D-Xylose: A Close Look at Origins, Properties, Uses, and Challenges
Historical Development
Back in the late 19th century, curiosity drove German scientist Emil Fischer to isolate D-xylose from wood. Biochemists soon realized this simple sugar played a hidden role in the structure of plant cell walls. As industries looked for new raw materials, D-xylose’s presence in hemicellulose made it a target. By the 1970s, the food and beverage sector saw potential in D-xylose’s low-calorie sweetness, and its production ramped up. What started as a chemist’s oddity turned into a staple for folks seeking sugar alternatives, but with every new application, researchers needed to answer new questions about safety, purity, and function.
Product Overview
D-xylose often shows up in food science as a naturally occurring pentose sugar. Chemically, it’s a monosaccharide with five carbons, found in fruits, vegetables, wood, and straw. Refined D-xylose typically appears as a white, crystalline powder. Its faint sweetness draws manufacturers looking for low-calorie sweeteners, but it brings something else: prebiotic value for gut health. In my lab days, keepers of pilot plants would brag about D-xylose’s ability to sidestep spikes in blood sugar, bringing extra attention from anyone working with diabetic or ketogenic formulations.
Physical & Chemical Properties
D-xylose melts around 114°C and dissolves easily in water. It’s less sweet than sucrose, about 40% as sweet by weight, which suits it for subtle flavor backgrounds. In its D-form, it rotates polarized light to the right—a signature check for quality control staff. D-xylose ferments if left with the right bacteria or yeast, splitting into ethanol and CO2. For anyone who’s looked inside a sugar refinery, that fermentation pathway doesn’t just limit D-xylose’s shelf life; it opens up biofuel research, too. On the chemical front, its five-carbon backbone lets it play in more reactions than the familiar six-carbon glucose.
Technical Specifications & Labeling
Regulations carve out strict lanes for D-xylose in food and pharma. Food-grade D-xylose needs to be at least 99% pure, with moisture below 1%. Heavy metal content—think lead, arsenic, mercury—stays under strict limits, in line with Codex Alimentarius and FDA language. On ingredient panels, D-xylose often pops up as “wood sugar” or by its scientific name. Labels flag that it comes from corn cobs, birch, or other plant sources, answering demand from allergen-conscious shoppers. In pharmaceutical use cases, documentation trails get longer, with full CoA (Certificate of Analysis) provided for GMP auditing. From my time consulting, the burden always lands on the manufacturer to show the chain of custody from biomass to bagged product.
Preparation Method
Extraction takes patience and detailed process engineering. Most D-xylose enters the market through acid hydrolysis, targeting xylan-rich plant material: corn cobs, straw, hardwood chips. The plant matter undergoes a dilute acid treatment, breaking xylan into D-xylose monomers. Refining the product means running multiple crystallization and filtration steps, then purifying with activated carbon. Some of the best yields I’ve seen came from small changes in acid strength and residence time—a classic industrial balance between cost, purity, and environmental impact. Enzymatic methods, using xylanase to cut up xylan chains, look easier on harsh chemicals, though scale-up has lagged behind in many places.
Chemical Reactions & Modifications
The backbone of D-xylose gives chemists room to play. You can hydrogenate D-xylose to get xylitol, a sugar alcohol that gives chewing gum its famed tooth-friendly kick. Under mild oxidation, D-xylose converts to xylaric acid, a molecule with growing value in specialty synthetic applications. Dehydration leads to furfural, a platform chemical useful for resins and bio-based solvents. In the biotech world, D-xylose drives fermentation projects aimed at producing lactic acid, ethanol, or specialty enzymes. Those early breakthroughs in D-xylose bioconversion made their mark in field stations where every fraction of a percent yield mattered. If you work with polymers or fine chemicals, you likely know D-xylose as a building block, modified further to fit into more complex structures.
Synonyms & Product Names
D-xylose sometimes sits on ingredient lists as “wood sugar,” an echo of its industrial roots. Chemists refer to it as “D-xylopyranose” or “D-xyloside,” depending on the ring or open-chain form present. In commercial portfolios, you’ll find it branded according to the raw material source—corn-based versus birch-based D-xylose—or as a pharma grade, signaling extra purity for intravenous solutions and oral preparations.
