D-Galactal: A Commentary on Its Role and Future
Historical Development
Scientists unraveled the puzzle of D-Galactal long before modern labs, tracing its origins from curiosity about rare sugars. Chemists looking to understand the building blocks of biomolecules focused on how slight shifts in sugar structure could mean huge differences in biological roles. In the nineteenth and early twentieth centuries, sugar chemistry drew sharp minds, driving breakthroughs in isolation, crystallization, and purification. D-Galactal came out of that history, shaped by pressure from industry and academia to get to grips with carbohydrate chemistry. Its story follows advances in reaction control, chromatographic separation, and structural determination—which eventually gave researchers a focused look at its unique double bond and utility in synthesis.
Product Overview
D-Galactal stands out because it serves both as a research tool in academic settings and finds use in specialized chemical industries. The molecule is not mass-produced for broad commercial markets; rather, it addresses precise needs in pharmaceutical synthesis, biochemical research, and the design of glycosylated drugs. People working in synthetic chemistry appreciate D-Galactal for its ability to provide access to more complex carbohydrate molecules. The shape and functionality make it a practical precursor in the hands of a skilled chemist, serving as both a substrate and a building block. In my experience, it usually arrives as a white crystalline powder in tightly sealed bottles, often with detailed technical pamphlets on handling and purity metrics.
Physical & Chemical Properties
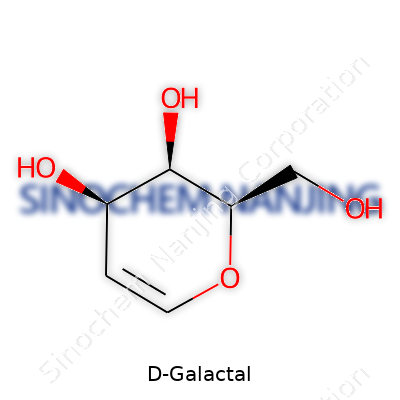
D-Galactal carries a formula of C6H10O5. The structure features a cyclohexene core with one end double-bonded, a key difference from fully saturated analogues like galactose. Density lands it on the lighter side, and it shows decent solubility in water and polar organic solvents. Don't expect it to have much of a smell or color. D-Galactal’s reactivity sets the stage for its popularity in organic synthesis; the enol ether function serves as both a target and a handle for chemical transformations. Melting points hover in the range familiar to seasoned bench chemists—typically well above room temperature, yet sensitive enough that direct sunlight or excess moisture would spoil a good batch.
Technical Specifications & Labeling
Suppliers mark their bottles with batch numbers, precise molecular weight, physical state, melting point, and a recommended storage temperature, usually advising cold, dry environments. Labels include the chemical names, hazard pictograms, risk phrases, and emergency protocols. Product sheets usually display purity percentages, with leading brands showing figures above 98 percent. My own lab work has shown how much difference a single percent of impurity can make on downstream reactions, with chromatographic purity tests backing up these labels.
Preparation Method
The pathway to D-Galactal starts most often from D-Galactose. Chemists dry galactose under vacuum, convert it to its corresponding acetylated or halogenated forms, and then treat it with strong bases or elimination reagents under controlled temperature. Getting high yield without byproducts comes from practice, close attention to time, and pH controls. My practical experience with this process underscores the importance of clean glassware and proper temperature ramps—cutting corners leads to lower yields and purification headaches.
Chemical Reactions & Modifications
D-Galactal shines for chemists interested in glycosylation. The enol ether region acts as a reactive site, taking on nucleophilic additions, oxidation, or electrophilic activation. This property has opened up doors for new carbohydrate drugs as well as for studies of how sugars interact on cell surfaces. Adding protecting groups or shifting substituents alters function and reactivity, letting researchers build up libraries of analogues for screening. Experiments I’ve run show that minor changes in catalysts or reaction time can spell the difference between a productive run and a tanked reaction. A sharp eye for minor side products makes or breaks scale-up.
