstream from manufacturing cities, with egg shell thinning in birds and fertility drops in mammals. Regulatory agencies now put it on their watch lists alongside better-known toxins like PCBs and dioxins. Humans face exposure from dust, old furniture, carpet padding, or workplace air. Risks aren’t always clear or immediate, but neglect only piles up the cost.
Looking Ahead: Where to Next?
Society finds itself in a bind: remove hazards, protect those exposed, and fix past mistakes without causing new ones. Some researchers push for large-scale remediation—cleaning up soils, rivers, and dumping grounds with cutting-edge chemistry. Community organizers urge wider medical screening in at-risk towns. Lawmakers write stricter rules for legacy waste and restrict future use. Industry partners, some pressured by public campaigns, shift to safer substitutes or reinvest in green chemistry. The fight for environmental justice continues, with residents near old plants demanding cleanups and comprehensive health tracking. The challenge demands honesty, scientific rigor, and a commitment to put health ahead of convenience. Trust only returns with proof of progress—something earned one careful step at a time.
What is Decabromobiphenyl used for?
A Look at Why Decabromobiphenyl Shows Up in Everyday Life
People who make electronics, textiles, and construction materials have long turned to flame retardants for added safety. Decabromobiphenyl is one of those chemicals with a mouthful of a name, but it has played a big role in the way some products resist fire. If you’ve picked up an old TV or an older office chair with foam padding, you’ve probably handled something treated with this substance. Its main job? Slow things down when materials catch a spark, giving everyone more time to react.
Growing up in a household with older electronics, I ran into the distinct odor that sometimes gave away the presence of fire retardants. Decabromobiphenyl stands out as a classic example among polybrominated biphenyls (PBBs). It helped meet fire safety standards in places with strict rules about flammable materials. The same properties that make plastics and textiles tougher against flames have kept this chemical in industry for decades. Factories favored it for easy mixing and a record of stretching out ignition times, especially in items molded with plastics like ABS or polystyrene.
Looking Past Safety: The Health and Environmental Trade-Off
Ask a parent with young kids crawling on carpets why chemical flame retardants set off alarm bells, and you’ll get a quick answer: what helps in a fire often raises questions about safety. Recent research highlights how PBBs, including decabromobiphenyl, leak into dust and can spread beyond the products themselves. I recall my own worries years ago when an old office chair started fraying and I realized why it never quite smelled fresh—flame retardant particles linger long after manufacture. The Environmental Protection Agency and international agencies flag these chemicals as persistent in both the environment and our bodies.
Animal studies have found that high levels of exposure affect thyroid function, create problems for the liver, and may cause cancer. In people, the evidence isn’t always crystal clear, but long-term risks give public health experts a reason to pay attention. PBBs don’t break down quickly. They travel through water and air, finding their way into soil, wildlife, and even our food.
Finding Safer Solutions and Cleaner Alternatives
Regulations have started to catch up: Decabromobiphenyl now falls under bans or heavy restrictions in places like the European Union. Looking for better options, companies explore safer flame retardants or rethink product design altogether. My experience watching the rollout of new eco-friendly electronics showed that it’s possible to cut down on risky chemicals by using different plastics or barrier materials.
Real progress will rely on transparency. Shoppers, builders, and recyclers deserve better information about what goes into everyday products. Efforts like product labeling, public databases, and third-party certifications push industries to lean on science for safer picks. I’ve seen parents come together in community groups and demand furniture makers reveal what chemicals they’re using. Good data and open conversation lead to smarter choices, both in factories and at home.
By turning toward science-backed materials, following strong regulations, and pushing for honest labeling, society can do a better job protecting health without giving up fire safety. Clearer information, continued research, and advocacy keep future generations safer from the hidden risks in the products we trust.
Is Decabromobiphenyl hazardous to human health?
What Is Decabromobiphenyl?
Decabromobiphenyl belongs to a group called polybrominated biphenyls (PBBs). These chemicals once played a big role in fireproofing plastics, textiles, and electronic equipment. Nobody can deny the usefulness of keeping household goods from going up in smoke, but the hidden costs come from the persistent bits left behind in materials and in the environment.
How Decabromobiphenyl Enters Human Life
Every time old electronics or furniture break down, PBBs like decabromobiphenyl can escape. Dust and small particles get around in the home and outdoors. People face risk not only from direct contact but also from eating food grown in contaminated soil. Researchers found these compounds don’t go away quickly. Instead, they travel through water, air, and food chains, stacking up in fat tissues over time.