Safety & Operational Standards
Production remains tightly regulated. Exposure limits protect workers from inhaling too much dust, a lesson learned in older, poorly ventilated sugar plants. Food safety agencies, including EFSA and FDA, recognize D-xylose as generally safe when used in foods, but purity always stays center stage. Each production batch wears a traceability badge: manufacturer IDs, lot codes, and expiration dates. Quality teams track all inputs, confirming the final product meets published specifications for ash, heavy metals, and microbial contamination. Training for staff doesn’t stop at the basics—emergency protocols require regular review. In practice, that means donning masks, gloves, and eye protection, especially where D-xylose dust ends up airborne around conveyer systems.
Application Area
D-xylose crosses several sectors. In food, it serves as a controlled-sweetness additive, especially in diabetic-friendly products and low-calorie syrups. Bakers use it in browning reactions, taking advantage of the Maillard effect to boost color and aroma in bread and cookies. Beverage makers slip it into prebiotic drinks, leveraging its ability to support beneficial gut bacteria. In the drug world, doctors use D-xylose tests to assess intestinal absorption, particularly in diagnosing malabsorption syndromes. Biofuel researchers ferment D-xylose-packed feedstocks into ethanol, pushing for higher economic returns from what was once labeled as agricultural waste. The pulp and paper field also sees D-xylose both as a challenge—since it can gum up processes—and as a potential profit center when isolated and sold.
Research & Development
Academic labs and private R&D groups continue to tinker with both production and novel applications. Genetic engineering opens new routes: yeasts and bacteria now process D-xylose more efficiently, which could mean less reliance on harsh chemicals and lower costs. Analytical chemists design more sensitive detection tools, making sure that D-xylose levels measured in foods and feeds stay accurate. Pharmaceutical research explores how D-xylose interacts with human metabolism, a sharp focus for anyone designing better diagnostic panels. In green chemistry, D-xylose forms the linchpin of new bio-based polymers and solvents. Startup companies often tout “cradle-to-cradle” pathways, extracting D-xylose without fossil-fuel inputs, drawing on funding set aside for next-generation circular economies.
Toxicity Research
Human studies show D-xylose passes through the digestive tract without triggering large blood sugar spikes. Some folks, especially those with rare metabolic disorders, may struggle to absorb or process it, which is why medicine tests use D-xylose loading to check for those problems. Toxicologists in the 1980s looked hard for side effects from high doses—so far, results suggest little risk for cancer or chronic disease. Still, long-term, high-dose exposure carries the risk of gut discomfort and diarrhea, much like other sugar alcohols. The safety picture stays clearer on the environmental side, where D-xylose breaks down rapidly and doesn’t linger in soil or water like synthetic chemicals.
Future Prospects
Demand for plant-based and low-calorie products keeps D-xylose in the innovation spotlight. Industrial biotechnology expects enzymes that work faster, cleaner, and under milder conditions, which would cut emissions from sugar factories. Food manufacturers see D-xylose as a partner for new prebiotic and functional products, especially in markets caring more about gut health and metabolic syndrome. The push for green chemicals makes D-xylose a sustainable building block, rich with possibilities in resins, biodegradable films, and natural flavor agents. Policy shifts toward climate neutrality add pressure to recycle more agricultural waste, potentially making D-xylose one of the pillars of the next circular bioeconomy. Research builds on decades of know-how, banking on that small five-carbon chain to spark new answers to energy, health, and sustainability challenges.
What is D-Xylose used for?
D-Xylose: More Than Just a Sweetener
People often overlook simple sugars like D-Xylose, but its applications reach far beyond a nutrition label. D-Xylose comes from natural sources such as wood, fruits, and corn cobs. At its core, D-Xylose provides a mild sweetness with fewer calories than table sugar, making it a staple for many food companies aiming to develop products for diabetics and those watching their sugar intake. With diabetes and obesity on the rise, especially in the United States, these alternatives can make day-to-day life easier for folks needing to manage their diets without giving up the flavor of sweetness.