Synonyms & Product Names
People in labs or ordering catalogs often see D-Galactal listed under alternate names such as 2,3,4,6-Tetrahydroxyhex-1-en-5-al, Hex-1-enopyranose, or simply as Galactal. A few distributors stamp their own brand names or short codes, but chemists working at the bench mostly stick to systematic or accepted trivial names to avoid confusion during ordering or synthesis planning.
Safety & Operational Standards
D-Galactal doesn’t jump off the table as a high-hazard compound, but standard chemical hygiene remains fully in play. Eye protection, gloves, and fume hoods guard against any inhalation or accidental skin contact. Safety data sheets detail fire compatibility, spill protocols, and compatibility warnings—particularly important since strong bases or acids can decompose the compound and create side products. I also pay attention to waste management and avoid putting any leftover material down regular drains. Labs with ISO certification run handling and storage procedures by the book, conducting risk assessments before scaling up experiments.
Application Area
Most D-Galactal finds its way into the world as a synthesis intermediate for specialized glycosylated molecules, especially in drug and vaccine research pipelines. It serves as a platform for making modified sugars that mimic pathogens’ surface markers, feeding into both basic research and applied immunology. Analytical labs use it to probe enzyme specificity or develop standards for carbohydrate analysis. My time in research groups has shown growing demand for custom derivatives in oncology and immunology, with partners in pharma keen on the way sugar decorations alter drug function and targeting.
Research & Development
Scientists push the boundaries with D-Galactal by tweaking synthetic pathways for cleaner, greener reactions, and by exploring completely new transformations at the enol ether site. A flood of literature covers novel catalysts, new chiral auxiliaries, and more sustainable solvent systems. University-industry partnerships drive these efforts by seeking ways to scale up without losing purity or selectivity. Graduate students and postdocs contribute by running new reactions every week, logging side products and successes in the hope of publishing or patenting advances that shave time and money off the synthetic process.
Toxicity Research
Researchers have tested D-Galactal through cell culture and animal models, searching for signs of acute or chronic toxicity. So far, the compound does not trigger alarm bells—especially at the low concentrations common in research work. Still, chronic exposure or accidental ingestion has not been ruled out for all use cases, so labs rely on safety data and close monitoring. Studies continue so regulatory agencies can set tight controls and prevent unexpected side effects in scaled-up industrial settings. Most chemists, myself included, take a “better safe than sorry” approach—maintaining vigilance even with low-risk compounds, tracking disposal, and logging exposure according to best practice guidelines.
Future Prospects
Looking ahead, chemists and biotech companies aim to unlock more efficient D-Galactal syntheses, with eyes on renewable feedstocks and greener reagents. The role it plays in medicinal chemistry, especially for designing carbohydrate drugs and vaccines, seems likely to grow as precision medicine carves deeper paths into individual treatment. Analytical tools will probably improve detection and manipulation of D-Galactal derivatives in complex samples. As automation and AI-driven synthesis planning push deeper into labs, D-Galactal stands out as a nimble candidate for rapid prototyping and reaction screening—something younger scientists and seasoned professionals alike can appreciate for its versatility and reliability at the bench.
What is D-Galactal used for?
What Makes D-Galactal Worth Noticing?
Walk into any decent biochemistry lab and someone will be talking about sugars. D-Galactal stands out in this conversation. This rare sugar isn’t something you’ll spot at the grocery store or sprinkle on cereal, but it quietly supports a huge chunk of life science research and industry. My own time working with carbohydrate chemistry made that clear. D-Galactal often gets called the “starting block” when scientists want to build more complicated molecular structures or understand the details of glycoscience.
Where D-Galactal Shows Up in Research
The first thing people learn about D-Galactal: it acts like a special building block for making oligosaccharides. Oligosaccharides decide a lot about how cells stick together, communicate, and react to disease. Researchers love D-Galactal because of its double bond between carbon 1 and 2. That makes it more reactive for certain chemical reactions. So, if you want to create special sugars for studying immune responses or investigating how viruses recognize cell surfaces, you need D-Galactal.