What Science Says About Health Impacts
My interest in environmental health started from watching family and neighbors live near waste sites. Reports of animals with birth defects and farmers with strange illnesses have a way of making the problem real. Decabromobiphenyl is not just a technical term; health agencies have tracked clear effects. Animal studies show that high amounts cause liver and thyroid problems, and they mess with hormone signals. The World Health Organization and other big health groups do not downplay these connections. For humans, too much exposure leads to similar trouble: thyroid disease, problems with memory, growth delays in children, and possible links to some cancers.
The Evidence: Not Just Theory
Facts don’t come only from the lab. In the 1970s, a huge mistake in Michigan added PBBs to cattle feed by accident. After that, meat, milk, and other products spread chemicals throughout homes in the region. Families paid the price for decades. Cases like this give a face to what might otherwise read as technical jargon. Health studies from these decades point to more babies born underweight, changes in behavior, and a buildup of toxins in people’s blood.
What Can Be Done
Governments learned some hard lessons. Many countries stopped using PBBs like decabromobiphenyl in the 1970s and 1980s. Some places support regular checks for contaminated sites, especially where people grow food or let kids play. At home, people throw out old electronics and furniture properly, instead of burning or dumping them, to limit new exposure. On a large scale, there is pressure for clearer labeling of chemicals in products. If factories substitute dangerous flame retardants with safer ones, lives improve over the years.
Nobody can erase mistakes from the past, but sharing information and pushing for clean-up and safer products help guard families now and in the future. Access to simple facts about chemicals like decabromobiphenyl shouldn’t be the privilege of scientists only. Everyday people deserve to know what they face and what works to protect their health.
What are the physical properties of Decabromobiphenyl?
Staring Down at a White, Crystalline Solid
In the world of industrial chemistry, plenty of substances try to stay out of the spotlight. Decabromobiphenyl comes with a subtle reputation, mostly sitting on shelves as a white, fine crystalline powder. You can spot it by its snowy color and almost waxy texture. It doesn’t give off much smell, which, honestly, always brings some reassurance in the lab. The powder flows gently between your fingers, never sticky, never clumpy. It’s dense, but pour it into a beaker and you’ll see just how easily it settles. That comes from its high molecular mass, weighing in at a hefty 963.2 g/mol.
Melting Point and Heat Worries
The heat resistance of decabromobiphenyl stands out. It won’t even think about melting until around 345 degrees Celsius. This resilience keeps it stable when blended into plastics or other materials working under tough conditions. If you ever get the urge to see it burn, prepare for disappointment. Its flash point sits above 250 degrees Celsius, which means house fires or standard industrial mishaps won’t bother it much. That high melting threshold is exactly what led manufacturers to use the stuff in making flame retardants for electronics and furniture.
Living with Solubility and Density
Toss a handful in water and nothing happens. Water beads off decabromobiphenyl like rain off wax. This insolubility sounds harmless, but it means spills linger in the environment. If it winds up in lakes or soil, you can forget about a rainy day washing it away. I remember a case where a factory worker lost a bag of this powder in a spill near a storm drain. That small mistake turned into a months-long cleanup because standard solvents wouldn’t budge it.
In denser liquids—organic solvents like toluene or acetone—you’d have to work hard to force it to dissolve. This stubborn behavior comes from its tightly bound bromine atoms clinging to the biphenyl core. It practically hugs onto those atoms, refusing to break apart in most chemical baths. Density checks in near 3.2 g/cm³, which makes it heavier than many organic compounds and ensures it always sinks to the bottom, whether it’s a beaker, a vat, or a pond.
Hardiness in Light and Air
Some chemicals break down just from sitting in sunlight or air. Decabromobiphenyl ignores both. That hardheaded attitude gives it staying power wherever it’s placed. On one hand, this property keeps its flame-retardant effect going strong inside old furniture foams and electrical insulation. On the other hand, it means the environment struggles with it. Once out in the open, ultraviolet rays barely scratch the surface, and even oxygen can’t really get inside to break it apart.
Why Physical Properties Matter for Health and Safety
People sometimes forget that the density, melting point or insolubility of a substance aren’t just tags for a textbook—they shape how we deal with it. I’ve seen colleagues slack off on wearing masks during powder handling. Turns out, tiny bits can get airborne despite the heft, and factory dust collectors struggle with powders that don’t clump. A dense, non-volatile powder seems less threatening, but inhalation or dermal contact build up over time.