How D-Xylose Influences Food and Health
Bakers and beverage makers use D-Xylose as a low-glycemic option. It doesn't spike blood sugar the same way regular sugar does, so it’s gained a dedicated following among people with type 2 diabetes. In my own experience, having relatives who are diabetic, ingredients like D-Xylose allow them to share in celebrations without feeling left out or risking health problems.
D-Xylose also stands out for its prebiotic potential. This sugar encourages the growth of good gut bacteria, supporting digestive health. Many nutritionists point out that a balanced gut often translates to better immunity and overall well-being. While most know about probiotics, fewer talk about what feeds those helpful microbes—D-Xylose steps in here, giving the gut bacteria the support they need.
From Lab to Everyday Life
D-Xylose supports innovation on several fronts, especially in biotechnology. Scientists rely on it to measure the absorption ability of the intestines. Physicians often use a D-Xylose test to help diagnose problems related to nutrient absorption—such as celiac disease or certain types of gut infections. Early diagnosis can shape the long-term health of someone who has unexplained digestive issues, which is something doctors deal with every day.
Another interesting use is in the production of xylitol, a sugar alcohol known for its dental benefits. Chewing gums and toothpaste manufacturers like xylitol for its ability to help prevent tooth decay, and all of it starts with D-Xylose. As parents, we look for ways to keep our children’s teeth healthy—in products featuring xylitol, we see D-Xylose working behind the scenes.
Challenges and Solutions
Not everything about creating and using D-Xylose is straightforward. One ongoing issue involves the cost and efficiency of extracting it from natural materials. Efficient processing methods can drive prices down, but that often depends on stable raw material supplies. Looking to the future, researchers are searching for newer, cleaner, and greener extraction techniques, hoping to make D-Xylose more accessible across different regions—especially where sustainable sourcing matters.
Some critics point out that not every claim around D-Xylose holds up under scrutiny. There’s a gap in research on long-term effects, especially if people use it daily in large amounts. This echoes a bigger need for transparency and quality control within the food industry. Independent lab testing and government regulation can help make the benefits of D-Xylose widely available without hidden risks.
Building Trust and Making Informed Choices
People deserve clear information about the foods and ingredients they use. D-Xylose won’t solve every health problem, but it offers choices for both consumers and manufacturers. As research progresses, it’ll become easier to separate genuine benefits from overhyped marketing claims. For now, anyone who wants to eat healthier or add variety to their diet can look at D-Xylose as a possible option, backed by science and guided by practical experience.
Is D-Xylose safe for human consumption?
What is D-Xylose?
D-Xylose pops up in ingredient lists on some processed foods—often in low-calorie sweeteners, baked goods, and diabetic-friendly items. It’s a naturally occurring sugar found in plants like corn cobs, birch, and even fruits such as strawberries. Researchers discovered it over a century ago, but interest has grown as food makers look for ways to cut calories without losing flavor.
Looking at the Evidence
A lot of research on D-Xylose has roots in dietary science. The U.S. Food and Drug Administration gives it "Generally Recognized As Safe" (GRAS) status, which carries its own weight. European authorities also allow its use in foods as an additive. Regular food-grade forms show up on shelves in Asia too. Studies in labs and on people show it digests differently from table sugar. Most of it doesn’t get absorbed—it moves through the body and ends up in urine instead of spiking blood sugar, which makes it popular for diabetic products.
D-Xylose sometimes gets used in nutritional tests. When doctors check if the small intestine absorbs nutrients well, they sometimes measure D-Xylose in urine after a drink—proof it can pass through the gut with little fuss. Side effects seem mild in these controlled settings, usually tied to unusually large doses. Some people report a bit of stomach upset or diarrhea at very high intakes—roughly triple what anyone eating regular food would run into.
Who Should Be Cautious?
Even with a good safety record, there are always exceptions. Kids usually do well with foods carrying D-Xylose, but infants and people with rare metabolic conditions should talk to their doctors before trying foods that lean heavily on specialty sweeteners. High consumption, mostly from supplements or pure powders—not the tiny amounts in an energy bar—could cause cramping or loose stools. Folks with kidney problems may want to ask about any new food ingredient. People taking certain medicines should check in with their clinician, just to avoid surprises from unexpected interactions.