Enzyme research often leans on D-Galactal too. Glycosyltransferases help stitch sugars together. By using D-Galactal to test enzyme functions, labs get clues on how these processes work in living cells. These insights have real impact. Take vaccines, for example. Fine-tuning the sugars attached to proteins can change how our immune systems react. D-Galactal helps tune this process in the lab, laying groundwork for next-generation vaccine design.
Pharmaceuticals and Diagnostics
When pharmaceutical scientists talk about D-Galactal, they’re usually interested in how it helps make “glycosylated” drugs. Many drugs today mimic natural molecules, especially complex sugars. Some cancer therapies, for instance, depend on sugar molecules that can only be built with the help of building blocks like D-Galactal. Without it, some drugs just wouldn’t work the same. I once spent weeks troubleshooting a synthesis that always stalled—until someone suggested using D-Galactal to kickstart the reaction.
Diagnostics can’t ignore D-Galactal either. Modern test kits in hospitals sometimes need sugars modified in just the right way to stick to disease markers. Using D-Galactal helps make those test molecules sharp and selective. For diseases like cancer or rare metabolic disorders, test accuracy can decide treatment plans. The details matter at this level.
Making D-Galactal Sustainable and Affordable
One challenge: D-Galactal is tough to produce at scale. Most of it starts as D-galactose from dairy or plant material and then gets chemically tweaked. This process can leave behind a trail of waste and eat up lots of energy. Green chemistry fans—including myself—think about new enzyme processes or better catalysts to produce D-Galactal with less impact. The dream? Factory-style setups using renewable materials, slashing costs, and shrinking the environmental footprint.
Looking Ahead with Smart Collaboration
D-Galactal’s story isn’t just about chemistry. Universities and health tech companies have reason to cooperate so new production methods actually move from the lab into regular manufacturing. Grants, open data, and transparent patent strategies help push things along, making D-Galactal more available for breakthroughs in research and medicine. If history tells us anything, it’s that collaboration beats isolation—especially for molecules that punch above their weight.
What are the main benefits of D-Galactal?
Why D-Galactal Catches Attention in Research and Industry
D-Galactal doesn’t show up in grocery store aisles or trend on nutrition blogs. Still, for anyone invested in science or medicine, it stands out. Researchers like it because it packs a rare blend of reactivity, accessibility, and versatility. In labs, D-Galactal serves as a gateway for making complex sugars—valuable in pharmaceuticals, vaccines, and diagnostics.
The backbone of D-Galactal, a six-carbon sugar with a unique double bond, makes it a prized starting material. Chemists reach for it to build glycoconjugates: molecules that often play central roles in drug delivery and immune system research. Manipulating this molecule opens doors for developing new therapies for cancer and infectious diseases. Looking at cancer vaccine research, scientists break apart and recombine sugars similar to D-Galactal to create mimics of tumor markers, hoping to train the immune system to spot and destroy cancer cells.
A Building Block That Saves Time and Money
Anyone who’s spent hours (or days) in an organic lab appreciates how a reactive molecule can speed up tough syntheses. D-Galactal’s structure can help a chemist avoid long, fiddly preparation steps that often bog down research. Instead of jumping through hoops to build up a sugar from scratch, researchers can start off close to their target. This cuts down both the number of chemical steps and the risk of losing yield along the way. I remember running a reaction with galactal back in grad school—the “Aha!” moment happened when the product formed in a single, smooth motion, no desperate column chromatography needed. In tight budgets and short timelines, that’s gold.
Medicine Needs Sugars—The Right Sugars
Sugars aren’t just about sweet flavors. Our own cells—and the bugs that attack them—use specific sugar patterns to communicate and hide from the body’s defenses. D-Galactal helps researchers make identically shaped sugars (so-called “glycosides”), which are crucial for designing vaccines, antibiotics, and other advanced medicines. For example, many bacterial surfaces host rare sugars. To create vaccines or diagnostics against these bacteria, scientists need to build those rare sugars in the lab. Trying to harvest them from natural sources isn’t practical on a large scale, so D-Galactal becomes an entry point for these complex syntheses.