Decabromobiphenyl’s resistance to breaking down means families and wildlife near old industrial sites still find traces years after industries move away. Scientists started noticing high bromine residues in some rivers back in the 1980s. These cases show why understanding these physical traits helps build smarter safety plans, tighter regulations, and pushes the search for safer substitutes that don’t stick around so long or hit health so hard. Investing in serious dust management, regular monitoring, and research for less persistent alternatives isn’t a luxury—it’s something we’ve learned the hard way.
How should Decabromobiphenyl be stored and handled?
The Risks We Live With
Decabromobiphenyl does a real job in fire prevention, helping to lower risks in electronics and textiles. Destined for wide industrial use, it is one chemical that demands solid respect. Breathing it in, letting it touch your skin, or eating anything with residue can set off a series of health problems—think irritation, or worse, long-term impacts on the nervous system. I once talked to a safety manager who said the only mistake made on a site with this chemical is not taking it seriously. That message deserves repeating.
Keep It Locked Down
Every storage site I’ve visited trusts common sense—secure storage means a tight-lidded container, kept out of direct sunlight or heat. Think cool and dry, about the same conditions where you’d put a can of paint you plan to keep for years. Store away from food, animal feed, or break areas. You never want this powder being tracked through a building or wafting around an unfiltered warehouse. Someone once tried stashing it too close to an oxidizer, not realizing how much those two love to wreak havoc. Separate chemicals with a reputation for mischief, and use secondary containment just in case of leaks.
Transportation Takes a Team
Trucking this stuff around isn’t as simple as tossing it into a delivery van. Anyone loading or unloading should suit up—gloves, a proper respirator with P3 filters, goggles, and a lab coat or Tyvek suit. I’ve seen decent workplaces skip the face shield, and that’s when I hear about eye problems. If you spill powder, sweep it up without stirring the dust, use a HEPA vacuum if possible, and never wash it down a drain. No matter how rushed things get, spills get flagged to a supervisor immediately. Cleanup kits need to remain nearby, checked monthly for expiration or missing parts. Documents travel with the shipment, including Material Safety Data Sheets, so no handler is left in the dark.
Personal Safety on the Line
In one shop, we ran a safety drill that saved a worker’s eyesight. Wash stations and showers aren’t decorations—they’re lifelines, and they must sit close enough for someone to reach in seconds, not minutes. Training should echo through the halls. Workers learn exactly how to spot warning signs in themselves or others—skin reddening, breathing trouble, or dizziness. Training sticks when leaders show up, repeat, and live out safety protocols. A company owner who briefly let his team relax their gear rules found out through health monitoring just how fast problems can develop.
Legal and Reputation Risks
Some countries restrict decabromobiphenyl under programs like REACH or TSCA. Failing an inspection leads to fines and a public image mess. Transparent labeling, routine record audits, and up-to-date permits are the best insurance. Ignore the rules, and the phone can start ringing with angry regulators or, worse, lawyers.
Better Practices, Real Results
If you’re improving an old site, start with ventilation upgrades. Airflow changes everything, lowering what workers and neighbors breathe. Substitute nonhazardous chemicals where science allows, and put worker health at the front. Invite local fire departments for walk-throughs, so in a crisis, no one loses time to confusion. Smart storage and sharp training carry real weight, not just for regulatory boxes but because people matter far more than paperwork.
Is Decabromobiphenyl regulated or restricted in any countries?
What Is Decabromobiphenyl and Why Should We Care?
Decabromobiphenyl—a name that hardly rolls off the tongue—usually lives inside plastics, electronics, and older textiles. Chemically, it belongs to a group called polybrominated biphenyls (PBBs), used to reduce flammability. Around the late 1970s and ‘80s, researchers and everyday workers started noticing odd health issues appearing in places that used these chemicals. Birth defects, cancer risk, learning problems, and immune disorders started showing up—especially in communities close to factories using PBBs. Scientists soon realized that these substances don’t break down easily and can travel through food, water, and even breast milk. Once people began to understand just how far decabromobiphenyl could reach, governments finally took action.
How Has the World Responded?
Some countries acted quickly when risks became known. The United States banned manufacturing and use of PBBs, including decabromobiphenyl, in the late 1970s, after one of the worst food contaminations in Michigan’s history. Decabromobiphenyl and other PBBs got mixed into cattle feed, sickened livestock, and contaminated tons of food. That crisis pushed federal action which mostly eliminated PBBs from U.S. products. Europe later moved under the REACH Regulation and the Stockholm Convention—both restrict or ban the intentional use of decabromobiphenyl. Sweden and Germany called for action even before EU-wide bans, putting human and environmental safety first. Japan treated it as a substance of high concern and backed it out of production standards.
Why Do These Bans Matter?