Why Safety Still Matters
Food labels often say “natural” or “plant-based” in big letters, but not every plant sugar belongs in every diet. Even substances with clear safety records could pose risks to people dealing with allergies or underlying illnesses. Sometimes companies slip new sweeteners into products without fully explaining what long-term, steady intake does to the body over many years. That’s not a conspiracy theory—food science simply takes time to catch up with everyday habits.
I’ve learned to trust small steps: trying new foods in reasonable servings, reading labels, and asking questions about any unfamiliar ingredient. In the age of wellness trends and sugar alternatives, knowledge and moderation win out over hype. D-Xylose stands out as one of the more studied plant-based sugars, but nobody should assume "safe" means "perfect."
Where Do We Go From Here?
Regulators need to keep an eye on how people use D-Xylose as diets evolve and food technology moves forward. Doctors and nutritionists can report side effects they notice in their patients. Food producers should keep sharing data on ingredients, including long-term safety studies, not just short-term tests in healthy volunteers. In the end, people benefit most from honest labels and open communication—something that feels more important now than ever.
How does D-Xylose differ from other sugars?
What Makes D-Xylose Different?
D-Xylose pops up as an ingredient in all sorts of foods, especially in products meant for people keeping an eye on their blood sugar. Unlike regular table sugar, which sends blood glucose higher just moments after eating, D-Xylose avoids causing those sharp spikes. This helps health-conscious folks feel a bit more at ease about their dessert or drink choices.
The best thing about D-Xylose: the body doesn’t treat it quite like other sugars. D-Xylose belongs to the group of “pentose” sugars, which means it only has five carbon atoms in its molecular chain instead of the six tucked inside glucose and fructose. That small difference shapes much more than you’d expect. When I tried baking with D-Xylose, I noticed the dough didn’t brown the way it does with table sugar. Fewer Maillard reactions means lighter bakes, which points to its different structure and how it interacts with proteins and heat.
What Happens Inside the Body
Most people who eat D-Xylose find it passes through their digestive system without a fuss. The small intestine only absorbs a small portion, and most of the sugar exits unchanged. For people like me who have diabetic relatives, choosing a sweetener that doesn’t nudge up insulin or blood sugar counts matters a lot. Studies show that D-Xylose barely registers on the glycemic index, so it doesn’t create the rollercoaster effect of common sugars.
There’s also less risk of tooth decay with D-Xylose. Bacteria in the mouth struggle to use it as fuel. Regular sugar, on the other hand, gives mouth bacteria everything they need to churn out acids and carve out cavities. D-Xylose takes away that power, leaving smiles safer in the long run.
Where D-Xylose Comes From
Most D-Xylose comes from plant sources—typically wood, straw, or corncobs. Instead of getting squeezed out of sugar cane or beet, D-Xylose is gently separated from xylan-rich plants. Looking at the bigger picture, using agricultural byproducts helps keep waste down. That gives it a sustainability edge compared to many sweeteners, making it an appealing choice for folks who put the planet high on their list of priorities.
Room for Growth and Caution
Though D-Xylose is generally safe, eating too much at once can lead to mild stomach upset, especially in kids. A handful of studies also note that people with certain digestive or genetic issues may process it differently. Like most things in modern nutrition, moderation stays as the golden rule.
As more shoppers look for blood sugar–friendly alternatives, food companies are starting to mix D-Xylose into low-calorie candies, baked goods, and drinks. That option gives people more freedom, especially those watching their diet closely.
Digging Deeper Into Sugar Options
Everyone wants to enjoy treats without paying the price with their health. D-Xylose offers a way to keep the sweet moments happening while watching the impact on blood sugar and teeth. For anyone shopping for alternatives that come from renewable sources and skip the blood sugar bounce, D-Xylose stands out as a reliable pick.
Can D-Xylose be used by diabetics?