Food, Function, and the Path Forward
Some might think these sugars are only useful in sterile labs, far away from real-life problems, but that’s not true. Sugar-based molecules designed from D-Galactal form part of new treatments and rapid diagnostic tools. They’re showing up in next-generation vaccines, such as those targeting meningitis and pneumonia. For anyone tracking antimicrobial resistance, the ability to innovate new antibiotics by tweaking sugar molecules is a major perk. Research teams at top universities and biotech startups are pushing D-Galactal derivatives further—into enzyme inhibitors and antiviral compounds too.
Possible Solutions to Roadblocks
Despite the promise, high purity D-Galactal isn’t always easy to source or store. It also needs careful handling to avoid unwanted reactions. Scaling up its production takes technical know-how and investment. There’s room for companies to create greener, more efficient manufacturing routes: using renewable feedstocks or enzyme-catalyzed processes could improve sustainability and drive prices down. Training more chemists in advanced sugar chemistry will spread access and spark more discoveries. Public funding and open-source research would help small labs jump into the game, keeping innovation healthy and moving forward.
How should D-Galactal be stored?
Understanding the Basics
D-Galactal isn’t just a random sugar, it has value in organic synthesis and sometimes in biochemical studies. If left out in the open without care, it loses reliability fast. D-Galactal doesn’t act up like a teenager, but it won’t last if each step of storage isn’t done intentionally.
Direct Experience: What Goes Wrong Without Thoughtful Storage
I remember working in a university lab where someone grabbed a bottle off a shelf, no label, and the once-fine white powder now looked clumpy, with yellow streaks. No one could trust it. D-Galactal, like so many sugars, draws in moisture from the air and turns sticky, then starts breaking down. Once that happens, accurate measurements become a guessing game, and research loses its backbone.
Temperature and Moisture: The Details Often Overlooked
D-Galactal stands best at cooler temperatures. Refrigeration beats room temperature. Science backs this up: heat speeds up reactions, including those that sap the quality of sensitive reagents. That means a fridge set around 2 to 8°C offers much more predictable shelf life.
Desiccators work well for labs lacking cold storage. Toss in fresh silica gel or a good desiccant packet inside the container, and moisture’s less likely to creep in. Putting D-Galactal in a tightly sealed glass vial or amber bottle cuts down on unexpected air exposure. Plastic bags or loose caps open the door to deterioration. If you see caking or notice odors, it’s time to toss that batch instead of risking mistakes in the project.
Light and Oxygen: Quiet Threats to Your Chemicals
Light-sensitive compounds always seem eager to make trouble, and in the presence of oxygen, degradation speeds up. Whenever possible, amber vials or aluminum-wrapped bottles help block UV rays. Short exposure here and there may not ruin an entire supply, but extended shelf life depends on consistency.
Oxygen exposure chips away at purity over time. After each use, cap the container tightly and keep it upright. Vacuum-sealing small portions pays off for longer-term storage—especially if you only use D-Galactal now and then. Every time a lid opens, the clock ticks a bit faster for the rest of the batch, so splitting larger stocks into smaller containers makes sense.
Supporting Data
Research shows that D-Galactal stored at 4°C in a well-sealed, moisture-free container, out of direct light, keeps its form and reactivity far beyond a month. Stability studies for sugars confirm what practice already teaches: each misstep in storage shaves off some of the product’s potential. Data from chemical catalogs and peer-reviewed studies back up these details, with consistent findings for related sugars.
Simple Solutions Work Best
Every lab has its own rhythm, but forethought about D-Galactal storage saves time and money in the long run. Small, airtight, labeled vials stay in a designated fridge or desiccator, set aside from more reactive chemicals. Never store open containers near strong acids or bases—they can trigger unwanted reactions. Handwritten records near the fridge log the date each lot came in and when it was last opened, so everyone knows what’s still good.