People often forget how chemicals sneak past regulations in many countries. Old electronics sent overseas for recycling can leak decabromobiphenyl into the soil and rivers. Children and workers in low-income countries pay the price. Studies in Nigeria and India revealed high levels of these chemicals in soil and even in breast milk samples. My own work in community health clinics has shown that “legacy” chemicals keep returning, long after industries leave a region. Recycling centers often sit near neighborhoods, spreading risks to families. Taking on those old stocks and waste imports calls for stronger international rules and support.
Lessons Learned—and What Needs to Happen Next
No law works without real enforcement. Plenty of places still use or import secondhand plastics filled with banned flame retardants. Bans can’t just sit on paper. Education for small businesses, community warning systems, stricter waste export checks, and upgraded lab testing make a real difference. The Stockholm Convention led to new monitoring and screening projects, but every country needs to follow through. For people living near e-waste processing hubs—from the outskirts of Accra to villages in Southeast Asia—this isn’t abstract policy, it’s daily life. Letting polluters evade responsibility punishes the families least able to fight back.
Building Safer Alternatives
Safe replacements exist. More manufacturers now pick halogen-free flame retardants with better human and environmental profiles. Those changes only stick if governments reward safer innovation, back up bans with resources, and share how these chemicals move through trade routes. Repair shops and recycling cooperatives need clear instructions and protective gear. Transparency and accountability transform rules into actual progress. For anyone who has had to clean up after a chemical crisis, these details can mean the difference between a safe neighborhood and years of sickness.
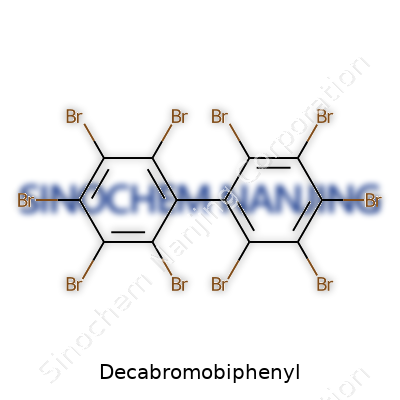
| Names | |
| Preferred IUPAC name | 5,5′,6,6′,7,7′,8,8′,9,9′-Decabromo-1,1′-biphenyl |
| Other names |
Decabromodiphenyl Biphenyl, decabromo- DecaBB Decabromobiphenyl (DBB) |
| Pronunciation | /ˌdiː.kəˌbroʊ.moʊ.baɪˈfiː.nɪl/ |
| Identifiers | |
| CAS Number | 13654-09-6 |
| Beilstein Reference | 12060595 |
| ChEBI | CHEBI:83418 |
| ChEMBL | CHEMBL43055 |
| ChemSpider | 21502 |
| DrugBank | DB14485 |
| ECHA InfoCard | 03b6bde8-51fc-4e38-8e8c-36bd4a894046 |
| EC Number | 201-236-9 |
| Gmelin Reference | 1200227 |
| KEGG | C18541 |
| MeSH | D003662 |
| PubChem CID | 6616 |
| RTECS number | KN0350000 |
| UNII | Y4D51Q8ME6 |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C12Br10 |
| Molar mass | 628.228 g/mol |
| Appearance | White to off-white crystalline powder |
| Odor | Odorless |
| Density | 3.36 g/cm³ |
| Solubility in water | Insoluble |
| log P | 8.9 |
| Vapor pressure | 1.13E-7 mm Hg at 25 °C |
| Acidity (pKa) | 14.95 |
| Magnetic susceptibility (χ) | -1300.0e-6 cm³/mol |
| Refractive index (nD) | 1.770 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 0.713 kJ·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | ΔfH⦵298 = 63.3 kJ/mol |
| Pharmacology | |
| ATC code | V09AX04 |
| Hazards | |
| Main hazards | May cause damage to organs through prolonged or repeated exposure |
| GHS labelling | GHS02, GHS07, GHS08, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H351: Suspected of causing cancer. |
| Precautionary statements | P261, P273, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-1-0-NA |
| Flash point | > 316°C |
| Autoignition temperature | > 482 °C |
| Lethal dose or concentration | LD50 (oral, rat): >15 g/kg |
| LD50 (median dose) | LD50 (median dose): > 5,000 mg/kg (rat, oral) |
| NIOSH | SN9000000 |
| PEL (Permissible) | 1 mg/m3 |
| REL (Recommended) | 0.02 mg/m³ |
| IDLH (Immediate danger) | NE (Not Established) |
| Related compounds | |
| Related compounds |
Polybrominated biphenyl Hexabromobiphenyl Polychlorinated biphenyl |