Understanding D-Xylose and Blood Sugar
D-Xylose keeps showing up in sugar-free foods, which has a lot of people with diabetes asking questions. It comes from wood and some fruits, and the food industry grabs it because it’s a sugar with less impact on blood sugar. The thing is, not all sweeteners work the same in the body. Diabetes means paying attention to everything eaten, even what goes into a cup of tea.
Digestion and Absorption
Unlike regular table sugar, the body only absorbs some of the D-Xylose in the gut. A chunk of it gets flushed out, so blood sugar spikes less than with glucose or sucrose. Studies show most people won’t see huge blood sugar jumps after a D-Xylose snack, but that doesn’t mean there’s zero effect. The remaining D-Xylose finds its way to the bloodstream, so the pancreas still does a bit of work.
Why Label Claims Matter
Supermarkets carry plenty of snacks boasting “sugar-free” on the label, sometimes using D-Xylose to get that claim. The FDA and other health bodies look at glycemic index (GI) for context. D-Xylose has a low GI, around 7, while table sugar is up around 65. That sounds reassuring until you realize labels sometimes skip all the details. Real-world eating doesn’t happen in a lab, and snacks rarely come with a chart explaining what happens next in the bloodstream. People with diabetes know a safe sweetener for one person won’t always be a free pass for someone else.
Personal Experience and Trust in Brands
I remember testing my blood sugar with different sweeteners out of caution. My numbers didn’t climb much after a drink sweetened with D-Xylose, but I noticed effects could stack up if I mixed in other carbs the same day. The body sometimes surprises us. I learned to scan ingredient lists for hidden sources of starches or sugars, since just one can shift everything. Friends in the diabetes community swap notes on how a new sweetener works for them; trust comes from experience more than from marketing.
The Research So Far
The science teams studying D-Xylose count a lot on short trials. They give a dose, watch blood sugar, and publish numbers. One review in the European Journal of Clinical Nutrition found that people taking D-Xylose instead of glucose had tamer blood sugar rises. The American Diabetes Association also finds low-GI sweeteners useful, but only as part of an overall plan. Long-term effects get less attention. If a person’s body handles D-Xylose in a certain way, that’s meaningful, but healthcare still needs more studies, especially around regular use and kidney health.
Practical Solutions and Takeaways
For diabetics, D-Xylose offers another tool—like stevia, monk fruit, and erythritol—but nobody can treat it as magic. Testing with a glucose meter, sharing results with doctors, and checking for new research all matter. Companies need to label D-Xylose honestly. If food makers want trust, they can share clear, GI-tested info, not just marketing claims. Health care providers help by teaching patients to experiment safely and look out for symptoms. D-Xylose expands choices, but learning how it works in a real body, meal by meal, sets the real limit.
What are the side effects of D-Xylose?
What Is D-Xylose Used For?
D-Xylose draws attention in the health field for good reason. It often comes into play during breath tests doctors use to check digestive function, especially when looking for malabsorption or issues with carbohydrate digestion. The body normally absorbs it quickly, so any changes in how much shows up in your urine point to a possible problem with your small intestine. Some supplement makers also use it as a sweetener with fewer calories than sugar.
Possible Side Effects of D-Xylose
Most people tolerate D-Xylose quite well. The body handles small amounts, like those found in the typical test dose, without trouble. Still, every chemical has a flipside because we all process things differently. After talking with healthcare providers and reading medical studies, side effects tend to be mild but worth knowing.
Digestive upset is the most common issue. Some folks end up with bloating, gas, or stomach cramps after taking D-Xylose in test doses. Loose stools or diarrhea can happen if you take more than your body can handle. I’ve met a few patients who felt queasy or needed the bathroom urgently during a hospital xylose test—nobody had a serious threat, but it can still be rough at the time. If the gut is already sensitive, symptoms could show up faster or feel more intense.
Blood sugar swings rarely show up because D-Xylose isn’t metabolized the same way as regular glucose, but people with diabetes notice mild increases after taking it. Its caloric load might be lower, yet it’s always smart to check with a healthcare provider before adding anything new to your daily routine, especially with a medical condition in play.