Safe storage practices for D-Galactal reflect common sense more than complexity: low temperature, dry air, protection from light, limited oxygen, and clear labeling. Fewer mistakes, clearer data, and less wasted budget—all from caring a little more where and how you keep your reagents.
Is D-Galactal safe for human consumption?
Understanding D-Galactal
D-Galactal sounds like something straight out of a biochemistry lecture. This sugar isn’t part of our typical diet. People might hear about it as a rare monosaccharide related to galactose, which you’ll find in milk and many processed foods. D-Galactal comes up most often in labs, not in kitchens or supermarkets. Its unique structure lands it in scientific research, especially in the chemistry of carbohydrates.
The Safety Conversation: What We Know
Safety questions always start with basic human trials and a big stack of paperwork from toxicologists and food scientists. D-Galactal doesn’t show up in food safety regulations on either side of the Atlantic—not in the European Food Safety Authority (EFSA) lists, nor in the U.S. Food and Drug Administration (FDA)’s GRAS notifications. Without these approvals, calling it safe would be reckless. Food scientists must show in animals and people that a compound doesn’t harm living tissue, mess with organs, or get involved in nasty metabolic paths.
Galactose, which looks like D-Galactal but isn’t the same thing, can cause problems in folks with galactosemia—a rare disorder. If D-Galactal behaves anything like its cousin, those worries may be worth considering. Data is thin, though, so hard numbers on absorption, breakdown in the gut, or long-term buildup don't exist. Eating rare monosaccharides in meaningful quantities hasn't been part of anyone's daily habits, so evidence just isn’t there.
Why Safety Matters
People might figure, “If it’s a sugar, it can’t be dangerous.” That gets risky fast. Even common sugars, taken in excess, do a number on our health. Anything out of the ordinary in the chemistry world, especially without robust food-grade testing, belongs nowhere near a lunchbox.
Mistakes in the past prove this point. Cyclamate, once touted as a safe substitute for sugar, led to cancer concerns and bans. Olestra, a fat substitute, caused stomach troubles for plenty of unlucky snackers. Just because a compound comes from a “natural” source or shares a name with safe substances doesn’t mean it comes without pitfalls.
Weighing the Evidence
Right now, peer-reviewed papers on D-Galactal cover mostly plant or microbial fermentation, or its utility as a building block for more complex molecules. No serious food safety studies stand up to scrutiny in databases like PubMed or ScienceDirect. The substance stays in test tubes and out of the everyday diet. That’s a gap that anyone hoping to introduce it to snack foods or supplements must close, fast.
Some folks might feel tempted to experiment with this rare sugar. Curiosity in chemistry can drive innovation, but eating something because it’s new or different doesn’t usually end well—especially without any published data to say that humans can break it down safely.
Smarter Paths Forward
If food companies or researchers see value in D-Galactal—maybe for sweetening, maybe for special functions—they’ve got to start with animal trials, move on to controlled, peer-reviewed human studies, and report everything transparently. Regulators look for evidence of no toxicity, how the body handles it, and how much people can safely consume.
People deserve clear labeling and honest information about what’s inside their food. Until then, D-Galactal belongs in the lab, not on the plate.
What is the purity level of the D-Galactal product?
Getting Real About Chemical Purity
D-Galactal pops up as a topic in research circles, from pharmaceutical labs to glycoscience platforms. The purity level plays a huge role in every result. For scientists, getting an impurity-riddled compound is like cooking with spoiled ingredients—the final outcome never matches the plan. Most expect that D-Galactal bought for synthesis or structural studies boasts a purity over 95%. More often, high-end providers offer 98% or even better. These numbers carry weight well beyond a tiny certificate or digits printed on a shipping label.
Purity Isn't Just a Fancy Metric
When a chemist measures D-Galactal, purity means less background noise and fewer contaminants that foul up sensitive reactions. Glitches in an NMR spectrum aren’t just annoying; they can throw off the interpretation and real progress screeches to a halt. Once, I spent days chasing down a “weird peak” until the supplier confirmed a batch with less-than-promised purity—wasted time and resources. Those who run analytical work know the headache when a 90% batch brings along ghost sugars, water, or organic leftovers.
In the pharmaceutical world, the stakes hit even higher. Traces of unwanted sugars or byproducts can alter biological assays or enzyme reactions. Testing for critical endpoints turns blurry. So a stated purity of 98% isn’t just a selling point—it's critical for reproducibility and real trust in the research. Think about emerging fields like glycobiology, where a single unexpected molecule can seriously mislead pathway discovery or vaccine design.
How Purity Gets Measured
Reliable suppliers check D-Galactal purity using detailed approaches. NMR spectroscopy helps spot structural imposters. HPLC lays out the real picture, showing how much of the sample fits the expected retention time. Mass spectrometry gives the final thumbs up by checking for the real molecular weight. Good providers publish their data, not just a sticker. Looking past the catalog sheet means reading spectral data or even asking for the report, because some sellers round up purity numbers in their marketing.
I always respect companies willing to share their batch-by-batch data with clear numbers. Sourcing D-Galactal from vendors with transparency means less stress for users in deep research settings. They also flag any possible sources of error—hydroscopic water, side products from incomplete synthesis, or breakdown from rough packaging. In my experience, even the temperature used in shipping changes the water content on delivery day.
How Users Can Side-Step Problems
Scientists, libraries, and startups don’t have to cross their fingers and hope. There’s power in running a quick check in the lab. NMR and TLC take only a Saturday morning and offer facts about the specific bottle in hand. Connecting with suppliers with strong reputations for open paperwork prevents headaches before a single pipette even moves.
The smart move: partner with vendors invested in batch traceability and consistent documentation. Don’t skimp or gamble for a slight price drop. From personal lessons, low-purity D-Galactal multiplies work effort, can waste expensive reagents, and tanks experiments. For real science and scalable processes, 98% isn’t a luxury—it's part of the foundation.
| Names | |
| Preferred IUPAC name | (2R,3S,4R,5R)-2,3,4,5-Tetrahydroxyhex-1-enal |
| Other names |
1,2,3,4,6-Penta-O-acetyl-D-galactal D-Galactal, 2,3,4,6-tetraacetate D-Galactal tetraacetate |
| Pronunciation | /diː-ɡəˈlæk.tæl/ |
| Identifiers | |
| CAS Number | 2596-97-2 |
| 3D model (JSmol) | `3d_jmol('C1=CC(O)C(O)C(O)C1O')` |
| Beilstein Reference | 344306 |
| ChEBI | CHEBI:37672 |
| ChEMBL | CHEMBL1217387 |
| ChemSpider | 5464267 |
| DrugBank | DB02472 |
| ECHA InfoCard | 100.018.853 |
| EC Number | 207-063-3 |
| Gmelin Reference | 87738 |
| KEGG | C01743 |
| MeSH | D-Galactal MeSH: "D-Galactal |
| PubChem CID | 441431 |
| RTECS number | MA9575100 |
| UNII | P6T3H3F4YY |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C6H10O5 |
| Molar mass | 164.16 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.555 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -2.22 |
| Acidity (pKa) | 12.08 |
| Basicity (pKb) | pKb: 6.87 |
| Refractive index (nD) | 1.464 |
| Viscosity | Viscous syrup |
| Dipole moment | 2.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 321.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2347 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | D11AX10 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | [H][C@H]1O[C@@H](CO)[C@@H](O)[C@@H](O)[C@H]1O |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Flash point | 72.3°C |
| LD50 (median dose) | LD50 (median dose): >5 gm/kg (oral, rat) |
| REL (Recommended) | 20°C |
| Related compounds | |
| Related compounds |
Galactose D-Galactose L-Galactal L-Galactose D-Glucose D-Mannose |