Allergic reactions seem rare. In theory, an allergy could happen—itching, rash, or even swelling. I haven’t seen one myself, and there aren’t many reports in the medical literature, but it’s never out of the question. Anyone with a known allergy to sugars or sugar alcohols should mention it to their doctor before testing.
Taking high doses, far beyond what you find in a regular breath test, hasn’t been studied much. Large quantities may bring unknown risks, especially for the kidneys and liver, since those organs process chemical leftovers. The FDA lists D-Xylose as generally recognized as safe (GRAS) when used as a sweetener, but more research would help clarify its long-term safety, especially if it becomes popular in food products.
Why Side Effects Matter
Doctors rely on D-Xylose tests for diagnosing real problems—like celiac disease or pancreatic disorders—so knowing the risk profile matters. If you can’t handle the sugar, the test results may not be valid, or you may end up with unnecessary discomfort. Clear conversations with your doctor help steer things in the right direction. Mention allergies, gut sensitivities, or other conditions before the test. For anyone considering sweeteners for weight control, a slow approach works best. Start with small doses and see how your body responds. If unusual symptoms show up, get medical advice right away.
Researchers still watch for rare effects as doctors use D-Xylose in bigger populations. The medical community updates guidance as soon as new risks or benefits surface. Sticking to recommended doses, staying honest with your healthcare team, and tracking your body’s response all protect your health while you learn more about this sugar.
| Names | |
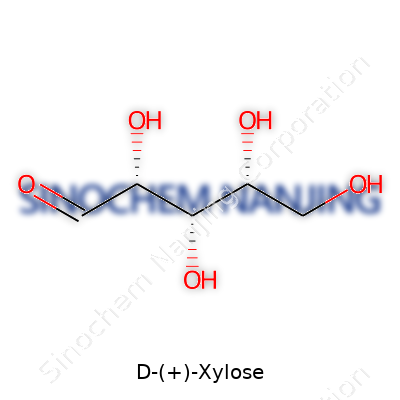
| Preferred IUPAC name | (2R,3R,4R)-2,3,4,5-tetrahydroxypentanal |
| Other names |
D-Xylose Wood sugar Xylo-pentose Xylopyranose Xylofuranose |
| Pronunciation | /ˈdaɪ.zaɪ.loʊs/ |
| Identifiers | |
| CAS Number | 58-86-6 |
| Beilstein Reference | 1720249 |
| ChEBI | CHEBI:28694 |
| ChEMBL | CHEMBL1231318 |
| ChemSpider | 5367 |
| DrugBank | DB00155 |
| ECHA InfoCard | 100.038.257 |
| EC Number | EC 200-416-4 |
| Gmelin Reference | 60759 |
| KEGG | C00181 |
| MeSH | D-Xylose |
| PubChem CID | 135191 |
| RTECS number | ZE0175000 |
| UNII | Z8IFR0R69U |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID3023083 |
| Properties | |
| Chemical formula | C5H10O5 |
| Molar mass | 150.13 g/mol |
| Appearance | White crystalline powder |
| Odor | sweet |
| Density | D = 1.520 g/cm3 |
| Solubility in water | 1 g/mL (20 °C) |
| log P | -2.00 |
| Vapor pressure | <0.01 hPa (20 °C) |
| Acidity (pKa) | pKa = 12.15 |
| Refractive index (nD) | 1.638 |
| Viscosity | 1.185 mPa.s (25 °C, water solution, 10%) |
| Dipole moment | 2.56 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -971.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2341 kJ/mol |
| Pharmacology | |
| ATC code | A09AA02 |
| Hazards | |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P264, P270, P301+P312, P330 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Flash point | 93 °C |
| Autoignition temperature | 410 °C |
| Explosive limits | Explosive limits: 7–36% |
| Lethal dose or concentration | LD50 oral rat 10,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 10,000 mg/kg (Rat, oral) |
| NIOSH | SR2975000 |
| PEL (Permissible) | 15 mg/m³ |
| REL (Recommended) | 0.09 g/kg bw |
| Related compounds | |
| Related compounds |
Arabinose Xylulose Ribose Lyxose Glucose Mannose |

